Abstract
DNA amplification by PCR detects KIT exon 11 internal tandem duplications in canine mast cell tumors (MCTs). Tissue-specific inhibitors often contaminate DNA extracted from formalin-fixed, paraffin-embedded (FFPE) canine MCTs, blocking PCR amplification and, consequently, preventing mutation detection. We used a commercial kit to extract DNA from FFPE canine MCTs. Two independent PCR assays, each with one primer set, were used to amplify target genes (HPRT and KIT) directly after FFPE DNA extraction. PCR amplification failed with at least one primer set in 153 of 280 samples (54.6%, 95% CI: 48.8–60.5%). One or 2 DNA washing steps were required to remove PCR inhibitors in 130 of 280 (46.4%) and 23 of 280 (8.2%) of these cases, respectively. DNA concentration and quality (A260/A280 and A260/A230) either pre- or post-washing were not associated with ability of the samples to be amplified by PCR using both HPRT and KIT primer sets. Low-grade and subcutaneous MCTs were less likely to amplify directly after DNA extraction and without any washing steps compared to high-grade MCTs using KIT gene primers.
Dogs have a unique risk of developing cutaneous mast cell tumors (MCTs). Internal tandem duplications (ITDs) in exon 11 of the KIT proto-oncogene cause constitutive activity of the Kit protein, 10 contribute to mast cell malignancy, and are prevalent in up to 50% of high-grade MCTs.5,16 Veterinary diagnostic laboratories use PCR and gel electrophoresis to test DNA from high-grade MCTs for exon 11 ITDs. Dogs who harbor a KIT exon 11 ITD in their MCT, and are unresponsive to conventional chemotherapeutic treatments, or whose MCT is not amenable to wide surgical excision, are suitable candidates for treatment with tyrosine kinase inhibitors. Tyrosine kinase inhibitors block the abnormal autophosphorylating activity of the Kit protein caused by ITDs in exon 11 of KIT.11,14
Formalin-fixed, paraffin-embedded (FFPE) tissue archives are a valuable resource for retrospective studies of a plethora of diseases given the economic and space-efficient storage requirements of blocks in comparison to frozen materials. However, PCR amplification of DNA extracted from FFPE tissues can be problematic because of the variable and often high level of DNA degradation induced by formalin fixation. This limits amplification for most FFPE samples to small fragments of <250 base pairs (bp).1,4 Various techniques have been investigated to optimize extracted DNA integrity and quality for downstream molecular analysis. Type of fixative, length of fixation, extraction method, length of enzymatic tissue digestion, and age of block impact the quality of the DNA end product, and all have been targets for optimization.1,6,8,15,17 Moreover, extraction of high-quality DNA from either FFPE or frozen tissue samples has been reported as peculiarly problematic for MCTs in comparison to other tumor types and normal tissues, most likely as a result of the co-extraction of unknown cell-specific PCR inhibitors.6,16,17
We investigated poor DNA amplification from 280 FFPE canine MCTs. FFPE canine MCT tissue blocks were sourced from the Veterinary Diagnostic Laboratory at the University of Adelaide (Roseworthy, SA, Australia; n = 16) and from Gribbles Veterinary Pathology commercial laboratories in Clayton (VIC, Australia; n = 175) and Glenside (SA, Australia; n = 89). A commercial kit (QIAamp DNA FFPE tissue kit; Qiagen, Hilden, Germany), which has been previously determined to yield good quality FFPE-extracted DNA in comparison to other techniques, 7 was used to extract DNA from a 20-µm thick section of each FFPE MCT block. Xylene was used for deparaffinization of FFPE tissue sections, and DNA was extracted according to the manufacturer’s instructions, eluted in 60 µL of a 0.1 mM EDTA buffer supplied in the kit, and stored at 4°C for later use. The DNA was extracted between 2015 and 2016, and the DNA quantity (concentration) and quality (A260/A280 and A260/A230 absorbance ratios) were measured (NanoDrop ND-1000 spectrophotometer; Thermo Fisher Scientific, Waltham, MA). At the time of DNA extraction, 68 blocks were ≤1 y old, 189 blocks were 1–2 y old, and 23 blocks were 2–4 y old. Sample fixation times are unknown.
Each sample underwent PCR amplification using 2 different primer sets (Table 1). Primers for the reference gene hypoxanthine-guanine phosphoribosyltransferase (HPRT) were used to determine whether the DNA was of sufficient quality to generate a small PCR product. Previously published primers were used to identify ITDs in exon 11 of the KIT gene. 5 PCR conditions were identical for both sets of primers, and reactions were performed as described previously. 16 Approximately 10–50 ng of sample DNA was added to each 20-µL PCR reaction. DNA extracted from frozen normal canine testis and water were used as positive and negative controls for PCR amplification, respectively. Resulting PCR products were analyzed on 2% agarose gel by electrophoresis.
Nucleotide sequences of primers used in PCR assays of canine mast cell tumors.
DNA that failed to amplify by PCR using either one or both primer sets underwent a “washing” step (DNA clean and concentrator kit; Zymo Research, Irvine, CA) per the manufacturer’s instructions. In brief, DNA samples were mixed with DNA binding buffer at a ratio of 5:1 and then transferred to a spin column in a collection tube, before centrifugation at 13,000 × g for 30 s. The columns were washed twice with 200 µL of DNA wash buffer, and each wash was followed by 13,000 × g centrifugation for 30 s. DNA was eluted in 40 µL of the 0.1 mM EDTA elution buffer supplied in the Zymo kit and the concentration measured on the NanoDrop. The samples then underwent a second PCR amplification test. Samples that still failed to produce a PCR amplicon underwent a second DNA washing procedure, a direct repeat of the first wash, followed by a third PCR test.
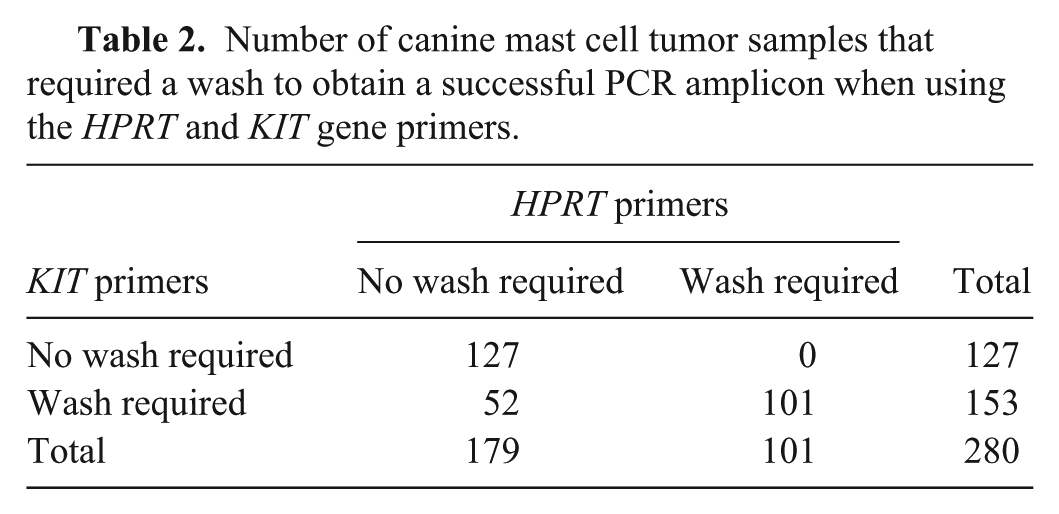
Using the HPRT primers, 179 of 280 (63.9%) samples were successfully amplified directly after DNA extraction. Eighty-six (30.7%) samples required a single wash, and 15 (5.4%) samples required 2 washes before amplifiable DNA was obtained. Using the KIT primers, 127 of 280 (45.4%) samples required no washing, 130 (46.4%) samples required a single wash, and 23 (8.2%) samples required 2 washes in order for amplifiable DNA to be obtained. Overall, 153 of 280 (54.6%, 95% CI: 48.8–60.5%) MCT DNA samples required at least 1 wash for successful PCR of both HPRT and KIT (Table 2).
Number of canine mast cell tumor samples that required a wash to obtain a successful PCR amplicon when using the HPRT and KIT gene primers.
Data were analyzed using SPSS statistical software (v.25; IBM, Armonk, NY). As determined by Pearson chi-square test, MCT DNA samples that required washing for successful PCR amplification by one primer set were predictably more likely to require washing for PCR amplification success when using the other primer set (χ² [1, n = 280] = 131, p < 0.001). The association between HPRT with KIT primer pairs for washing requirements was strong (φ = 0.684). There was no significant relationship between age of block and washing requirements when using either the KIT primers (p = 0.074) or HPRT primers (p = 0.262) as determined by a Pearson chi-square test.
Post-washing DNA concentrations and the A260/A280 and A260/A230 absorbance ratios varied from being more diluted to more concentrated than the original sample (Supplementary Table 1). There was no statistically significant relationship between ability to amplify the DNA using both HPRT and KIT primer sets and DNA quantity (concentration) or quality (absorbance ratios; p = 0.700), as determined by binary logistic regression analysis fitted with a Hosmer–Lemeshow model.
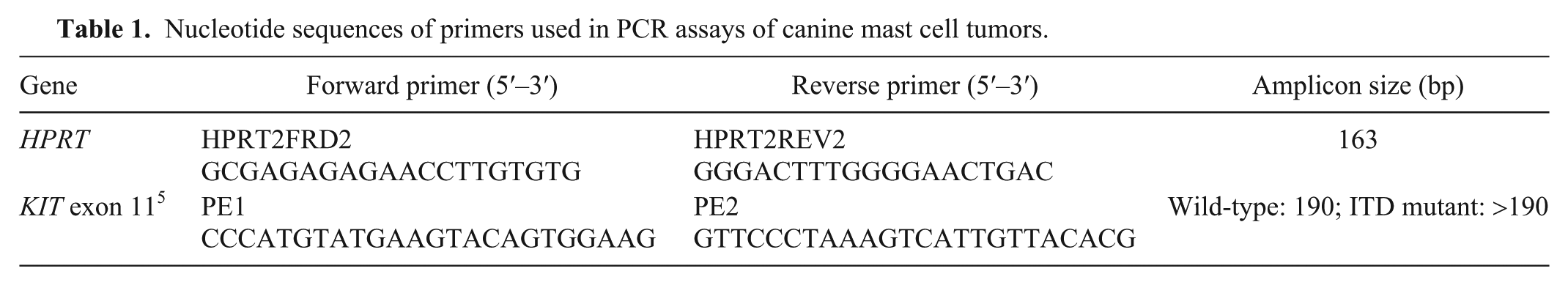
To further investigate the possibility that co-extraction of PCR inhibitors interfered with DNA amplification, we conducted HPRT PCR reactions on MCT DNA samples that required at least one wash for successful PCR amplification. Two PCR assays were prepared from a single master mix solution. The first assay was conducted according to the standard HPRT protocol in which 1 µL of 10–50 ng of MCT DNA was added to each reaction. The second assay was identical to the first, but each reaction was “spiked” with 1 µL of canine control DNA (concentration of 12 ng/µL) in addition to the 1 µL of MCT sample DNA. PCR products were analyzed on 2% agarose gel by electrophoresis (Fig. 1). Amplification of the positive control was inhibited in the unwashed but not the washed DNA. This supports a conclusion that contaminants existing in the MCT DNA solutions inhibit amplification by PCR but are successfully removed using the DNA washing method described herein.
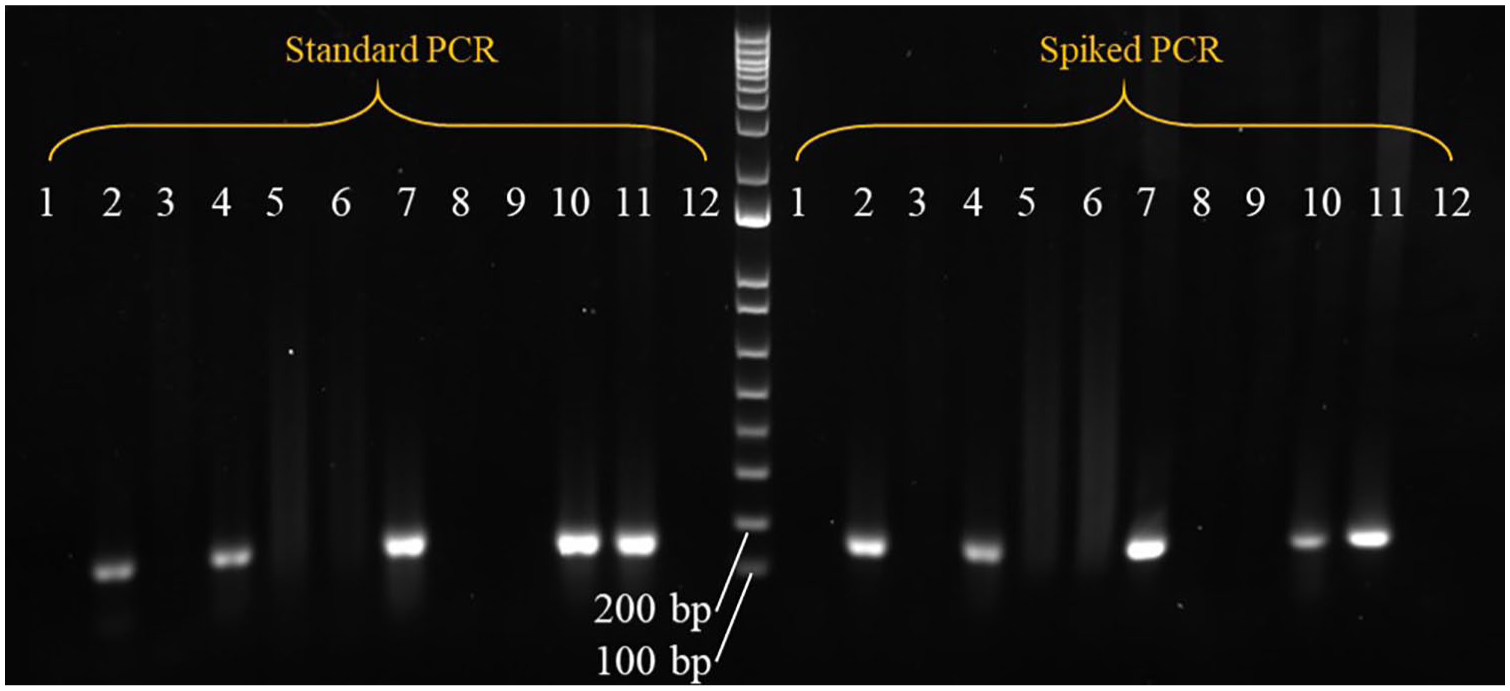
Agarose gel electrophoresis of genomic DNA extracted from 4 formalin-fixed, paraffin-embedded canine mast cell tumor (MCT) blocks, amplified by PCR using HPRT reference gene primers and run against a 1-kb DNA ladder marker. Expected product size is 163 bp. Two PCR assays were performed from a single master mix solution. The first assay amplified canine MCT DNA according to the “standard” PCR protocol. The second assay was exactly the same as the first except that each reaction, excluding the negative control, was “spiked” with wild-type canine testis positive control DNA in addition to the tumor sample DNA. Lanes 1, 2 = sample A86 with no wash and after one wash, respectively. Lanes 3, 4 = sample M186 with no wash and after one wash, respectively. Lanes 5–7 = sample M127 with no wash, after 1 wash, and after 2 washes, respectively. Lanes 8–10 = sample U47 with no wash, after 1 wash, and after 2 washes, respectively. Lane 11 = positive control (wild-type canine testis DNA). Lane 12 = negative control (water). Sample identifiers as per Supplementary Table 1.
The 280 FFPE tumor specimens were histologically classified as subcutaneous (n = 41, 14.6%) or cutaneous (n = 239, 85.4%) MCT by 2 board-certified veterinary pathologists (EC Dobson and L Woolford). The cutaneous tumors were graded according to 2 systems widely used in veterinary pathology.9,13 According to the Patnaik grading scheme, the cutaneous MCTs included 13 grade I (5.4%), 181 grade II (75.7%), and 45 grade III (18.8%) tumors. All Patnaik grade I and III tumors were graded as Kiupel low- and high-grade, respectively. Of the Patnaik grade II tumors, 171 of 181 (94.5%) were classified as Kiupel low-grade and the remaining 10 (5.5%) as Kiupel high-grade. Given the low numbers representing the Patnaik grade I class, only the Kiupel grading scheme was investigated in the subsequent statistical analyses.
A KIT exon 11 ITD was observed in 22 of 55 (40%) high-grade tumors and 2 of 184 (1%) low-grade tumors, with an overall prevalence in cutaneous MCTs of 10% (24 of 239; 95% CI: 6.2–13.9%). Additionally, 1 cutaneous low-grade tumor harbored a 21-bp exon 11 deletion (Fig. 2), and one of the 41 subcutaneous MCTs carried an ITD. A KIT exon 11 ITD mutation in a subcutaneous MCT has not been reported previously, to our knowledge. ITDs ranged from 25 to 60 bp duplicates and resided in the 3′-end of exon 11, with 9 ITDs extending into intron 11 (Fig. 3). The largest ITD extended 35 nucleotides 5′ of the reverse primer and, therefore, ITD mutations are not suspected to impede primer ability to amplify the DNA.
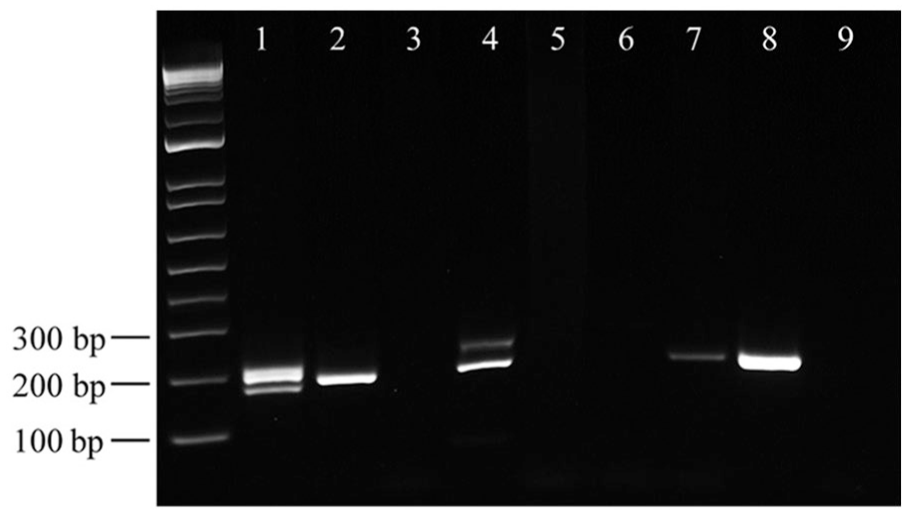
Agarose gel electrophoresis of genomic DNA extracted from 4 formalin-fixed, paraffin-embedded canine mast cell tumor blocks amplified by PCR using KIT gene primers and run against a 1-kb DNA ladder marker. Wild-type PCR product is 190 bp, internal tandem duplication PCR products are larger (represented by the upper bands in lane 4), and deletion mutant product is smaller (represented by the lower band in lane 1). Lane 1 = sample M29; 21-bp deletion. Lane 2 = sample A1; wild-type KIT. Lanes 3, 4 = sample M25 with no wash and after one wash, respectively; ITD mutant. Lane 5–7 = sample M107 with no wash, after 1 wash, and after 2 washes, respectively; wild-type KIT. Lane 8 = positive control (wild-type canine testis DNA). Lane 9 = negative control (water). Sample identifiers as per Supplementary Table 1.
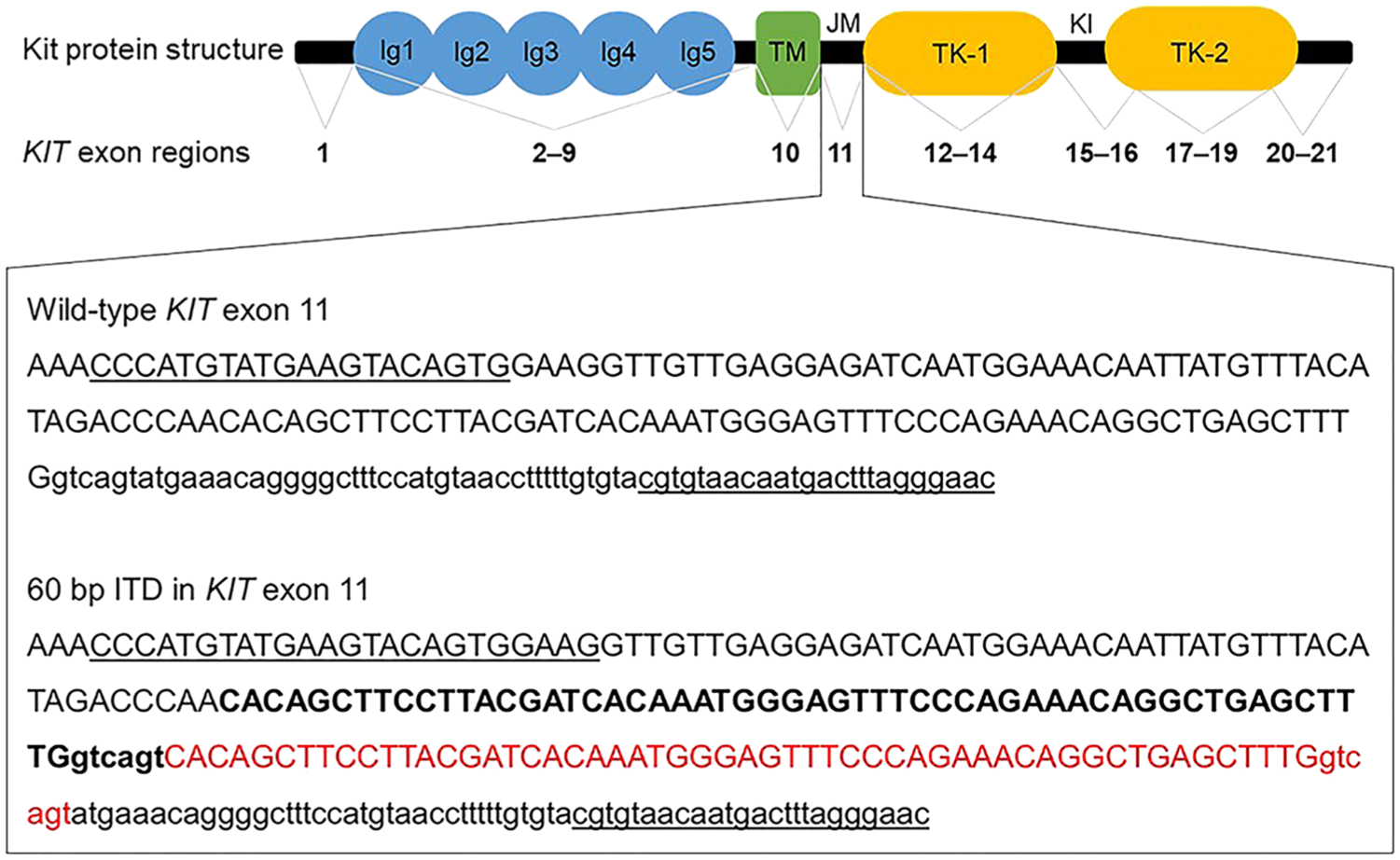
Structure of canine Kit protein and corresponding KIT gene exon regions. Noted are the immunoglobulin-like domains (Ig1–5), transmembrane domain (TM), juxtamembrane domain (JM), tyrosine kinase domains (TK-1, TK-2), and the kinase insert (KI). The zoomed-in textbox represents the wild-type DNA sequence of exon 11 (uppercase text) and part of intron 11 (lowercase text) from canine KIT gene. Primer sequences are underlined. Also shown is the DNA sequence of a 60-bp internal tandem duplication (ITD) detected in a mast cell tumor. The region of the KIT sequence that is duplicated is in boldface, and the duplication is in red.
When using HPRT primers, 17 of 41 (42%) subcutaneous MCTs, 71 of 184 (39%) cutaneous low-grade MCTs, and 13 of 55 (24%) cutaneous high-grade MCTs required at least 1 wash to obtain amplifiable DNA (Supplementary Table 1). A generalized linear logistic model with binary logistic regression fitted with a Wald chi-square analysis suggested no significant relationship between washing requirements for HPRT primers with tumor type or grade (p = 0.060). For KIT primer pair, 27 of 41 (66%) subcutaneous MCTs, 105 of 184 (57%) cutaneous low-grade MCTs, and 20 of 55 (36%) cutaneous high-grade MCTs required at least 1 wash to obtain amplifiable DNA. In pairwise comparisons, cutaneous low-grade (p = 0.005) or subcutaneous (p = 0.001) MCTs were significantly more likely than cutaneous high-grade MCTs to require at least one wash to obtain amplifiable DNA when using the KIT primer pair. Washing requirements for cutaneous low-grade MCTs and subcutaneous MCTs were not significantly different from each other (p = 0.167).
High-grade MCTs are commonly affected by local inflammation, ulceration, and tissue necrosis associated with spontaneous degranulation by tumor cells of bioactive molecules such as histamine, serotonin, serine proteases, eicosanoids, and cytokines. 3 It has been hypothesized that high-grade MCTs would experience greater PCR inhibition as a result of these factors. 2 However, our results do not support this hypothesis and instead show that the DNA from the more benign MCTs (i.e., cutaneous low-grade and subcutaneous MCTs) was more likely to require washing to enable successful PCR amplification of KIT. There is no clear explanation for this result. One possible explanation is related to the copy number of the KIT gene in high-grade tumors. MCTs with KIT mutations have been reported to have increased copies of the KIT locus on CFA 13. 12 An increased quantity of initial DNA templates might offset the effect of tissue inhibitors in some PCR reactions. However, this does not explain why HPRT but not KIT was amplified from 44 low-grade or subcutaneous samples without washing (Supplementary Table 1). In these cases, primer annealing properties or primary DNA structural properties in concert with PCR inhibitors may have contributed to the finding. For samples successfully amplified by HPRT primers directly after DNA extraction, 2-sample t-tests determined statistically significant differences in the mean DNA concentration (p = 0.016) and A260/A280 (p < 0.001) and A260/A230 (p < 0.001) absorbance ratios for samples requiring and not requiring a wash for successful KIT primer amplification. It is unclear whether post-washing DNA concentrations were the sole contributor to successful PCR amplification in these cases or if removing inhibitors also had an effect on outcome.
Our study is limited by lack of data regarding tissue sample processing. Time delay between surgical excision of the tumor and placement in formalin is unknown, and standard operating procedures regarding tissue processing likely varied between veterinary pathology laboratories. Discrepancies in these processes may have affected downstream molecular analyses. The thickness of FFPE sections taken for DNA extraction may have also contributed to the co-extraction of PCR inhibitors, although this has not been investigated. Nonetheless, the washing method described herein is effective in resolving this issue.
MCTs are the most common skin tumors in dogs, and high-grade tumors have a guarded prognosis given their aggressive biologic behavior. Detection of KIT gene mutants can guide therapy. Failure of PCR amplification using DNA from canine MCTs occurred in 54.6% of cases, presumably because of the combined effects of KIT gene copy numbers and the co-extraction of tissue-specific contaminants. This problem can be solved readily by our method described herein. The exact biological contaminants interfering with the PCR remain unknown, but our results suggest that they are more prominent in cutaneous low-grade MCTs and subcutaneous MCTs.
Supplemental Material
DS1_JVDI_10.1177_1040638719867743 – Supplemental material for DNA purification increases PCR-amplifiable DNA extracted from formalin-fixed, paraffin-embedded canine mast cell tumors for routine KIT mutation detection
Supplemental material, DS1_JVDI_10.1177_1040638719867743 for DNA purification increases PCR-amplifiable DNA extracted from formalin-fixed, paraffin-embedded canine mast cell tumors for routine KIT mutation detection by Vanessa S. Tamlin, Elizabeth C. Dobson, Lucy Woolford and Anne E. Peaston in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We are grateful to Gribbles Veterinary Pathology for supplying the FFPE MCT blocks, to Dr. Michelle Hebart for her statistical assistance, and to Dr. Cynthia Bottema for reviewing the manuscript. We also thank the anonymous reviewers for their insightful comments and suggestions.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
A grant from the Australian Companion Animal Health Foundation supported this work.
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
