Abstract
Dry-mount fecal cytology (FC) is a component of the diagnostic evaluation of gastrointestinal diseases. There is limited information on the possible effect of the sampling method on the cytologic findings of healthy dogs or dogs admitted with diarrhea. We aimed to: (1) establish sampling method–specific expected values of selected cytologic parameters (isolated or clustered epithelial cells, neutrophils, lymphocytes, macrophages, spore-forming rods) in clinically healthy dogs; (2) investigate if the detection of cytologic abnormalities differs among methods in dogs admitted with diarrhea; and (3) investigate if there is any association between FC abnormalities and the anatomic origin (small- or large-bowel diarrhea) or the chronicity of diarrhea. Sampling with digital examination (DE), rectal scraping (RS), and rectal lavage (RL) was prospectively assessed in 37 healthy and 34 diarrheic dogs. The median numbers of isolated (p = 0.000) or clustered (p = 0.002) epithelial cells, and of lymphocytes (p = 0.000), differed among the 3 methods in healthy dogs. In the diarrheic dogs, the RL method was the least sensitive in detecting neutrophils, and isolated or clustered epithelial cells. Cytologic abnormalities were not associated with the origin or the chronicity of diarrhea. Sampling methods differed in their sensitivity to detect abnormalities in FC; DE or RS may be of higher sensitivity compared to RL. Anatomic origin or chronicity of diarrhea do not seem to affect the detection of cytologic abnormalities.
Dry-mount fecal cytology (FC) is a component of fecal examination in dogs with gastrointestinal diseases.2,13 FC is especially useful for the observation of specific infectious agents (e.g., Prototheca sp., Cryptococcus sp.), as well as inflammatory and neoplastic cells.4,15,17 Fecal specimens are usually collected by digital examination (DE), rectal scraping (RS), or saline-based rectal lavage (RL), obtaining material mainly from the enteric lumen, the rectal mucosa, or from both. 2 In clinically healthy dogs, expected FC findings are a heterogeneous population of bacilli, a few clustered or isolated epithelial cells, a few spore-forming bacilli, and incidental yeasts of unknown clinical significance (e.g., Cyniclomyces guttulatus), whereas white blood cells are not typically seen.7,16 To our knowledge, no studies have been performed to investigate if the sampling method may affect the cytologic findings in healthy or diarrheic dogs, or if the FC abnormalities are associated with the anatomic origin or the chronicity of diarrhea.
We sought to (1) establish sampling method–specific (DE, RS, or RL) expected values of selected cytologic parameters (isolated or clustered epithelial cells, neutrophils, lymphocytes, eosinophils, macrophages, spore-forming bacilli) in clinically healthy dogs; (2) investigate if the detection of cytologic abnormalities differs among the sampling methods in dogs admitted with diarrhea; and (3) investigate if there is any association between the occurrence of cytologic abnormalities and the anatomic origin (small bowel or large bowel) or the chronicity (acute or chronic) of diarrhea.
A total of 71 (37 healthy and 34 dogs with diarrhea) client-owned dogs were presented to the University Small Animal Teaching Hospital (Aristotle University of Thessaloniki, Thessaloniki, Greece), from August 2014 through July 2015, and were prospectively enrolled in the study. Written informed consent was required from the owners for their dogs to be enrolled in the study.
Healthy dogs (group A) were presented for vaccination, elective surgery (spaying, neutering), or orthopedic (e.g., osteoarthritis) conditions. Inclusion criteria included: (1) dogs >2 mo of age and body weight >2 kg; (2) no historical evidence of general or gastrointestinal manifestations or exposure to drugs other than those intended for routine deworming for the last month prior to admission; (3) historically sufficient endoparasite control and no evidence of parasite elements upon fecal parasitologic examination; (4) no clinical evidence of rectal, anal, or perineal lesions; and (5) normal complete blood count (CBC). This group included purebred (15 of 37, 41%) and mixed-breed dogs (22 of 37, 59%), of which 19 (51%) were males and 18 (49%) were females, with an age range of 0.4–12 y (median: 2 y).
Group B dogs were patients admitted with a history of acute or chronic diarrhea, the result of a primary gastrointestinal disease. Inclusion criteria included (1) dogs >2 mo old of age and body weight >2 kg; (2) dogs in which the veterinarian could assess stool fluidity, volume, and daily frequency of defecations, based on history and/or physical examination 1 ; (3) no evidence of systemic diseases, which were reasonably excluded based on CBC and comprehensive serum biochemistry testing performed in all dogs; and (4) no historical drug exposure other than those intended for routine deworming prior to the occurrence of diarrhea.
Diarrhea was defined as chronic if the duration was >15 d. 8 When possible, the anatomic location of diarrhea was defined as mostly of small or of large intestinal origin, based on the historical or clinical occurrence of tenesmus, dyschezia or hematochezia, the presence of fecal mucus, the defecation frequency, and the approximate fecal volume per defecation.3,8 Ancillary tests, such as fecal parvoviral antigen testing (n = 3), fecal Giardia antigen testing (n = 6), colonoscopy (n = 6), and histopathology (n = 6), were performed at the discretion of the attending clinician for the establishment of a definitive or presumptive diagnosis. Group B dogs included purebred (23 of 34, 68%) and mixed-breed dogs (11 of 34, 32%), of which 27 (79%) were males and 7 (21%) were females, with an age range of 0.17–13 y (median: 2 y).
On admission, a fecal sample (1–2 g) was collected from the rectum of all dogs for parasitologic examination that included: (1) wet-mount saline preparation for the detection of motile organisms, 2 (2) zinc sulfate flotation method, 14 (3) formalin–ether sedimentation method, 12 and (4) modified Ziehl–Nielsen for the detection of Cryptosporidium sp. 5 Wet mount preparations were examined within 10 min of fecal sampling, whereas the rest of the fecal aliquots were examined within 3 d, after storage in a refrigerator.
Following the initial sampling for parasitologic examination, 3 sampling techniques (DE, RS, and RL) were used for the collection of material for FC. For DE, at least 2 thin smears were prepared by digital rectal examination, avoiding scraping the mucosal surface. 16 For RS, a saline-moistened, cotton-tipped non-sterile applicator was used to obtain material from the mucosal surface. The cotton tip of the applicator was gently rolled on a glass slide to prepare a direct thin film. This technique was repeated for the preparation of 2 smears. For RL, an 8F rubber catheter was passed through the anus up to 5–10 cm into the rectum. Ten mL of normal saline were infused through the catheter and aspirated 3 times to obtain a sample with mud-like consistency. 2 This sample was directly centrifuged (500 × g, 5 min), and at least 2 smears were prepared from the sediment with the blood smear technique. 2 The time for completion of the sampling procedure per method was recorded in all dogs. Slides were air-dried and stained with Giemsa (Merck, Darmstadt, Germany) for cytologic evaluation.
Cytologic evaluation was performed simultaneously and blindly (the examiners were unaware of the group allocation of the smears) by 2 of the authors (PS Frezoulis and ME Mylonakis) with a dual-headed microscope. Non-diluted or minimally hemodiluted microscopy fields were reviewed in all healthy dog cytologic specimens, although this was not possible in group B dogs admitted with bloody diarrhea. At least 10 consecutive monolayer areas (microscopy time: ~1–2 min) of the smears were reviewed (40× objective lens) for counting isolated or clustered (>3 cohesive cells) columnar epithelial or goblet cells (squamous cells were excluded) per 10 microscopy fields. The same number of fields (microscopy time: ~5–10 min) was reviewed (100× objective lens) for counting the absolute numbers of neutrophils, lymphocytes, macrophages, eosinophils, and “safety-pin”–like, spore-forming bacilli per 10 microscopy fields, or to evaluate for the presence or absence of gull-wing–shaped or spiral-shaped bacteria and any other protozoal or fungal organisms in the smear.
Comparisons of the FC evaluations among the 3 sampling methods (DE, RS, and RL) in groups A and B were made with the use of the Kruskal–Wallis test. Multiple comparisons were made with the Wilcoxon signed rank sum test and the McNemar test and were adjusted with the Bonferroni correction. The median values of the sampling time (between the insertion of the finger, applicator, or catheter in the rectum until procurement of material) among the 3 methods were compared with one-way analysis of variance. In this analysis, group A and group B were analyzed together and not as 2 different groups. The associations of the presence of fecal findings (overall leukocytes and epithelial cells) with diarrhea of small- or large-bowel origin and acute or chronic diarrhea were made with the McNemar test of symmetry (dogs with inadequate data on anatomic origin or chronicity were excluded from statistical analysis). Statistical analyses were made using Stata 13 (StataCorp, College Station, TX).
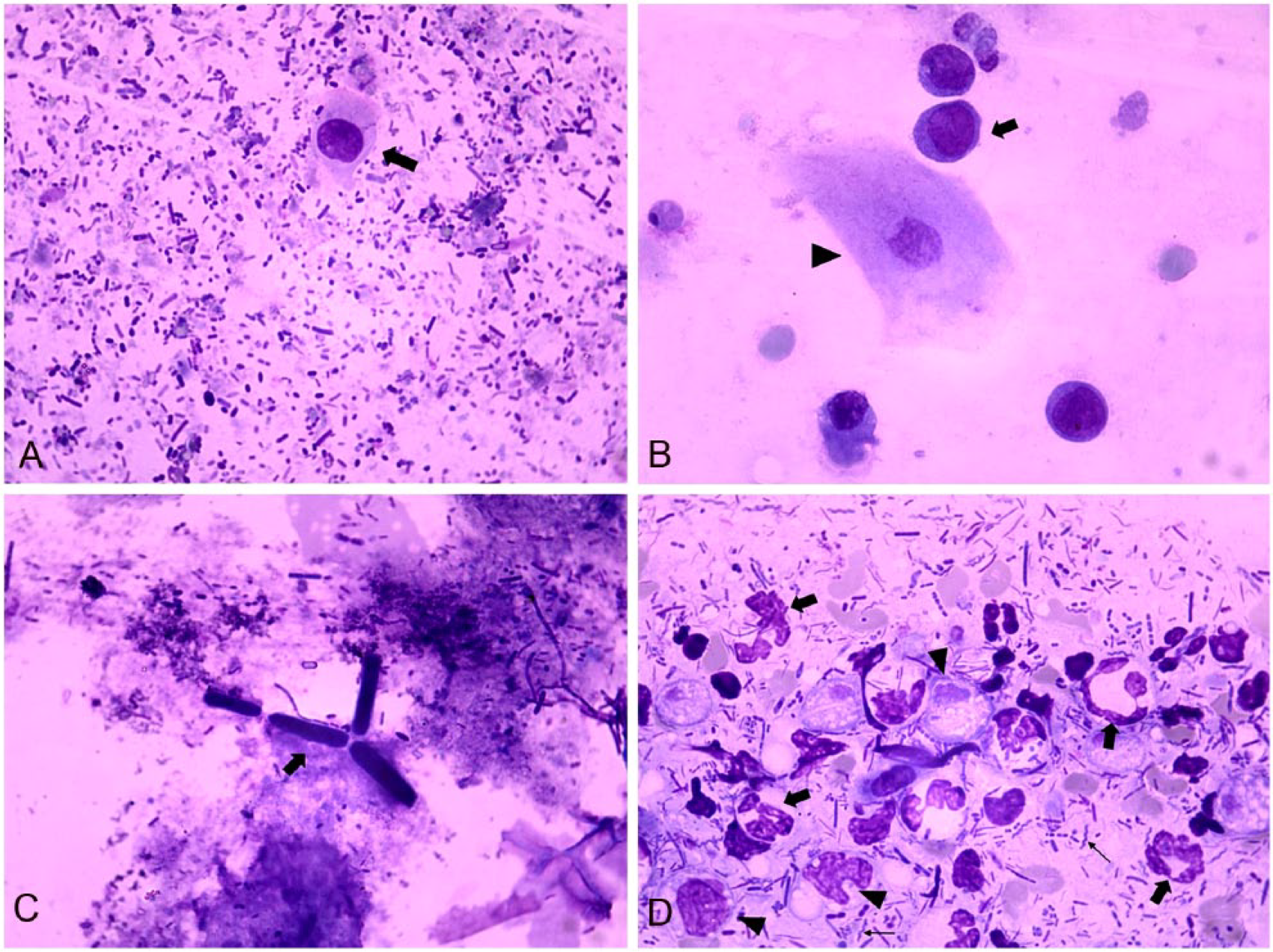
In healthy dogs, the median values of isolated epithelial cells (Fig. 1A), clustered epithelial cells, and lymphocytes differed significantly (p ≤ 0.05) among the 3 sampling methods, and were higher when fecal sampling was done by RS (Table 1).
Median values (per 10 microscopy fields) and percentages of positive dogs of selected fecal cytologic features in 37 healthy dogs (group A) based on different sampling methods.*
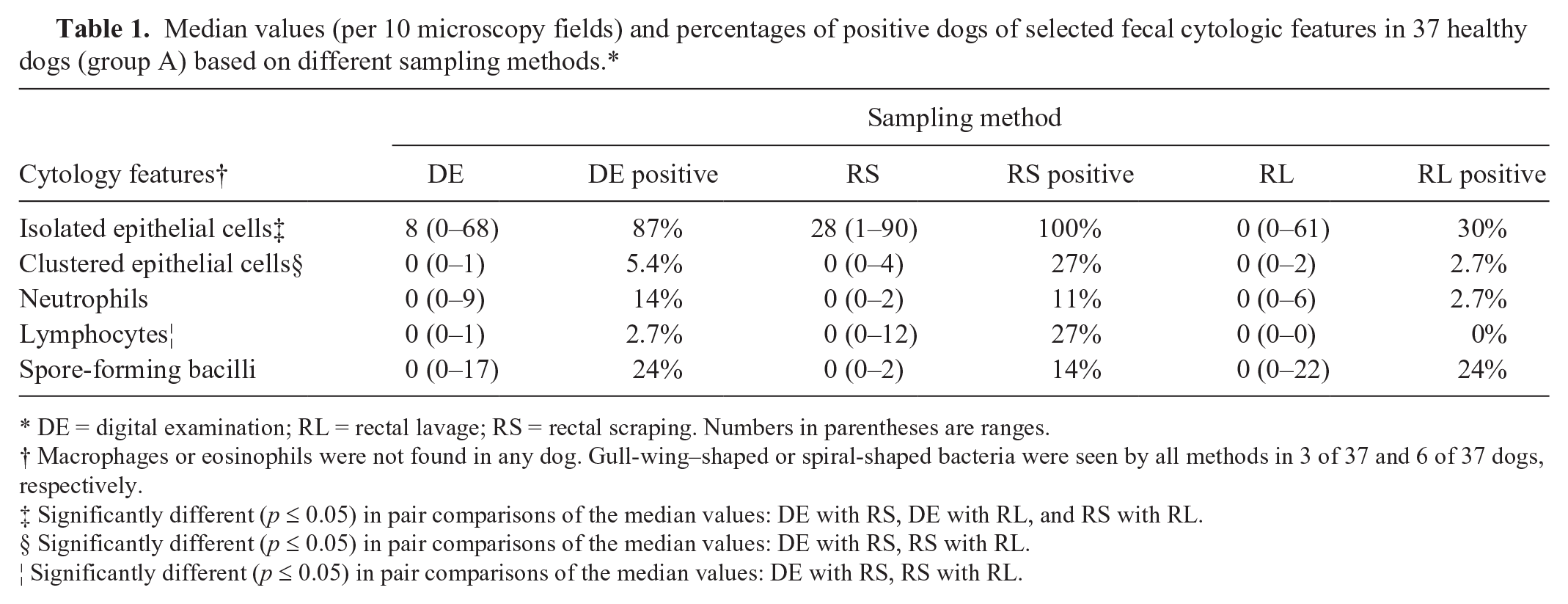
DE = digital examination; RL = rectal lavage; RS = rectal scraping. Numbers in parentheses are ranges.
Macrophages or eosinophils were not found in any dog. Gull-wing–shaped or spiral-shaped bacteria were seen by all methods in 3 of 37 and 6 of 37 dogs, respectively.
Significantly different (p ≤ 0.05) in pair comparisons of the median values: DE with RS, DE with RL, and RS with RL.
Significantly different (p ≤ 0.05) in pair comparisons of the median values: DE with RS, RS with RL.
Significantly different (p ≤ 0.05) in pair comparisons of the median values: DE with RS, RS with RL.
In the dogs admitted with diarrhea (group B), the definitive or presumptive diagnosis included parasitic enteritis (n = 9), acute hemorrhagic diarrhea (n = 5), food-responsive diarrhea (n = 5), inflammatory large bowel disease (n = 5), antibiotic-responsive diarrhea (n = 3), parvoviral enteritis (n = 2), high-grade rectal lymphoma (n = 1), leishmaniosis (n = 1), and distemper (n = 1); a diagnosis was not achieved in 2 dogs. Atypical lymphocytes (Fig. 1B) were seen in FC in the dog with rectal lymphoma by DE and RS methods, but not by the RL method. Parasitologic examination in group B revealed positive tests in 9 of 34 (27%) cases. Six of these dogs had single infections (2 with Toxocara canis and 1 each with Spirocerca lupi, Uncinaria stenocephala, Giardia spp., and Ancylostoma caninum); 3 dogs had mixed infections with 2 or more parasites (1 with T. canis and Isospora rivolta, 1 with U. stenocephala and Trichuris vulpis, and 1 with U. stenocephala, T. vulpis, and Toxascaris leonina). Two dogs were diagnosed with giardiasis based on fecal examination (cysts) and/or fecal antigen ELISA testing, but no Giardia trophozoites were detected in any of the FC techniques. C. guttulatus (Fig. 1C) was detected in one group B dog, with only one sampling method (RL).
In group B, the median values of isolated and clustered epithelial cells and the proportion of dogs with fecal neutrophils (Fig. 1D) differed significantly (p ≤ 0.05) among the sampling methods, and were higher when fecal sampling was done by RS (epithelial cells) or by DE or RS (neutrophils; Table 2).
Median values of the isolated and clustered epithelial cells and percentages of positive samples (all other cytologic findings) of 34 dogs admitted with diarrhea (group B) using 3 sampling methods.*
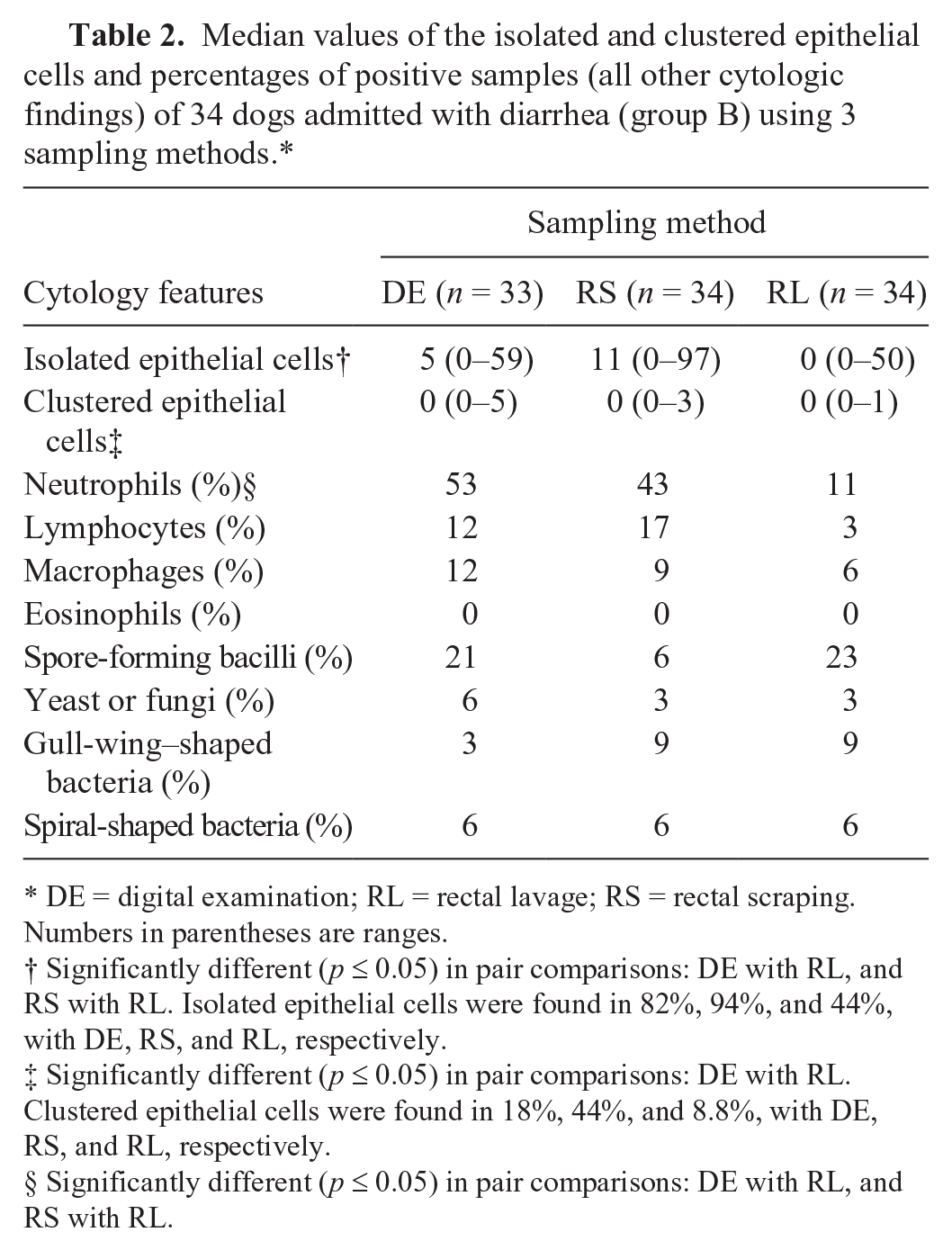
DE = digital examination; RL = rectal lavage; RS = rectal scraping. Numbers in parentheses are ranges.
Significantly different (p ≤ 0.05) in pair comparisons: DE with RL, and RS with RL. Isolated epithelial cells were found in 82%, 94%, and 44%, with DE, RS, and RL, respectively.
Significantly different (p ≤ 0.05) in pair comparisons: DE with RL. Clustered epithelial cells were found in 18%, 44%, and 8.8%, with DE, RS, and RL, respectively.
Significantly different (p ≤ 0.05) in pair comparisons: DE with RL, and RS with RL.
The mean (95% confidence intervals [CIs]) sampling time (in s) among the DE, RS, and RL sampling methods were 7 s (95% CI [4; 10]), 8 s (95% CI [5; 10]), and 29 s (95% CI [15; 60]), respectively. The mean values of the sampling time among the 3 methods differed significantly (p ≤ 0.05). The sampling time differed significantly between DE and RL (p ≤ 0.05) and RS and RL (p ≤ 0.05).
In 10 of 34 (29%) and 18 of 34 (53%) group B dogs, the anatomic origin of the diarrhea was assessed to be mostly from the small and large intestine, respectively; a valid determination of origin was not possible in 6 (18%) dogs. Diarrhea was evaluated as acute in 18 of 34 (53%) dogs, chronic in 15 of 34 (44%) dogs; it was indeterminate in 1 dog. No association was established between the presence of selected FC abnormalities (overall leukocytes and epithelial cells) and the origin (p = 0.8) or chronicity (p = 0.09) of diarrhea.
Our results indicate that sampling methods may significantly affect the numbers of certain cytologic features in healthy dogs, and they also differ in their sensitivity to demonstrate cytologic abnormalities in dogs with diarrhea. In addition, epithelial cells and leukocytes may be noticed on FC regardless of the anatomic origin or the chronicity of diarrhea.
The rationale behind generating expected values for selected FC parameters in healthy dogs is based on the fact that current knowledge on normal FC is based mostly on anecdotal information. 16 Importantly, although fecal leukocytes (neutrophils in particular) are thought to be absent from healthy dogs, 16 apparently healthy dogs in our study had cytologic evidence of neutrophils (2.7–14%) and lymphocytes (2.7–27%), as opposed to the invariably negative results for macrophages and eosinophils. Therefore, although a few fecal neutrophils and small lymphocytes may be interpreted with caution in diarrheic dogs, finding macrophages or eosinophils may be an indication of underlying mucosal lesions. Not unexpectedly, group A dogs had isolated (30–100%) or clustered (2.7–27%) epithelial cells. 16 RS yielded significantly higher numbers of lymphocytes and epithelial cells compared to the other methods, which may be attributed to the slightly more aggressive nature of this procedure, possibly exfoliating lymphocytes from the colonic lymphoid follicles and columnar epithelial cells from the mucosa more consistently. 16 Therefore, the sampling method–specific effect should be integrated into the numerical interpretation of lymphocytes and epithelial cells on FC; RS may be preferred when the deeper mucosal layers are to be sampled, such as for diagnosis of colonic lymphoma. In the sole rectal lymphoma case in group B, for example, atypical lymphocytes were found by both RS and DE methods but not with the RL method. In accord with previous studies, low numbers of spore-forming rods, but not in excess of the traditionally expected number of 3–5 spore-forming rods per 100× objective field, were also seen in 14–24% of the healthy dogs.9,10 Based on the results of our study, the number of spore-forming rods does not seem to be affected by sampling method. Observation of fecal endospores is a finding of arguable clinical relevance, as fecal endospore counts do not specifically indicate Clostridium spp. sporulation, and they do not consistently correlate with the occurrence of diarrhea induced by, or the presence of fecal enterotoxin produced by Clostridium spp.9 –11,16
As in the healthy dogs, isolated epithelial cells were recovered in higher numbers (and overall in higher frequency) with RS, but the median numbers of clustered epithelial cells did not differ between DE and RS methods. The clinical relevance of the visualization of fecal epithelial cells has yet to be elucidated. RL was significantly less sensitive in demonstrating fecal neutrophils (observed in 11% of the dogs) compared to DE (53%) and RS (43%), which makes it the least preferable method for documenting neutrophilic inflammation, especially in light of its time-consuming and labor-intensive nature compared to the other methods. No sampling method effect was documented in the remaining cytologic features, including lymphocytes, macrophages, yeast or fungal elements, spore-forming rods, and gull-wing– or spiral-shaped bacteria. Even though the rectal lymphoma was captured by FC, no attempt was made in our study to correlate the FC findings with the diagnosis or the clinical outcome of the dogs enrolled in group B, and future studies may be warranted to address these issues. Of comparative interest, a good correlation (specificity 97% and sensitivity 93%) was previously shown between exfoliative cytology obtained during endoscopy (brush or specimen touch) and subsequent endoscopic or surgical biopsy. 6
In our study, unlike the common notion that FC reflects mainly abnormalities of the distal intestinal segment, 16 epithelial and most importantly white blood cells were found in dogs with either small- or large-bowel disease. Although the latter finding may simply reflect diffuse rather than segmented pathologic processes, it can also be concluded that FC may be applicable regardless of the anatomic location of intestinal lesions. Chronicity of the diarrhea was also not shown to affect the outcome of FC in our study. A limitation of the present study is the relatively small number of dogs studied. A larger population might have allowed the establishment of reference intervals rather than expected values in the FC variables studied in healthy dogs, or the stronger evidence-based evaluation of the relative sensitivity of FC in diarrheic dogs.
Footnotes
Acknowledgements
We thank Dr. I Oikonomidis for technical support.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was financially supported by the Companion Animal Clinic, School of Veterinary Medicine, AUTh, Greece.
