Abstract
Our study was undertaken to assess the application of semiautomated methods available at the reference laboratory level for the evaluation of plasma protein and cholesterol via electrophoresis in samples from cownose rays (Rhinoptera bonasus). Three groups of animals were assessed: clinically normal, clinically abnormal, and parasitized with leeches. As reported previously, the albumin band was negligible; the protein electrophoretograms were dominated by a large beta-globulin fraction. While the group of samples from the leech-parasitized rays did not show any large differences, the abnormal group exhibited significantly elevated total solids and cholesterol levels. The latter was related to a significant increase in very low density lipoprotein levels. The results demonstrate the potential application of these laboratory methods in quantitation of plasma proteins and cholesterol fractions in subclass Elasmobranchii.
Introduction
The subclass Elasmobranchii includes more than 800 shark and batoid species, many of which are popular display species in public zoological institutions. The most recent (2008) American Elasmobranch Society census noted that cownose rays (Rhinoptera bonasus) are the most commonly displayed ray species worldwide (http://elasmo.org/census). This species and many others generally thrive under human care; however, health challenges for this and other elasmobranch species can include parasitism, infectious diseases, trauma, nutritional challenges, age-related changes, or other problems.4,15,27,36 Veterinarians and animal care staff rely on both individual and species-specific health parameters to provide adequate care for these animals.
Baseline reference intervals have been established for common hematology and chemistry analyses for some elasmobranchs including aquarium-maintained cownose rays, free-ranging southern stingrays, and dogfish and other sharks.6,12,13,29,30 While these intervals provide value for basic health assessment of these species, the applications of more advanced clinical pathology assays have not been investigated. In veterinary medicine, protein electrophoresis has been used widely to evaluate protein fractions in mammals and avian species.8,10 This information can more accurately quantitate albumin in some species and can provide a valuable snapshot to assess ongoing inflammatory processes.9,10 The applications of cholesterol electrophoresis have not been as widely studied in animal species as in humans where it has allowed for greater lipoprotein definition versus automated analyzer methods where a formula validated for human lipoprotein quantitation is often employed.5,31
The goal of our study was to investigate methods of protein and cholesterol electrophoresis in cownose rays. Initial steps were undertaken to define the respective electrophoretograms in this species, and preliminary analyses were completed to understand possible difference in electrophoretogram values based on the clinical condition of the study animals.
Materials and methods
Animals
All cownose rays included in this study were either subadult or adult rays of both sexes, housed in artificial saltwater systems with numerous other elasmobranch and teleost species. Salinity ranged between 32 and 34 parts per thousand, and water temperature ranged between 24 and 25°C. All rays were fed twice daily ad libitum.
Samples from 35 clinically normal cownose rays were analyzed in this study representing 33 females and 2 males. These animals were considered healthy based on results of clinical examination and routine bloodwork inclusive of a complete blood cell count and plasma biochemistry panel. The median total white blood cell (WBC) count was 9.9 × 103/µL (interquartile range [IQR]: 5.5–19.0), median fine eosinophilic granulocytes (FEG) was 2.1 × 103/µL (IQR: 1.0–6.9), and the median packed cell volume (PCV) was 25.5% (IQR: 24.0–27.5). A second group of 27 cownose rays were assigned to the clinically abnormal group. This was inclusive of 20 females and 7 males. Samples from these rays were obtained during physical examination after the appearance of wounds related to conspecific trauma. Versus the clinically normal cownose ray group, this group had a mild but not significant increase in total WBC count with a median 11.8 × 103/µL (IQR: 8.2–22.4, P = 0.20), mild significant increase in FEG with a median 4.7 × 103/µL (IQR: 2.8–8.1, P = 0.05), and a significant decrease in PCV with a median 18.8% (IQR: 16.3–22.0, P < 0.0001). There were no significant differences in plasma biochemistry analytes or other hematologic parameters. A third group of 21 cownose rays were assigned to the leech group. This was inclusive of 10 males and 11 females. These rays had heavy leech burdens primarily in the buccal cavity and, in some cases, leeches were found in the gill slits and spiracles. Heavily infested rays carried >50 leeches ranging from a few millimeters to 7 cm in size. Externally, some of these rays exhibited dermal blotchiness or pallor. Leeches could also possibly cause wounds or erosions at the site of attachment, which can led to secondary infections. In some cases, leeches occluded the esophagus and hindered mastication making feeding difficult. As against the the clinically normal cownose ray group, this group had a significant increase in total WBC count with a median 17.4 × 103/µL (IQR: 11.3–26.8, P = 0.02), significant increase in FEG with a median 9.3 × 103/µL (IQR: 5.9–15.5, P = 0.0007), and significant decrease in PCV with a median 14.8% (IQR: 10.5–18.3, P < 0.0001). There were no significant differences in plasma biochemistry analytes or other hematologic parameters. Several of these rays were concurrently infected with the external monogenean parasite Benedeniella posterocolpa, which were visible on the skin of the affected animals; the size averaged 1 cm in length. Heavy burdens were more often noted in more compromised animals and may have contributed to poor health as a result of irritation of the skin and changes in behavior and swim patterns, as rays typically spend more time rubbing on the substrate. It should also be noted that some rays (both healthy and unhealthy) had concurrent mild infection with the internal parasite Eimeria southwellii, as confirmed by coelomic fluid aspiration and cytologic evaluation of samples. This parasite was tolerated by healthy animals but could negatively impact animals that were compromised.
Samples
Blood was collected from individuals from a wing vessel, using a 23-gauge butterfly needle attached to a 3-mL syringe, while the ray was manually restrained in water. Samples were immediately transferred into lithium heparin. All samples were rocked gently until processed for 10–20 min postcollection when they were centrifuged at 3,000 × g for 7 min to generate plasma. Plasma was frozen and stored at −80°C until testing. All blood samples were examined by protein electrophoresis and, as sample size permitted, by cholesterol electrophoresis methods.
Protein electrophoresis
Samples were analyzed an electrophoresis analysis system a according to the manufacturer’s instructions using gels purchased to accompany the system, as previously described for use with avian samples. 7 Results were produced after gel scanning and analysis by manufacturer software. a Fraction delimits were placed using conventions used in mammals: albumin, alpha 1, alpha 2, beta-, and gamma-globulins, but identified as fractions 1–5, respectively. Percentages for each fraction were determined by this software, and absolute values (g/dL) for each fraction were obtained by multiplying the percentage by the total solids concentration. The coefficient of variation (CV) for fraction quantification was observed as follows: fraction 1, 7.5%; fraction 2, 7.8%; fraction 3, 8.2%; fraction 4, 2.1%; and fraction 5, 3.5%.
Cholesterol electrophoresis
Samples were analyzed according to the procedure provided by the electrophoresis analysis system a and the cholesterol profile kit. a Samples were run on an agarose gel, which was stained using 4-nitro blue tetrazolium. Results were produced after gel scanning and analysis by manufacturer software. Fraction delimits were placed using conventions used in mammals: high-density lipoprotein (HDL), lipoprotein(a) [Lp(a)], very low-density lipoprotein (VLDL), and low-density lipoprotein (LDL). Percentages for each fraction were determined by this software, and absolute values (mg/dL) for each fraction were obtained by multiplying the percentage by the cholesterol concentration. The CV for fraction quantification was observed as follows: HDL, 20.7%; Lp(a), 17.0%; VLDL, 1.5%; and LDL, 8.7%.
Other analyses
Cholesterol and other routine biochemistry analyses were determined using a chemistry analyzer. b Total solids was determined using a non–temperature compensated refractometer. c Albumin was determined using a chemistry analyzer d using the bromocresol green (BCG) methodology. Total protein was also determined by this analyzer using the biuret method. A complete blood cell count was conducted by Natt and Herrick’s methodology with a manual review of the blood smear.
Statistical analysis
Data was analyzed for normality using the Shapiro–Wilk test. As most of the data was not normally distributed, nonparametric analysis was conducted using the Mann–Whitney test. Data is presented as median and IQR. For comparisons of different methods of albumin determinations and total protein versus solids results, paired results were analyzed using the Wilcoxon signed rank test. Significance was set at P < 0.05. These analyses were conducted using statistical software. e Reference intervals were generated using the robust method. Outliers were detected by the Tukey method, and distribution was examined by the D’Agostino–Pearson method. Analyses were conducted using an alternate software. f
Results
Analysis of total protein and albumin levels
An excellent correlation between values for total protein and total solids obtained by the chemistry analyzer and refractometer, respectively, was found, r = 0.90 (P < 0.0001). The median value for the chemistry analyzer was 3.3 g/dL (95% confidence interval [CI]: 2.7–4.2) and for the refractometer was 6.0 g/dL (95% CI: 5.7–7.0). This represented a significant difference (P < 0.0001).
A poor correlation between values for albumin obtained by the chemistry analyzer and protein electrophoresis (fraction 1) was found (r = 0.17, P = 0.3890). The median value for the chemistry analyzer was 1.1 g/dL (95% CI: 0.9–1.4) and for electrophoresis was 0.54 g/dL (95% CI: 0.41–0.73). This represented a significant difference (P < 0.0001).
Protein electrophoresis
Based on protein electrophoresis conventions for fraction delimits in mammals, 5 fractions were defined (Fig. 1). The value for prealbumin was defined as zero and not included in the analysis. A fraction possibly corresponding to albumin was recognized based on comparison of fraction migration characteristics of mammals, birds, and reptiles; this was identified as fraction 1. Fraction delimits for alpha migrating globulins were ill-defined and assigned as fractions 2 and 3. Beta and gamma migrating fractions were clearly defined and assigned as fractions 4 and 5. Reference intervals are presented in Table 1.
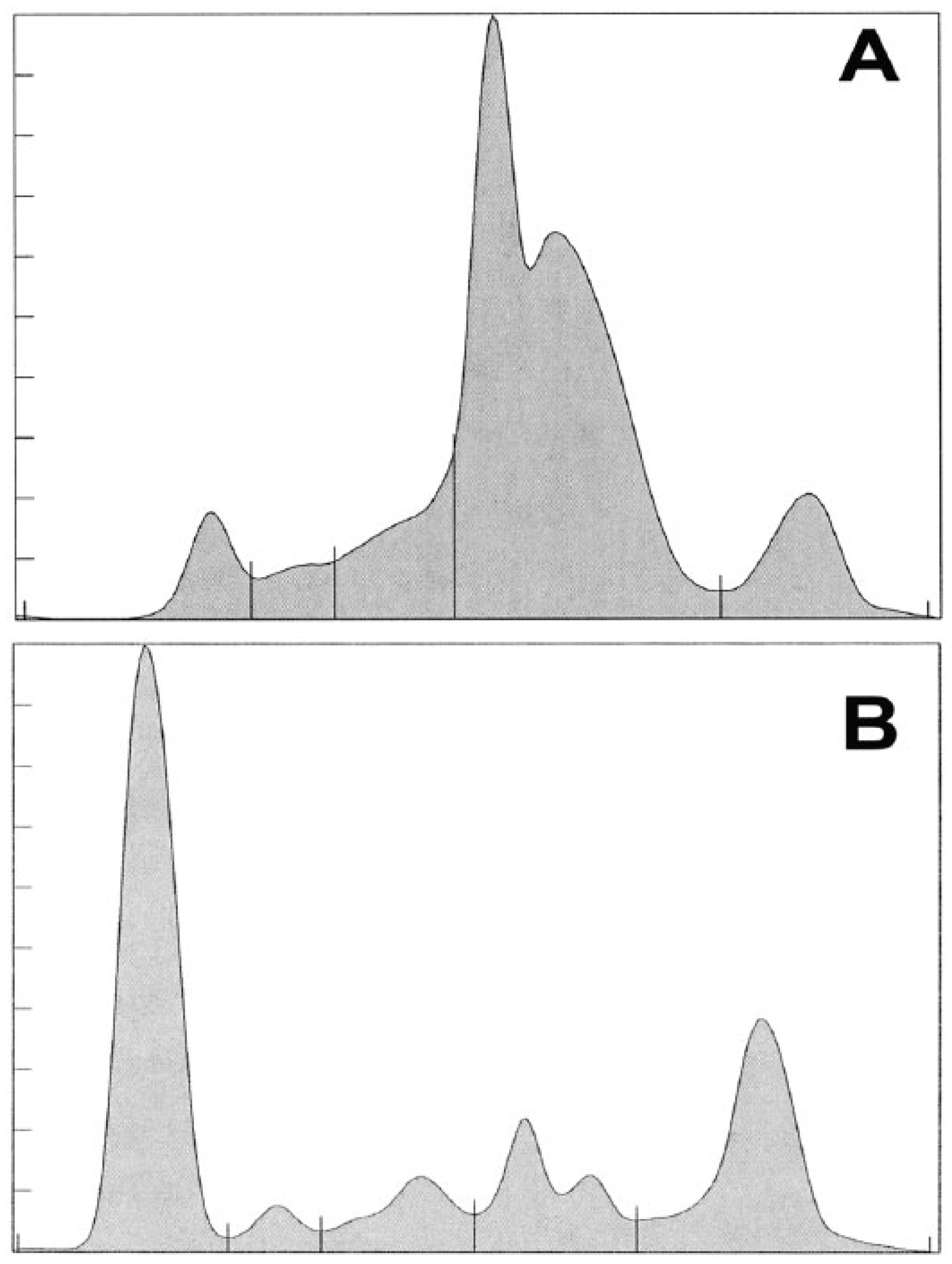
Representative protein electrophoretogram of a cownose ray (Rhinoptera bonasus;
Reference intervals for protein electrophoresis values of cownose rays (Rhinoptera bonasus).*
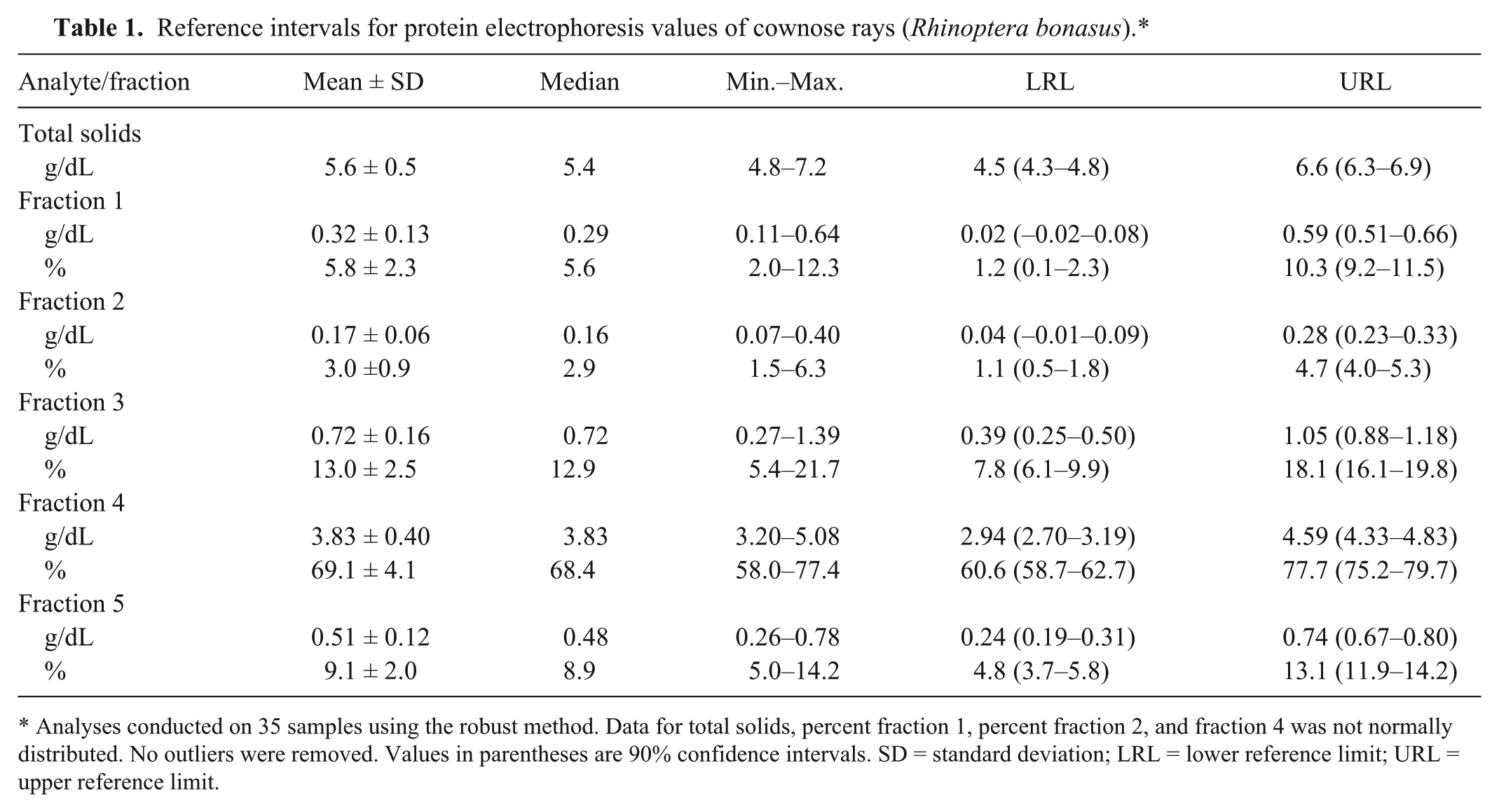
Analyses conducted on 35 samples using the robust method. Data for total solids, percent fraction 1, percent fraction 2, and fraction 4 was not normally distributed. No outliers were removed. Values in parentheses are 90% confidence intervals. SD = standard deviation; LRL = lower reference limit; URL = upper reference limit.
Cholesterol electrophoresis
Based on cholesterol electrophoresis conventions for fraction delimits, 4 fractions were defined (Fig. 2). The fraction for HDL was poorly defined in the ray samples. However, based on fraction migration of the control sample, fractions for Lp(a), VLDL, and LDL were defined. Reference intervals are presented in Table 2.
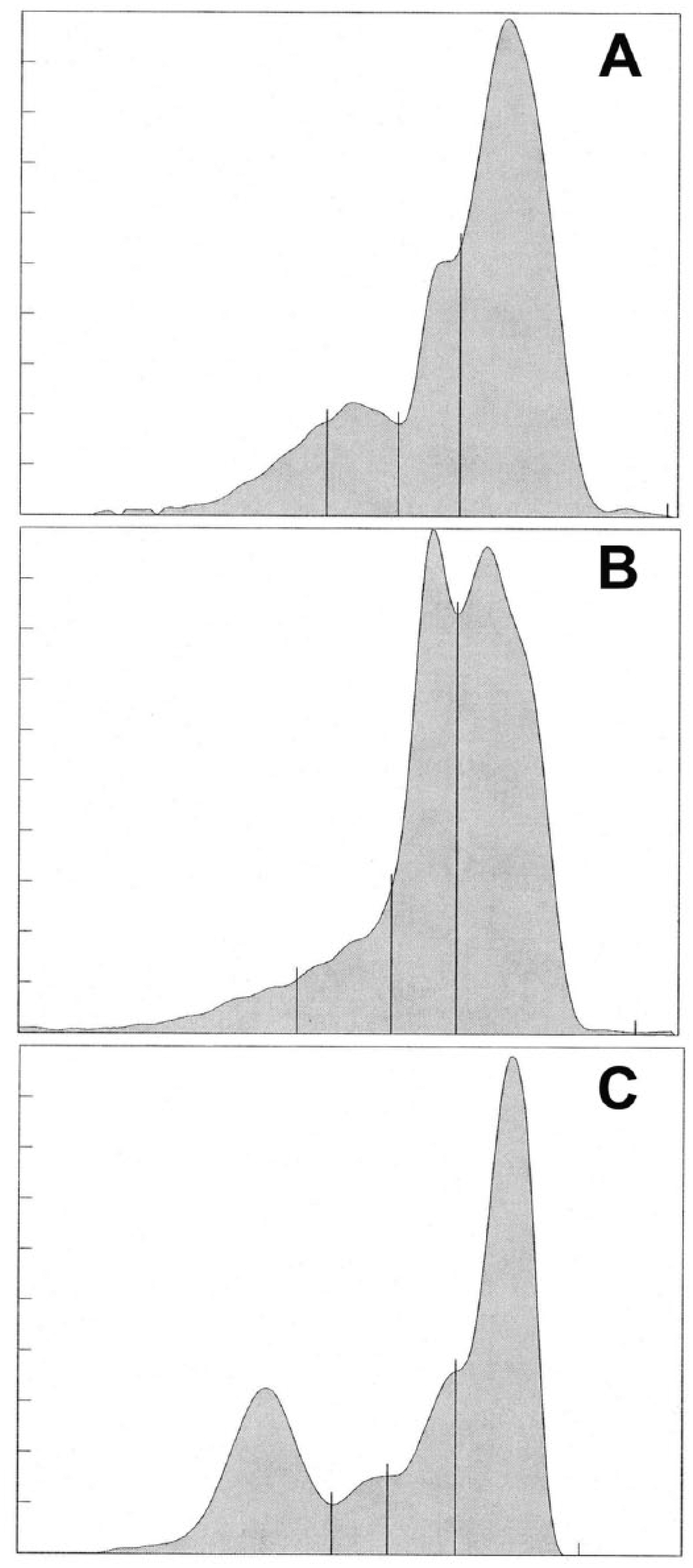
Representative cholesterol electrophoretogram of a cownose ray (Rhinoptera bonasus;
Reference intervals for cholesterol electrophoresis values of cownose rays (Rhinoptera bonasus).*
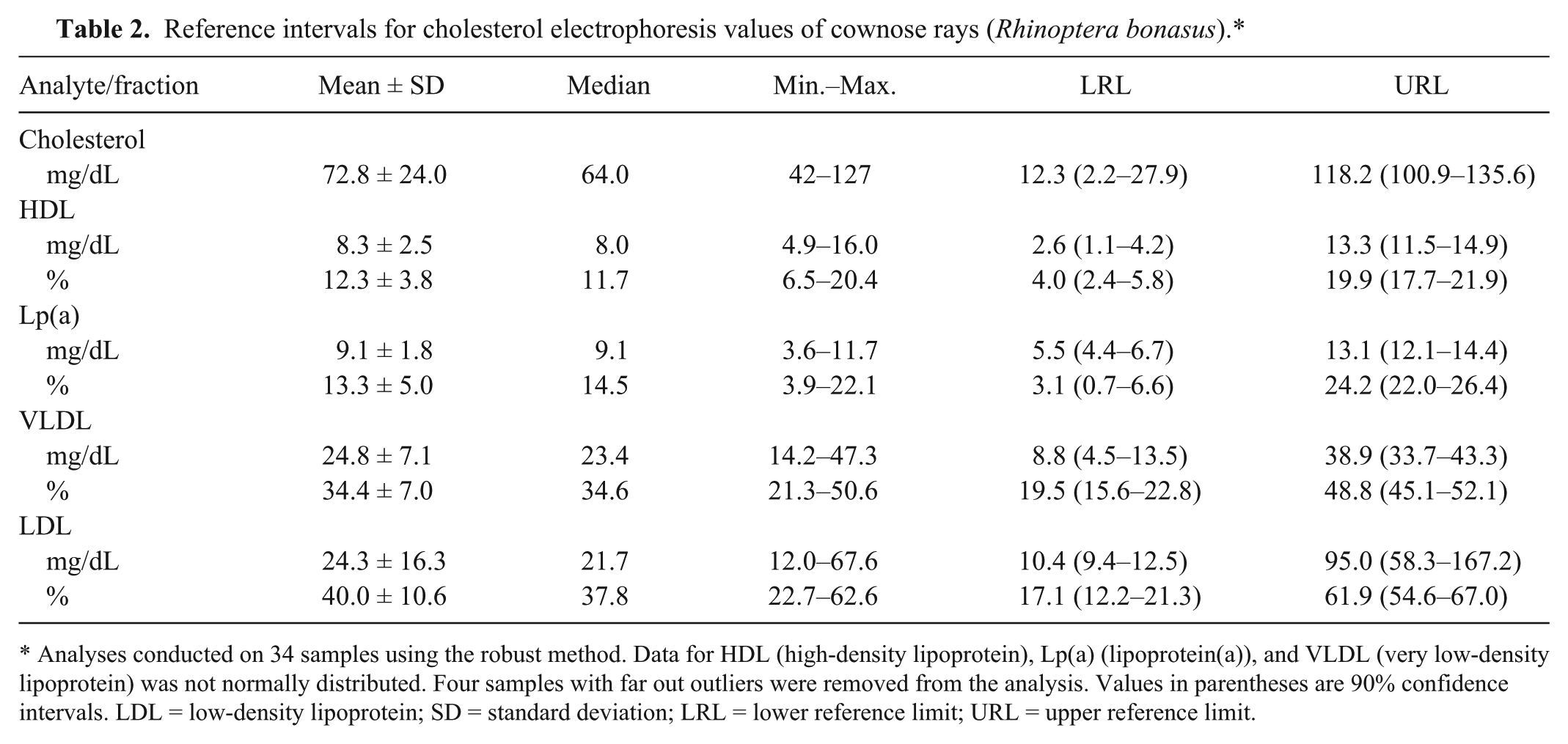
Analyses conducted on 34 samples using the robust method. Data for HDL (high-density lipoprotein), Lp(a) (lipoprotein(a)), and VLDL (very low-density lipoprotein) was not normally distributed. Four samples with far out outliers were removed from the analysis. Values in parentheses are 90% confidence intervals. LDL = low-density lipoprotein; SD = standard deviation; LRL = lower reference limit; URL = upper reference limit.
Comparison of clinically normal rays to clinically abnormal rays
In the analysis of the protein electrophoresis data of normal and abnormal groups, mild differences in fractions 1 and 3 were observed (Table 3). There was no difference in percent fraction 4 or 5. These changes were also observed in the leech-parasitized group. In the analysis between the clinically normal and abnormal groups, the median cholesterol level was more than 1.6 times higher in the abnormal group (Table 4). The level of VLDL was significantly increased.
Protein electrophoresis values from 3 groups of cownose rays (Rhinoptera bonasus).†
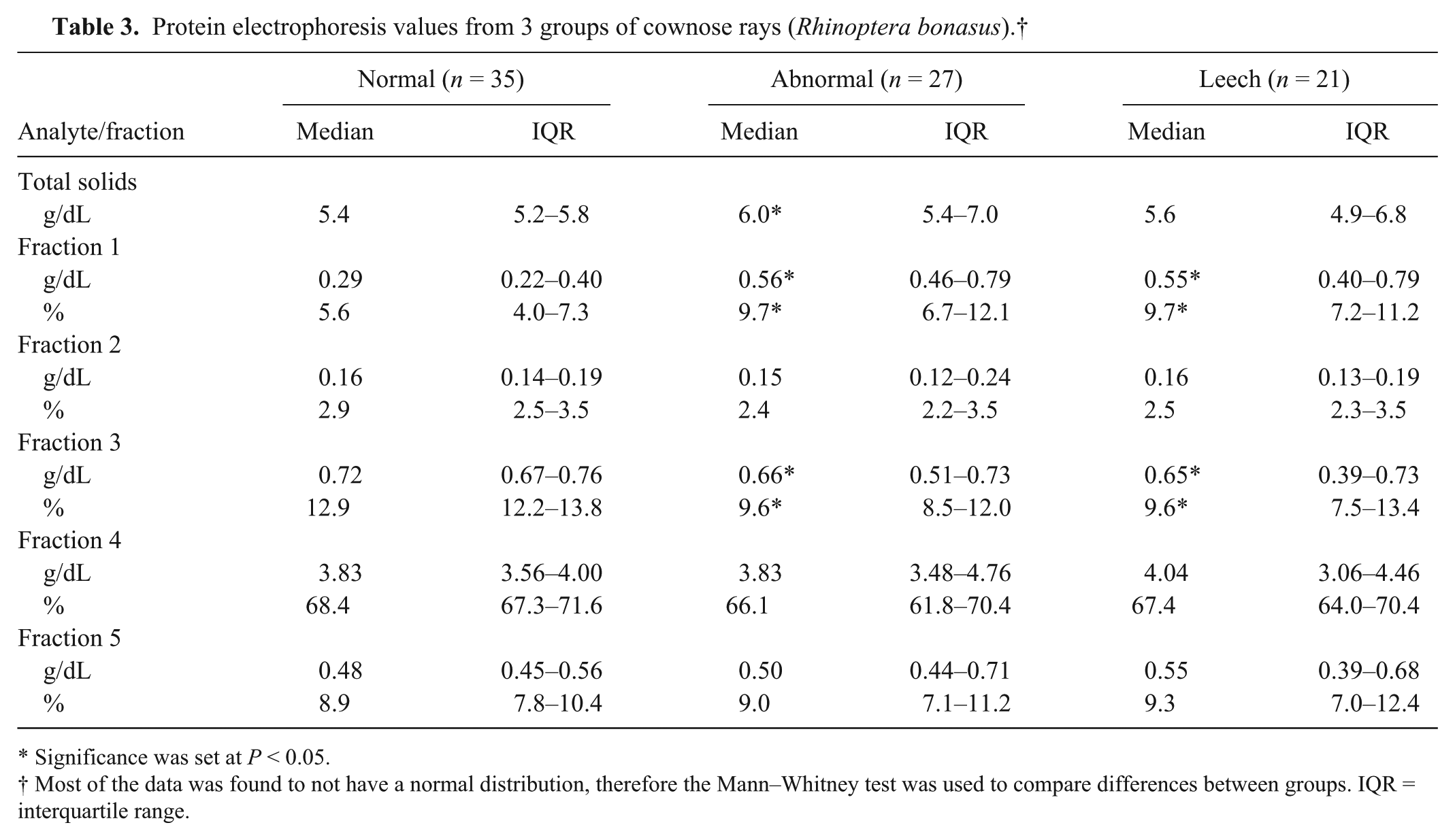
Significance was set at P < 0.05.
Most of the data was found to not have a normal distribution, therefore the Mann–Whitney test was used to compare differences between groups. IQR = interquartile range.
Cholesterol electrophoresis values from 3 groups of cownose rays (Rhinoptera bonasus).†
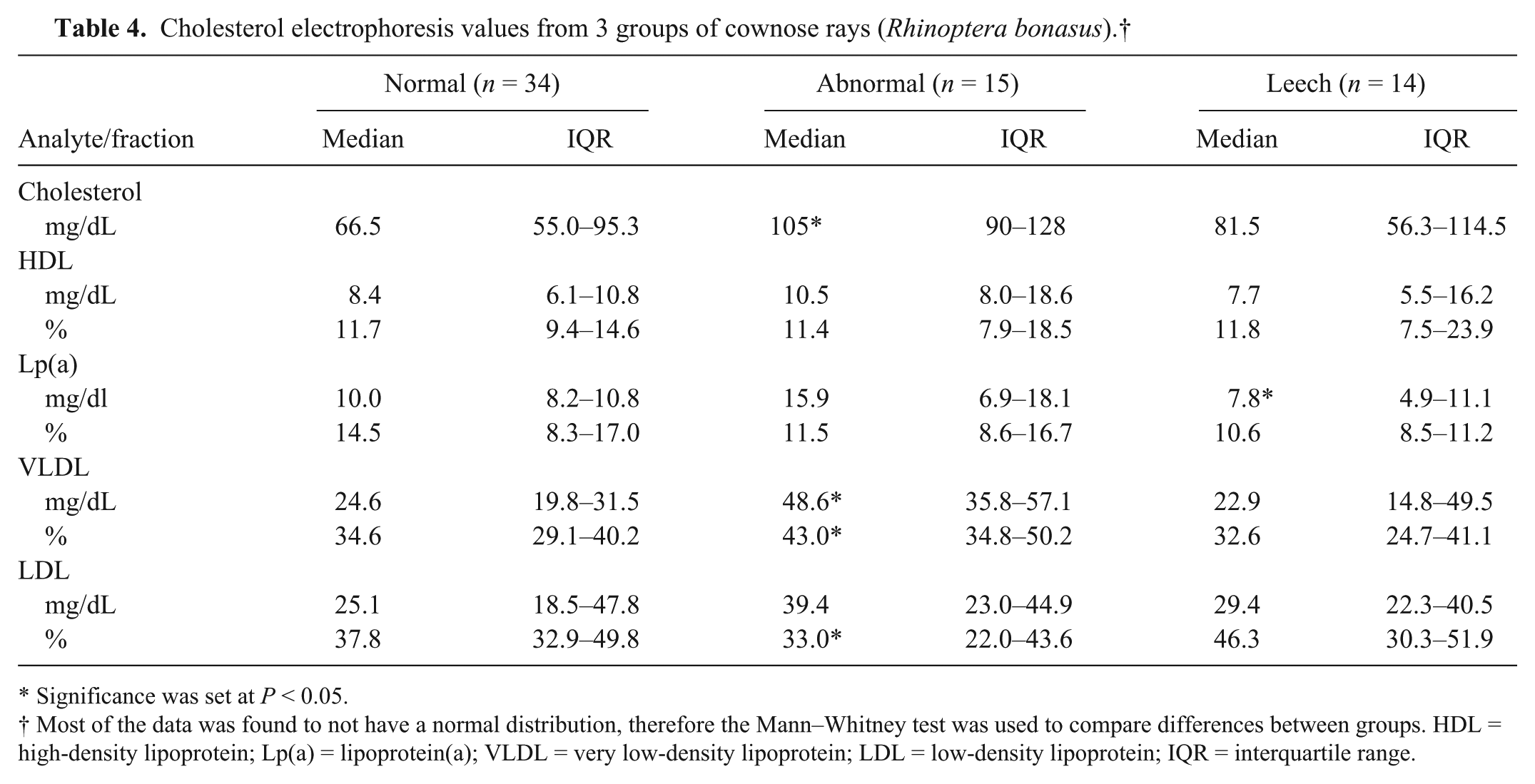
Significance was set at P < 0.05.
Most of the data was found to not have a normal distribution, therefore the Mann–Whitney test was used to compare differences between groups. HDL = high-density lipoprotein; Lp(a) = lipoprotein(a); VLDL = very low-density lipoprotein; LDL = low-density lipoprotein; IQR = interquartile range.
Discussion
Plasma protein electrophoresis revealed negligible levels of albumin in cownose rays. This observation is consistent with previous reports and reviews on elasmobranchs.3,25 While early precise studies demonstrated that most elasmobranchs did not have detectable levels of nonesterified fatty acids (and NEFA binding proteins), a study using more modern technology found albumin to be absent in a variety of representative marine class Chondrichthyes species including sharks and skates.11,25 The latter study was based on assessments of electrophoretic mobility, palmitate binding, and molecular mass studies. Earlier studies did report the presence of a protein that had BCG-binding properties, indicating the presence of <1% albumin in some elasmobranch species. 11 BCG is the dye commonly used in automated chemistry analyzers for the detection of albumin but has been demonstrated to produce erroneous results in birds and reptiles.9,28 The concept of a presence of albumin in some elasmobranchs has been potentiated by the use of this methodology while establishing reference intervals.12,14,30 This type of description should be disregarded. As demonstrated by correlation analysis in our study, albumin levels generated by BCG and electrophoretic methods of albumin quantitation are not equivalent.
In our study, a minor band was identified that migrates near to the mammalian albumin band. This was previously described to be consistent with HDL and was identified as fraction 1 in our study. 25 In review of the protein electrophoretograms of the cownose ray and other elasmobranchs (C Cray, personal observation), fractions 2 and 3 (generally referred to as alpha-globulins) are ill defined. Considerable protein was observed in the beta fraction range (fraction 4). This routinely was observed to be defined as 2 peaks and correlates with the pre-beta and beta migrating VLDL and LDL reported in other marine elasmobranchs. 25 The last highly reproducible fraction (fraction 5) migrated in the mammalian gamma fraction range. This band may represent the dominant immunoglobulin M response reported in other elasmobranchs.16,23 Overall, the fraction proportions observed in the cownose electrophoretogram correlate with the values of those reported as albumin, and alpha-, beta-, and gamma-globulins for Atlantic sharpnose, bonnethead, and spiny dogfish sharks. 13
Future studies of plasma protein electrophoresis may benefit from quantifying the 2 fractions within fraction 4 separately, and the composition of fraction 5 should be further defined. Moving forward, reference intervals for elasmobranchs should be established that are method oriented. In our study, a representative electrophoretogram is included to provide some level of consistency among diagnostic laboratories. It should be noted that absolute values for electrophoresis fractions have been calculated using total solids rather than total protein.
While an excellent correlation was found between these 2 methods, refractometry-derived values did reflect in a median 1.8-fold increase. This is similar to that reported previously for cownose rays. 12 Marine elasmobranch plasma osmolarity is slightly hyperosmotic to the environment because of the high concentration of plasma urea and trimethylamine N-oxide. The difference between plasma protein measured in a refractometer as total solids and by the biuret method as total protein has been attributed to the contribution of these solutes. 14
The data obtained from semiautomated methods for cholesterol electrophoresis used in our study well approximated that reported previously in elasmobranchs.2,3,25 HDL has been described to be in very low levels in class Chrondrichthyes.2,3,25 One study estimated it as <10% of the total lipoprotein, which is consistent with that observed in the present study. While perhaps more defined in the protein electrophoretogram, it could only be quantified as a region in the cholesterol electrophoretogram. In contrast, the fractions representing the VLDL and LDL were well defined. The dominance of these fractions is consistent with that reported in several chondrichthyan species, and higher VLDL levels was first reported in an early analysis of the spiny dogfish.20,25 By ultracentrifugation methods, the lipoproteins of this elasmobranch were described as 61% VLDL, 34% LDL, and 5–6% HDL. 26 The current method also resolved a fraction with the migration characteristics of Lp(a), which is composed of an LDL-like lipoprotein that is bound with apolipoprotein(a) and apolipoprotein(b). 24 This protein has not yet been described in elasmobranchs using modern techniques but apolipoproteins have been described in a variety of species of fish. 2 Future clinical studies should continue to document this fraction while basic science approaches should better define its composition.
The cholesterol levels reported in our study are similar to those reported previously for different species of free-ranging sharks and aquarium-maintained cownose rays.12,13 Differences in cholesterol levels in marine fish, including elasmobranchs, have been ascribed to age, nutrition, salinity, temperature, stress, and activity levels.2,18 While most factors were suggested to decrease cholesterol, salinity changes and stress were reported to increase cholesterol levels. In a study of dogfish, plasma cholesterol levels increased more than 2.5-fold with a 30% increase in salinity and decreased more than 2-fold with a 50% decrease in salinity; lipoprotein levels were not examined. 1 In shore crabs, beta lipoproteins were observed to increase with higher salinity and decrease with lower salinity. 33 Stress from handling was found to result in a maximal 33% increase in cholesterol over a 24-hr period in salmon. 37 Injection of high doses of adrenaline resulted in a 1.5-fold increase in cholesterol in the eel; increases continued to be present through the experimental endpoint of 24 hr. 19 In rat and human studies, higher glucocorticoid levels were associated with increased VLDL levels.17,35
In the group of clinically abnormal rays, the majority of the samples were taken from animals with wounds or trauma. Cholesterol was significantly increased in this group because of an increase in VLDL. Specific observations were not made regarding activity, anorexia, or weight changes in this group as they were housed in an extremely large multispecies exhibit. Acute phase responses have been observed in another species of ray with the injection of bacterial antigen; it could be proposed that the change in cholesterol was a stress-related response relative to inflammation involving the trauma. 22
The effects of leech infestation were examined in an experimental model using yellow stingrays. 21 During acute infestation, with mortality occurring within 3 days, animals exhibited anorexia, decreased PCV, and decreased total solids. While total WBC counts did not change, decreased hepatocellular lipid deposits were noted in addition to cutaneous ulceration and edema. Serum cholesterol was not measured. In the present study, only mild changes were observed in the electrophoretic values although a significant increase in total WBC count was observed. This may reflect a species difference or the level of leech burden and chronic processes related to this type of parasitism. As the liver is the primary site of energy storage in elasmobranchs, a severe, acute parasite infestation could quickly deplete hepatic reserves, while a more gradual but chronic burden could allow normalization of lipid metabolism. 34
Our study demonstrates the application of a modern semiautomated electrophoresis system for use with plasma samples from cownose rays. This system provided complementary results to basic science studies, which previously demonstrated a lack of albumin and a dominance of VLDL and LDL in elasmobranchs. 25 This preliminary investigation supports that electrophoretic methodology can provide quantitation of these analytes and that differences can be observed under varying conditions. In previous reports, fatty acid profiles have been proposed as a tool that could be used in the management of wild stingrays encountered in tourism experiences. 32 As additional studies are undertaken to study clinically abnormal animals, the value of the application of either plasma protein and/or cholesterol electrophoresis in the study or management of elasmobranchs will be evaluated.
Footnotes
Acknowledgements
We thank the Georgia Aquarium zoological operations and animal health staff and volunteers for their assistance in this project.
Authors’ contributions
C Field, A McDermott, L Leppert, and T Clauss contributed to conception of the study, and acquisition of data. C Cray and GD Bossart contributed to conception and design of the study, and analysis and interpretation of data. M Rodriguez contributed to the design of the study, and analysis of data. C Cray, C Field, A McDermott, T Clauss, and GD Bossart critically revised the manuscript. All authors drafted the manuscript, gave final approval, and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
SPIFE 3000 system, Split Beta gels, Vis cholesterol profile kit; Helena Laboratories Inc., Beaumont, TX.
b.
AU640e chemistry analyzer, Olympus Imaging America Inc., Center Valley, PA.
c.
Schuco, Japan.
d.
Vitros 250 chemistry analyzer, Ortho Vitros, Rochester, NY.
e.
Prism version 6.04, GraphPad Software Inc., LaJolla, CA.
f.
MedCalc version 14.8.1, MedCalc Software, Ostend, Belgium.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
