Abstract
Twenty-nine canine cutaneous mast cell tumors (MCTs) were morphometrically analyzed with regard to mean nuclear area (MNA) using cytopathology smears. The results showed a correlation between MNA and survival. When graded into 2 morphometrically different groups, there were statistically significant differences among high- and low-grade MCTs, regarding both Romanowsky-type stain and hematoxylin and eosin. Cytomorphometry could also separate histologic grade II tumors with better prognosis from the more aggressive MCTs. The results indicated that nuclear morphometry on cytopathology preparations can predict the biological behavior of cutaneous MCTs in dogs in an independent manner, yielding a rapid and reproducible diagnosis, which renders the method useful for veterinary oncology.
Introduction
Mast cell tumors (MCTs) are among the most frequent types of canine neoplasias, representing approximately 20% of the cutaneous tumors. Sexual predisposition has not been reported, and etiology has not been identified. The most commonly affected breeds are Boxer, Boston Terrier, Bulldog, and Labrador Retriever. The mean age of animals at the time of diagnosis is 8.5 years. 21,25,32
This neoplasm is regarded as potentially malignant because of its extremely variable biological behavior. Several types of treatments have been studied, taking into consideration the differentiation grades proposed in the most frequently used grading system 27 since they are related to survival. Intraobserver and interobserver variations are considered the main difficulties in histopathologic grading, with differences in 40–60% of the cases analyzed by different observers, 14,17,24,26,34 which is probably due to the use of various parameters in association with the grading of lesions.
In an attempt to predict the behavior of MCTs, other prognostic indicators have been evaluated. The best results have been achieved with proliferation markers 1,3,18,31,33 and KIT expression patterns. 10,15,19,20,30,36,37 Among the methods proposed to complement histomorphologic grading, computerized morphology is worthy of notice. This procedure has already proved useful in several types of human cancer, 6,9,11,13 and its application in veterinary medicine has been increasing. 7,22,23,34
To reduce subjectivity and increase reproducibility in grading MCTs, some researchers have studied tumor characteristics of potential interest morphometrically, such as nuclear dimension and shape, as well as lesion depth. A previous study 34 found cytopathologic differences in the nuclear sizes of neoplastic mast cells among histopathologic grades of malignancy. Those differences have been confirmed in histologic sections. 22 The results suggest that nuclear morphology can be helpful in grading MCTs. Lesion depth, one of the criteria used by conventional histologic grading, was of no prognostic significance when independently assessed, 16 but it was one of the best prognostic indicators for disease-free interval and survival time in other studies. 28
Cytopathologic examination is a low-cost, rapid, and simple method for the diagnosis of cutaneous neoplasms. 4,32,35 However, there are no objective criteria for the cytologic grading of MCTs alone regarding malignancy, which forces clinicians and surgeons to submit tissue fragments for histopathologic analysis and subsequent grading. The aim of the present work was to investigate the prognostic value of nuclear morphometry on cytologic examination as an independent prognostic indicator of survival and mortality and to establish morphometry as a simple, rapid, and more objective method for the grading of canine MCT malignancy.
Materials and methods
Tumors
Twenty-nine histologically confirmed canine cutaneous MCTs were examined. Specimens of the neoplasms were obtained from dogs presented to the veterinary hospitals of the Faculdade de Medicina Veterinária e Zootecnia da Universidade de São Paulo (FMVZ-USP; São Paulo, Brazil) and Universidade Bandeirante de São Paulo (UNIBAN; São Bernardo do Campo, Brazil).
The cytopathology smears were prepared by the same person using nonaspiration fine-needle biopsy followed by squash preparation. 35 Two of the smears were air dried and stained with Romanowsky-type stain, a and 2 other smears were fixed in absolute alcohol for 24 hr and stained with hematoxylin and eosin (HE). 29 The neoplasms were fixed in 10% formaldehyde and routinely processed for histopathology. 29 Two 5-μm sections were obtained from each tumor, stained with HE, and submitted to 3 experienced pathologists for histopathologic grading according to previously published guidelines. 27 The final diagnosis was established by consensus of 2 or more observers.
Nuclear morphometric analysis
The cytologic smears were analyzed with a trinocular microscope b coupled to a digital camera system of 5.1 megapixel c resolution and a computer equipped for image analysis. Image-Pro Plus software was used for image analysis. d The calibration of measurements was performed with the aid of a micrometer ruler and a 100× magnification oil-immersion objective.
For each neoplasm, 200 nuclei (100 stained by the Romanowsky-type method, 100 with HE) were outlined by tracing their margins with the help of a computer mouse. To avoid interobserver variation, all measurements were performed by the same pathologist, without any information about the neoplasm. The fields of view were randomly selected, and all mast cell nuclei that were not superimposed and had sharp margins were measured for each digital image. The nuclear parameter used in the present study was area, which was equivalent to the area of the outlined object. In the case of animals with multiple MCTs, the value recorded for study was the highest obtained among the various neoplasms.
Clinical information
The cases of MCT were evaluated by physical examination of the animals, information from hospital reports, and interviews with owners and veterinarians. Criteria for inclusion in the present study included the availability of cytopathologic and histopathologic samples for morphometric analysis, confirmation of the MCT diagnosis, treatment with surgery alone, and complete clinical follow-up for at least 60 days after surgery.
Statistical analysis
The relationship between mean nuclear area (MNA) and survival time was verified by Spearman correlation. After a cutoff had been established based on a receiver operating characteristic (ROC) curve, the relationship between the morphometric groups and histologic grade was determined by analysis of variance and Tukey-Kramer's multiple comparisons test. Survival times of the 2 resulting groups were analyzed using the Kaplan-Meier method, followed by a log rank test. Data were analyzed using GraphPad Prism e and BioEstat f software.
Results
Sixteen of the dogs were males (55.2%), and 13 were females (44.8%). The study population included 9 mixed-breed dogs (31%), 8 Boxers (28%), 4 Dachshunds (14%), 2 Brazilian Fila (7%), 1 Poodle, 1 Miniature Pinscher, 1 Schnauzer, 1 Doberman Pinscher, 1 Cocker Spaniel, and 1 Rottweiler. Thirtyfive neoplasms were analyzed, and multiple neoplasms were found in 6 dogs (6/29, 21%). The most common location for MCTs was the extremities (13/35, 37%), followed by the abdomen (9/35, 26%), thorax (6/35, 17%), inguinal region (4/35, 11%), and head and neck (3/35, 9%). The mean age of the dogs was 9.6 years. The clinical follow-up sessions varied from 81 to 2,334 days among the animals evaluated by Kaplan-Meier analysis (live animals at the end of the study, deaths unrelated to MCT, and interrupted clinical history were censored). Postsurgery survival among animals that died or were euthanized due to the MCTs varied from 3 to 751 days, with a median of 112 days.
The neoplastic mast cell MNA and the animals' survival were correlated both for Romanowsky-type stain (r = −0.6240, P = 0.0003) and HE (r = −0.6323, P = 0.0002). With a cutoff value for MNA measured in HE and determined by an ROC curve (cutoff = 52.29 μm2, sensitivity = 0.818, specificity = 0.722, accuracy = 75.84%), the neoplasms were separated into high-grade MCTs (with averages that were higher than the cutoff) and low-grade MCTs (with smaller averages). Survival analysis revealed statistically significant differences among groups (χ2 = 10.67; P = 0.0011; Fig. 1). Among MCTs regarded as low grade, 86% of the animals survived more than 2,334 days, while only 19% of those regarded as high-grade MCTs survived for a period longer than 1,231 days. Likewise, after the defined cutoffs for Romanowsky-type stain with an area of 62.39 μm2 (sensitivity = 0.818, specificity = 0.833, accuracy = 82.73%), the differences between high- and low-grade MCT groups were also found to be statistically significant (χ2 = 21.15; P < 0.0001; Fig. 2). Among MCTs regarded as low grade, 100% of the animals survived more than 2,334 days, while none of the animals with high-grade MCTs survived for periods longer than 751 days. The median survival was 207 days for high-grade MCTs both for Romanowsky-type and HE stains.
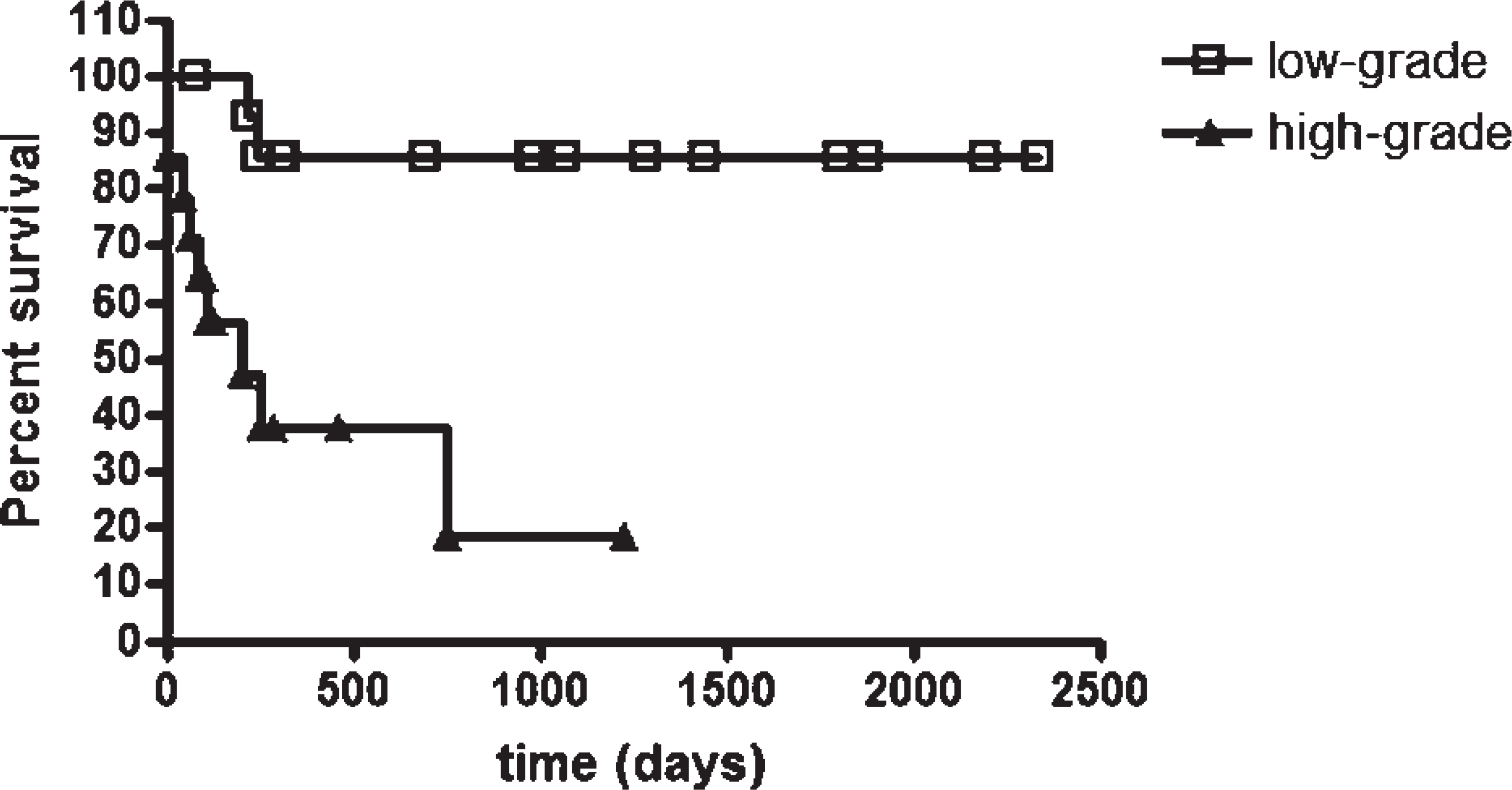
Survival curves for dogs with high-grade (≥52.29 μm2) or low-grade (<52.29 μm2) mean nuclear area values on hematoxylin and eosin-stained cutaneous mast cell tumors (χ2 = 10.67; P = 0.0011, Kaplan-Meier/log rank test). Points = censored data.
When nuclear area was compared to histopathologic grade, there were significant differences between grades II and III (P < 0.01 for HE; P < 0.05 for Romanowsky-type stain) and between grades I and III (P < 0.05 for HE; P < 0.001 for Romanowsky-type stain). Survival of dogs with histologic grade II tumors was compared using low- and high-grade morphometric groups, and statistically significant differences were detected for Romanowsky-type stain (χ2 = 4.624; P = 0.0315; Fig. 3) but not for HE (χ2 = 0.3255; P = 0.5683).
Discussion
The clinical information of animals in the present study was similar to previous reports; Boxers were the most frequent purebred dog with MCTs, and there was no sex predisposition for tumor development. 2,12,21,25,27,34 Moreover, the mean age of the affected animals and the location of the lesions, particularly on the trunk and perineal region, closely followed the veterinary literature. 12,16,21,34 However, the incidence of multiple MCTs in the present study was slightly higher (20%) than that reported in the veterinary literature (3–14%). 1,21,33
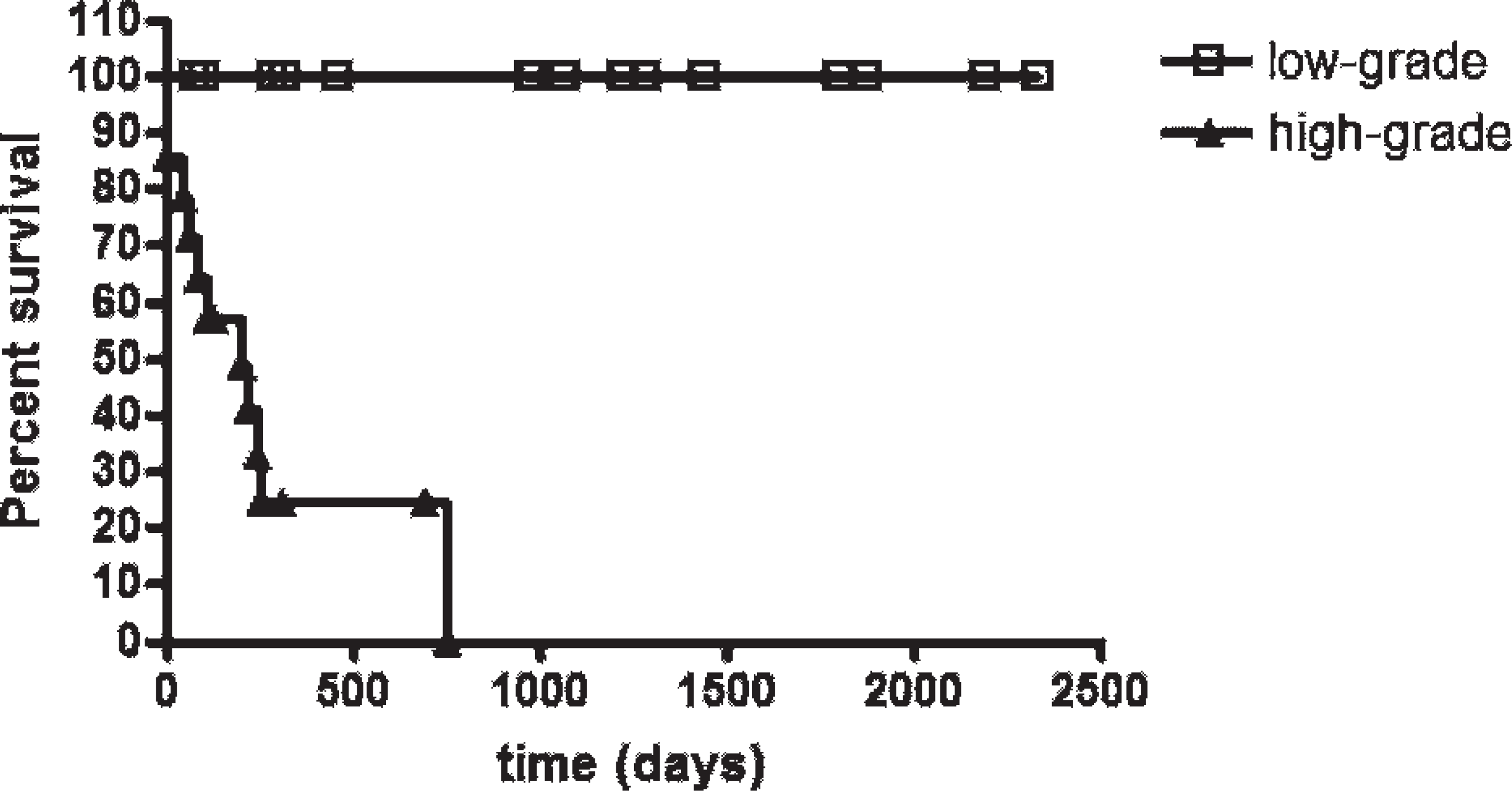
Survival curves for the dogs with high-grade (≥62.39 μm2) or low-grade (<62.39 μm2) mean nuclear area values on Romanowsky-type-stained cutaneous mast cell tumors (χ2 = 21.15; P < 0.0001, Kaplan-Meier/log rank test). Points = censored data.
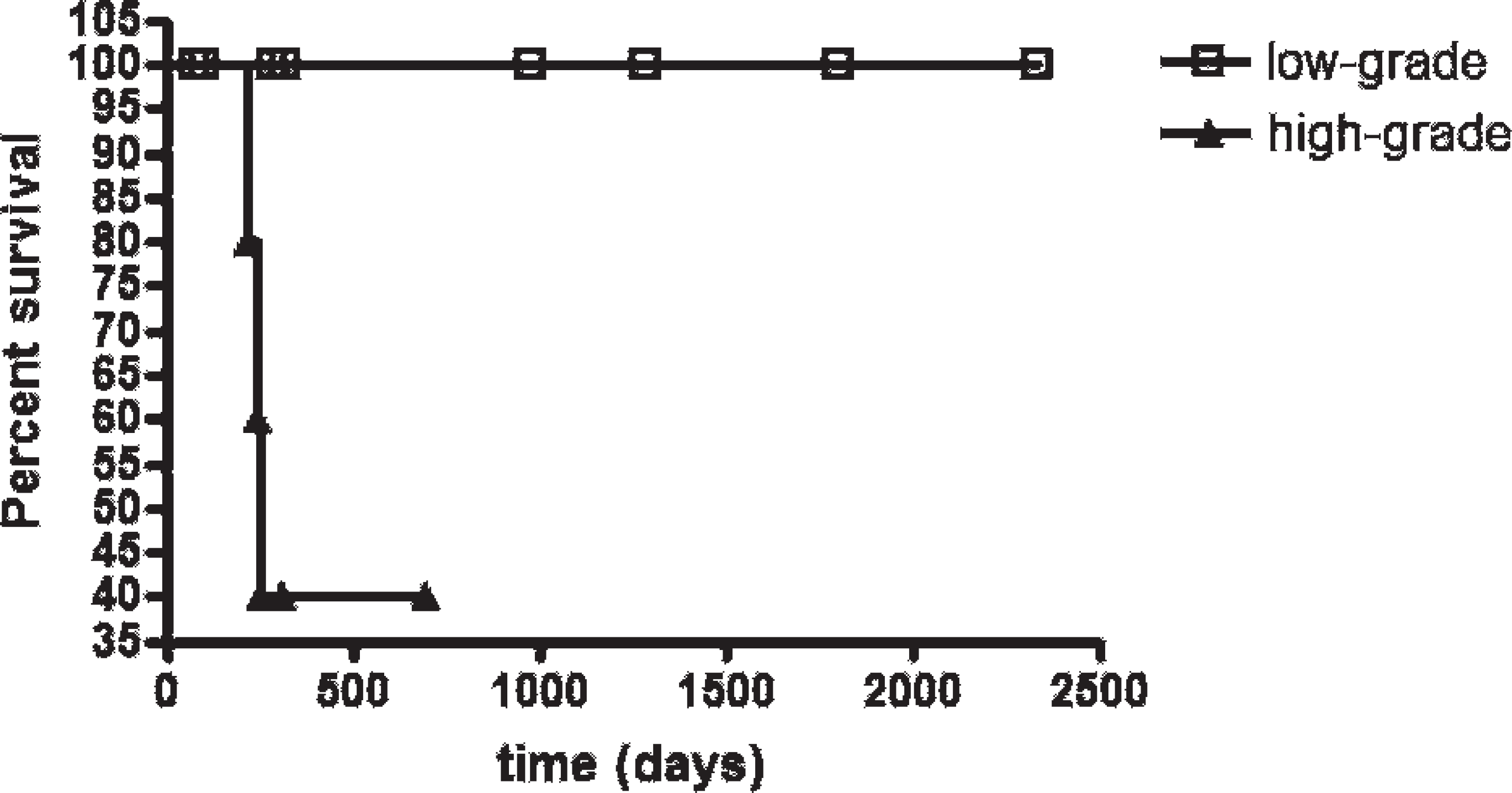
Survival curves for the dogs with histological grade II cutaneous mast cell tumors with high-grade (≥62.39 μm2) or low-grade (<62.39 μm2) mean nuclear area values on Romanowsky-type-stained preparations (χ2 = 4.624; P = 0.0315, Kaplan-Meier/log rank test). Points = censored data.
Despite the fact that several methods have been evaluated as prognostic indicators of canine cutaneous MCTs, no single method is more widely used than conventional histologic grading, 16,21,22,25,34 which is based on morphologic characteristics. Several generally subjective parameters must be considered in grading MCTs using the traditional classification method. 27 These variables may lead to interobserver and intraobserver variations in the grading of MCTs and other neoplasms, as verified by several authors. 14,17,24,26,34 Furthermore, tumor grading is largely based on histologic characteristics that require incisional or excisional biopsy, which adds time and relatively high costs to tumor diagnosis.
Cytopathologic examination, on the other hand, is a practical, rapid, and low-cost method for the diagnosis of tumors, especially those classified as cutaneous round-cell neoplasms. 4,8,32 Cells with poor cytoplasmic granulation, such as poorly differentiated MCTs, are more easily recognized cytologically than in histopathologic sections because of the higher resolution and metachromatic staining of the cytoplasmic granules. 8,32 Therefore, cytopathology is an important technique in the diagnosis of mast cell neoplasia. 21,25 Sampling lesions and preparing, fixing, and staining smears can be accomplished within 30 min with Romanowsky-type stains, and the final diagnosis can made rapidly thereafter. 4,32,35 Using cytologic preparations, cells have less alteration of their morphology related to fixation and staining. 6 Ancillary computerized morphometry can add to cytopathologic examination by providing several numerical parameters with increased objectivity. Therefore, computerized morphometry is more reproducible and increases its potential diagnostic value. 5,9,11,13
In the present study, the cytopathologic morphometric data indicated a correlation between MNA and patient survival; Kaplan-Meier analysis demonstrated that survival times were significantly shorter as nuclear area increased. These results corroborate previous data that demonstrate a relationship between morphometric analysis and histopathologic grading 22,34 and confirm the usefulness of cytomorphometry alone as a predictor of survival. Despite a low number of patients, the dogs in the present study were followed clinically for an extended period of time (average of 675 days and maximum of 2,334 days).
Histologic grading remains challenging in the evaluation of MCTs that are borderline between different grades. In the authors' experience, a number of grade II MCTs behave biologically as grade I neoplasms, while other MCTs behave as grade III neoplasms. This suggests that some MCTs are underdiagnosed with regard to malignancy. It has been suggested that a new grading system that categorizes MCTs as high- or low-grade malignancy would be more consistent and have a more meaningful prognostic value. 14,26
With the aim of determining the value of nuclear cytomorphometry in the prognosis of cutaneous MCTs in relation to survival, the analyzed cases of neoplasms were divided into 2 groups: low- and high-grade MCTs. The survival time of animals with high-grade MCTs was shorter than that of the animals with low-grade MCTs. Statistically significant differences were observed between survival curves with Romanowsky-type and HE staining. These results reinforce the need for simpler grading systems and present cytomorphometry as a precise and easily reproducible diagnostic technique.
In the present study, 2 staining methods were used. The HE application does not stain mast cell granules, which makes nuclear tracing easier. Romanowsky-type staining is routinely used in cytopathologic examination and is rapid and practical. Although mast cell granules may obscure nuclear detail, with Romanowsky-type staining, this problem was minimized by varying figure contrast during morphometric analysis, making the nuclear contour more visible. Furthermore, when histologic grade II MCTs were subgrouped by morphometry following Romanowsky-type staining, MCTs with more aggressive behavior could be identified. This observation suggests that this staining method is also suitable for morphometric analysis.
In conclusion, the results of the present study suggest that computerized nuclear morphometry may be a good prognostic indicator for canine MCTs. Cytomorphometric techniques can provide prognostic information about survival of dogs with cutaneous MCTs in an independent manner. Furthermore, this technique is rapid, accurate, reproducible, and low cost.
Acknowledgements
The authors thank Dr. Julia Maria Matera (Departamento de Cirurgia, FMVZ-USP) and Ana Paula Teixeira and Renata Achkar (Universidade Bandeirante de São Paulo) for submitting surgical samples, Dr. Fernando Ferreira (Departamento de Medicina Veterinária Preventiva e Saúde Animal, FMVZ-USP) for help in statistical analysis, and Fundação de Amparo à Pesquisa do Estado de São Paulo for financial support (grants 05/50407-3, 97/09459-7, and 96/4903-8). The present work is part of the doctorate presented by Ricardo De Francisco Strefezzi to the Departamento de Patologia, Faculdade de Medicina Veterinária e Zootecnia, Universidade de São Paulo, Brazil.
Footnotes
a.
HEMATOCOR®, Biolog®, Biológica Comercial Ltda., São Paulo, State of São Paulo, Brazil.
b.
Olympus® BX 50, Olympus Corp., Melville, NY.
c.
Evolution™ MP, Media Cybernetics Inc., Bethesda, MD.
d.
Image-Pro® Plus version 4.5 for Windows®, Media Cybernetics Inc., Bethesda, MD.
e.
Prism® version 4.02 for Windows®, GraphPad Software Inc., La Jolla, CA.
f.
BioEstat version 5.0, Universidade Federal do Pará, Pará, Brazil.
