Abstract
A 3.5-y-old Whippet mixed-breed dog was presented with a history of respiratory distress, exercise intolerance, and generalized demodicosis. Hematologic alterations included marked leukocytosis and neutrophilia. Radiographic examination showed a diffuse interstitial and mild peripheral alveolar lung pattern and pneumomediastinum. Because the cytologic examination of the bronchoalveolar aspirate was not diagnostic and a persistent perforation of the upper respiratory tract could not be ruled out, the dog was submitted to thoracoscopy, and subsequently the left cranial lung lobe as well as mediastinal and sternal lymph nodes were resected. Pulmonary pneumocystosis with spread to the thoracic lymph nodes was suspected after histologic investigation of lung and lymph nodes, which was confirmed by in situ hybridization, PCR, and subsequent Sanger sequencing. We document a rare, simultaneous occurrence of severe pulmonary and thoracic lymph node pneumocystosis with spontaneous pneumomediastinum in a dog. Definitive diagnosis was achieved through the use of Grocott methenamine silver staining, in situ hybridization, and PCR.
The genus Pneumocystis comprises a group of highly diversified opportunistic fungal organisms that attach primarily to pneumocytes of various mammalian hosts. 4 In dogs, clinical pneumocystosis occurs rarely, 18 and severe cases with development of spontaneous pneumomediastinum 14 or extrapulmonary pneumocystosis 16 are only observed sporadically. We describe a Pneumocystis carinii infection with thoracic lymph node involvement and severe pneumomediastinum in a Whippet mixed-breed dog.
A 3.5-y-old male neutered Whippet mixed-breed dog was presented to the University of Veterinary Medicine (Vienna, Austria) with ~2-mo history of respiratory distress. The owner’s consent for using samples and data was obtained on admission of the case, and no further ethics permission was required. The dog had been imported from Bulgaria, and its vaccination status was unclear. The dog had been pretreated with each of the following antimicrobial substances for 1 wk: amoxicillin–clavulanate, cefovecin, enrofloxacin, lincomycin, clindamycin, and azithromycin. As well as the therapy-resistant respiratory disorder, the dog suffered from generalized demodicosis and chronic diarrhea of unknown cause. Moreover, the dog had shown poor appetite and exercise intolerance. Physical examination revealed tachypnea (120 breaths/min) and dyspnea. On thoracic auscultation, the lung sounds were harsh.
A complete blood count, serum biochemical analysis, thoracic radiographs, as well as cytologic analysis of bronchoalveolar lavage fluid (BALF) were performed. Because of marked leukocytosis and neutrophilia, antibiotic treatment with azithromycin (10 mg/kg orally once a day) was started. Serum biochemical findings included increases in alanine aminotransferase (489 U/L; reference interval [RI]: <80 U/L) and alkaline phosphatase (377 U/L; RI: <130 U/L) activities. The examination of BALF revealed pyogranulomatous inflammation, with 2.4 × 108 cells/L, consisting mainly of ciliated epithelial cells and inflammatory cells (56% neutrophils, 44% mononuclear cells).
Thoracic radiography showed a diffuse interstitial and a mild peripheral alveolar lung pattern in the caudal part of the left cranial and the right medial lobe. A distinct gas compartment dorsal and ventral to the trachea extended over the mediastinum and led to the diagnosis of pneumomediastinum (Fig. 1A, 1B). Air within the axillary regions, the mediastinum, and contiguous with bronchovascular sheaths represented axillary emphysema, pneumomediastinum, and interstitial emphysema, whereas the patchy increased attenuation of the pulmonary parenchyma corresponded with pneumonia, edema, or hemorrhage. Sternal and bronchial lymph nodes were moderately enlarged.

Because the cause of the pneumomediastinum could not be determined, computed tomography (CT) was carried out. A persistent perforation of the upper respiratory tract could not be totally ruled out (Fig. 1C, 1D). For this reason, thoracoscopy was performed, but the cause of the pneumomediastinum still could not be detected. Despite artificial ventilation, the left cranial lung lobe was poorly ventilated and therefore resected. Histologically, the pulmonary architecture was effaced; pulmonary alveoli were diffusely filled with many small eosinophilic, foamy, spherical structures, which were predominantly free or phagocytosed by macrophages. The macrophages containing these organisms were variably sized and foamy, sometimes reaching 15–25 µm in diameter (Fig. 2A). The interstitium was moderately infiltrated by lymphocytes and histiocytes. Many of the numerous small congested interstitial blood vessels had prominent perivascular cuffs of lymphocytes and macrophages. There were moderate, multifocal areas of hemorrhage throughout the lung and moderate pleural fibrin. Lymphoid tissue of the mediastinal and sternal lymph nodes was depleted, and the architecture was loose. Marginal sinuses were enlarged, and free foamy structures aggregated focally directly under the capsule. The medullary sinuses showed blood resorption (Fig. 2B).
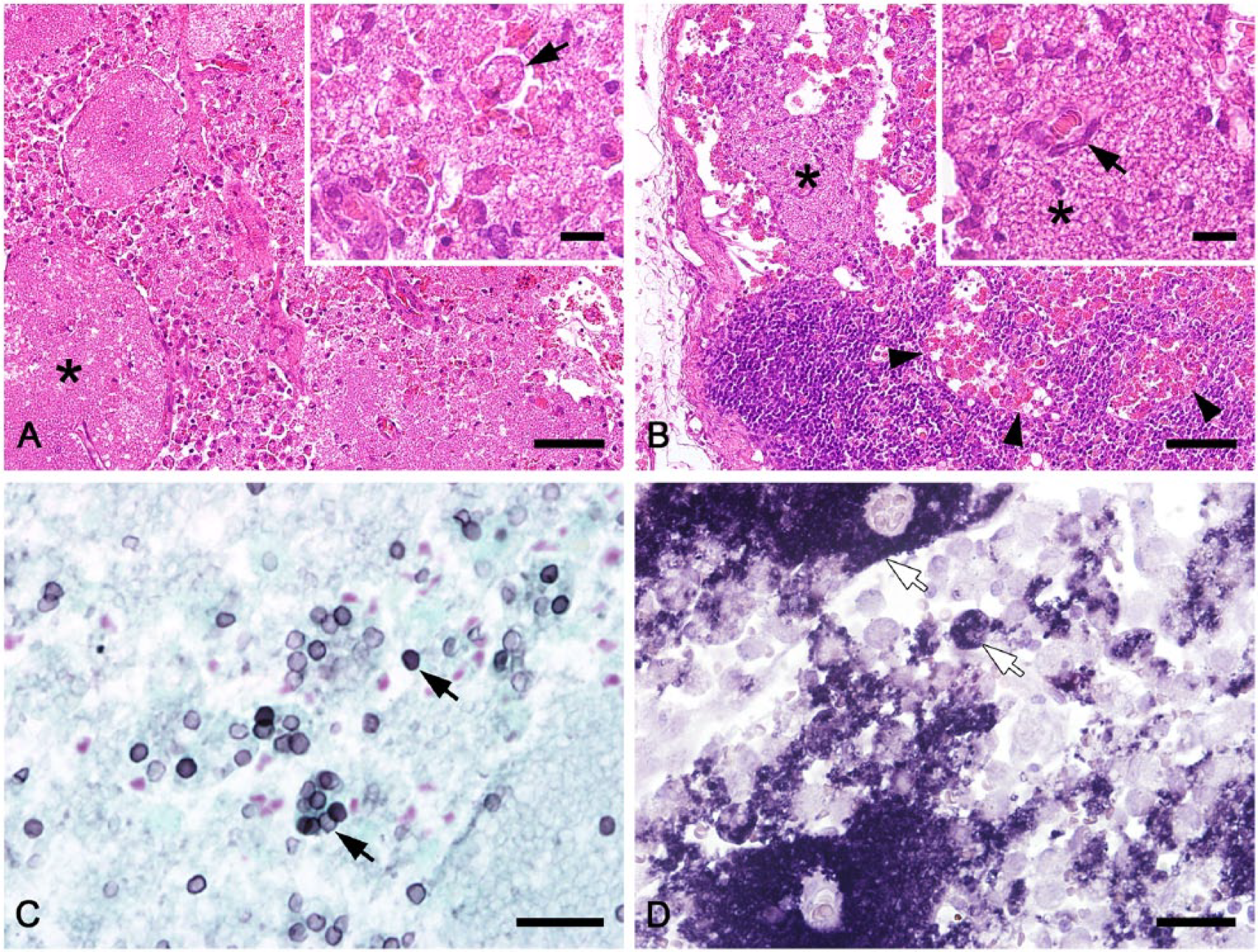
Pneumocystosis was suspected because of foamy structures in the pulmonary alveolar lumens and macrophages, and tissue sections were stained with Grocott methenamine silver (GMS). In the lung sample, only cystic developmental stages of Pneumocystis stained positive, and they comprised a minor part of all organisms present. The majority of the organisms were trophozoites, which remained unstained (Fig. 2C). In contrast, both developmental stages could be detected equally by in situ hybridization (ISH), which was carried out as described previously. 2 The lung sample was strongly positive; large colonies of organisms completely filled the alveoli, and signals were present free or within macrophages (Fig. 2D). In lymph nodes, ISH signals were localized focally directly under the capsule within the marginal sinuses or sporadically scattered within the medullary sinuses. The samples were also positive by PCR. 8 Prior to PCR, DNA was extracted from 5 formalin-fixed, paraffin-embedded lung tissue sections (10 µm thick; AllPrep DNA/RNA FFPE kit, Qiagen, Hilden, Germany). DNA was eluted in a volume of 60 µL of buffer. PCR was conducted with primers targeting the mitochondrial large subunit ribosomal RNA (mtLSU rRNA) gene of Pneumocystis carinii (pAZ102-H: 5’-GTGTACGTTGCAAAGTACTC-3’; pAZ102-E: 5’-GATGGCTGTTTCCAAGCCCA-3’). The PCR reaction master mixture consisted of 10 µL of master mix (HotMasterMix, 5Prime, Eppendorf, Vienna, Austria), 0.4 µM of each primer, 2 µL of template DNA (diluted 1:50 with distilled water), and distilled water to a total volume of 25 µL. The PCR temperature profile was 94°C for 2 min, followed by 40 cycles at 94°C for 30 s, 60°C for 30 s, and 72°C for 1 min. A final DNA elongation step was carried out at 72°C for 10 min. The PCR product had a size of 350 bp.
A phylogenetic tree was constructed using MEGA6 (www.megasoftware.net) with mtLSU rRNA sequences from the dog in our study (accession KX227609) and 28 representative human and animal species with DNA Data Bank of Japan accession numbers. Additionally, sequences labeled as English type A and B were included. 6 FJ385030 served as outgroup. The evolutionary history was inferred by using the maximum likelihood method based on the Hasegawa–Kishino–Yano model. The percentage of trees in which the associated taxa clustered together is shown next to the branches. The sequence identified in our study (accession KX227609) is located in one cluster with sequences from published canine and ferret Pneumocystis samples (Fig. 3). The similarity to LC009003 (dog) and the English type B sequence (dog) 6 was 100%; S42921 (ferret) had 87% similarity, and the English type A sequence (dog) 6 had an 84% sequence similarity.
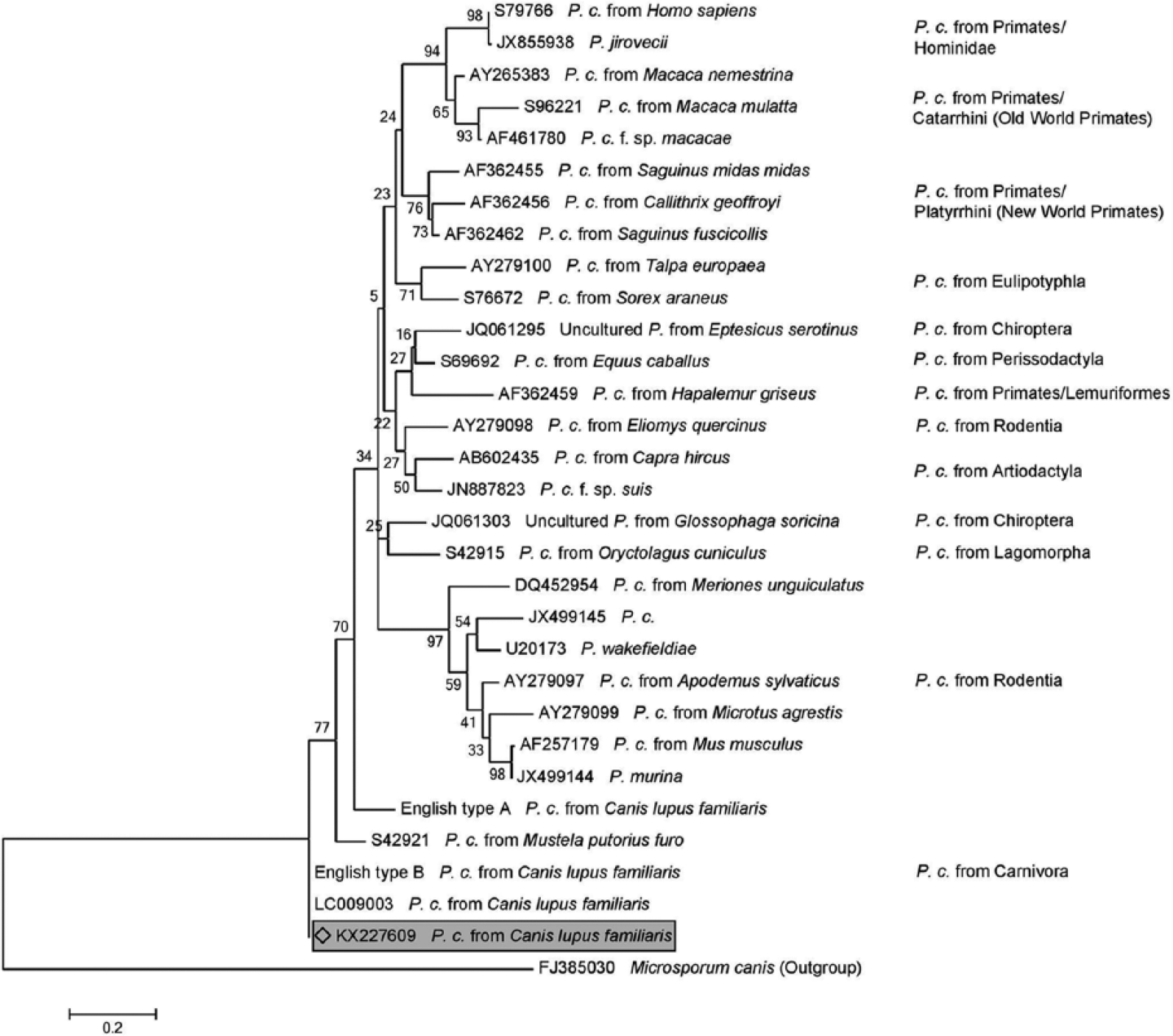
Phylogenetic tree constructed with mtLSU rRNA sequences from the Whippet mixed-breed dog of the present study (accession KX227609), 28 representative human and animal species with DDBJ accession numbers, and the sequences English type A and B. 6 FJ385030 served as outgroup.
During the diagnostic work-up, medical treatment with azithromycin, prednisolone (0.5 mg/kg), bronchodilators, and constant-rate infusion with Ringers lactate was started. Despite intensive care, including oxygen supplementation, suction of gas and fluid by thoracostomy tube, and close monitoring of blood gas analysis, the clinical status of the dog deteriorated within 3 d after thoracoscopy. Additional radiographic examination of the chest revealed severe pleural effusion and pulmonary edema in addition to pneumomediastinum. As progressive metabolic decompensation became apparent, the owner agreed to euthanasia. Postmortem examination was not performed.
Subclinical infections with Pneumocystis seem to be relatively common in mammals including dogs. 4 However, clinical signs associated with Pneumocystis pneumonia are observed quite rarely. 4 Depending on the severity of infection, pneumocystosis has variable non-pathognomonic respiratory signs ranging from cough 3 to tachypnea and dyspnea. 13 Chronic coinfections, such as demodicosis,7,8 to which Whippets are predisposed (https://goo.gl/DTYp6R), dermatitis, 9 or enteritis 18 can impair immunity and may indirectly promote the proliferation of Pneumocystis. The genetic heterogeneity of Pneumocystis species of various mammals can be high. 4 For dogs, only 2 genotypes have been described, 6 and the sequence of our study could be assigned to one of them. Given the low number of published genetic results, it remains unclear if genotypes differ with regard to virulence or pathogenicity.
Consistent with other authors, results of blood analysis were nonspecific in our case. Thoracic radiography is one of the cornerstones used to establish the tentative diagnosis of pneumocystosis, with a presentation of bilateral, diffuse, symmetrical reticular or granular opacities. 5 In our case, pneumomediastinum was diagnosed radiographically, as reported once previously. 14 The occurrence of spontaneous pneumomediastinum secondary to pulmonary disease resembling fibrosing interstitial pneumonia has been reported in Dalmatian dogs. 1 However, pneumothorax is a known complication of Pneumocystis pneumonia in human medicine with an incidence of 5–6%.5,12 Moreover, nearly half of patients develop spontaneous pneumothorax. 12 The so-called Macklin effect results when air from ruptured alveoli migrates to the mediastinum. 10 Other patients develop pneumothorax during mechanical ventilation or after an invasive procedure. 12
In humans, most antemortem cases of Pneumocystis infection involve only the lungs. Extrapulmonary pneumocystosis occurs in ~2% of cases with Pneumocystis pneumonia, but may also be present without concurrent pneumonia. 11 The fungus spreads via both lymphatic and hematogenous routes and mainly affects lymph nodes, spleen, liver, and bone marrow. 15 Patients with a combination of pulmonary and extrapulmonary pneumocystosis usually die rapidly. In dogs, extrapulmonary pneumocystosis has been observed in lymph node and heart tissue. 16 As in our case, severe pulmonary lesions may lead to involvement of lymph nodes.
The diagnosis of pneumocystosis in dogs is based upon direct visualization of the Pneumocystis organisms in respiratory tract specimens. Unfortunately, Pneumocystis cannot be detected reliably in BALF samples by direct visualization. 18 The evaluation of Giemsa-stained cytologic slides requires experience, especially when the number of Pneumocystis organisms is low. As a consequence, the typical eosinophilic foamy structures can be missed easily. GMS stain can be useful and has been described to be 7.6% more sensitive than Giemsa stain. 19 The disadvantage of GMS stain is that only cysts are stained positive whereas the greater proportion of Pneumocystis organisms, the trophozoites, remain unstained, which could be a problem in low-grade cases. ISH identifies cysts and trophozoites equally but can only be performed on tissue sections and not on cytologic smears. Furthermore, Pneumocystis organisms may not be visualized in a high number of lavage samples. In humans, this occurs in 32–80% of cases. 17 An alternative could be the determination of Pneumocystis nucleic acid in BALF by highly sensitive PCR methods, as has proved useful in pigs. 20
The nonspecific clinical signs and the very rare occurrence of pneumocystosis make a fast and correct clinical diagnosis difficult; inappropriate treatment is associated with high mortality. 14 In chronic therapy-resistant dogs with a history of chronic respiratory disorders and generalized demodicosis, Pneumocystis infection should be considered as one of the most probable differential diagnoses.7,8 If Pneumocystis cannot be determined cytologically in BALF samples, a more sensitive PCR analysis can be recommended in order to avoid invasive interventions. In the case of a non-diagnostic lavage sample, the collection of lung biopsy samples can be considered as a final option for definitive diagnosis.
Footnotes
Acknowledgements
We thank Karin Fragner, Nora Nedorost, and Klaus Bittermann for their excellent technical support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
