Abstract
Free-roaming chickens on Caribbean islands are important sentinels for local avian diseases and those introduced by birds migrating through the Americas. We studied 81 apparently healthy unvaccinated free-roaming chickens from 9 parishes on St. Kitts, an eastern Caribbean island. Using commercial ELISAs, no chickens had antibodies against avian influenza virus, West Nile virus, or Salmonella Enteritidis, although seropositivity was high to infectious bursal disease virus (86%), infectious bronchitis virus (84%), Mycoplasma (37%), and avian avulavirus 1 (Newcastle disease virus, 31%). Examination of small and large intestinal contents revealed cestodes in 79% and nematodes in 75% of the chickens. Although ectoparasites and endoparasites were common (74% and 79%, respectively), only a few chickens had lesions at postmortem examination, mainly intestinal serosal nodules (12%) and feather loss (6%). Histologic examination of 18 organs from each bird revealed lesions in high percentages of organs, mainly the liver (86%), lung (75%), spleen (60%), small intestine (56%), skin (42%), and kidney (40%). Lesions included degenerative, reactive, inflammatory, and neoplastic, and were not correlated with the serologic status of the chickens except in one case of infectious bursal disease. Microscopically, Paratanaisia bragai was seen in the kidneys of 3 chickens and intestinal coccidiasis in 1 chicken. Pulmonary silicate aggregates were common, were present in intestinal serosal nodules, and were suggestive of environmental exposure.
Introduction
Chicken is a major ingredient in many Caribbean food dishes and comprises >80% of the animal protein consumed in the region (http://www.agricarib.org/products/poultry). Overall, poultry is the major agro-industrial business in the Caribbean, exceeding sugar production. Although large-scale poultry farms are found on the larger islands, there is considerable scope for poultry production by small business enterprises and backyard producers. As is the case in other developing areas, in particular Africa, 17 increased local small-scale poultry production could help alleviate the plight of economically deprived people in the region. Although management methods and production strategies have been established to enable successful small-scale and backyard chicken production, such enterprises represent substantial capital outlays for low-income people, and disease outbreaks can have dire consequences. Further, disease reservoirs and outbreaks in small-scale producer flocks can be sources of infection for nearby large-scale producers.
There is only fragmentary information on poultry pathogens in the Caribbean. 7 Studies on Trinidad & Tobago have provided strong evidence for the circulation of important poultry viral and bacterial pathogens in commercial 9 and backyard poultry, 5 including infectious bronchitis virus (IBV; species Avian coronavirus), Mycoplasma gallisepticum (MG), Mycoplasma synoviae (MS), avian influenza virus (AIV; species Influenza A virus), avian avulavirus 1 (formerly Newcastle disease virus, NDV), infectious bursal disease virus (IBDV), and Salmonella enterica serovar Enteritidis (SE). Furthermore, there is evidence for West Nile virus (WNV) infections in sentinel chickens in Puerto Rico, 33 chicken astroviruses in Grenada, 30 and Campylobacter spp. in Grenada 32 and Barbados. 34 Salmonella spp. have been characterized in chickens in Trinidad & Tobago, St Lucia, and Grenada, 1 as has NDV, which was responsible for an outbreak in the Dominican Republic in 2008. 12 AIV has been described in the Dominican Republic and Haiti, 28 and evidence for infections of wild birds has been provided from Trinidad & Tobago. 8 To date, there are only minimal data on ecto- and endoparasites of chickens, with mixed infections with Eimeria sp. having been shown to be common in broilers on Trinidad. 6 We found no reports of non-infectious causes of lesions in chickens from the Caribbean.
To develop control strategies for avian and zoonotic diseases, data on conditions of domestic poultry, wild and migratory birds, and other species, including humans, is important. The Caribbean island of St. Kitts represents an ideal location to study such interactions given that it is on a migratory bird flight path (Caribbean Island/Western North Atlantic Route,24,25 Fig. 1) and possesses all of the above-mentioned groups of birds, many of which live in close proximity to humans. In particular, free-roaming backyard chickens are very common and are ideal sentinels for avian diseases in this area. We report herein our results of serology, parasitology, and pathology investigations of conditions affecting free-ranging backyard chickens from St. Kitts.
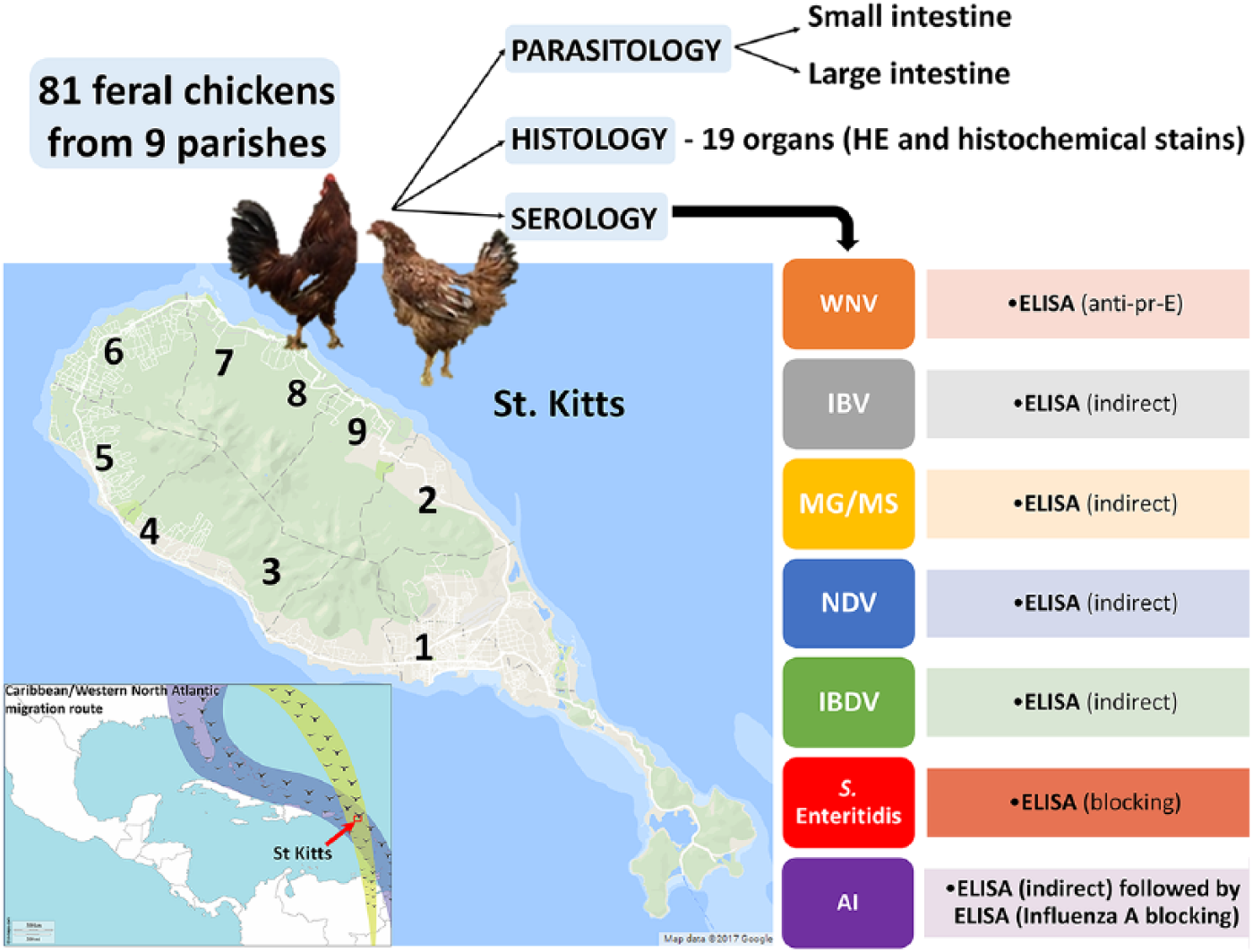
Study design, location, sampling, and testing strategy. Free-roaming backyard chickens originated from parishes 1–9 throughout St. Kitts, Leeward Islands. Inset: location of the study (arrow) on the Caribbean/Western North Atlantic pathway of migratory birds (green and blue shades).
Materials and methods
Sample collection, ecosystem, and study location
The island of St. Kitts (Fig. 1) has an area of 174 km2, a human population of ~35,000, and an estimated 1,500 free-roaming backyard chickens. Our study used 9 random free-roaming chickens obtained from each of the 9 parishes (Fig. 1) and was approved by the Institutional Animal Care and Use Committee of Ross University School of Veterinary Medicine (RUSVM; St. Kitts). The total study population was 81 chickens (~5% of the estimated total population), comprised of males (40%) and females (60%), mostly adults, apparently healthy, and with no known previous vaccination or treatments.
Serology
Sera were obtained immediately before euthanasia and stored at −20°C until tested with commercial enzyme-linked immunosorbent assays (ELISAs) for antibodies against WNV (multispecies ID Screen; IDvet, Montpellier, France), IBV (IBV Indirect; IDEXX, Westbrook, Maine), MG and MS (MG/MS Indirect; IDEXX ), NDV (NDV Indirect; IDEXX), IBDV (IBD-XR Indirect; IDEXX), and SE (SE blocking; IDEXX). For AIV, an initial screening assay (AI Indirect; IDEXX) was used, followed by a confirmatory assay (Influenza A blocking; IDEXX) with testing of the initial positives. Each ELISA was performed in duplicate at RUSVM, in compliance with the manufacturer’s instructions, and all readings were performed on the same microplate reader (ELx800; software Gen5 v.3.02; BioTek, Winooski, VT). To calculate the confidence level (CL), we used the following formula: p ± 1.960√(p(1 − p)/n), where p is proportion and n is number of observations.
Tissue collection and preparation
Routine postmortem examinations were performed post-euthanasia, and samples from 18 organs were collected from each chicken and processed with standard methods for histology: liver, kidney, lung, esophagus, proventriculus, small intestine (duodenum and jejunum), ceca, spleen, gonads, skin, heart, pancreas, brain, bursa of Fabricius, adrenal gland, thymus, trachea, and skeletal muscle. Tissues were fixed in 10% neutral-buffered formalin for at least 48 h at room temperature, processed routinely, stained with hematoxylin and eosin, and in some cases with other histochemical stains.
Parasitology examination
To assess gastrointestinal helminths, following intestinal sampling for histology, the intestines (from the gizzard to the cloaca) were removed, and the small intestine separated from the large intestine and ceca. These specimens were opened longitudinally and stored refrigerated (2–5°C) in saline (0.9%) at ~30°C for up to 3 h and then refrigerated (2–5°C). Within 24 h, the contents in saline, and the mucosae that were gently stripped from underlying tissue, were passed through a 50-µm sieve. The contents of the sieve were stored in 2–5% formaldehyde until parasites were identified by standard morphologic features using a stereomicroscope for scanning and a compound microscope to confirm detailed characteristics.
Results
Serology results
All sera tested negative for antibodies against WNV, AIV, and SE. Four sera were positive in the initial screening assay for AIV, but these samples were negative with the confirmatory blocking ELISA. Antibodies against IBV and IBDV were the most commonly detected in the study population (84% and 86%, respectively) with lower prevalences found for NDV and MG and MS (31% and 37%, respectively). There were no significant differences in the prevalence of seropositive chickens in the different parishes using ANOVA and t test in Excel (Microsoft, Redmond, WA).
Postmortem examination
External parasites, most likely Menopon spp. and Menacanthus spp., based on their location and previous studies 35 (Supplementary Fig. 1A, 1B), were seen on 60 chickens (74%), although 5 chickens had feather loss. Only a few gross anatomic lesions were seen in the birds, principally <5 mm, white-to-yellow, serosal nodules in 10 chickens (12%), mainly on the surface of small and large intestines. Intestinal cestodes and nematodes were noted, and are reported below. Only one chicken, IBDV positive, had an enlarged bursa of Fabricius, with edema and hemorrhages on the surface mucosa, confirmed histologically. The bursa was not evident in some chickens, but most of the birds examined were adults and the bursa normally regresses with sexual maturity. 26
Parasitology
Most of the chickens (n = 66, 82%) had at least 1 helminth species, and 55 (68%) had coinfections with cestodes and nematodes (Figs. 2–9). Cestodes were present in 64 chickens (79%), mainly Raillietina echinobothrida followed by R. tetragona and Amoebotaenia cuneata (A. sphenoides), with Choanotaenia sp. seen less frequently. Nematodes were also highly prevalent (n = 61; 75%); Capillaria spp. were most common (n = 51; 63%) followed by Heterakis gallinarum (n = 29; 36%), and Ascaridia galli (n = 10; 12%). In 40 chickens it was possible to speciate the Capillaria; 7 of 40 (18%) had mixed infections. The primary species was C. obsignata (n = 29, 72%), followed by C. anatis (n = 16, 40%), and C. bursata tentatively identified in 2 of the chickens (5%). All mixed infections were C. obsignata and C. anatis. Using ANOVA and the t test in Microsoft Excel, no differences were found in the prevalences of parasites in chickens from different parishes.

Specimens preserved in formalin were used for morphologic identification of the parasites present in the small and large intestine using a stereomicroscope or a compound microscope.
Microscopic lesions
Lesions were most commonly seen in the livers of the chickens (n = 70, 86%; Supplementary Table 1). Nodules (periportal, centrilobular-to-random) composed of a mixture of lymphocytes, plasma cells, and macrophages were the most common histologic change seen. Well-formed hepatic granulomas (Fig. 10A) had a necrotic center, surrounded by a rim of multinucleate giant cells, epithelioid macrophages, lymphocytes, and plasma cells with some also containing heterophils. To rule out bacterial or fungal etiology, special stains were performed: acid fast (modified Ziehl–Neelsen), Gram (Brown & Brenn, and Brown–Hopps), periodic acid–Schiff (PAS), and Grocott methenamine silver (GMS). In one of the portal granulomas, within macrophages in the lesions and outside of lesions, intracellular organisms were detected (suspected microsporidial spores), 2–4 µm in length, that were gram-positive (Fig. 10B), with a PAS-positive polar granule within spores, and acid-fast negative.
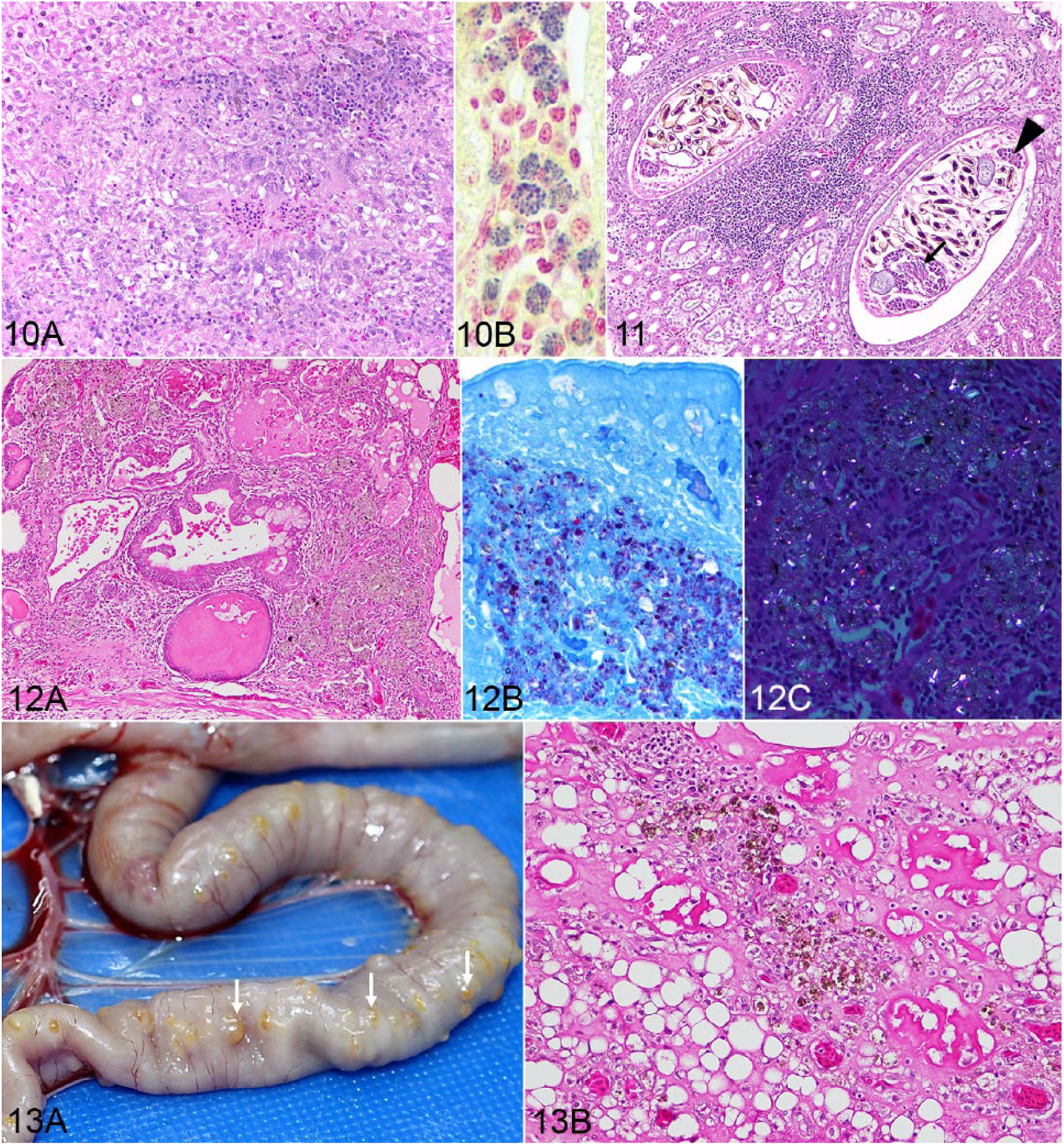
Hepatic granuloma in a chicken.
Renal lesions were seen in 32 chickens (40%), more commonly lymphoplasmacytic interstitial nephritis (n = 29); 25 of these (86%) were seropositive for IBV, suggesting that the nephrotoxic strain of IBV could be responsible for the lesions. However, the lesion may have been caused by a different agent, such as avian nephritis virus (species Avastrovirus 2) or NDV (24% of interstitial nephritis cases were NDV-seropositive). In one chicken, cystic dilation of renal tubules was observed with metaplasia of renal epithelium to ciliated epithelium (Supplementary Fig. 2). This can occur in the mammalian kidney, probably as a consequence of faulty cell division. 14 In the dilated collecting ducts and pelvis of 3 chickens, an adult trematode (Paratanaisia bragai) was detected (Fig. 11), and confirmed by PCR of the paraffin-embedded renal tissues (data not shown).
All chickens with pulmonary lesions (n = 61; 75%) had aggregates of environmental silicates (confirmed by energy dispersive X-ray analysis). The lesions were multifocal, predominantly surrounding parabronchi, and forming nodules or granulomas consisting of accumulations of macrophages and heterogeneous, yellow-brown-black pigmented, partially birefringent, smaller non-crystalloid or larger crystalloid material, up to 8 µm diameter, free or intracytoplasmic within macrophages (Fig. 12A). No fibrosis was observed. Some cases also had an infiltrate of lymphocytes, with lesions extending into the interatrial septa or perivascularly. The particulate material was easier to observe with the modified Ziehl–Neelsen stain (Fig. 12B), being variably acid-fast positive (the smaller particles, not the crystalloid structures) and was partially birefringent under polarized light (Fig. 12C). On opening the coelomic cavity, in 10 chickens that also had silicate aggregates, white-to-yellow, sometimes coalescing, raised, dense 5-mm diameter nodules were noted on the serosal surface of small and large intestine (Fig. 13A). Histologically, these were foci of fat necrosis and a granulomatous reaction around the same type of particulate material (silicates) described in the lungs (Fig. 13B).
Microscopic changes of the gastrointestinal tract included, in the proventriculus of one chicken, a tubular duct distended by an adult nematode (1.5 mm diameter) that, based on morphology and location, appeared to be a spirurid (Tetrameres sp.; Supplementary Fig. 3). The free-roaming chickens in our study had easy access to water crustaceans or cockroaches, which are known intermediary hosts for Tetrameres spp. 20 Forty-five chickens (56%) had microscopic changes in the small intestine; 33 had metazoan parasites visible free in the lumen or embedded in the mucosa, mainly cestodes (Supplementary Figs. 4A, 4B), 1 had lesions compatible with intestinal coccidiasis, and 2 had a plerocercoid infestation.
Splenic changes were seen in 49 chickens (61%), mainly reactive hyperplasia of the germinal centers of lymphoid tissue (n = 35). The spleen was involved as part of a more generalized lymphoproliferative disorder (likely lymphoid leukosis) in 13 chickens. In these birds, neoplastic cells were also seen intravascularly (predominantly) and extravascularly in the serosa or mesentery (n = 8), liver (n = 6), heart (n = 3), lung (n = 3), bursa of Fabricius (n = 3), pancreas (n = 4), kidney (n = 2), thymus (n = 1), and trachea (n = 1).
In 2 testes, the tunica albuginea was focally infiltrated by seminiferous tubules, with thinning of the tunic. The tubules were functional in one case (active spermatogenesis), and in the other case, entrapped within the tunica albuginea, isolated from the rest of the testis, and only composed of Sertoli cells, lacking spermatic line cells (Supplementary Fig. 5). The presence of seminiferous tubules within the tunica albuginea has been described previously in people, and can mimic neoplasia. 2
Discussion
Our study documents that free-ranging chickens on St. Kitts experience a wide variety of conditions, knowledge of which is important to poultry producers and animal health workers in the region. Serology revealed exposure to a variety of common poultry pathogens, which is consistent with other reports from the region, mainly Trinidad 3 and Grenada. 17 Vaccines against these pathogens appear to be advisable in commercial flocks in the region.
Our finding that chickens were seronegative to AIV and WNV has also been reported from Trinidad. 3 Chickens were reported to be seropositive for AIV in Grenada (10%), the Dominican Republic, and Haiti, 29 and AIV-infected wild birds have been reported in Trinidad & Tobago. 7 The usefulness of animal surveillance, including chickens, in the early detection of WNV has been proven.11,22 Equids on St. Kitts have antibodies against influenza A virus and WNV, which may or may not be associated with vaccination. 4 WNV-positive sentinel chickens have been found in Puerto Rico, 22 and WNV-seropositive chickens and horses have been found in Guadeloupe. 19
There was no clear association between the presence of antibodies and lesions in most organs. Similarly, lesions did not appear to be related to the parish from which the chickens were obtained. There was only one case in which the serologic status of the chicken correlated with lesions compatible with infectious bursal disease grossly and histologically. Overall, our study showed that histology represents an excellent additional tool for the detection of environmental conditions (pulmonary silicate aggregates and intestinal serosal granulomas), and early, subclinical proliferative or parasitic diseases.
We found that gastrointestinal parasites are common in free-ranging chickens on St. Kitts, with species and prevalences similar to those previously reported from the Caribbean.3,23 The prevalence of helminth infections on St. Kitts is similar to the 67% found in free-roaming chickens on Grenada. 23 The primary species (in descending order) on Grenada were Raillietina tetragona, Gongylonema ingluvicola, Ascaridia galli, Heterakis gallinarum, and Capillaria contorta. 23 The primary differences between the chickens on Grenada and St. Kitts are the observations of G. ingluvicola on Grenada and the diversity of cestodes and Capillaria spp. on St. Kitts. Poultry breeds have been shown to have different levels of natural resistance to A. galli infection, with younger birds having higher infections and repeat exposure resulting in some resistance to reinfection.13,21,27 Adapted breeds and inclusion of only adult chickens might explain the limited infection intensity seen in our study, even with the ideal climatic conditions for egg survival in the environment. Although the infection intensity of individual helminth species was low, the overall worm burdens, diversity of species, and prevalence of infection suggest that helminths could have a significant impact on weight gain and result in unthrifty and weak birds more susceptible to other diseases. 16 The likely level of environmental contamination with infective stages and consumption of paratenic and intermediate hosts by scavenging free-roaming birds makes control of infection challenging, even with strategic deworming programs.
Differential diagnoses for hepatic nodules or granulomas in birds include avian tuberculosis, coligranulomatosis, pullorum disease, other Salmonella infections, Staphylococcus infection, fowl cholera, aspergillosis, Tetratrichomonas gallinarum, and neoplasia. 18 In our case, no acid-fast bacilli were detected in lesions. To our knowledge, the only microsporidian species reported in chickens is Enterocytozoon bieneusi, which is a zoonotic pathogen that can cause chronic diarrhea in humans. 31 Lymphoid foci in the liver can represent chronic immune stimulation related to other diseases such as toxoplasmosis, which was reported by our group in the same 81 chickens, with 41% prevalence on serology and PCR. 15
The high prevalence of pulmonary silicate aggregates and coelomic serosal granulomas seen in our chickens is suggestive of environmental exposure, most likely of topsoil origin and related to the physiology of the avian respiratory tract, either the long stagnation in abdominal air sacs, with decreased clearance, or transmigration through the intestinal wall during normal elimination through the gastrointestinal tract after mucociliary clearance from the respiratory tract. 10
Supplemental Material
DS1_JVDI_10.1177_1040638719843638 – Supplemental material for Infections and pathology of free-roaming backyard chickens on St. Kitts, West Indies
Supplemental material, DS1_JVDI_10.1177_1040638719843638 for Infections and pathology of free-roaming backyard chickens on St. Kitts, West Indies by Pompei Bolfa, John J. Callanan, Jenifer Ketzis, Silvia Marchi, Trista Cheng, Hieuhanh Huynh, Tiffany Lavinder, Kenneth Boey, Clare Hamilton and Patrick Kelly in Journal of Veterinary Diagnostic Investigation
Footnotes
Acknowledgements
We thank Dr. Tracey Challenger for support in documenting the population data, Candita Chapman for helping during postmortem examinations, Dave Hilchie for histology and histochemistry support, Dr. Tim Littlewood for molecular biology support on identifying Paratanaisia bragai, and Professor Chris Oura for technical advice on the serology and avian influenza virus testing.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplementary material
Supplementary material for this article is available online.
Funding
This work was supported by the RUSVM Research Center “Integrative Mammalian Research” under grant 770511-65119.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
