Abstract
An undifferentiated renal tubular carcinoma was diagnosed in a juvenile male olive baboon (Papio anubis). The animal suddenly appeared depressed and refused to eat. During physical examination, a firm, palpable mass in the left abdominal area and flank pain were detected. Clinical pathology findings included mild anemia, hypoalbuminemia, hyponatremia, and mildly increased serum creatinine and urea concentrations. Radiographs revealed a large mass in the left abdominal area. Exploratory laparotomy disclosed a 10 cm × 15 cm multilobulated mass involving the left kidney and adjacent organs. Because of a poor prognosis, the animal was humanely euthanized, and necropsy was performed. Tissue samples of the neoplasm were taken for histopathological examination. Immunohistochemical staining was done using vimentin, cytokeratin, S-100 protein, Ki-67, α-actin, and desmin-specific primary antibodies. Microscopically, elongated and irregular tubules were lined by 2 or more layers of atypical epithelial cells. Anisocytosis, anisokaryosis, and frequent mitotic figures were also observed. Following immunohistochemical staining, the cytoplasm of neoplastic cells was positive for cytokeratin, vimentin, and S-100 protein and negative for α-actin and desmin. Positive nuclear staining for Ki-67 was observed. The neoplasm was diagnosed as an undifferentiated renal tubular carcinoma.
The baboon has become an increasingly important animal model for biomedical research, especially in areas such as xenotransplantation. In humans, renal cell carcinoma is the third most common urologic malignancy and the seventh most common neoplasm overall. 17 Spontaneous benign and malignant neoplasms have been reported in nonhuman primates, but the overall frequency of neoplasms in captive primates has been low. 1,2,9,14,15,18 The urogenital tract is the third most affected system, but only 9 cases of spontaneous renal neoplasia have been reported in the baboon. 3 A positive correlation between advancing age and incidence of cancer has been well documented in several nonhuman primate species including baboons. 3,19
The current study describes an undifferentiated renal tubular carcinoma that originated spontaneously in a juvenile olive baboon (Papio anubis). This species reaches puberty at 6 years of age. 13 The clinical, hematological, and pathological findings in this juvenile baboon are described.
An abdominal mass was detected in a 4-year-old male olive baboon (weight 9 kg). The animal was part of a colony of 40 breeding baboons maintained in the Primatology Unit of the University of Murcia (Murcia, Spain). The colony provided animals for a xenotransplantation research program, and the baboons were not subjected to any experimental procedure up to the moment of tissue transplantation. The colony was founded with animals from Kenya and South Africa, but the animal in the current report was born at the University of Murcia facilities. The colony was housed in 2 parks containing 20 animals in each. Husbandry and maintenance were provided in accordance with the recommendations listed in the Guide for the Care and Use of Laboratory Animals. 20 The baboons were fed with simian commercial feed and supplemented with fresh fruit, dry seeds, and vegetables, which varied depending on the growing season. Routine serological monitoring was negative for Hepatitis A, B, and C virus; Simian alphaherpesvirus 8; Primate T-lymphotropic virus 1 isolate Simian T-lymphotropic virus 1; and Simian immunodeficiency virus. Testing for the latter virus was done by enzyme-linked immunosorbent assay. The health protocol included semiannual treatment of external and internal parasites and tuberculin testing as well as a yearly complete blood cell count and biochemical profile for each animal.
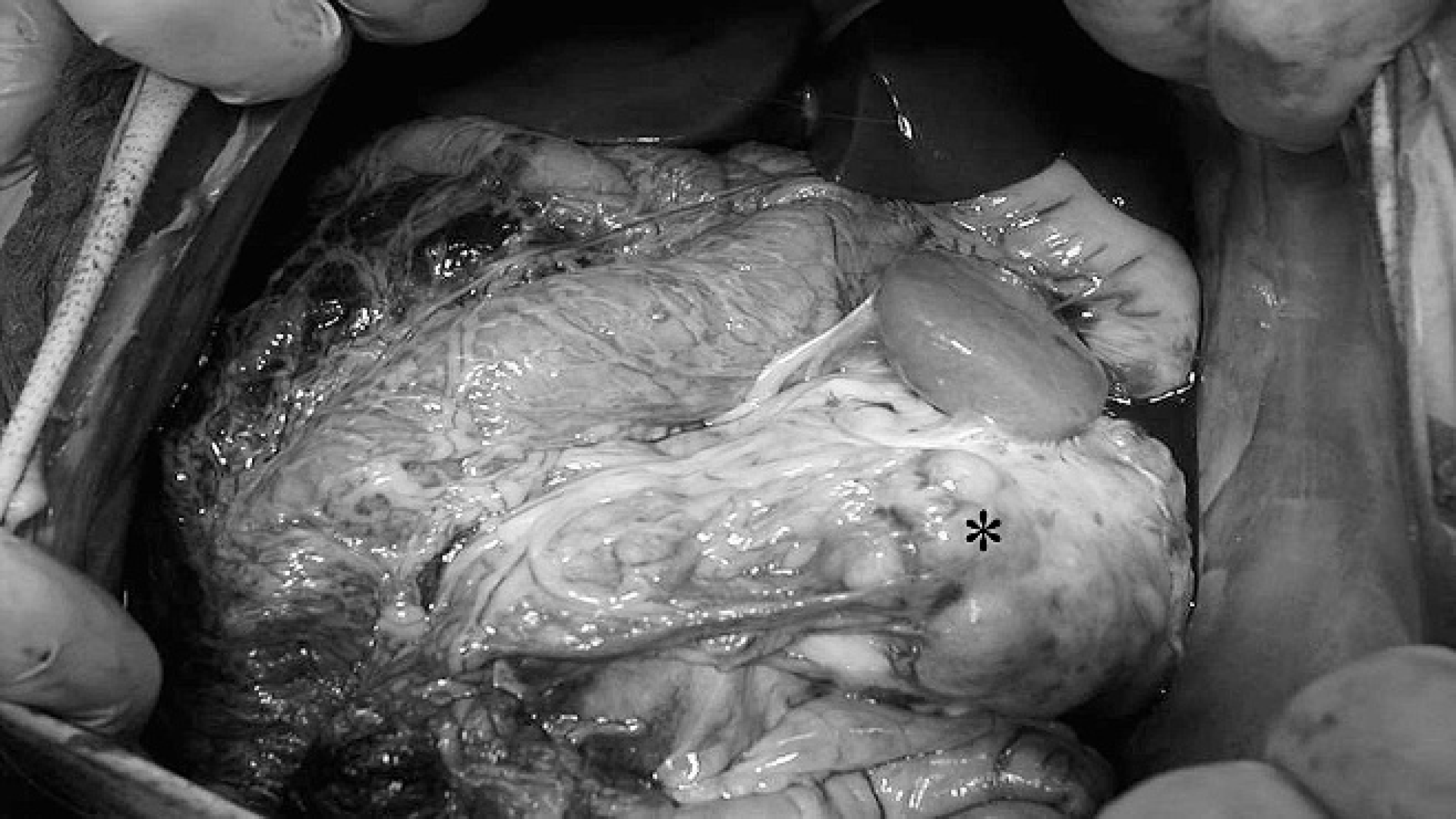
Multilobulated, white to yellow mass (*) effacing approximately 80% of the left kidney and involving the adjacent intestine and spleen.
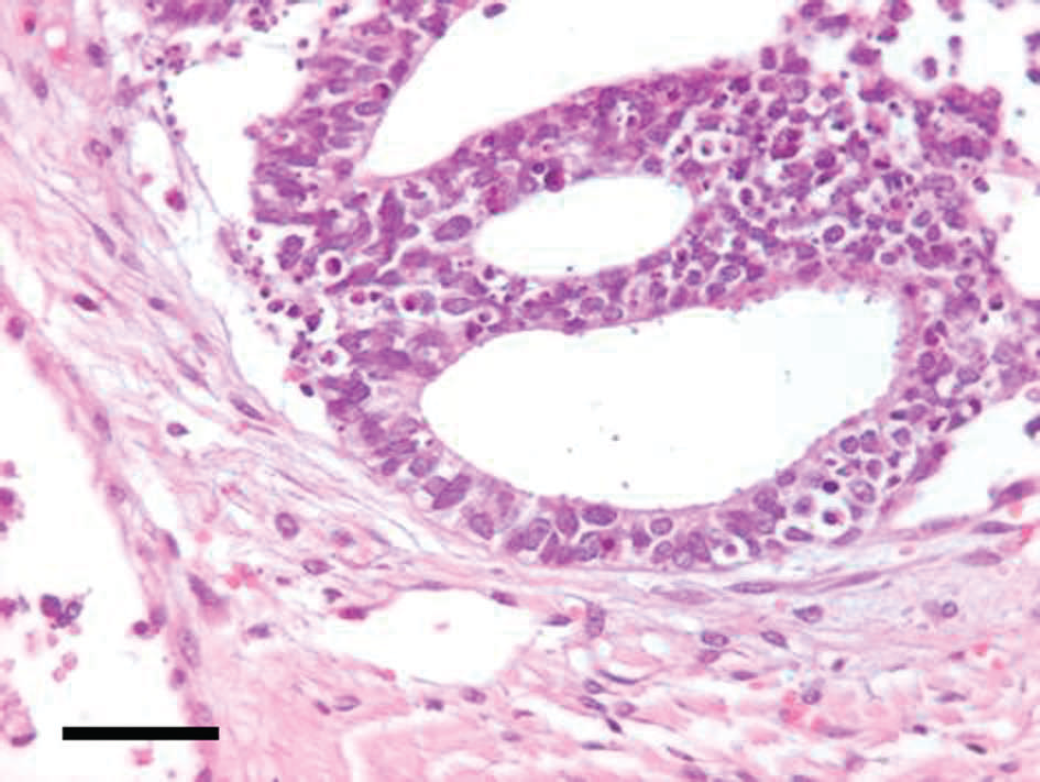
Irregular tubules lined by neoplastic cells that exhibit anisocytosis and anisokaryosis and contain prominent euchromatic nuclei, eccentric nucleoli, and finely granular eosinophilic cytoplasm. Occasional clear cells also are present. Numerous apoptotic cells are scattered within the neoplastic cell population. Hematoxylin and eosin. Bar = 45 μm.
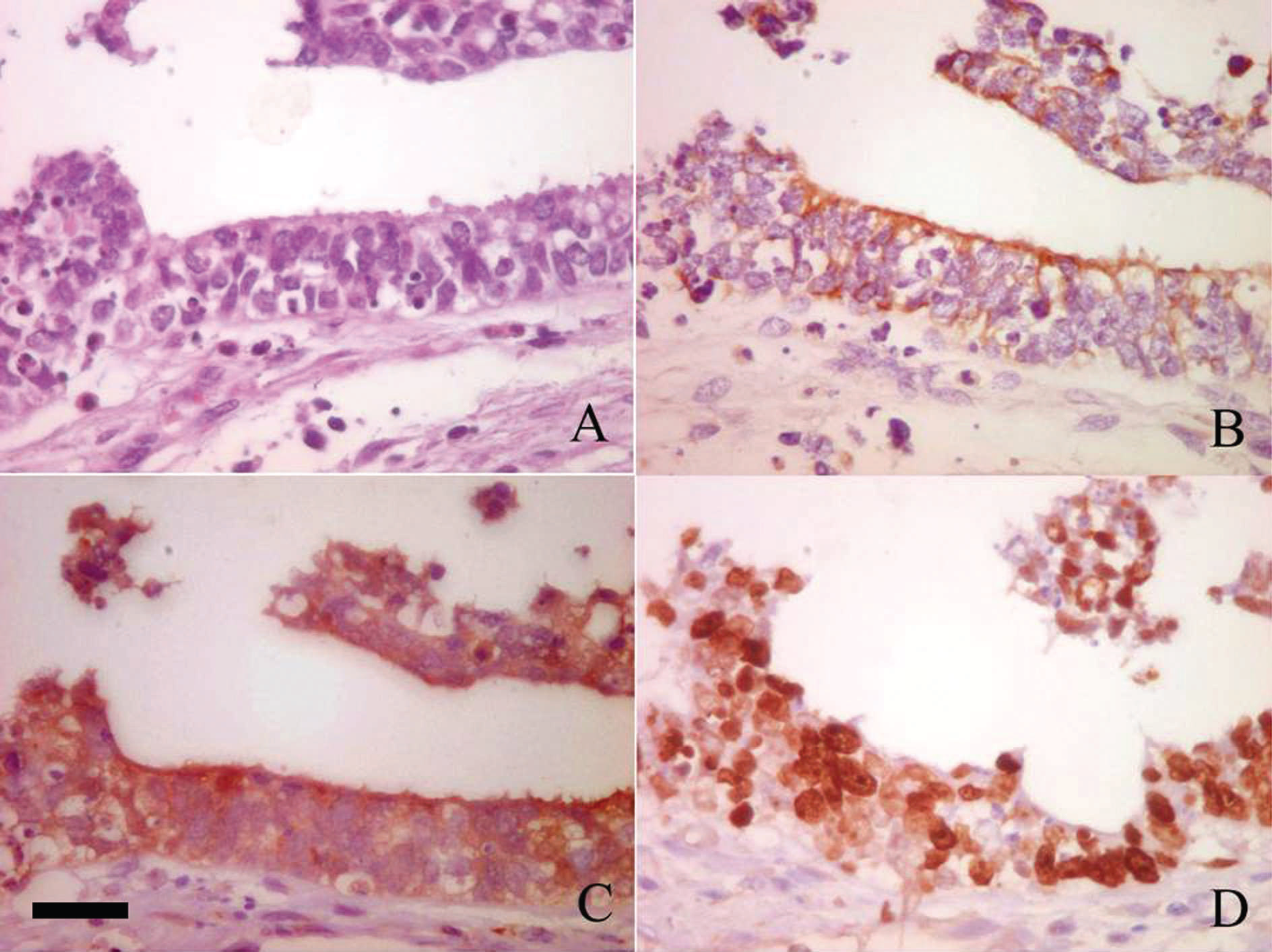
Detail of neoplastic cells. A, hematoxylin and eosin stain. B, positive cytoplasmic staining for cytokeratin within the neoplastic cells. C, positive cytoplasmic staining for S-100 protein within the neoplastic cells. D, positive nuclear staining for Ki-67 protein within the neoplastic cells. Bar = 30 μm (applies to all figure panels).
Characteristics of primary antibodies used for immunohistochemical staining in the current study.
The juvenile baboon of the current report was home reared because of early maternal rejection and was added to the colony when he was 1 year old. The animal's medical history was devoid of any clinical, biochemical, or hematological abnormalities. The clinician first noted that the baboon was depressed and refused to eat. A physical examination revealed a firm palpable mass in the left abdominal area and flank pain. Biochemical and hematological evaluations were relatively nonspecific and included mild anemia (3.4 × 1,012 RBC/l, reference [ref.] interval: 4.8–6.3 × 1,012 RBC/l), 11 mild hypoalbuminemia (2.7 g/dl, ref. interval: 2.9–4.2 g/dl), 11 and mild hyponatremia (135 mg/dl, ref. interval: 143–158 mg/dl). 11 Serum urea nitrogen (57 mg/dl, ref. interval: 9–25 mg/dl) and creatinine (1.6 mg/dl, ref, interval: 0.8–1.4 mg/dl) concentrations were mildly increased. 11 The low reticulocyte percentage (0.5%) indicated nonregenerative anemia. A urine specimen was not available for laboratory analysis. The laboratory findings were suggestive of renal disease, but this is generally rare in most juvenile animals. Prerenal azotemia was also a considered because the baboon had inappetence and was not drinking water. Survey abdominal radiographs were suggestive of a large mass in the left abdominal cavity. Exploratory laparotomy, performed through a midline incision under isoflurane anesthesia, disclosed a large renal mass involving the inferior vena cava and the adjacent intestine and spleen. Firm adhesions were present between capsular surface of the kidney and adjacent organs (Fig. 1). Because of a poor clinical prognosis, the animal was euthanized, and a complete necropsy was performed.
At necropsy, a 12 cm × 15 cm, white to yellow, soft, multilobular mass was observed in the abdominal cavity. The mass appeared heterogeneous on the cut surface and effaced approximately 80% of the parenchyma of the left kidney. Tissue samples of the mass were taken for histopathological examination. Macroscopic lesions were not observed in other organs; however, samples of the lungs, spleen, heart, liver, and brain were also taken for histopathology to exclude the presence of micrometastasis. The tissue samples were fixed in 10% neutral buffered formalin, processed routinely, embedded in paraffin, sectioned, stained with hematoxylin and eosin, coverslipped, and examined microscopically. The mass involving the left kidney consisted of elongated, irregular tubules that were lined with 2 layers of atypical epithelial cells. Individual cells had indistinct cellular borders and were tightly compressed, with nuclear displacement. Tubular epithelial cells also exhibited anisocytosis and anisokaryosis. Individual neoplastic cells had a prominent, euchromatic nucleus with an eccentrically located nucleolus. The cytoplasm was eosinophilic and finely granular. A few clear cells also were observed (Fig. 2). Forty-five mitoses were in ten 400× fields of view and some mitoses appeared atypical. Apoptotic cells and moderate desmoplasia also were observed. Scattered mononuclear infiltrates were present in the interstitium. The differential diagnosis for this renal mass included renal tubular carcinoma, nephroblastoma, undifferentiated sarcoma, and oncocytoma. Based on the histologic features of this neoplasm, a tentative diagnosis of poorly differentiated renal tubular carcinoma was made based. Microscopic examination of the lungs, spleen, heart, liver, and brain failed to reveal micrometastases.
Anticipated immunohistochemical staining pattern of neoplastic cells in 4 renal neoplasms.*
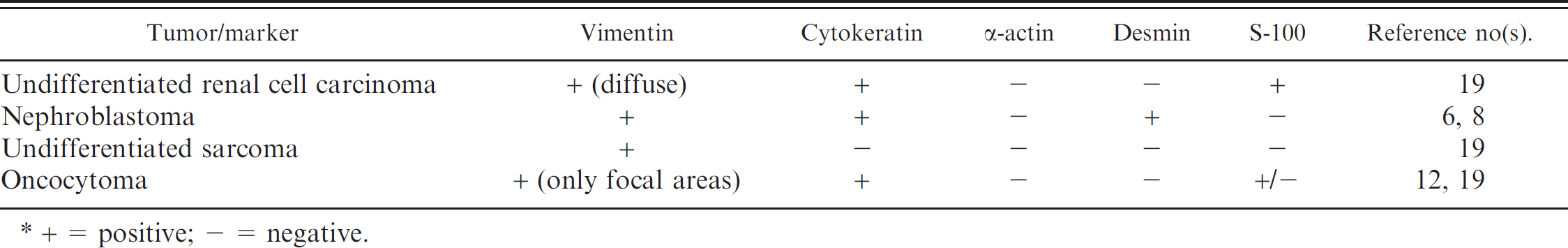
+ = positive; - = negative.
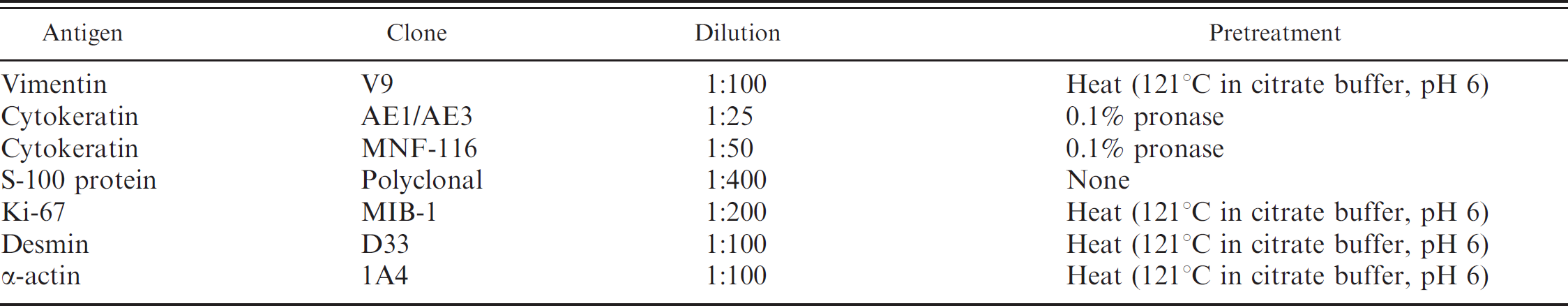
Immunohistochemistry was performed to establish a definitive diagnosis. Replicate 4-μm tissue sections were stained using the avidin-biotin-peroxidase complex (ABC) technique and several primary antibodies. a The primary antibody clone, primary antibody dilution, and pretreatment regimen for antigen retrieval are presented in Table 1. The anticipated immunohistochemical staining pattern for various renal neoplasms considered in the differential diagnosis is presented in Table 2. Although mitotic activity has been used as a prognostic factor for many neoplasms, the mitotic index of renal carcinomas is usually too low to provide meaningful information about tumor staging and grading in individual neoplasms. 5 Therefore, nuclear Ki-67 expression was evaluated to determine the degree of cellular proliferation. 5 Evaluation of Ki-67 expression also has been suggested as a useful marker to differentiate renal tubular carcinoma from oncocytoma. 21 After dewaxing, rehydration, and pretreatment for antigen retrieval, the tissue sections were incubated with 0.5% hydrogen peroxide in methanol for 30 min, washed in 0.05 M Tris-buffered saline solution (TBS), and blocked for 30 min with normal serum b diluted 1:100 in TBS to minimize nonspecific binding of the primary antibodies. The slides were then incubated with the primary antibody in a moist chamber for 60 min. After incubation, the slides were washed in TBS, incubated with the biotinylated antibody b b for 30 min, washed once more in TBS, and incubated with the ABC b reagent for 30 min. Sites of primary antibody binding were visualized using a chromogen solution of 3-3′-diaminobenzidine tetrahydrochloride (0.05%) c and hydrogen peroxide (0.03%) in TBS with a 5-min incubation period. The reaction stopped after rinsing the slides in tap water. The slides were counterstained in Mayer hematoxylin for 3 min, rinsed in tap water, dehydrated, and coverslipped. The negative tissue control was normal kidney from an olive baboon. Sections of a human renal cell carcinoma were used as positive controls.
Following immunohistochemical staining, the neoplastic cells were positive for vimentin, cytokeratin, and S-100 protein. In addition, >70% of the neoplastic cells expressed Ki-67 protein (Fig. 3). The neoplastic cells were negative for desmin and α-actin. A definitive diagnosis of undifferentiated renal tubular carcinoma was based on the histologic and immunohistochemical findings.
Spontaneous neoplasia has been documented across all subspecies and most hybrid combinations of baboon. 3,4,15 Neoplasms involving the urogenital tract account for 21 % of all primate tumors, but only 9 spontaneous neoplasms have been reported in baboons. 3 Renal adenoma, renal carcinoma, and nephroblastoma have been reported previously in baboons, 3,4 but the low incidence of spontaneous neoplasms is probably because most nonhuman primates are euthanized at a young age in most research settings.
Previous investigations have reported a positive correlation between age and incidence of spontaneous neoplasia in baboons. 3,4,19 The neoplasm in the baboon of this report is unusual in that it occurred in a juvenile animal. To date, only nephroblastoma has been described in young baboons. 4 Renal adenoma and renal carcinoma have been reported in baboons only older than 15 years. 3,4
In humans, renal cell carcinoma may remain clinically occult for most of its clinical course. The classic triad of flank pain, hematuria, and presence of a flank mass is uncommon (10%) and usually indicative of advanced disease. Twenty-five percent to 30% of human patients are asymptomatic, and the renal cell carcinomas are detected incidentally during radiographic studies.
In the baboon of the current report, the neoplastic renal cells expressed cytokeratin and vimentin. If the neoplasm is positive for cytokeratin and vimentin, a diagnosis of renal carcinoma can be made. In contrast, sarcomas are positive for vimentin and negative for cytokeratin. Diagnosis of nephroblastoma is based on the histologic observation of embryonic glomeruli and blastemal proliferation, often with muscular and osseous metaplasia. 10 However, these histologic features were not found in the neoplasm of the present report. Desmoplasia is a common feature in nephroblastomas and undifferentiated carcinomas 19 but may also be observed in renal carcinomas. Immunohistochemically, human nephroblastomas are reportedly positive for vimentin, 7 cytokeratin, 6 and desmin 8 ; however, the neoplasm in this baboon was negative for desmin and other muscle markers such as α-actin. Therefore, a diagnosis of nephroblastoma was excluded. Although the S-100 staining is a useful marker necessary to differentiate some types of renal carcinoma from oncocytoma, 16 vimentin stain was useful for this distinction in the neoplasm reported in this baboon. Expression vimentin in oncocytomas has been controversial. Recent studies of human oncocytomas document focal expression of vimentin in some neoplastic cells, 12 but more uniform, diffuse, cytoplasmic expression of vimentin was observed in the neoplastic cells of this baboon. Expression of Ki-67 is much lower in oncocytomas than in renal cell carcinoma. 21 Based on the histologic features and immunohistochemical staining of the neoplasm in the baboon of the current report, a diagnosis of oncocytoma also was excluded.
More than 70% of the neoplastic cells in the renal tubular carcinoma from this juvenile baboon had intense nuclear expression of Ki-67, indicating marked cellular proliferation. Although this degree of Ki-67 expression suggests moderate malignant potential, metastases were not detected in other tissues and organs that were examined. Only 30% of human renal carcinomas metastasize to other organs. 17 Thus, the definitive diagnosis was undifferentiated renal tubular carcinoma.
Footnotes
a.
Dako North America Inc., Carpinteria, CA.
b.
Vector Laboratories Inc., Burlingame, CA.
c.
Sigma-Aldrich, St. Louis, MO.
