Abstract
A 9-y-old, zoo-housed, male red panda (Ailurus fulgens) became progressively lethargic and inappetent over a 1-wk period. Physical examination was unremarkable. A complete blood count showed mild normocytic, normochromic, non-regenerative anemia with the presence of trypomastigote organisms, consistent with a Trypanosoma sp. The organism was confirmed later as Trypanosoma cruzi lineage TcI via PCR and genome sequencing. The panda was initially treated supportively; however, its clinical status within 24 h from presentation deteriorated, and euthanasia was elected. Autopsy showed severe systemic T. cruzi infection with the presence of amastigotes in the heart, brain, peripheral nerves, skeletal muscles, tongue, liver, and testes. We used genome sequencing and serology in identifying the agent.
The red panda (Ailurus fulgens) is an endangered procyonid that is at risk of contracting infectious diseases commonly reported in domestic dogs. Chagas disease, caused by the protozoan Trypanosoma cruzi, is endemic in humans and domestic dogs in South and Central America, and cases in both species have been reported in the southern United States.1,2,4,9,11 The vectors of Chagas disease, triatomine insects, particularly Triatoma and Paratriatoma spp., are present in the western, southern, and southeastern United States. 8 Only one case of Chagas disease has been reported previously in a captive red panda from a facility in the southwestern United States in 1975. 3
A 9-y-old intact male, captive-born red panda housed in a zoologic collection in northeastern Kansas was examined because of a 1-wk history of lethargy and progressive inappetence. The panda was housed in a medium-sized outdoor exhibit with a grass yard and wooden cage furniture with access to a concrete indoor enclosure with wood nest boxes. Vegetation in the outside exhibit included honeysuckle bushes (Lonicera spp.). It was housed with an adult female panda and their 2 offspring. On visual examination, the patient had bilateral pelvic limb weakness with mild right pelvic limb lameness. The patient was placed under general anesthesia and maintained using isoflurane (Akorn Animal Health, Lake Forest, IL). Physical examination and whole-body radiographs were unremarkable, aside from moderate dental disease. A whole-body computed tomography (CT) scan revealed generalized lymphadenopathy, sacralization of lumbar vertebra 6, protrusion of the intervertebral disc between lumbar vertebrae 5 and 6, and a hypoattenuating liver. An inflammatory leukogram and mild normocytic, normochromic, non-regenerative anemia were noted on a complete blood count. Blood smear examination revealed numerous trypomastigotes and a single macrophage containing numerous amastigotes (Fig. 1). A serum chemistry panel showed mild hypoalbuminemia, mild hyponatremia, and hypocholesterolemia.
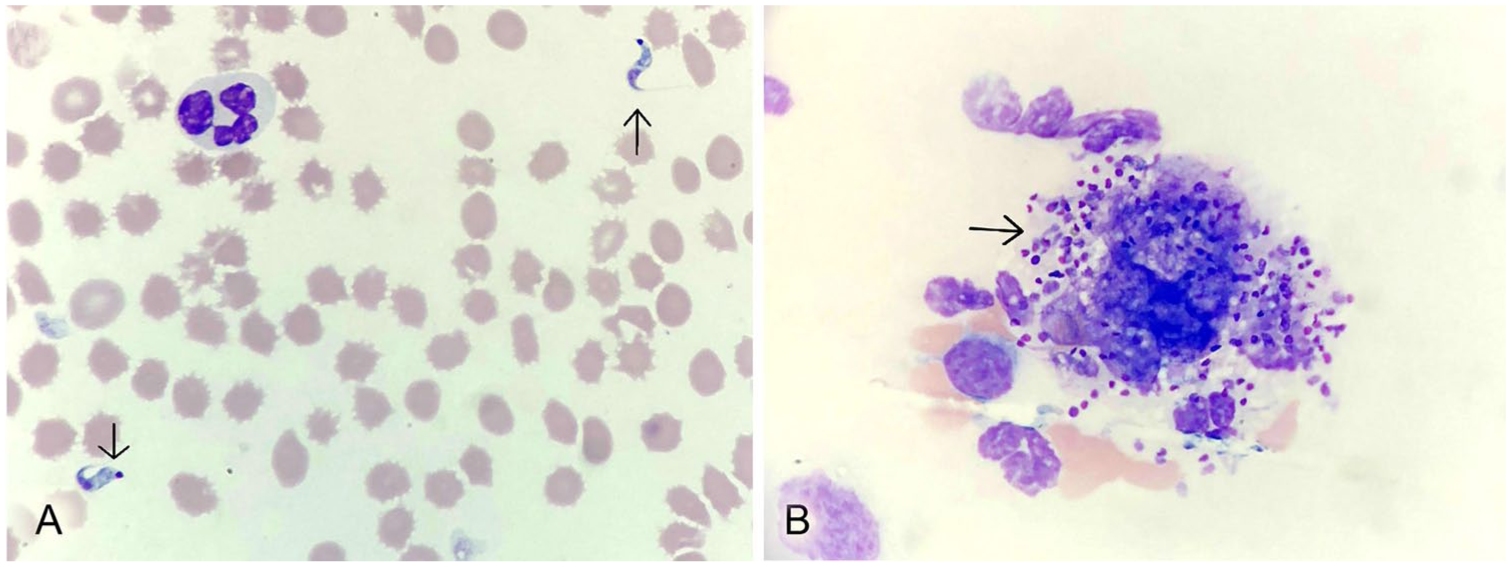
Trypanosoma cruzi stages in a peripheral blood smear from a zoo-housed red panda.
Pending test results, the panda was treated supportively with subcutaneous lactated Ringer solution (45 mL/kg; Baxter Healthcare, Deerfield, IL), buprenorphine (0.03 mg/kg SQ; Par Pharmaceutical, Rochester, MI), meloxicam (0.1 mg/kg SQ; Unichem, Memphis, TN), and cefovecin sodium (8 mg/kg SQ; Convenia; Zoetis, Kalamazoo, MI).
Over the course of the next 24 h, the panda became minimally responsive. Given the panda’s rapid clinical decline and the clinical suspicion of T. cruzi, euthanasia was elected. On postmortem examination, gross lesions included generalized mucous membrane pallor, pinpoint gastric ulcers, melena, and a pale markedly enlarged liver. Additionally, the heart was moderately globose, with a dilated right atrium and ventricle.
Protozoal organisms were detectable histologically in almost every tissue examined, including skeletal muscle, tongue, heart, testis, connective tissue, fat, liver, kidney, endo- and perineurium of peripheral nerves, cerebrum, submucosal and myenteric plexuses, and within cells adhered to the adventitial surface of vessels (Fig. 2). Changes were most prominent within the skeletal and myocardial musculature. Distending the sarcoplasm of the skeletal myocytes and occasional cardiomyocytes were numerous, 60-µm pseudocysts composed of numerous 2–4 µm diameter, round-to-oval protozoal amastigotes with a distinct basophilic nucleus and an adjacent rod-shaped kinetoplast. Within the myocardium, amastigotes were most often present in interstitial cells, which were desmin-negative (muscle marker) but vimentin-positive (mesenchymal cell marker; Fig. 2D).
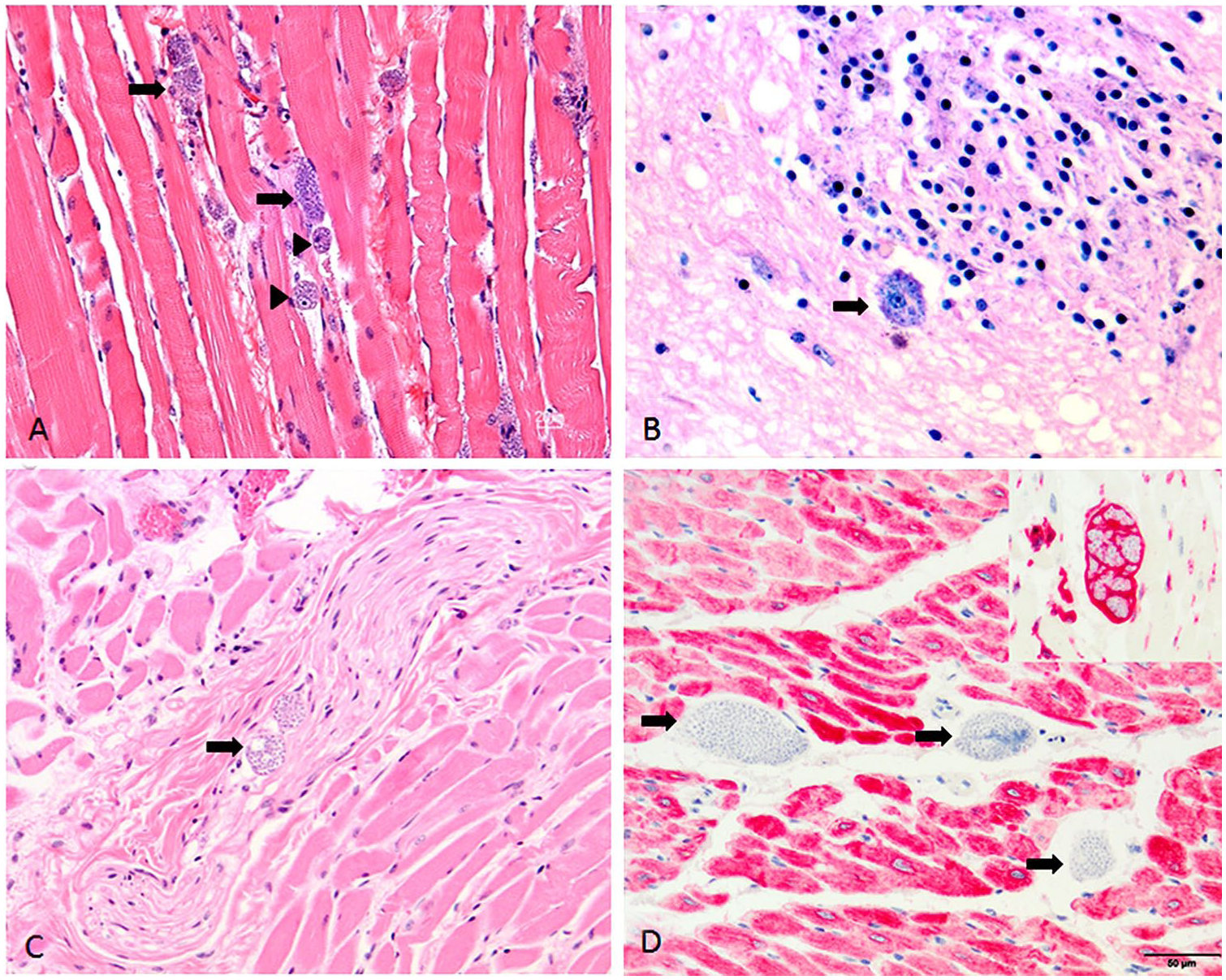
Trypanosoma cruzi amastigotes within multiple tissues from a zoo-housed red panda. Amastigotes were numerous within the tissues, including:
Frequently in all examined tissues, these protozoal organisms were associated with cellular degeneration and necrosis, with occasional fibrosis and multifocal-to-coalescing inflammation composed of histiocytes, lymphocytes, and plasma cells. Protozoal pseudocysts were also frequently present within tissue histiocytic cells, and rarely extracellularly. Large numbers of organisms were found inside nucleated cells that were located within and surrounding peripheral nerves, and were associated with multifocal neuritis.
EDTA whole blood was submitted to the University of Georgia (UGA; Athens, GA) for T. cruzi PCR testing and sequencing. DNA extracted from whole blood was tested for the size-variable D7 divergent domain of the 24Sα gene of T. cruzi using primers D75 and D76 as described previously. 5 The amplicon was extracted from the gel, purified, and bidirectionally sequenced at the Georgia Genomics Laboratory (UGA) to confirm lineage. Based on the size of the 24Sα gene amplicon, the panda was infected with T. cruzi lineage TcI. Sequence analysis confirmed infection with this lineage given that it was 97–99% similar to TcI lineages in GenBank (CP015675, CP015657, L22334). 5 A water template was used as a negative control for the PCR; a sample of the T. cruzi raccoon strain was used as a positive control. In the United States, lineage TcI is found in humans and opossums, whereas infections in domestic dogs, raccoons, and skunks are most commonly lineage TcIV. 12 Serum was submitted to Texas A&M University (College Station, TX) for serologic testing. Serum was analyzed using 2-fold serial dilutions from 1:20 with an indirect fluorescent antibody test (IFAT). Following dilution, the serum was incubated on a slide containing fixed T. cruzi organisms. Anti-canine, anti-raccoon, anti-feline, and anti-ferret IgG conjugates with fluorescein isothiocyanate were applied to slide wells and incubated. Canine sera of known positive and negative antibody status were used as positive and negative controls, respectively. The sample was positive for T. cruzi when using canine (1:80), feline (1:320), and raccoon (1:80) antibodies, but negative with ferret antibodies. The female conspecific of our case tested negative for T. cruzi via PCR after a brief illness characterized by anorexia, lethargy, and vomiting. However, routine screening for T. cruzi for other animals in the collection has not been implemented as of time of publication.
Chagas disease is endemic in Central and South America and has been reported in the United States in humans, dogs, and a number of wild reservoir species, including raccoons (Procyon lotor), armadillos (Dasypus novemcinctus), opossums (Didelphis virginiana), skunks (Mephitis mephitis), and wood rats (Neotoma micropus).1,2,4,6,9 Raccoons are most commonly tested for T. cruzi antibodies, and throughout the southeastern United States, 0–66% of raccoons have tested positive. 2 In Kansas, ~57% of adult and 25% of juvenile raccoons had antibodies to T. cruzi, and antibody- and PCR-positive dogs have been described in eastern Oklahoma 4 (Everson TD, et al. Hemoparasites of urban raccoons (Procyon lotor) in Manhattan, Kansas. Abstract 63. Proc 46th Am Assoc Vet Parasitol Ann Meeting; July 2001; Boston, MA. Available from: https://www.aavp.org/documents/2015/06/aavp-2001-annual-meeting-proceedings.pdf/). The organism is transmitted by triatomine insects, mainly via wound contamination with insect feces, but also through vertical transmission, blood transfusion, organ transplants, and ingestion of contaminated bugs.1,2,11,13 In our case, no similar insects were reliably reported near this animal.
The life cycle of T. cruzi is complex, with the organism transitioning through 3 different forms.1,2,11,13 The trypomastigote is found in blood and lymph of the host, is ingested in blood meals by the insect vector, and transforms into epimastigotes, which change into trypomastigotes and are expelled in the insect’s feces. Upon entering the host either through contamination of a wound with infected feces or through ingestion of the insect vector, the organisms invade host cells and differentiate into amastigotes.1,2,11,13
Chagas disease in humans and dogs is divided into acute, latent, and chronic phases.1,2,13,14 The acute phase usually results in mild, nonspecific clinical signs or is asymptomatic in humans. 14 Following an asymptomatic latent phase, the disease progresses to either the cardiac form, characterized by dilated cardiomyopathy, cardiac arrhythmias, and congestive heart failure, or the digestive form, which results in either megaesophagus or megacolon. 14 The clinical signs in our case were consistent with the lethargy and anorexia demonstrated in the acute phase observed in dogs and humans; the abnormal hindlimb gait and suspected discomfort may have been the result of peripheral neuritis.
In humans, amastigotes may be found in the heart, connective tissue, smooth and skeletal muscle, liver, kidneys, peripheral nerves, and glial cells. 14 In the previously reported case of Chagas disease in a red panda, diffuse lymphoplasmacytic myocarditis was noted, as well as organisms present in skeletal muscle and the tongue. 3 The marked difference in parasite tropism in our case compared to the previous red panda case may be the result of a different route of infection, such as ingestion of T. cruzi–infected insects, which causes more severe disease in humans. 11 The difference may also be attributed to a difference in T. cruzi lineage given that the lesions in our case are more similar to the lesions in humans, whereas lesions in the previous case aligned more closely with lesions reported in dogs.11,13,14
Chagas disease can be diagnosed by detecting trypomastigotes in blood during the acute phase, in combination with a clinical history of potential exposure to the insect vector and corresponding clinical signs. 1 Serology, most commonly IFAT and ELISA, can be used to confirm diagnosis during the latent and chronic phases; however, there can be cross-reactivity with Leishmania spp. in dogs.1,13 PCR can be used for detection of parasites in the acute phase, and is more sensitive than microscopic detection, but sensitivity decreases during the latent and chronic phases because parasitemia decreases over time. 13 However, PCR assays are not readily available through commercial, diagnostic laboratories for this agent.
Chagas disease is reportable in several southern states; we reported our case to the Centers for Disease Control and Prevention (CDC) and the Kansas Department of Health and Environment.2,4,9 There were reported cases of human Chagas disease in Kansas in 2015 and 2016, both of which were likely travel associated.7,8 With the wide variety of species potentially exposed to T. cruzi and the presence of triatomine insects in the United States, zoo animals are at risk of infection. 15 The risk of disease transmission may vary depending on the specific triatomine vector present in each state. For example, triatomine species, Triatoma lecticularia and T. sanguisuga, located in Kansas, do not defecate during feeding, which limits transmission to ingestion of infected insects. 10 The previously reported red panda case was diagnosed postmortem, whereas, as demonstrated in our case, blood smear evaluation, serology, and PCR can be used for antemortem diagnosis in the red panda.
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
