Abstract
Saksenaea erythrospora is a filamentous fungus belonging to the order Mucorales. Cases of cutaneous mucormycosis caused by Saksenaea spp. have previously been reported in immunocompetent and immunosuppressed people. A premature, 1-day-old bull calf from Texas with numerous plaque-like and ulcerative lesions in the skin was found at necropsy to have multiple areas of mycotic dermatitis and abomasitis. Fungal culture of the skin followed by morphological characterization and genetic analysis identified the etiologic agent as S. erythrospora.
The genus Saksenaea is a member of the subphylum Mucoromycotina, order Mucorales. Historically, this genus contained the single species Saksenaea vasiformis, a filamentous fungus characterized by flask-shaped sporangia with oval sporangiospores. Following the original isolation of S. vasiformis from soil in India, this organism was identified as a causative agent of human cutaneous zygomycosis.3,12,15 There are 2 previous reports of veterinary cases of S. vasiformis infection. In one report, a cow in Australia developed weight loss, ptosis of the left eyelid, drooping of the left ear, and difficulty swallowing as a result of infection of the left cranium and dura mater with S. vasiformis. 5 In 2002, 2 cases of infection of marine mammals in Texas were documented. 13 In 2010, 2 new species of Saksenaea were proposed. 1 The case described herein yielded 1 of the 2 isolates of Saksenaea erythrospora used in the formal proposal of the species. 1
A 1-day-old, mixed-breed bull calf, estimated to be 1 month premature, was presented to the Food Animal Emergency Service at the Texas A&M University Veterinary Medical Teaching Hospital (College Station, TX) because of weakness, inability to ambulate, and the presence of multiple, circular, plaque-like skin lesions on the neck and front legs. The calf weighed 11 kg on presentation and had a body temperature of 34.1°C. Supportive care including intravenous fluids, and oral colostrum was provided. The following morning, the calf became increasingly weaker and more lethargic and had a minimal suckle reflex. The body temperature continued to decrease, and the patient died during the second day of hospitalization.
On postmortem examination, large, firm, raised plaques were observed in the haired skin over the lateral aspect of the neck and shoulders (Fig. 1A). The lateral aspects of the rear limbs had multiple, slightly raised, alopecic and ulcerative areas that were occasionally covered with a dried crust or serosanguineous exudate (Fig. 1B). The mucosa of the abomasum contained multifocal to coalescing, irregular, slightly raised, 0.5–2.0 cm red areas with a thin white rim (Fig. 1C).
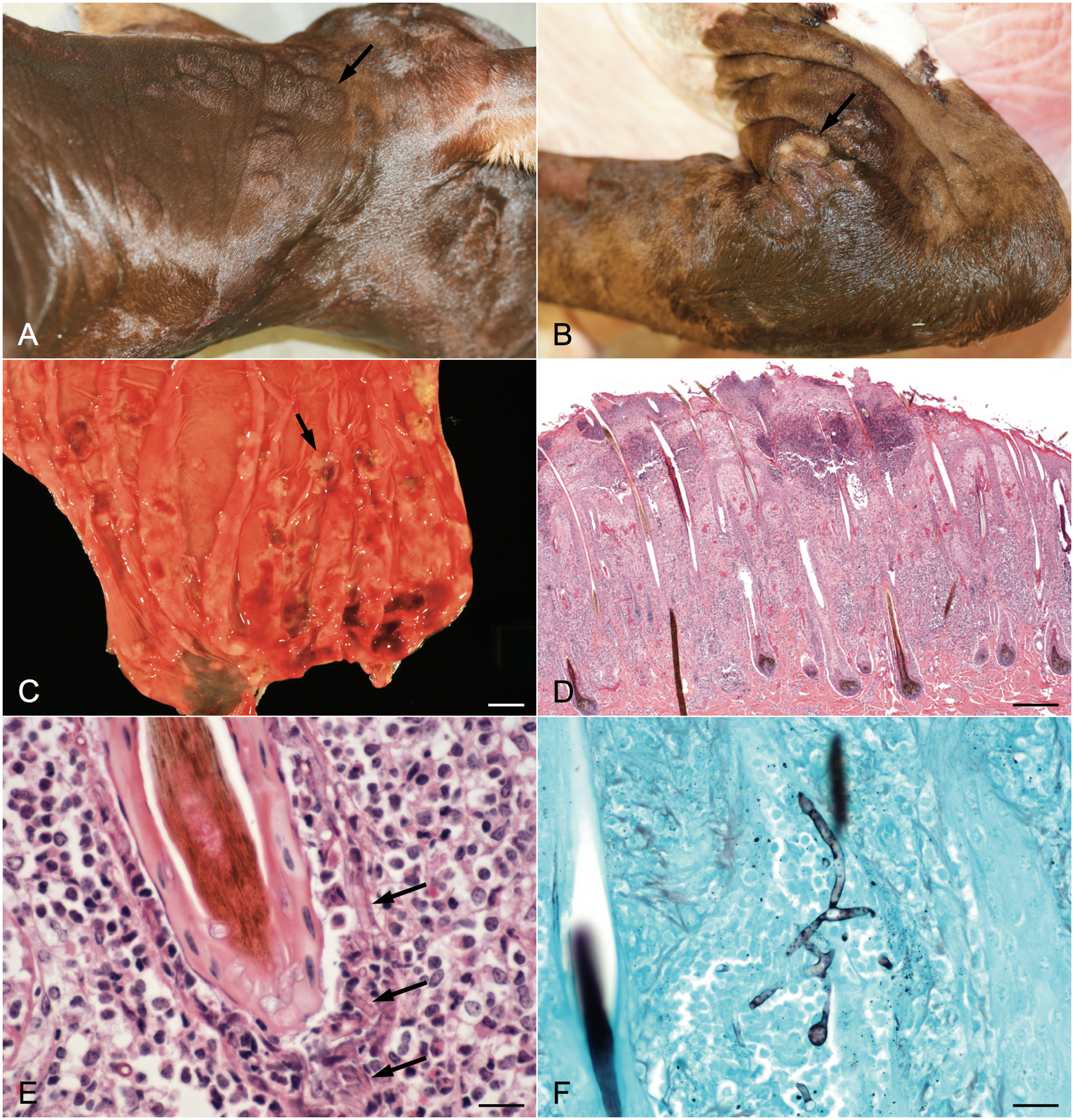
Histologically, the cutaneous plaques consisted of locally extensive areas of severe neutrophilic and necroulcerative dermatitis with multifocal areas of folliculitis and furunculosis (Fig. 1D). Numerous, 5–9 µm in diameter, nonseptate, basophilic fungal hyphae with parallel walls and dichotomous to irregular branching infiltrated and replaced hair shafts and the follicular infundibulum (Fig. 1E). Fungal hyphae showed strong positive staining with Grocott methenamine silver stain (Fig. 1F). Occasionally, there were multifocal epidermal and subcorneal pustules containing low numbers of fungal hyphae. Vasculitis was observed primarily in the superficial dermal blood vessels, with scattered areas involving deep blood vessels. Additional findings included dermal hemorrhage and fibrin, multifocal areas of orthokeratotic hyperkeratosis, and scattered superficial coccoid bacterial colonies.
In the abomasum, there were multifocal areas of neutrophilic inflammation throughout the mucosa and submucosa, with scattered areas of necrosis within the gastric glands. A severe necrotizing vasculitis with vascular thrombosis, marked edema, and fibrin deposition was observed mainly in the submucosa, with fewer areas of vasculitis and inflammation in the muscularis and rare areas in the serosa. Fungal hyphae, similar to those in the skin, were within the affected mucosa, submucosa, and the lumen and wall of blood vessels. The ulcerated abomasal mucosa was covered with a thick layer of neutrophils and numerous colonies of bacterial cocci and coccobacilli. Similar areas of necrotizing inflammation and vasculitis were found in the small intestine. The calf also had a severe fibrinosuppurative omphalophlebitis with abundant intraluminal bacterial cocci and coccobacilli. Additional histologic findings included mild pulmonary hemorrhage and congestion with scattered intra-alveolar mixed bacterial colonies and minimal neutrophilic and eosinophilic pericholangitis.
Abomasal fluid was submitted for bacterial culture, and a piece of affected skin and a sample of abomasal fluid were submitted for fungal culture. Culture of the abomasal fluid on tryptic soy agar with 5% sheep’s blood at 35°C in air supplemented with 5% carbon dioxide yielded Escherichia coli, Streptococcus bovis, and an alpha hemolytic Streptococcus sp. that was not identified further. Culture of the abomasal fluid on Sabouraud dextrose agar a and potato dextrose agar a was negative for fungal growth. Culture of the skin yielded a uniform population of rapidly growing, white fungal colonies on both Sabouraud dextrose agar and potato dextrose agar at 25°C in room air. The colonies grew within 7 days. The fungal colonies were initially identified phenotypically as a mucoralean organism but failed to sporulate. The isolate was subcultured to a Sabouraud dextrose slant and submitted to the Fungus Testing Laboratory at the University of Texas Health Science Center (UTHSC; San Antonio, TX) where it was accessioned as UTHSC 08-3606. The isolate remained sterile on a variety of media including a water agar culture, 11 hay infusion agar, 17 and Czapek–Dox agar,1,10 all prepared in-house and used to induce fruiting in mucoralean genera that are reluctant to fruit on Sabouraud dextrose or potato dextrose agar, namely Apophysomyces and Saksenaea. Temperature studies indicated good growth at 37ºC but a lack of growth at both 45ºC and 50ºC. A 24-hr culture on Sabouraud dextrose agar was used to prepare template DNA for polymerase chain reaction (PCR) assay by adding a loopful of hyphae to 50 µl of commercial reagent b in a 0.5-ml microfuge tube. The suspension was vortexed for 60 sec then heated for 15 min at 100°C. The suspension was centrifuged at 16,000 × g for 5 min, and 3 µl of supernatant was used directly for PCR. The D1/D2 region of the large ribosomal subunit was amplified and sequenced using the NL-1 1 (5’-GCATATCAATAAGCGGAGGAAAAG-3’) and NL-4 (5’-GGTCCGTGTTTCAAGACGG-3’) primers as described. 14 Briefly, PCR assays were performed in a 50-µl volume, using 3 µl of template DNA, 5 µl of 10× PCR buffer, 5 µl of a 10-µM stock solution of each primer, 1.5 µl of 10 mM deoxyribonucleotide triphosphate, c and 5.0 U of Taq DNA polymerase. d The PCR reactions were run in a thermocycler e with a temperature profile of 2 min at 94°C followed by 35 cycles of 20 sec at 94°C, 20 sec at 60°C, and 1 min at 72°C. The 35 cycles were followed by 5 min at 72°C. An aliquot was checked on 0.7% agarose gel, stained with ethidium bromide, and the remaining reaction was cleaned with a commercial kit f and then sequenced on both strands at the UTHSC Advanced Nucleic Acids Core Facility (San Antonio, TX). The sequence was used to search GenBank using the BLASTn algorithm (http://www.ncbi.nlm.nih.gov/genbank/), and results were sorted according to % identity, with identities ≥97% considered significant. The 3 highest ranked hits all corresponded to S. vasiformis (accession no. EU644756.1 [99%], accession no. AY234872.1 [98%], and accession no. AF113483 [95%]), with the third hit considered nonsignificant, although it also corresponded to S. vasiformis.
In a subsequent polyphasic study of the genus Saksenaea, examining a diverse collection of strains (including the isolate described herein) by means of internal transcribed spacer (ITS), 28S ribosomal RNA gene D1 and D2 domains, and elongation factor 1α (EF-1α) sequence data as well as by various morphological and physiological features, S. vasiformis was shown to encompass additional species. 1 From this study, S. vasiformis was redefined as a complex, and 2 new cryptic species were identified, S. erythrospora and S. oblongispora. The strain isolated from the patient described in the current case report has been designated as the holotype for the species and has been deposited into the Centraalbureau voor Schimmelcultures under accession number CBS H-20472. 1 Antifungal susceptibility results for the S. vasiformis complex as performed using the Clinical Laboratory Standards Institute M38-A2 method 4 were reported previously as a range. 1 The susceptibility results for this isolate, reported in µg/ml, suggested in vitro resistance to amphotericin B, voriconazole, caspofungin, anidulafungin, and micafungin (4, 8, >16, >16, and >16, respectively), and potential susceptibility to posaconazole, itraconazole, and terbinafine (0.25, 0.5, and 0.03, respectively). These results are in agreement with the collective data reported for the 9 strains evaluated in the taxonomic study. 1
Fungal infection is a well-recognized cause of abortion and neonatal death in cattle. 8 The causative agents are often Aspergillus spp. and fungi in the order Mucorales. 8 Contamination of feed during winter months is often cited as a potential source of infection, and hematogenous spread from the rumen or respiratory tract to the placenta may result in necrotizing vasculitis and abortion.7,9,16 Abortions often occur late in gestation, and premature birth of an affected calf may also occur as in the case presented herein.2,7 Cutaneous lesions in the aborted fetus or neonatal calf, consisting of thick, elevated, hairless plaques around the eyes, shoulders, back, and sides, are often observed, along with occasional cases of bronchopneumonia. 8 Typically, fungi can be recovered from the placenta, stomach contents, or skin lesions.2,8,9 The most common mucoralean fungi identified include the following genera: Mucor, Absidia, Rhizopus, Rhizomucor, and Mortierella. 8 Angiotropism is common, and lesions caused by these highly invasive fungi are typically associated with vasculitis.6,8,9 In histopathologic sections, hyphae of mucoralean fungi often lack septa, unlike those of Aspergillus spp.6,9 Otherwise, there is little to distinguish them from one another, making fungal culture often necessary. 6 The lack of prior published cases of Saksenaea infection as a cause of bovine neonatal death is likely due to the difficulty of inducing these fungi to sporulate for identification. The adoption of DNA sequencing of sterile fungi will undoubtedly increase the number of cases attributed to this genus.
The present case documents isolation of the newly described S. erythrospora from a newborn calf. The cause of death was attributed to mycotic abomasitis and dermatitis along with bacterial omphalophlebitis. The severe mycotic abomasitis and dermatitis were presumed to be the result of in utero S. erythrospora infection, which likely contributed to the calf’s premature birth. In addition to other agents of mucormycosis, S. erythrospora should be considered as a possible etiologic agent of abortion and premature birth in cattle. Nucleic acid sequencing is a useful test for identification of sterile fungi and should be attempted as an adjunct to phenotypic identification in difficult cases.
Footnotes
a.
Remel Inc., Lenexa, KS.
b.
PrepMan® Ultra reagent, Applied Biosystems, Foster City, CA.
c.
Invitrogen Corp., Carlsbad, CA.
d.
TripleMaster® Taq DNA polymerase, Eppendorf North America, Hauppauge, NY.
e.
Eppendorf Master thermocycler, Eppendorf North America, Hauppauge, NY.
f.
QIAquick® PCR Purification Kit, Qiagen Inc., Valencia, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
