Abstract
Superabsorbent sodium polyacrylate polymeric hydrogels that retain large amounts of liquids are used in disposable diapers, sanitary napkins, and other applications. These polymers are generally considered “nontoxic” with acute oral median lethal doses (LD50) >5 g/kg. Despite this favorable toxicity profile, we identified a novel toxic syndrome in dogs and rats following the ingestion of a commercial dog pad composed primarily of a polyacrylic acid hydrogel. Inappropriate mentation, cerebellar ataxia, vomiting, and intention tremors were observed within 24 h after the ingestion of up to 15.7 g/kg of the hydrogel by an adult, castrated male Australian Shepherd mix. These observations prompted an experimental study in rats to further characterize the toxicity of the hydrogel. Adult, female Sprague Dawley rats (n = 9) were assessed before and after hydrogel ingestion (2.6–19.2 g/kg over 4 h) using a functional observation battery and spontaneous motor activity. Clinical signs consistent with neurotoxicity emerged in rats as early as 2 h after the end of hydrogel exposure, including decreased activity in an open field, hunched posture, gait changes, reduced reaction to handling, decreased muscle tone, and abnormal surface righting. Hydrogel-exposed rats also had reduced motor activity when compared with pre-exposure baseline data. Rats that ingested the hydrogel did not develop nervous system lesions. These findings support the conclusion that some pet pad hydrogel products can induce acute neurotoxicity in animals under high-dose exposure conditions.
Introduction
Polyacrylic acid is extremely hydrophilic and can absorb over 100 times its weight in water, producing a “hydrogel.” 2 Hydrogels are polymeric materials that swell and retain a significant fraction (>20%) of water within their structure when they are placed in water. 21 This absorbent property has led to widespread commercial applications in medicine, engineering, environmental remediation, and the pharmaceutical industry. 21 Polyacrylic acid is also used in diapers, sanitary napkins, hand sanitizers, mascara, hair-styling products, and moisturizers. 21
One advantage of these superabsorbent polymers is that they are relatively nontoxic, with acute oral median lethal doses (LD50) often >5 g/kg. 21 An acute oral LD50 of >40 g/kg has been reported for Rhotex GS, a 15:85 mixture of sodium polyacrylic acid and water in rats. 3 Hydrogels also have relatively low toxicity following repeated exposure. For example, rats given a structurally related cross-linked polyacrylic acid polymer ≤5% in their diet for 32 d or ≤3 g/kg/d in their diet for 93 d did not exhibit signs of overt toxicity.5,9
Although these polymers are considered nontoxic, significant adverse effects can be seen following massive ingestion. In 2015, a 3-y-old, 22.3 kg, castrated male Australian Shepherd mix was referred by a local veterinarian to the North Carolina State University Veterinary Hospital (NCSU-VH) for evaluation of acute onset of clinical signs (ataxia, vomiting, anxiety, intention tremors, and tachypnea) occurring within 24 h after the ingestion of ~0.35 kg piece of sodium polyacrylate–based hydrogel found within a commercial dog pad (estimated exposure = 15.7 g/kg). The owner reported that the outer plastic shell of the pet pad was torn open by the dog and the interior hydrogel material was missing. Visual examination of a vomitus sample collected shortly after ingestion verified the presence of the hydrogel material, thus confirming that exposure occurred. A complete blood count and serum biochemical analysis performed by the referring veterinarian showed azotemia and slightly elevated liver enzyme activities (Table 1). Initial neurologic examination at NCSU-VH revealed the presence of inappropriate mentation characterized by agitation and increased sensitivity to sound, profound ataxia and hypermetria in all 4 limbs, wide-based posture, intention tremors, bilateral absence of the menace response, and diffuse discomfort on palpation of the thoracolumbar spine. Based on the neurologic examination, the most likely neuroanatomic diagnosis was a multifocal process involving the forebrain and cerebellum. Although toxicity was strongly suspected, other differential diagnoses were considered, including infectious meningoencephalitis and neoplasia.
Summary of abnormal biochemical results observed in a dog after ingestion of a pet pad hydrogel.
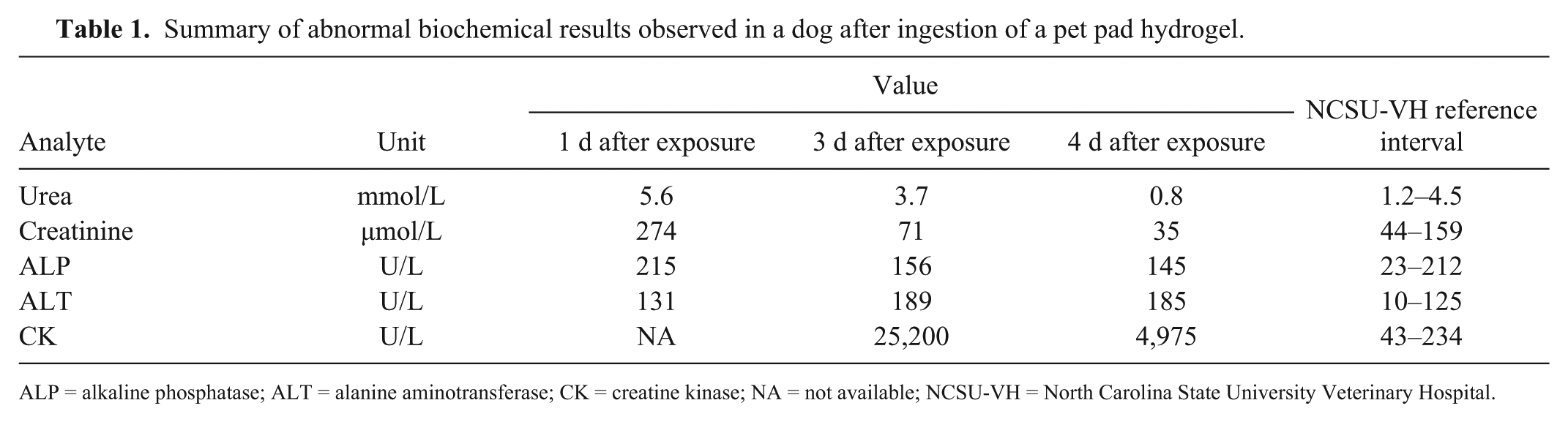
ALP = alkaline phosphatase; ALT = alanine aminotransferase; CK = creatine kinase; NA = not available; NCSU-VH = North Carolina State University Veterinary Hospital.
Laboratory tests for this dog explored potential toxic, infectious, and neoplastic etiologies. An ethylene glycol exposure test was negative. Magnetic resonance imaging (MRI) studies did not reveal any morphologic changes in the central nervous system (CNS), although focal ill-defined areas of contrast enhancement were noted in the left splenius, rectus capitis, and longus capitis muscles. These muscle lesions were felt to be consistent with inflammation secondary to muscle spasm. Cerebrospinal fluid (CSF) collected from the cerebellomedullary cistern after MRI was found to be within normal limits. A PCR-based panel evaluating CSF and whole blood for canine distemper virus (species Canine morbillivirus), West Nile virus, Borrelia burgdorferi, Neospora caninum and N. hughesi, Toxoplasma gondii, Anaplasma phagocytophilum, Ehrlichia canis, and Rickettsia spp. as well as serology for N. caninum were negative. Creatine kinase (CK) was markedly elevated by the following day (Table 1). Urinalysis revealed amber-colored urine with marked hematuria (250 × 109 RBC/L) and proteinuria (1.0 g/L). The dog was treated with intravenous fluid therapy, and within 3 d the dog’s neurologic conditions had improved significantly, with only minimal residual ataxia, and the hematuria had grossly resolved allowing discharge of the dog. Two wk after ingestion, the owner reported that the dog had recovered fully and resumed normal activity. Laboratory tests did not reveal common toxic and infectious etiologies or a CNS mass as the cause of this dog’s acute neurologic crisis, suggesting that the likely cause was consumption of the hydrogel material in the commercial dog pad. The limited availability of information about the potential toxicity of polyacrylic acid hydrogels in this commercial product prompted our follow-up rat study, which tested the hypothesis that oral exposure to this polyacrylate-based hydrogel can elicit toxicity in mammals.
Materials and methods
Animals
Our study was conducted under federal guidelines for the care and use of laboratory animals and was approved in advance by the NCSU Institutional Animal Care and Use Committee. 16 Nine adult (12-mo-old) female Sprague Dawley rats (Charles River, Kingston, NY) were used in our study and housed under conditions reported previously. 4 Rats were given water and pelleted chow (Purina 5001 diet, Purina Mills, St. Louis, MO) ad libitum except as otherwise noted.
Test material and dosing
The pad used in this project (Cool pet pad, The Green Pet Shop Enterprises, Northbrook, IL) was a second unused and undamaged pad that was originally purchased by the owner of the Australian Shepherd mix dog. The product material safety data sheet (MSDS) reported that it contained the sodium salt of polyacrylic acid (30%) and water (70%) and had an oral rat LD50 of >40,000 mg/kg. Consistent with the described super-absorbent nature of the polymer, the hydrogel expanded when placed in water (pH = 7) or acidified water (pH = 2).
Beginning ~10 d before exposure, the rats were acclimated to consuming food over a short time period by restricting the time food was available to 4 h/d. Some rat chow was also flavored with a small coat of peanut butter to promote consumption of the rat chow in the 4-h time frame. Food was withheld from the rats for ~24 h prior to the day of exposure (day 0). Rats were transferred from their domiciliary cage to an empty dosing cage supplied with water at ~0900 h. Each rat was then given a pre-weighed hydrogel sample, ~32 cm2 (mean weight = 12.11 ± 0.58 g), which was covered with a small quantity (<5% of the weight of the hydrogel) of peanut butter as a flavoring agent. Rats licked off the peanut butter coating and then voluntarily consumed the hydrogel over a 4-h period. At the end of the 4 h period, all remaining hydrogel was collected and weighed. The difference in the original weight of hydrogel presented and the remaining weight at the end of the 4-h feeding period was used to estimate the exposure. Introduction of the hydrogel was considered to be the start of the study (designated as time 0). Afterwards, the rats were returned to their domiciliary cage and provided 4 untreated pieces of rat chow.
Clinical and neurologic assessments
Body weights were measured on days −5, 0, and +1. A functional observation battery (FOB) was performed in the afternoons (1400–1600 h) of days −5, 0, and +1. Quantitative and descriptive measures of the FOB have been described previously and include evaluation of the animal’s posture, gait, clinical signs, and response to stimuli.7,14,15 Briefly, clinical signs were assessed while the animal was in their domiciliary cage and then during a 2-min observation period in a 75 × 38 × 20 cm open field. The 2-min open-field period was followed by an assessment of visual approach response, response to a novel odor (vanillin), acoustic startle response, tail pinch response, visual placing, and surface-righting reflex. Hind-leg splay was determined by painting the middle toe pads of the hind paws with a watercolor paint and dropping the rat from a height of 30 cm onto a sheet of white unlined paper. Hindlimb and forelimb grip strengths were measured using force gauges (Chatillon DFIS-10, Wagner Instruments, Greensboro, NC). Grip strength and splay evaluations were performed twice (average value was used), except that these evaluations were not performed on severely affected rats.
Motor activity was measured on the mornings (0900 h) of days −6 and +1 using an infrared system (Actitrack, Panlab, Barcelona, Spain). Each Plexiglas arena (45 × 45 × 30 cm) was equipped with 2 frames with photocells located at 2 and 12 cm above the cage floor. Each animal was initially placed in the center of the arena, and spontaneous behavior was tracked for 15 min. During the test, the horizontal locomotor activity and vertical activity (number of rearing episodes breaking the photocell beams of the upper frame) were recorded. At the end of the session, animals were returned to their home cage, and arenas were wiped with water to remove residual olfactory cues.
Postmortem examination and neuropathology
Rats (n = 6; Table 2) were prepared for neuropathology assessment using methods published previously. 1 Rats were anesthetized with sodium pentobarbital (150 mg/kg given by intraperitoneal injection) and perfused with ~0.15 L of a heparinized saline flush solution followed by fixation with ~0.25 L of 4% methanol-free formaldehyde (reconstituted from paraformaldehyde powder) in 0.05 M sodium phosphate buffer (pH 7.4). Solutions were maintained at 4°C until immediately prior to perfusion. The brain, spinal cord, and one sciatic nerve with its main branches were exposed, examined grossly, and immersed in fresh fixative at 4°C for at least 24 h prior to histologic processing for neuropathology evaluations. Dorsal root ganglia; spinal nerve root fibers; brain (olfactory bulbs, cerebral cortex [associative, motor, and sensory areas], striatum, hippocampus, thalamus, hypothalamus, cerebellum, pons, and medulla oblongata); spinal cord (2 blocks from the lumbar intumescence); and sciatic nerve (adjacent to the sacrum and just proximal to the stifle) from one hindlimb were embedded in paraffin and sectioned serially at 5–6 μm to permit examination by bright-field light microscopy. Sections were stained with hematoxylin and eosin (to permit an assessment of general architecture) or anti-glial fibrillary acidic protein (to reveal any early glial reactions).
Estimated exposure of individual rats consuming a pet pad hydrogel.
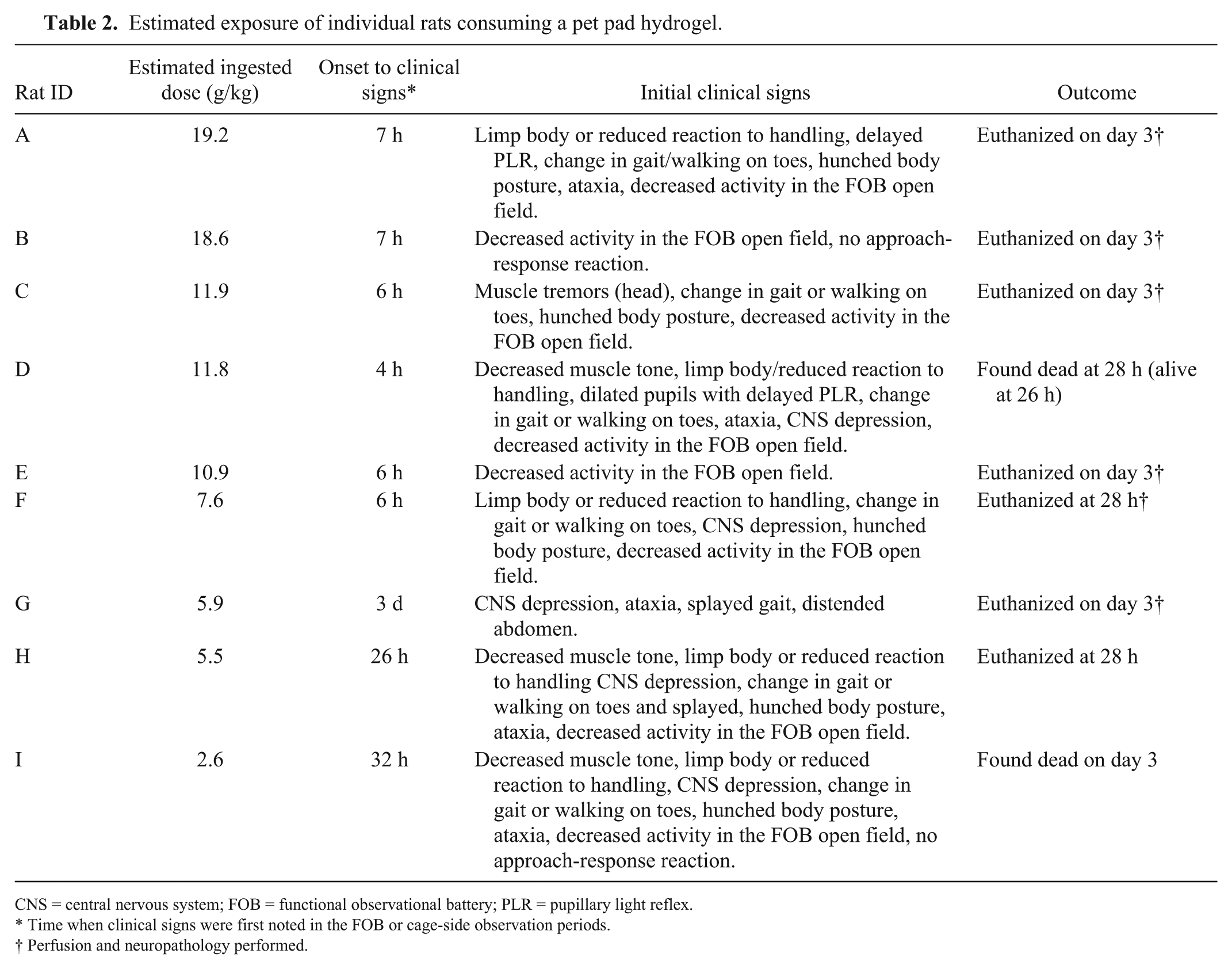
CNS = central nervous system; FOB = functional observational battery; PLR = pupillary light reflex.
Time when clinical signs were first noted in the FOB or cage-side observation periods.
Perfusion and neuropathology performed.
Chemical characterization of the hydrogel
Swollen gel samples, as well as gel extracts and reference sample (polyacrylic acid sodium salt, Sigma Aldrich, St. Louis, MO) were analyzed (360 MHz 1H-NMR spectrometer, Bruker, Billerica, MA). Polyacrylic acid (0.1 g) was lyophilized and re-dissolved in 0.02 L of deuterium oxide (D2O; Sigma Aldrich) and subsequently analyzed by 1H-NMR spectrometry. Hydrogel extracts were obtained by immersing 0.1 g of the hydrogel in 0.02 L of D2O for 24 h. Afterward, samples of the hydrogel and a 0.005-L aliquot of the liquid phase were analyzed separately by 1H-NMR spectrometry. Additional 0.1-g samples of the hydrogel and the polyacrylic acid reference samples were lyophilized (FreeZone 2.5 L benchtop freeze dry system, Labconco, Kansas City, MO) for 28 h prior to infrared spectroscopy. Infrared analysis of the dried powder was conducted using a Fourier-transform infrared (FT-IR) spectrometer (Nicolet 380, Thermo Fisher Scientific, Waltham, MA) fitted with a single reflection attenuated total reflection (ATR) system (Silver Gate evolution ATR, Specac, Orpington, England).
Statistics
Pre- and post-exposure quantitative data were compared using a paired-samples t-test. Categorical FOB data were analyzed using a nonparametric test (Pearson chi-square). All data were analyzed using JMP Statistical Software (SAS, Cary, NC). Unless otherwise noted, the significance level for any given statistical test was set at p ≤ 0.05, and mean ± standard deviation values are reported.
Results
All pre-dosing FOB evaluations (on day −5) were within normal limits. All rats licked the peanut butter off the hydrogel immediately upon presentation. Afterwards, all rats consumed portions of the hydrogel starting at 2 h after the start of the feeding trial. The estimated mean amount of hydrogel material ingested was 10.4 ± 5.7 g/kg (range: 2.6–19.2 g/kg; Table 2). The most common neurologic abnormalities observed during day 0 FOB included decreased activity in the FOB open field, changes in posture and reactivity to handling, and change in gait or walking on toes (Table 2). Two rats that consumed 5.9–18.6 g/kg of the hydrogel had no abnormal findings during day +1 FOB. All other rats demonstrated one or more abnormalities during day +1 FOB. Hydrogel ingestion was also associated with decreased motor activity, reduced numbers of rears, decreased body temperature, and decreased body weight on day +1 (Table 3).
Quantitative functional data in hydrogel-exposed rats.
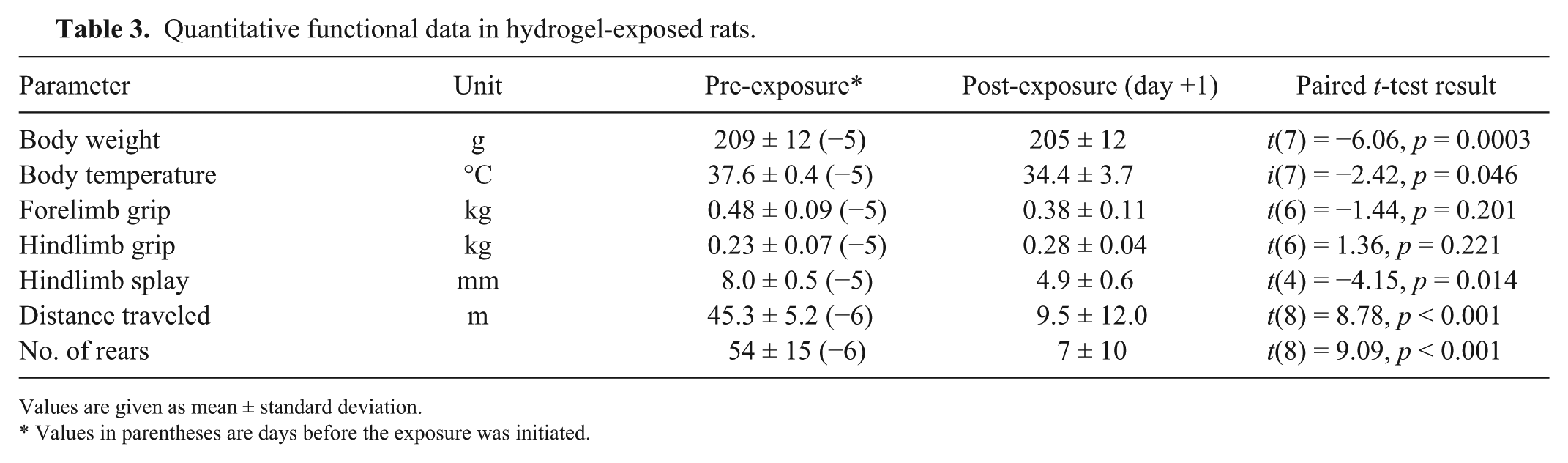
Values are given as mean ± standard deviation.
Values in parentheses are days before the exposure was initiated.
Histologic structures within examined sections of the nervous system for all 6 rats that survived to day +3 or 1 of 2 rats that were euthanized on day +2 (Table 2) were all within normal limits.
The 1H-NMR spectrum of the hydrated hydrogel is consistent with the polyacrylic acid reference sample but also shows peaks in the 6–8 ppm range that do not appear in the polyacrylic acid reference sample (Fig. 1). These peaks are also seen in the 1H-NMR spectrum of the D2O extract. The IR spectrum of the lyophilized gel also suggests the presence of alkenes and/or amino groups (Fig. 2).
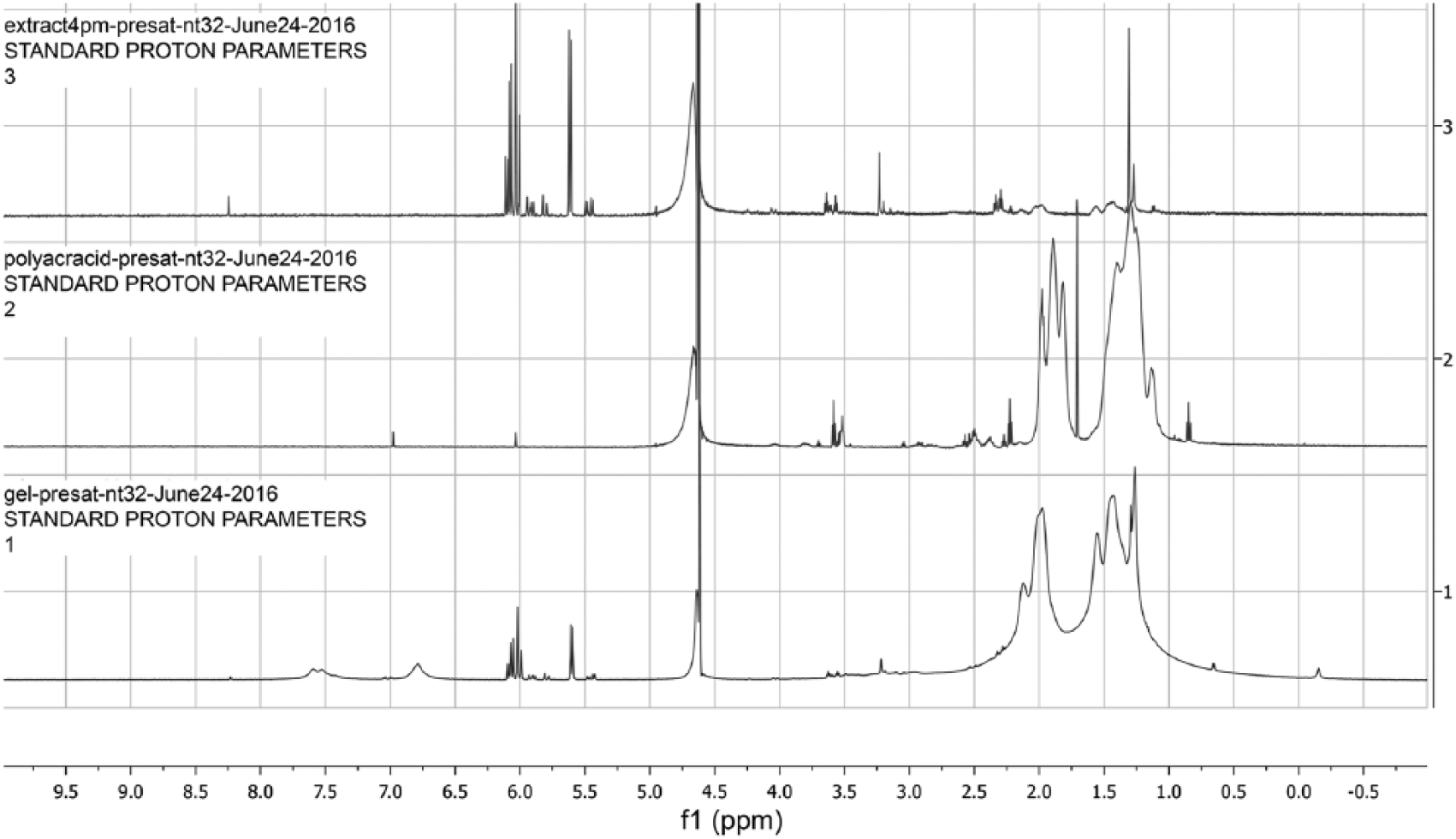
1H-NMR spectra of the deuterium oxide extract of the hydrated hydrogel from the commercial dog pad (top), polyacrylic acid reference standard (middle), and the hydrated hydrogel from the commercial dog pad (bottom).
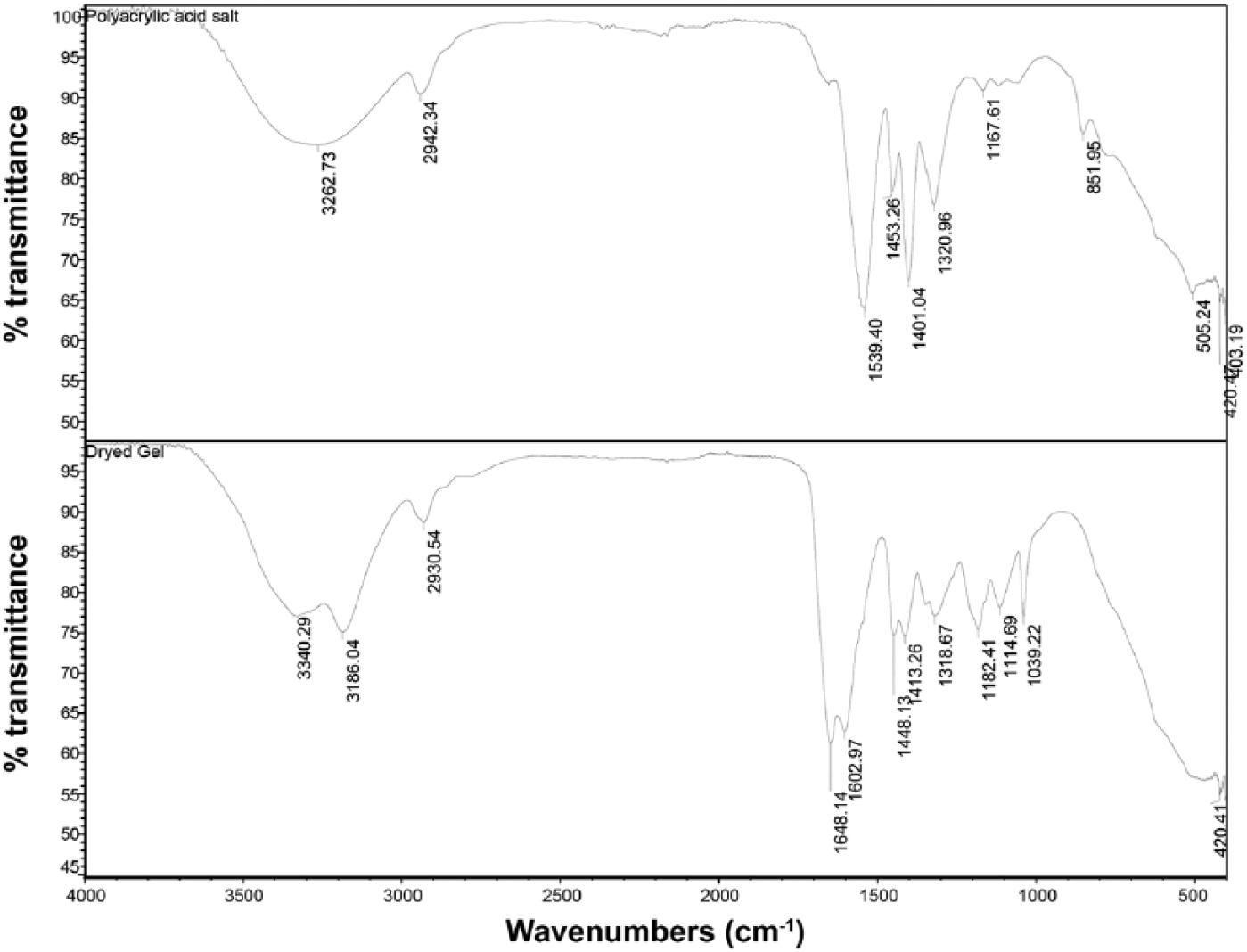
Fourier-transform infrared spectra of polyacrylic acid salt reference standard (top) and lyophilized gel sample taken from the commercial pet pad (bottom).
Discussion
An adult, male Australian Shepherd mix that was presented to NCSU-VH had developed vomiting, altered mentation, ataxia, intention tremors, and tachypnea within several hours following the ingestion of ≤15.7 g/kg of the hydrogel filling a commercial pet pad. Neurologic signs consistent with suspected acute forebrain and cerebellar neurotoxicity persisted for at least 96 h before the dog began to recover slowly. Data from the ASPCA Animal Poison Control Center indicate that a mixture of gastrointestinal (e.g., vomiting, diarrhea) and neurologic (e.g., ataxia, tremors, hyperesthesia) signs are often observed in dogs consuming hydrogels (Dr. T. Wismer, pers. com., 2017). When considered together, these findings suggest that polyacrylic acid hydrogel ingestion may induce clinically significant gastrointestinal irritation and neurotoxicity in dogs.
Evidence of acute neurotoxicity was also observed in rats that ingested the pet pad hydrogel. The onset of neurologic signs in rats appeared to be dose- and time-dependent. Rats ingesting >7 g/kg of the hydrogel developed behavioral changes or abnormal neurologic signs within 6–12 h after ingestion, whereas rats ingesting smaller amounts of hydrogel (2.6–5.9 g/kg) had no abnormal clinical or neurologic findings 12 h after ingestion. Ingestion of hydrogel by rats was often associated with one or more abnormalities on the neurotoxicity test battery used. The endpoints used in our study (FOB, motor activity, and neuropathology) are commonly incorporated in rodent neurotoxicity test batteries.8,15,17 Abnormal responses seen in rats during the post-exposure FOBs predominantly involved changes in neuromuscular function and coordination. Despite finding reduced muscle tone during the post-exposure FOBs, changes in grip strength were not observed during the study. This result is somewhat surprising given that hindlimb grip strength measures digital flexor muscle responses initiated by the grasp reflex and can be correlated with muscle tone.12,18 Decreased spontaneous motor activity and reduced rearing behaviors were also seen in the hydrogel-exposed rats. Spontaneous locomotor activity is an apical test of neuronal function and is a sensitive measure of nervous system effects.15,19 Although decreased motor activity is often associated with chemicals that affect the nervous system, changes in motor activity cannot be attributed to specific neuronal substrates. 15 Previous investigators have reported that poor clinical condition is not always associated with decreased motor activity in rodents, suggesting that the hydrogel may have direct effects on the nervous system. 10 Ataxia, splayed gait, slowed surface righting, and slowed pupillary light reflexes were also observed in some affected rats, which would further support the conclusion that the hydrogel is neurotoxic. None of the rats that ingested the hydrogel developed anatomic lesions in brain indicative of acute neurotoxicity (e.g., neuronal necrosis, increased glial reactivity). This finding is consistent with the lack of structural brain MRI changes seen in the dog that ingested hydrogel. Additional studies are needed to determine whether changes in brain neurochemistry could account for the observed neurologic effects.
Ingestion of the hydrogel may have additional effects on other organs or systems. Some of the changes in posture (hunched appearance) and gait (walking on tiptoes) seen in hydrogel-exposed rats during the post-exposure FOBs may be indicative of abdominal discomfort and pain in rats. 11 If pain was present, then the most likely cause is marked abdominal distension resulting from expansion of the hydrogel in the rat’s stomach as a result of water absorption. Expanded hydrogel resulting in gastric dilation was observed in several animals that died or were euthanized within 48 h of hydrogel ingestion. This gastric dilation effect may be more prominent in rats given that, unlike dogs, they cannot vomit. 6 Gastric impaction from the hydrogel may have contributed directly to some clinical signs and the death of exposed rats. Fatal gastrointestinal impaction has previously been reported in birds that consumed a polyacrylamide-based hydrogel. 13
One of the limitations associated with our case is identification of the neurotoxic chemical found in the pet pad hydrogel. Based on the MSDS provided by the manufacturer, the hydrogel contains a 30:70 mixture of polyacrylic acid and water. Chemical analyses (nuclear magnetic resonance and IR spectrometry) of the gel from our case confirmed the presence of polyacrylic acid along with other unidentified compounds. For example, the presence of the 1H-NMR peaks seen at 6–8 ppm suggests that leachable short carbon chain (3–8 carbons) molecules in the gels contain carbon–carbon double bonds, oxygen, and possibly nitrogen. Although the oxygen seen in the NMR can be associated with some polyacrylic acid chains present in the extracts, the short carbon–carbon chains with double bonds and amino groups are likely to be external contaminants. Although the shift of the carbonyl peak in the IR spectra from 1.539 (carboxylic acid salt) to 1.645 m-1 is consistent with gel formation, 20 the additional peaks at ~1.6 m-1 indicate the presence of alkene and/or amine groups, and the peaks at ~3.3 m-1 are indicative of primary amino groups. The Chemical Abstract Services (CAS) number assigned to the product (CAS 9003-04-7) is used for a broad family of structurally related polymers composed of the sodium salt of polyacrylic acid. In general, CAS registers polymers based on their structural repeating unit (e.g., in our case, the repeating unit is [C3H4O2]x•xNa). The exact composition and physicochemical properties associated with these polymers can vary based on polymer length, crosslinking, and other factors. 21 Thus, conclusions regarding hydrogel toxicity drawn from our study are applicable to the formulation found in the commercial pet pad product that was tested in our laboratory and may not be applicable generally to other products that contain polyacrylic acid–based hydrogels. Further work will be required to evaluate whether the clinical picture produced by this hydrogel is specific to its chemical composition or may be ascribed more generally to its physical properties as a hydrogel.
Footnotes
Acknowledgements
We thank Dr. Tina Wismer of the ASPCA Animal Poison Control Center for her assistance with this project.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received financial support for this research from the NCSU College of Veterinary Medicine. Additional undergraduate funding was provided through an NCSU Undergraduate Research Grant Award.
