Abstract
Cell structures morphologically consistent with Blastocystis were aspirated from a subcutaneous facial swelling in a 13-mo-old pet duck. On PCR analysis and sequencing, the organism was confirmed as Blastocystis sp. subtype 7. Blastocystis is a single-celled protist that is found in the intestinal tract of many species, including mammals, birds, reptiles, amphibians, and insects. A complete understanding of the lifecycle and pathogenesis of the parasite remains elusive. Blastocystis has been implicated in human and animal disease; however, its role is controversial given that it is commonly found among healthy gut microbiota. Infection with Blastocystis outside the intestinal tract has been reported only rarely in humans. Our case of subcutaneous Blastocystis infection in a duck is a novel presentation of a ubiquitous, generally asymptomatic, parasite or commensal of the intestinal tract.
A 13-mo-old duck was presented to the referring veterinarian for an acutely developing subcutaneous swelling of the left side of the face, caudal to the beak and ventral to the left eye (Fig. 1). The duck was otherwise healthy, and it was the only affected duck in a small flock. Treatment with tylosin did not result in significant improvement, and the duck was presented to the veterinarian again 1 mo later. The lesion was soft and fluctuant; a small quantity of cloudy, yellow viscous fluid was aspirated.

A soft tissue swelling is present between the left eye and the beak of a duck.
Cytologic examination of direct smears and a cytocentrifugation preparation revealed large numbers of round protist cysts, 10–15 µm in diameter (Fig. 2). The structures had a large single or occasionally 2–4 smaller central, clear to faintly basophilic vacuole(s) and a peripheral rim of mid-basophilic grainy cytoplasm. In the cytoplasm was a small oval aggregate of nuclear material. Large numbers of bacteria, a mixture of cocci and bacilli, were present, but no inflammatory cells were observed. The protists were morphologically compatible with the vacuolar form of Blastocystis.
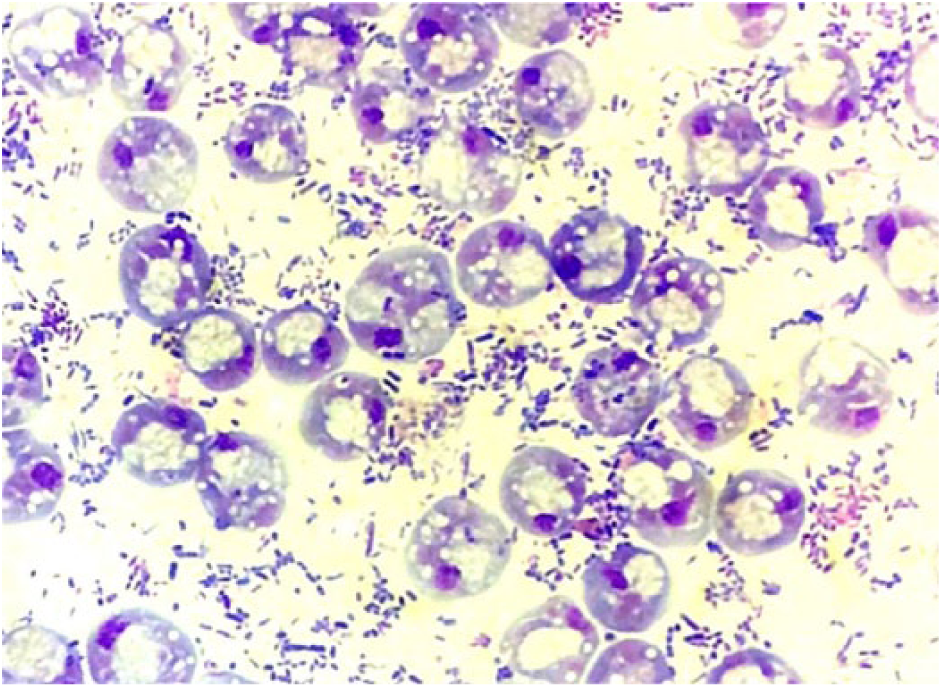
Numerous vacuolar forms of Blastocystis and large numbers of bacteria (predominantly bacilli) in an aspirate from the facial swelling in a duck. Wright–Giemsa. 50×.
The sample was submitted for confirmation by PCR and sequencing. A Blastocystis sp. primer set (RD5 and BhRDr) was used with a method described previously12,13 that targeted the small-subunit 18S ribosomal RNA gene (SSU-rDNA) of Blastocystis. The amplification of a PCR product supported the presence of Blastocystis DNA. Gene sequencing was performed on the amplicon and the sequence compared to a database of different Blastocystis sp. subtypes (www.pubmlst.org/blastocystis). Blastocystis sp. subtype 7 was confirmed.
Following diagnosis, the duck was treated for 1 mo with potentiated sulfonamides, and the facial mass resolved. The swelling recurred 1 mo post-therapy, but responded to a repeated course of potentiated sulfonamides. There has been no evidence of relapse subsequently.
Blastocystis is a single-celled protist found in the intestinal tract of humans, other mammals, birds, reptiles, amphibians, and insects. 1 Blastocystis belongs to the branch of Eukarya known as Stramenopiles or Heterokonta, which includes other organisms such as brown algae, water molds, and diatoms. It is an obligate anaerobe with several morphologic forms: cyst, vacuolar, granular, and amoeboid, which may correspond to different phases of the lifecycle. There is no animal model for Blastocystis, and multiple theories about its lifecycle, morphological forms, and mechanisms of cell division have been proposed. Infection with Blastocystis may occur by ingestion of the environmentally resistant cyst form.14,16 Blastocystis excysts in the intestine to the vacuolar form; the vacuolar form, as seen in our case, is most frequently encountered clinically. 14
Blastocystis is assumed to reproduce by binary fission during the vacuolar stage. 14 The vacuolar form may develop into a granular or an ameboid form; these stages are seen infrequently and are of uncertain significance. Vacuolar and possibly granular forms can encyst by synthesis of a protective thick cell wall, and the cyst is passed in the feces. The cyst is environmentally resistant and thought to allow fecal–oral transmission (Fig. 3).
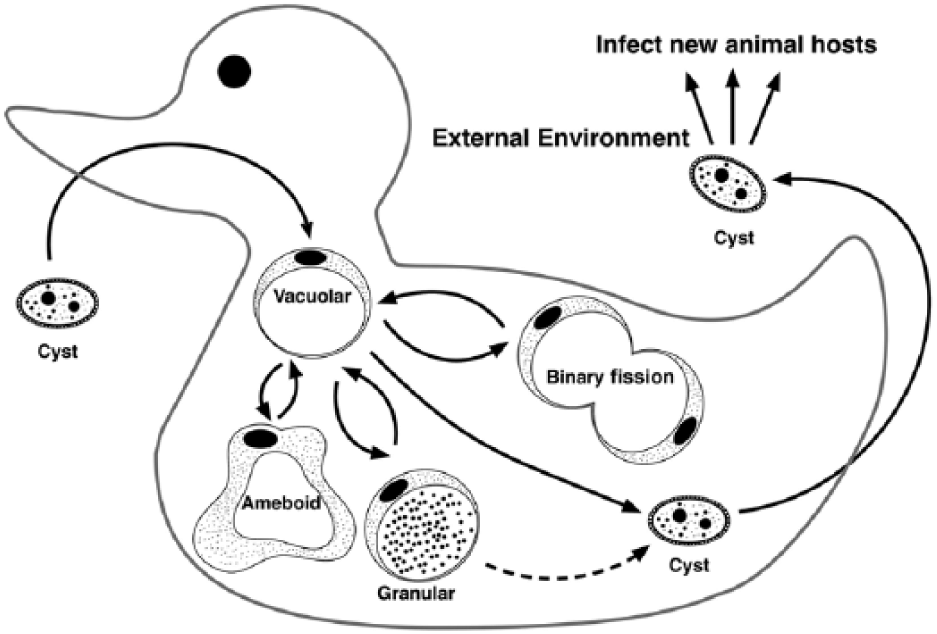
Schematic of the proposed lifecycle of Blastocystis. Dashed line represents possible encysting of granular form.
Historically, the taxonomy of Blastocystis reflected the host species, because it was believed that Blastocystis was host specific (Blastocystis sp. found in humans was termed Blastocystis hominis). Phylogenetic analysis of the species complex has revealed 17 genetically distinct subtypes with variable host ranges, and hence speciation by host specificity has been discontinued. Subtypes cannot be differentiated morphologically and require genetic sequencing.
The Blastocystis isolated from this duck is classified as Blastocystis sp. subtype 7. Blastocystis sp. subtypes 6 and 7 are considered “avian specific” 3 ; they are only isolated occasionally from non-avian hosts.
In humans, Blastocystis subtypes 1–4 are most prevalent, but low rates of infection with other subtypes, including subtype 7, are reported. 1 Human infection with “exotic” subtypes may indicate zoonotic transmission from birds, given that the subtypes are genetically very similar. 7 Close contact with animals is a risk factor for Blastocystis infection in people. 9
The role of Blastocystis as a cause of gastrointestinal disease remains controversial; it is detected frequently in the gastrointestinal tract of non-symptomatic humans and animals. 14 In humans, Blastocystis has been suggested as a cause of various diseases and signs and symptoms including inflammatory bowel disease, irritable bowel syndrome, chronic diarrhea, abdominal pain, and urticaria. 14 The low incidence of symptoms with infection suggests that there might be a complex of interactions between pathogen, host, and environment. 12 Significant factors may include the Blastocystis subtype, isolate virulence, host genotype, host immune function, parasite burden, and concurrent infections. 11
Reports of Blastocystis causing disease outside the gastrointestinal tract are rare. In humans, Blastocystis has caused septic arthritis in an immunosuppressed patient, 6 and organisms have been aspirated from hepatic abscesses5,8 and splenic cysts. 10 Dissemination is presumed to follow intestinal mucosal invasion, possibly in combination with immunosuppression. Mucosal invasion has been demonstrated in mice; the organism was identified in the lamina propria and muscle layers of the colon. 4
Blastocystis has been isolated from the feces of domestic ducks, chickens, and geese,3,15 but it has only been definitively demonstrated to cause disease in experimentally infected rats. 16 It has been identified in samples from cynomolgus macaques and dogs with diarrhea.2,19 To our knowledge, extra-enteric Blastocystis infection in animals has not been reported previously; Blastocystis organisms and bacteria in aspirates from the facial swelling of the duck in our report may have been the result of fecal contamination of a wound. The absence of inflammatory cells in the fluid may indicate that the lesion was “walled-off” and chronic. Hematogenous spread from the gastrointestinal tract is unlikely given the location and focal nature of the lesion. The duck may have been immunosuppressed, but there was no clinical evidence to support that speculation.
Metronidazole and potentiated sulfonamides have anti-protozoal activity and have been used to treat Blastocystis infections. Metronidazole-resistant strains of Blastocystis are reported 17 ; the cyst form appears to be drug resistant. 18 In our case, the lesion was not resampled on recurrence, therefore the precise cause of the swelling cannot be determined, and the reappearance of the mass cannot be confirmed to be the result of Blastocystis treatment failure.
Footnotes
Acknowledgements
We thank Dr. Pauline Scanlan (APC microbiome Institute, University College Cork, Éire) for performing the PCR and sequencing on this sample and reviewing the manuscript, and Iain Peters (TDDS, Exeter, UK) for extracting the DNA.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
