Abstract
An adult female captive-born Atlantic guitarfish (Rhinobatos lentiginosus) was found acutely moribund on exhibit and died soon after presentation. Abnormalities on autopsy were focal cutaneous erythema on the tail, a small liver, many variably sized friable ovarian follicles, and coelomic effusion. Histologic examination revealed systemic bacterial embolization, and yolk coelomitis with minimal associated inflammation and some mineralization. Bacterial culture of blood and coelomic effusion grew a rapidly growing Mycobacterium species that was further identified as Mycobacterium chelonae by PCR amplification and sequencing of the RNA polymerase subunit beta (rpoB) gene of isolated genomic DNA. Concurrent reproductive disease may have caused immunosuppression, thus predisposing to the mycobacterial infection. At another institution, an adult male wild-caught Atlantic guitarfish was found dead on exhibit with no premonitory signs. Abnormalities on autopsy were a thin body condition, small liver, and coelomic effusion. Histologic examination revealed acute mycobacterial septicemia. M. chelonae was also identified in this fish by PCR amplification and sequencing. Mycobacteriosis has rarely been reported in captive elasmobranchs. Guitarfish may have greater susceptibility to mycobacteriosis than other elasmobranchs, and acute and chronic manifestations of the disease may exist in this species.
Nontuberculous mycobacteria (NTM) are ubiquitous environmental bacteria found in water and soil worldwide. They are gram-positive, pleomorphic, non-motile, acid-fast, aerobic rods. 8 Mycobacteriosis of fish, also known as fish tuberculosis, is a very common disease of teleosts, having been documented in many freshwater and saltwater species.8,14 There are several NTM species that cause disease in teleosts, with the most commonly recognized being Mycobacterium marinum, M. chelonae, and M. fortuitum. Mycobacteriosis is typically characterized as a chronic progressive granulomatous disease, usually systemic in nature, either as a primary or single disease process or a secondary process with concomitant diseases.8,14 Acute fulminant mycobacteriosis is common in sygnathids. 4 Although common in teleosts, there have been no reports of mycobacteriosis in elasmobranchs until very recently. 7 M. chelonae was identified in a yellow stingray (Urobatis jamaicensis) and Atlantic guitarfish (Rhinobatos lentiginosus), and M. avium identified in an epaulette shark (Hemiscyllium ocellatum).2,6,10 We describe herein acute systemic M. chelonae infection in 2 adult Atlantic guitarfish. Unlike the previous Atlantic guitarfish case in which only the spleen was analyzed, 2 we performed multi-organ histologic assessments and cultures.
Case 1
On July 9, 2014, an ~2-y-old, 1.1-kg, female captive-born Atlantic guitarfish (R. lentiginosus) was acutely moribund, in dorsal recumbency, and weakly ventilating. The guitarfish had been exhibited in a 102,000-L natural saltwater, semi-open, recirculating, mixed-species touch tank housing several other elasmobranch species and American horseshoe crabs (Limulus polyphemus). Prior to being exhibited, the fish had completed routine quarantine including treatment with 5 ppm praziquantel (Fishman Chemical, Vero Beach, FL) immersion for 20 d in 2012. She was moved onto exhibit in May 2013 upon reaching an appropriate size for the exhibit. The aquarist reported that the fish had been eating the day before presentation. Physical examination revealed mildly thin body condition and a few 3-cm foci of erythema and petechiae on the left dorsolateral aspect of her tail. The animal was administered 1.2 mg of dexamethasone sodium phosphate (4 mg/mL, Dexium-SP, Bimeda LeSueur, Le Sueur, MN) intramuscularly (IM) and 30 mg of florfenicol (300 mg/mL, Nuflor injectable solution, Intervet, Summit, NJ) IM; then, ventilation ceased. Two rounds of emergency resuscitative drugs were immediately administered—6 mg of doxapram (20 mg/mL, Dopram injection, West-Ward Pharmaceutical, Eatontown, NJ) intravenously (IV), 6 mg of doxapram applied topically to the gills, 0.054 mg of atropine sulfate (0.54 mg/mL, Med-Pharmex, Pomona, CA) IV, and 0.16 mg of atropine sulfate applied topically to the gills—with no response. Echocardiography identified cessation of cardiac activity. Two soft egg cases were expelled from the vent immediately upon death.
The carcass was immediately refrigerated and an autopsy was performed 1.5 h later. Blood was collected at autopsy and placed on a culture swab in Amies medium without charcoal (BBL CultureSwab Plus, BD, Franklin Lakes, NJ) for aerobic bacterial culture. The gills were mildly reddened and coated by a moderate amount of mucus. Cytologic examination of skin scrapings revealed no abnormalities. There were two 3-cm diameter foci of cutaneous erythema on the left dorsolateral aspect of the tail and patchy erythema on the most caudal dorsal fin. The liver appeared relatively small, tan, and mottled, with a 2 × 1 cm area of gray coloration on the left ventral surface. There were many ovarian follicles bilaterally, 0.5–3.0 cm diameter. The ovaries filled the majority of the coelomic cavity. There was a small volume of straw-colored coelomic effusion that was collected for aerobic bacterial culture (BBL CultureSwab Plus, BD) and minimum inhibitory concentration (MIC) antimicrobial susceptibility testing. The remaining organs appeared grossly normal. The blood and coelomic fluid cultures were submitted to IDEXX Laboratories (North Grafton, MA), and cultures were performed as described previously. 9 Multiple tissues were fixed in 10% neutral-buffered formalin (Thermo Scientific, Richard Allan Scientific, Kalamazoo, MI) for histologic examination.
A rapidly growing presumptive Mycobacterium species was isolated from both blood and coelomic effusion cultures. The blood culture also grew Staphylococcus hominis subspecies hominis. The coelomic fluid isolate was forwarded to the National Jewish Health Advanced Diagnostic Laboratories (Denver, CO) for Mycobacterium species identification and MIC antimicrobial susceptibility analysis.
Genomic DNA from the coelomic fluid colonies was isolated with a DNA extraction kit (PrepMan Ultra sample preparation reagent, Life Technologies, Foster City, CA). PCR amplification of a segment of RNA polymerase subunit beta (rpoB) gene was then performed using rpoB-specific primers (forward 5’-GGCAAGGTCACCCCGAAGGG-3’and reverse 5’-AGCGGCTGCTGGGTGATCATC-3’). 1 The thermocycling conditions for the rpoB PCR reaction were denaturation at 95°C for 5 min and then 40 cycles at 95°C for 15 s, 61.8°C for 20 s, and 72°C for 75 s, with a final extension step of 72°C for 2 min. The amplified DNA product was visualized via gel electrophoresis (FlashGel system, Lonza Rockland, Rockland, ME). DNA was sequenced (BigDye Terminator kit v1.1, Life Technologies, Carlsbad, CA) and evaluated (3730xL DNA analyzer, Applied Biosystems, Foster City, CA). rpoB sequence assembly and DNA alignment was performed using bioinformatics software (BioNumerics v.6.6, Applied Maths, Austin, TX; Totten et al. Species level identification of organisms within the Mycobacterium abscessus group. Proc 113th Gen Meeting Am Soc Microbiol; May 2013; Denver, CO). The obtained consensus sequence was then analyzed using Basic Local Alignment Search Tool (BLAST) through the National Center for Biotechnology Information (http://blast.ncbi.nlm.nih.gov/Blast.cgi) and the isolate was identified as M. chelonae. The isolate was susceptible to amikacin, kanamycin, tobramycin, gentamicin, ciprofloxacin, clarithromycin, azithromycin, tigecycline, and clofazimine.
Histologic examination showed bacterial thromboembolization present throughout the gills (Fig. 1A), spleen, heart (Fig. 1B), rectal gland, and mesentery. Affected vessels sometimes had fibrinoid necrosis or edema of the wall, endothelial necrosis, or were cuffed by very small zones of granulocytes. The liver had moderate lipid depletion and mild lymphocytic inflammation in some of the portal tracts. The ovary had focal follicular rupture, granulocytic inflammation in the adjacent mesovarium ligament, and free yolk material on coelomic surfaces. The intestine, epigonal organ, colon, skin, and uterus were within normal histologic limits. The erythematous skin lesion and kidneys were not evaluated histologically. Fite acid-fast stain identified large numbers of acid-fast bacilli within thromboemboli in all tissues (Fig. 1C, 1D).
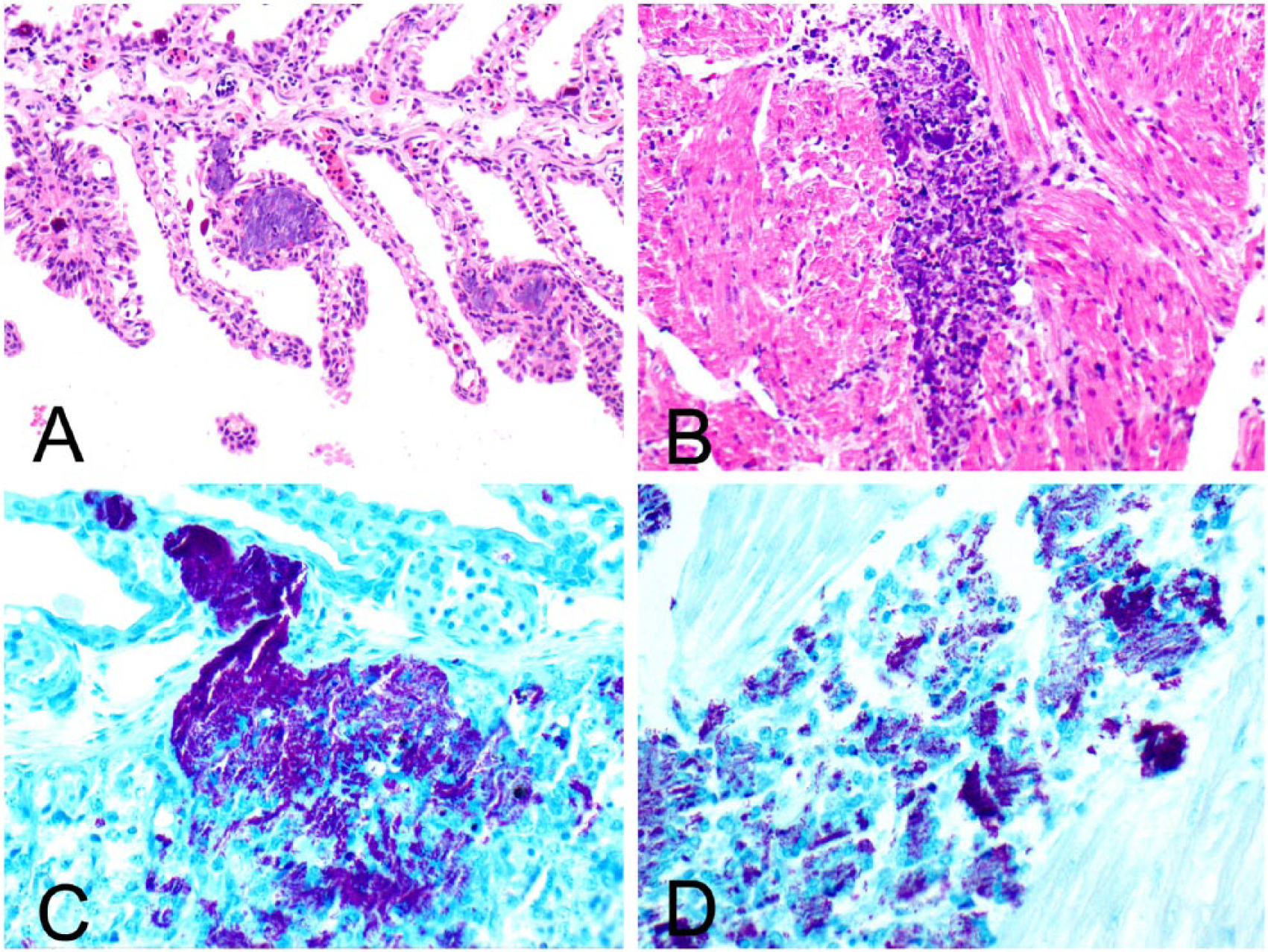
Systemic mycobacteriosis in an Atlantic guitarfish. Bacterial emboli in gill
Case 2
On October 15, 2014, an ~5-y-old male wild-caught Atlantic guitarfish was found dead on exhibit at a second institution with no history of premonitory signs or previous health issues. The guitarfish had been exhibited in a 20,060-L, artificial saltwater, closed, mixed elasmobranch species touch tank. The fish had been placed directly on exhibit and treated with 4 ppm praziquantel (Diamondback Drugs, Scottsdale, AZ) immersion baths of <4 h duration every 7 d for 3 treatments upon arrival in 2012. At the time of death, the animal was immediately refrigerated, and an autopsy was performed 30 min later. Autopsy revealed no external lesions, slightly thin body condition, light serous coelomic effusion, and a small liver. Multiple tissues were fixed in 10% neutral-buffered formalin (Labsco Advantage, Louisville, KY) for histologic examination.
Histologically, bacterial emboli were identified throughout the gills, heart, and liver. Embolization was associated with fibrin deposition, necrosis, and neutrophilic inflammation. The brain, eye, thyroid, pancreas, testis, stomach, skin, and spleen were histologically normal. Fite acid-fast stain identified large numbers of acid-fast bacilli within foci of bacterial embolization. To identify the agent, DNA was extracted from the formalin-fixed, paraffin-embedded tissues (QIAamp DNA mini-kit, Qiagen, Valencia, CA), amplified using universal Mycobacterium genus–specific primers for the 16S-23S ribosomal RNA ITS gene, directly sequenced, and analyzed as previously described.11,15 ITS gene sequence alignment revealed 100% sequence identity between the Atlantic guitarfish sample sequence and banked M. chelonae sequences (DQ866772 and AJ294537). The Atlantic guitarfish sequence was <94% similar to the ITS sequences from the next most closely related species, Mycobacterium salmoniphilum (DQ866764).
In the first case, mycobacteriosis was diagnosed histologically and by cultures of blood and coelomic fluid and the species identified by sequence analysis. Similarly, the second case was diagnosed histologically, and the organism identified to species by sequence analysis from the paraffin block. Primers targeting the rpoB region used for Mycobacterium identification of case 1 are not specific to Mycobacterium species, and although these primers are suitable for a pure culture, they are not suitable for identification from fresh or fixed tissues where mixed bacterial flora are present. Fresh tissues for culture were not available for case 2, therefore Mycobacterium genus–specific primers were used to identify the bacteria from formalin-fixed, paraffin-embedded tissues. Ideally DNA from the case 1 isolate would be amplified using the Mycobacterium genus–specific primers targeting the 16S-23S ribosomal RNA ITS gene that were used in case 2 for comparison of the 2 strains. However, DNA from the case 1 isolate was no longer available for this analysis.
In both cases, the clinical course and histologic findings were consistent with an acute, fatal, systemic infection. There were large numbers of organisms with relatively mild inflammatory responses, mirroring the course and morphologic features of mycobacteriosis described in sygnathids,3,4 whereas in teleosts the disease tends to be chronic and granulomatous.7,11 Fish can display a wide range of clinical signs, from no outward signs of illness to severe changes leading to death. Unlike these 2 cases, chronic granulomatous disease similar to what is described in teleosts8,14 was observed in an epaulette shark with M. avium 10 and an Atlantic guitarfish with M. chelonae found in the spleen. 2 Reproductive abnormalities were present in one of our cases and the previously reported guitarfish, although their exact causes are unknown. 2 Although concurrent disease such as yolk coelomitis and suboptimal nutritional status seen in case 1 could have predisposed the patient to mycobacteriosis by stressing the immune system, case 2 had no other disease processes. These findings indicate that M. chelonae in these fish was likely a primary pathogen.
The source of the mycobacterial infections in these 2 guitarfish was possibly ingestion of contaminated water or food or introduction through a traumatic injury. M. chelonae is a zoonotic species that can cause local cutaneous and/or systemic disease in humans, usually in immunocompromised individuals and/or those with preexisting wounds.5,12 Thus, it is important to have proper hand disinfection stations and protocols available for use after participation in a touch tank exhibit. 13 Although M. chelonae has zoonotic potential, it is ubiquitous in the environment and therefore depopulation or closure of the exhibit is unwarranted. Mycobacteriosis is a possible differential diagnosis to be considered in an ill elasmobranch.
Footnotes
Acknowledgements
We thank Dr. C. Innis for manuscript review.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
