Abstract
Cecal infarction is an uncommon lesion in calves that results in localized peritonitis and, on occasion, perforation with secondary diffuse peritonitis and death. This lesion in calves has not been described previously. We reviewed the postmortem cases of cecal infarction in dairy calves ≤30 d of age that had been submitted over the course of 5 y to the Tulare branch of the California Animal Health and Food Safety Laboratory System. The area of cecal infarction and the associated lesion margins were examined histologically. Ischemic necrosis of the mucosal side of the cecal wall with various degrees of neutrophilic inflammation of subjacent tissues was found consistently, and thrombosis and vascular occlusion within the areas of necrosis and inflammation was found in 21 of 34 cases. Cecal infarction cases were then compared to controls using a retrospective matched case-control study design. Cases (n = 34) and controls (n = 86) were compared with respect to bacteremia (as defined by pure culture of a single bacterial agent from lung and/or liver), species of bacteria isolated, and for the presence of K99 Escherichia coli (calves ≤5 d), attaching and effacing E. coli, Cryptosporidium (calves ≥5 d of age), Salmonella isolation from the intestine, rotavirus, Bovine coronavirus, and Bovine viral diarrhea virus. In addition, the presence of rumenitis or abomasitis, and omphalitis were compared between cases and controls. There were no significant differences in ruminal, abomasal, or umbilical cord tissue inflammation, or pathogen test–positive status between cases and controls.
Keywords
Cecal infarction (CI) is a rare condition in human medicine and can be a result of occlusive or nonocclusive ischemic necrosis of antimesenteric portions of the cecum. Nonocclusive infarction has been seen following open heart surgery, 4 hemodialysis, 2 and chronic heart disease. 5 Occlusive disease has been seen with atherosclerotic 1 or thromboembolic occlusions of the cecal arteries. 3 Within the Tulare branch of the California Animal Health and Food Safety Laboratory (CAHFS-Tulare), we have diagnosed CI in calves ≤30 d of age for many years, but we have been unable to locate reports of bovine CI in the peer-reviewed literature. Herein we describe the results of postmortem and histologic examination in a series of calves ≤30 d of age with findings consistent with CI. As well, in a matched case-control study, we compared CI cases to controls with respect to test-positive status for gastrointestinal pathogens.
The laboratory information management system at CAHFS-Tulare was queried for all submissions with 1 or more calves ≤30 d of age in which the diagnosis included the term “cecal infarction” during the period from July 1, 2007 to June 30, 2012. A case was defined as a calf diagnosed with CI on postmortem examination. Matching was performed using controls, calves without CI on gross postmortem, from the same laboratory submission (hence matched on premises and submission date), when possible. If the submission from which a case was identified had only 1 calf, then controls were selected from the immediately subsequent calf submission with controls matched by age.
Routine postmortem and histologic examinations were performed on all animals. Aerobic bacterial cultures were performed on lung and/or liver, and intestinal content was tested for Salmonella by culture, K99 Escherichia coli (calves ≤5 d of age) by antigen agglutination, a rotavirus by enzyme-linked immunosorbent assay (ELISA), b Cryptosporidium (calves ≥5 d of age) by acid-fast staining of smears of intestinal content, and attaching and effacing E. coli (AEEC) infection was determined by routine histologic examination of hematoxylin and eosin–stained colonic sections. In addition, frozen sections of colon were tested for both Bovine coronavirus and Bovine viral diarrhea virus by fluorescent antibody staining methods. The presence of omphalitis, and rumenitis and/or abomasitis was determined by postmortem examination and histopathology.
The odds ratio for each of the exposures (predictors) bacteremia, rumenitis and/or abomasitis, or omphalitis, and ancillary test results were estimated using conditional logistic regression. Between July 1, 2007 and June 30, 2012, all 34 Holstein calves with a postmortem diagnosis of CI at CAHFS-Tulare were selected. Calves in the case group were submitted from 10 different dairy calf ranches (25 calves) and 3 dairy farms (5 calves). As controls, 86 calves submitted from the same premises on the same day or the next submission of similar age calves were selected. Calves in the control group originated from 12 dairy calf ranches (81 calves) and 3 dairy farms (5 calves). The predominance of calf ranch submissions is consistent with the overall submission pattern for CAHFS-Tulare. Treatment and vaccination records were often incomplete and therefore were not included in this study. Case–control matching resulted in 29 case–control sets consisting of 1 or 2 cases, and 1–6 controls. The mean age of cases (10.6 d, SE = 0.8) and controls (8.9 d, SE = 0.6) was not significantly different. Table 1 summarizes the distribution of CI cases by gross postmortem, histopathology, or other test results. Univariate models showed no significant association between any of the predictors and CI (p > 0.05). A multivariable model similarly showed no significant associations between any of the predictors and CI (p > 0.05).
Testing frequency and percent positive for gross postmortem, histopathology, or laboratory tests performed on calves diagnosed with cecal infarction (cases) at postmortem, and a submission and age-matched sample of calves without cecal infarction (controls) submitted to the California Animal Health and Food Safety Laboratory, Tulare branch between July 1, 2007 and June 30, 2012.*
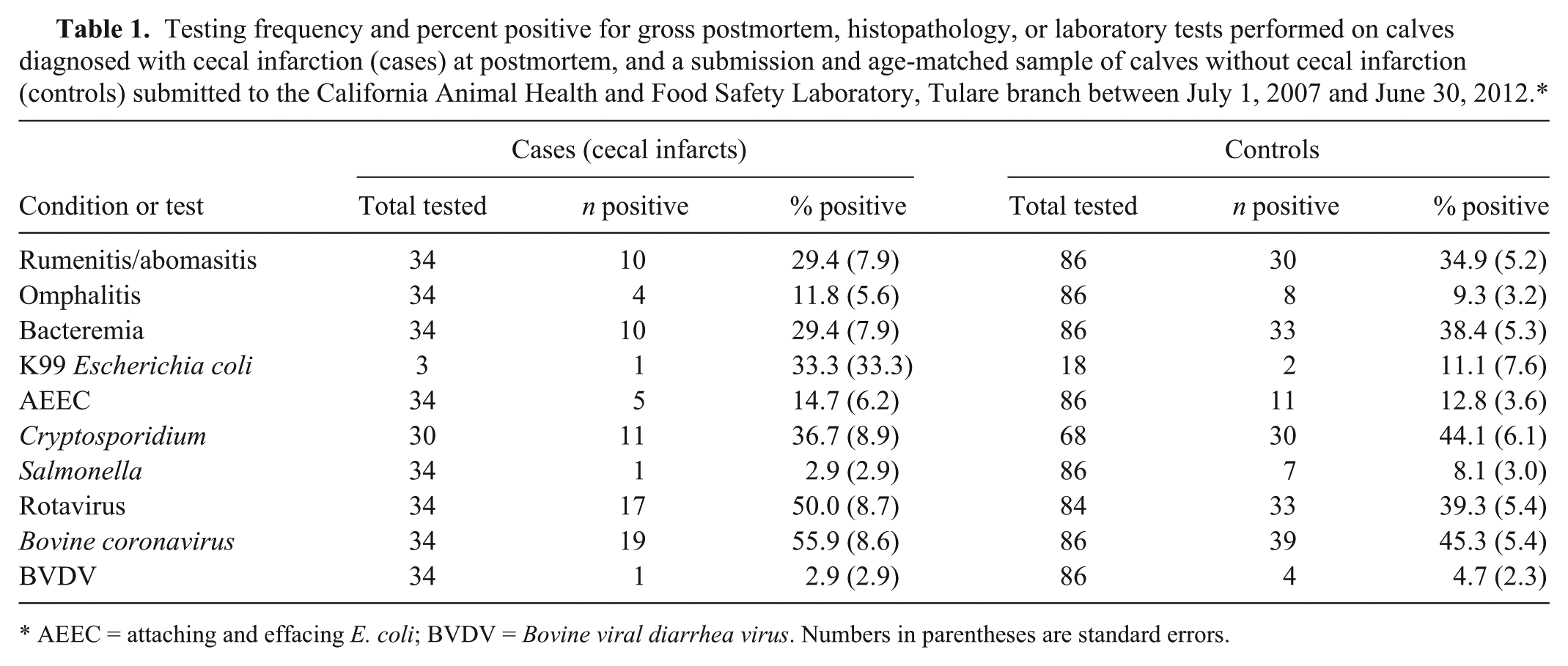
AEEC = attaching and effacing E. coli; BVDV = Bovine viral diarrhea virus. Numbers in parentheses are standard errors.
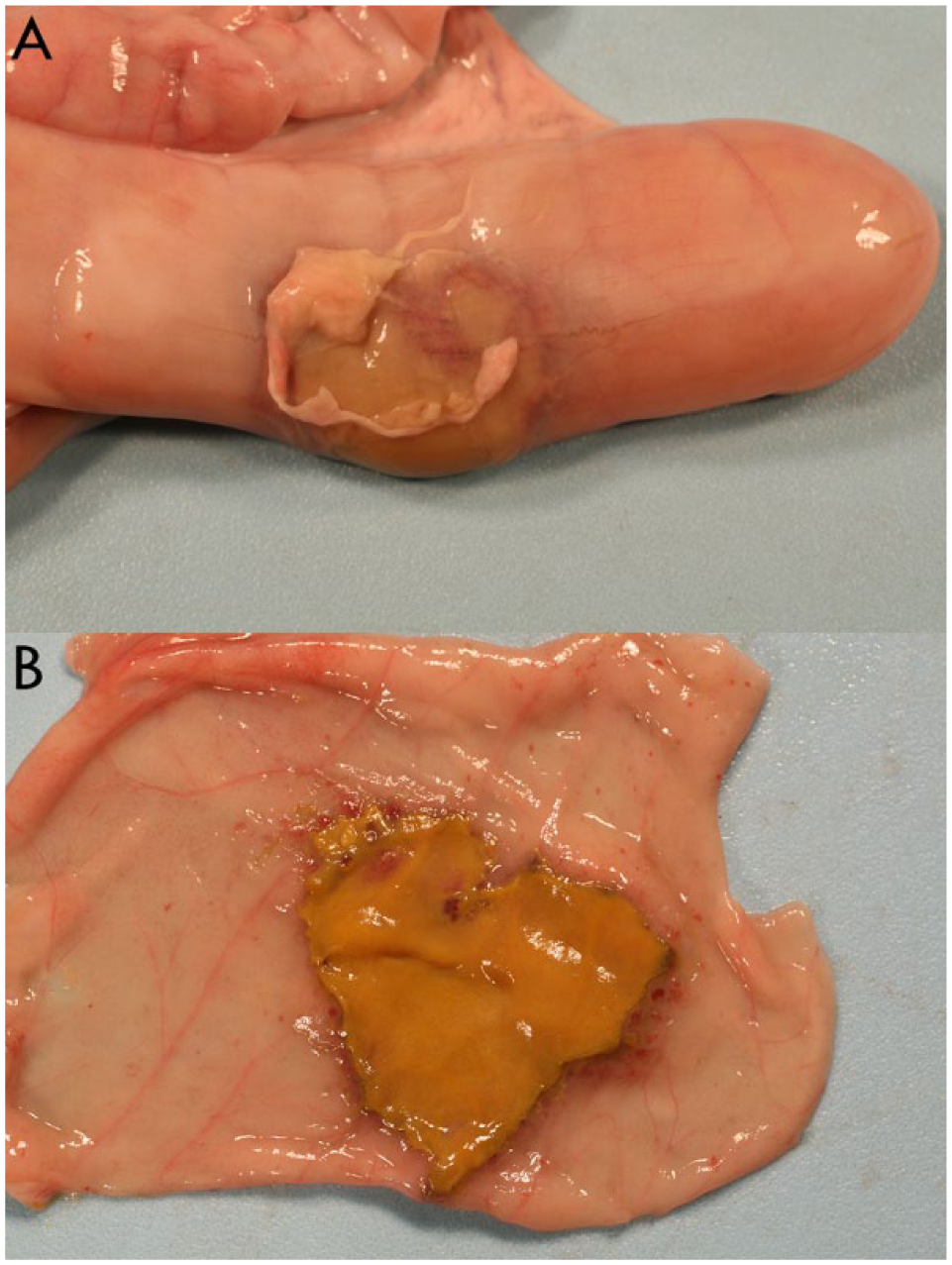
The gross postmortem lesion was most often near the midpoint of the cecum and consisted of a clearly demarcated, variably sized, discolored area of necrosis, usually on the antimesenteric side of the cecum. Although usually focal, the lesion was nearly circumferential in a small number of cases, and on occasion there was perforation at the center of the lesion. Localized to diffuse peritonitis with fibrin was common (Fig. 1A). The mucosal surface was often discolored tan to gray-black with a slightly raised red margin. Occasionally, a fibrinonecrotic membrane was adherent to the mucosal surface (Fig. 1B).
Histologically, the lesions consisted of a well-demarcated area of coagulative necrosis involving the mucosa that extended, to variable degrees, in a roughly wedge-shaped manner into the subjacent submucosa, muscularis, and serosal layers. At the deep and lateral margins, there were often abundant, usually degenerate, neutrophils. The necrotic tissue often contained large numbers of mixed bacteria, but there was minimal inflammation. There were variable amounts of fibrin adherent to the serosal surfaces and this corresponded to the severity of the necrosis in the cecal wall.
In the relatively unaltered viable submucosal tissues adjacent to the band of neutrophils, there was variable, often mild, vascular congestion and occasionally there were small arteries with fibrinoid necrosis or, rarely, mild vasculitis with neutrophils, and luminal fibrin thrombi. In some cases, the submucosa and muscularis adjacent to infarcts exhibited mild to moderate interstitial neutrophilic infiltrates and mucosal necrosis with abundant neutrophils. In many cases, there was also fibrin with small numbers of neutrophils adherent to the mucosal surfaces.
In humans, the anterior and posterior cecal arteries may form a dual blood supply for the cecum if they arise from a vascular arcade or loop and, if so, there may be some protection against vascular occlusion because blood can be supplied from both ends of the arcade. If, however, these arteries arise from the colic branch of the ileocolic artery, they may form only a single blood supply thus rendering increased susceptibility to infarction. 8 In cattle, the cecal artery is an arcade with each end fed by the anti-mesenteric ileal branch of the ileal artery. However, there are also branches off the cecal artery arising at the mesenteric border of the cecum and extending toward the antimesenteric margin. 7 If 1 or more of these branches are occluded, there is a possibility for localized CI. In nonocclusive CI in humans, it is thought that a low-pressure vascular state triggers reactive vasoconstriction in the intestinal arteries to result in underperfusion and necrosis. 9 A similar mechanism could also be hypothesized to exist in calves.
Our retrospective study focused on calves ≤30 d, but this lesion has not been seen in adult cattle in the CAHFS-Tulare laboratory. This is in contrast to humans in which the lesion is usually encountered in aged, often debilitated individuals. The common factor may be the degree of physiologic impairment in both aged humans and calves. In humans, bacterial superinfection has been suggested as a potential cofactor in the pathogenesis of isolated cecal necrosis. 9 In this scenario, the site of initial ischemia and damage to the mucosal barrier is invaded by bacteria from the cecal lumen. These bacteria, along with inflammatory cells, cause additional damage to the cecal wall, including the microvasculature to propagate the lesion. Because of the duration of the lesions and, in some cases, the degree of autolysis, it was difficult to determine whether the cecal infarcts in these calves were the result of occlusive or nonocclusive disease. We found that approximately a third of the cases exhibited some degree of thrombosis histologically and this may have been the cause of the infarction. Alternatively, the infarction occurred first and the damage to tissue and blood vessels promoted thrombosis. Coagulopathy is common in acute sepsis. 6 In the series reported here we did not find a higher rate of bacteremia in the calves with CI compared to the controls, suggesting that thrombosis is not the inciting cause for the lesion. Often the cases did not have a good history with regard to the clinical course of disease and therefore we were not able to investigate the possibility that the animals were suffering from low vascular flow as a possible cause of the lesions. The possibility of an alternative anatomic vascularization being a predisposing factor for CI in calves, as has been suggested in humans, cannot be ruled out, but we were unable to find any anatomic description of cecal vascularization that differed from the one referenced here.
Footnotes
Authors’ contributions
JM Adaska contributed to conception and design of the study; contributed to acquisition and interpretation of data; drafted the manuscript; and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. RB Moeller and PC Blanchard contributed to acquisition of data. SS Aly contributed to analysis and interpretation of data. All authors critically revised the manuscript and gave final approval.
a.
Escherichia coli antigen test kit, K99 Pilitest; VMRD, Pullman, WA.
b.
Rotaclone, Rotavirus ELISA; Meridian Bioscience, Cincinnati, OH.
Declaration of conflicting interests
The author(s) declared that they have no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Support for the research, authorship and publication of this article was provided by the California Animal Health and Food Safety Laboratory System, School of Veterinary Medicine, University of California, Davis.
