Abstract
Two adult dogs with the same owner were intoxicated by ingestion of fertilizer composed of residual plant material of the castor bean plant (Ricinus communis L.). Both dogs died within 2 and 3 days, respectively, after the first signs of vomiting and abundant hemorrhagic diarrhea. Toxicologic and histopathologic examinations were performed on different organs. Histopathologic examination of the kidneys revealed tubular degeneration and necrosis and membranous glomerulonephritis. Additionally, myocardial degeneration with localized inflammation, lymphoid necrosis, and depletion in the spleen and mesenteric lymph nodes and hemorrhagic ulcerative gastroenteritis were found. The 2 cases could be used to elucidate the lethal dose of ricin and the histopathologic lesions in dogs.
Ricinus communis L. (i.e., castor bean, mole bean, Palma Christi, wonder tree, African coffee tree, Mexico weed) is an ornamental plant introduced from the West Indies. It produces oil that is used for industrial and medicinal purposes. 1,19 The residual plant material from the oil extraction, castor bean cake, is largely used as fertilizer. The plant contains ricin, a phytotoxin (lectin) that is considered one of the most potent plant toxins and is listed as a Category B bioterrorism agent by the Centers for Disease Control and Prevention. 15 All parts of the plant are toxic, but the highest levels of ricin can be found in the hard tick-like seeds, which need to be crushed or broken to release the toxic component. 20 The toxic lectin inhibits protein synthesis, leading to cell death, and can affect different species of domestic mammals and poultry, but also humans. 3,7,13
A 6-year-old male Australian Shepherd (dog 1) and a 12-year-old male Golden Retriever (dog 2) from the same owner and living in the same household were presented for postmortem examination (24 and 48 hr after death, respectively, with a suspicion of ricin poisoning). The carcasses were kept at a temperature of approximately 15°C before postmortem examination. Both animals were brought to the veterinarian with symptoms of sudden prominent uncontrollable vomiting followed by abundant hemorrhagic diarrhea after the gardener had fertilized the plants using a commercially available product composed entirely of castor bean (Ricinus) residual plant material after extraction of castor oil. In spite of intensive therapy, the animals died 48 and 72 hr, respectively, after the first clinical signs leading to shock.
Macroscopic lesions consisted of prominent hemorrhagic, ulcerative gastroenteritis and parenchymal congestion. The lesions found in the dog that died after 48 hr (dog 2) were more pronounced than those in the other dog (dog 1). Samples of the myocardium, intestines, kidneys, liver, spleen, and lymph nodes were taken for histopathologic examination, and samples of stomach, intestinal (cecum) content, liver, and kidney were taken for toxicologic examination.
The most prominent histopathologic lesions were in the kidneys. Multifocal renal tubular degeneration and necrosis were present. In the glomeruli, the main feature was the presence of eosinophilic fluid and debris surrounding the glomerular capillaries in both dogs. Focal thickening of the basement membrane of Bowman's capsule, characteristic of a membranous glomerulonephritis, was observed with the use of periodic acid—Schiff staining. Several glomeruli also were shrunken and had solidification of the tufts. Phosphotungstic acid—hematoxylin (PTAH) staining failed to reveal deposits of fibrin (Fig. 1).
Histologic examination of the jejunal mucosa revealed congestion and hemorrhage, erosion and disruption of villi, infiltration by degenerative leukocytes, and abundant nuclear debris. The crypt epithelial cells were focally necrotic, and the submucosa was diffusely hemorrhagic with congestion of capillaries. The myocardium contained multiple degenerative myofibers with localized limited lymphocytic infiltrates. Occasional foci of hepatic degeneration were also noticed. Follicles in the spleen and the lymph node had numerous lymphocytes with pyknotic or karyorrhectic nuclei, resulting in depletion or total absence of the follicles. Thrombi were not seen in the organs examined in the present study.
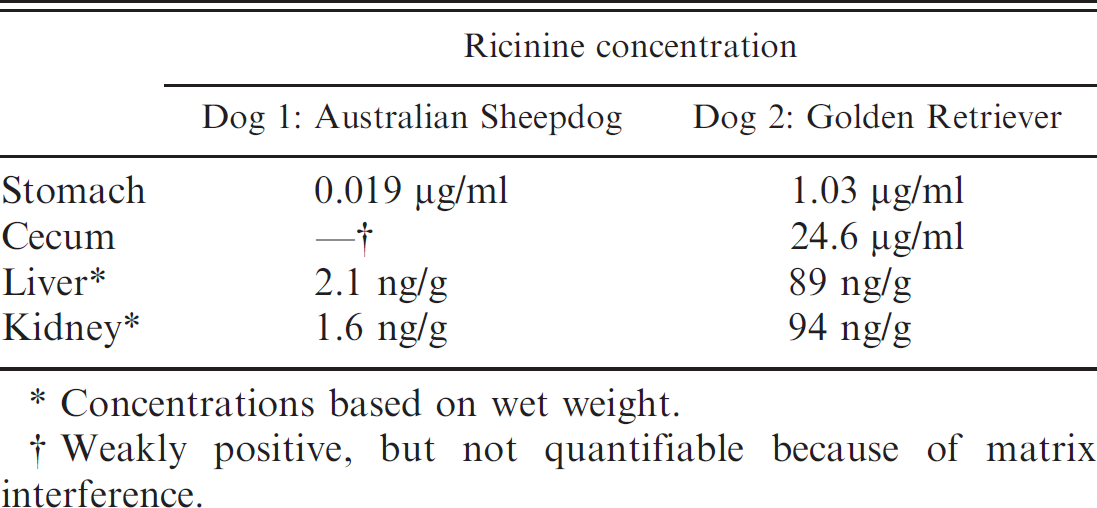
Exposure to ricin was determined by identification of the biomarker ricinine in the gastric and intestinal (cecal) content, as well in the liver and kidneys, by solid phase extraction and liquid chromatography—tandem mass spectrometry, with the electrospray source in positive ionization mode. Multiple reaction monitoring was used for specific detection. 4 The levels of ricinine contained in the tissues of these dogs were prominently different. Tissues from the Golden Retriever, which died first, contained much higher levels of ricinine (see Table 1). Ricinine, described as a biomarker for ricin exposure, is a piperidine alkaloid toxin that is present in small amounts in castor beans and leaves. This biomarker also is coextracted with ricin. 12,15,19 The toxic principle of R. communis seeds is the toxoalbumin ricin, a potent inhibitor of protein synthesis. Two glycoproteic chains, A and B, constitute the ricin molecule. The B chain allows endocytosis of ricin through binding to the cell surface via galactoside-containing proteins. The A chain enters the cell, blocks protein synthesis by disruption of 28S ribosomal nucleic acid, and ultimately causes cellular death. 16,17 When ingested, the castor bean is nontoxic as long as the hard, water-impermeable seed coat remains intact. Toxicity depends on the level of mastication or maceration of the seeds. 2 It is known that ricin is metabolized quickly before excretion and is heavily localized to the area of exposure. Thus, ricin may be missed in some clinicopathologic samples. 6,18 The difference in times of death after exposure of these 2 dogs to ricin could be related to different initial exposure levels rather than differences in metabolism, distribution, or excretion of the toxin. The main advantage of ricinine biomonitoring stems from the small size of the molecule (164 g/mol), which can easily be extracted and quantitated. An animal study verified that ricinine can be monitored in urine up to 48 hr after exposure. 5,12 It may be that the dog with the lower concentration of ricinine at the time of death had a lower initial exposure to the toxin. However, it also seems plausible that the dog could have metabolized or excreted more of the biomarker because it lived longer postexposure. However, details on the kinetics of ricinine metabolism are still unknown.
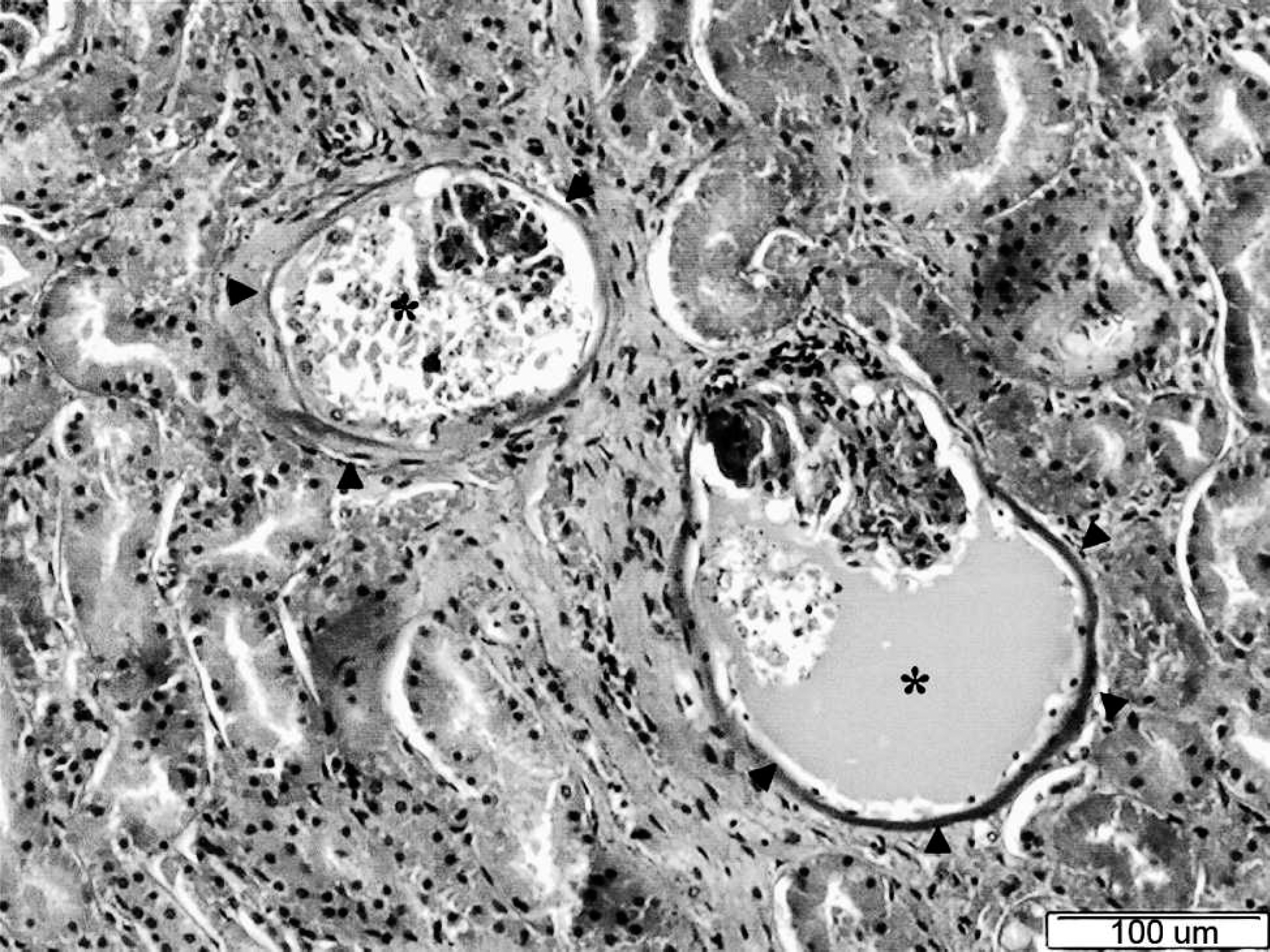
Kidney, cortex (dog 1). Prominent diffuse glomerular lesions are noted characterized by thickening of the Bowman's capsule (arrowheads) and filling of Bowman's spaces with different quantities of eosinophilic fluid containing detached endothelial cells and necrotic cellular debris (*). Additional swelling and necrosis of the epithelial cells of the proximal tubules also are present. Hematoxylin and eosin. Bar = 100 μm.
Ricinine concentrations determined with the use of solid phase extraction and liquid chromatography—tandem mass spectrometry in different tissues of 2 dogs.
Concentrations based on wet weight.
Weakly positive, but not quantifiable because of matrix interference.
Ricin intoxication in dogs has been described in the veterinary literature, but reports of histopathologic lesions in dogs associated with such intoxication are scarce, 1,15,21 probably because only 9% of toxicoses actually end in death or euthanasia. 1 Clearly, similar lesions can be seen in different species, but some of them are only described in laboratory models. 9,22,23 In experimental model studies with rats and mice, the major lesion associated with a lethal dose of ricin was impairment of renal function with a typical histopathologic lesion called “glomerular thrombotic microangiopathy.” This ricin-induced lesion has been proposed as a model for hemolytic uremia syndrome. 9,22 In both dogs in the current study, several renal changes could be detected, but the presence of fibrin thrombi could not be demonstrated in the glomerular capillary loops, perhaps because of species differences, the possibility that the rodents were sacrificed after advanced toxicosis occurred, or both. 9,22,23 Another explanation for the absence of fibrin deposits could be fibrinolysis. 14 In humans, fibrinolysis occurs in vivo and in postmortem conditions. 11,14 However, unlike the situation in humans, primary disorders of the fibrinolytic system have not been characterized yet in domestic animals, although alterations of the fibrinolytic system associated with inflammatory conditions are frequently recognized as a prelude to diffuse intravascular coagulation. 10 The current study presents the histopathologic lesions of the kidney associated with lethal ricin toxicosis in dogs. Given that these animals had vomiting and hemorrhagic diarrhea, some of these histologic changes could be attributed to shock, as described in human beings. 1 Additionally, the present study also presents the concentrations of ricinine, a biomarker for ricin, in various tissues. The information concerning histologic lesions and concentrations of ricinine could help in determining the lethal dose of this compound in dogs, which is currently unclear. 8
Acknowledgements The staff of the Unit of Pathology (CODA/CERVA) is thanked for technical assistance. Christian Puttevils (Ghent University) is acknowledged for his help in performing the PTAH staining. The authors also thank the owners of the dogs for their cooperation in this investigation.
