Abstract
Classification of body cavity effusions is an important step in the investigation and diagnosis of disease in cats. Feline inflammatory effusions are often highly proteinaceous and viscous, which can cause clumping of white cells and subsequently inaccurate nucleated cell counts (NCCs) using automated and manual methods. Microscopic assessment of cellularity can also be difficult given the variable thickness of smears and cell clumping, which skews white cell distribution. The ADVIA 120 uses 2 white cell–counting channels, the basophil/lobularity (WBC/baso) and differential/peroxidase channels, which can provide quite different results in highly viscous feline samples and often disagree with smear assessment of cellularity. We investigated the effects of pre-incubation of feline effusion samples with hyaluronidase and its effects on NCCs and cellularity assessment. NCCs were obtained by automated analysis using the ADVIA 120 and by manual counting methods. Agreement was assessed using a Bland–Altman chart. Pretreatment of samples with hyaluronidase resulted in good agreement between the ADVIA basophil channel and manual counting methods in all samples in the study. However, improvements in NCCs after hyaluronidase treatment were significantly greater in clumped samples, and cell distribution of these samples on direct smears was also improved. Therefore, when nucleated cell clumping is observed on a direct smear, pretreatment of the sample with hyaluronidase prior to analysis on an automated analyzer is advised, with the WBC/baso channel displaying the most accurate NCC.
Classification of body cavity effusions is an important step in the investigation and diagnosis of disease because it can give an indication of the underlying mechanism causing the abnormal fluid accumulation. 8 Classification is achieved by measuring the protein concentration and the nucleated cell count (NCC) of the effusion. NCCs are normally measured by using an automated hematology analyzer or manually by hemocytometer. However, obtaining accurate NCCs from feline effusions is often problematic given their high viscosity and the tendency for white cell clumping, and previous studies have indicated only moderate correlation between automated and manual methods when analyzing fluid samples from this species. 6
Manual cell counting using a hemocytometer is the traditional method for obtaining fluid NCC. However, this method is time consuming and possesses inherent error. 11 Clumping of cells can also contribute to inaccurate cell counts using this method. 7 In contrast, automated hematology analyzers provide rapid results that are less labor intensive to obtain compared with traditional cell counting methods. Such analyzers also provide more accurate NCCs provided no nucleated cell clumping is present. 11 The ADVIA 120 a hematology analyzer measures white cells by 2 methods: the basophil/lobularity (WBC/baso) and the differential/peroxidase (WBC/perox) channel. The WBC/baso channel provides a total NCC; the WBC/perox channel provides both a differential NCC and a total NCC. Large discrepancies are often found between the 2 channels when analyzing feline effusions, and this has also been reported when analyzing feline effusions using the Sysmex XT-2000i. 5 In our experience, examination of a direct smear of an effusion can indicate that neither NCC is accurate; however, microscopic assessment of the NCC from the direct smear is complicated by the thick nature of these smears as well as by cell clumping, which skews the white cell distribution.
Problems with obtaining accurate NCCs have been encountered with other fluids that are of high viscosity. In synovial fluids, this has been overcome by preincubation of samples with hyaluronidase prior to analysis by automated analyzers.3,9 This technique has also been demonstrated to have utility in forensic medicine when analyzing vitreous humor. 4 The viscous nature of synovial fluid is caused by high levels of hyaluronic acid. 10 Incubation with hyaluronidase causes the hydrolysis of hyaluronan, which decreases the viscosity of the sample. Hyaluronic acid has been shown to be increased in induced cases of peritonitis in rabbits, 2 and it is possible that this mechanism may contribute to the high viscosity of feline inflammatory body cavity effusions. We therefore hypothesized that pre-incubation of feline body cavity effusions with bovine testicular hyaluronidase will decrease the viscosity of the samples, allowing more accurate NCC results by automated hematology analyzers and better distribution of cells on direct smears making assessment of cellularity easier.
To test this hypothesis, feline body cavity effusions submitted to the clinical pathology laboratory of the University of Glasgow (Glasgow, UK) were analyzed both with and without preincubation with hyaluronidase. The effusions were submitted to the laboratory in EDTA tubes and were analyzed on the day of arrival. Direct smears of the untreated samples were made and stained using the May-Grünwald/Giemsa method. These smears were examined to determine cell distribution and the presence or absence of cell clumping.
Automated NCCs were then obtained by using the ADVIA120, and manual NCCs were obtained by using a Neubauer counting chamber. To perform the manual NCC, 20 µL of sample were added to 380 µL of 1% acetic acid in order to lyse erythrocytes (methyl violet was added to stain nuclei). The sample was rotated gently for 2–3 min and then transferred to the Neubauer counting chamber using a capillary tube. The counting chamber was left in a moist chamber for 10 min to let cells settle before cell counting using a light microscope. 1
After initial analysis of the untreated sample, a 250-µL aliquot of sample was added to 250 µL of hyaluronidase (150 U/mL) and incubated at 37°C for 10 min. The hyaluronidase solution of 150 U/mL was prepared by dissolving 10 mg of bovine testicular hyaluronidase b solution (439 U/mg) in 30 mL of saline buffer (0.9 g/L). After 10 min of incubation with hyaluronidase, the treated sample was run through the ADVIA 120 to obtain automated NCCs. Manual NCCs on the treated sample were performed as described above. Results obtained were then corrected for the dilution caused by hyaluronidase added to the sample. A direct smear was also made immediately after incubation to assess white blood cell distribution and presence of clumping. Samples were thoroughly mixed in all instances before performing any procedures.
To demonstrate the action of hyaluronidase and confirm that any changes in the results obtained were not caused solely by dilution, an equal amount of normal saline was added to an aliquot of untreated sample. The mixture was incubated at 37°C for 10 min and NCCs obtained using the ADVIA120 and manually with the hemocytometer. Fluid protein measurements on untreated samples were obtained using the Biuret method. c
Results from the ADVIA 120 and manual counts were compared using Pearson correlation (r) and Bland–Altman plots. Fluid protein levels in clumped and nonclumped samples were compared using Student t-test. The manual NCC obtained from hyaluronidase-treated samples was considered the benchmark result because the presence or absence of clumping could be ascertained at the time of analysis. Statistical significance was established at (p < 0.05). Data were analyzed using commercial software. d
Repeatability of NCC from the ADVIA120 in untreated samples was assessed on 2 samples with no obvious nucleated cell clumping and 1 sample with clumping observed by collecting 5 consecutive measurements of each on the same day and then calculating the coefficient of variation (CV) with the following formula: CV = standard deviation (SD)/mean × 100%. Precision of the analyzer was found to be adequate when analyzing nonclumping samples with WBC/baso channel CVs <5%. However, the CVs from the sample in which clumping was present were much greater (Table 1).
Precision data for nucleated cell counts (NCCs) from untreated feline effusions using the ADVIA 120 basophil/lobularity (WBC/baso) and differential/peroxidase (WBC/perox) channels.*
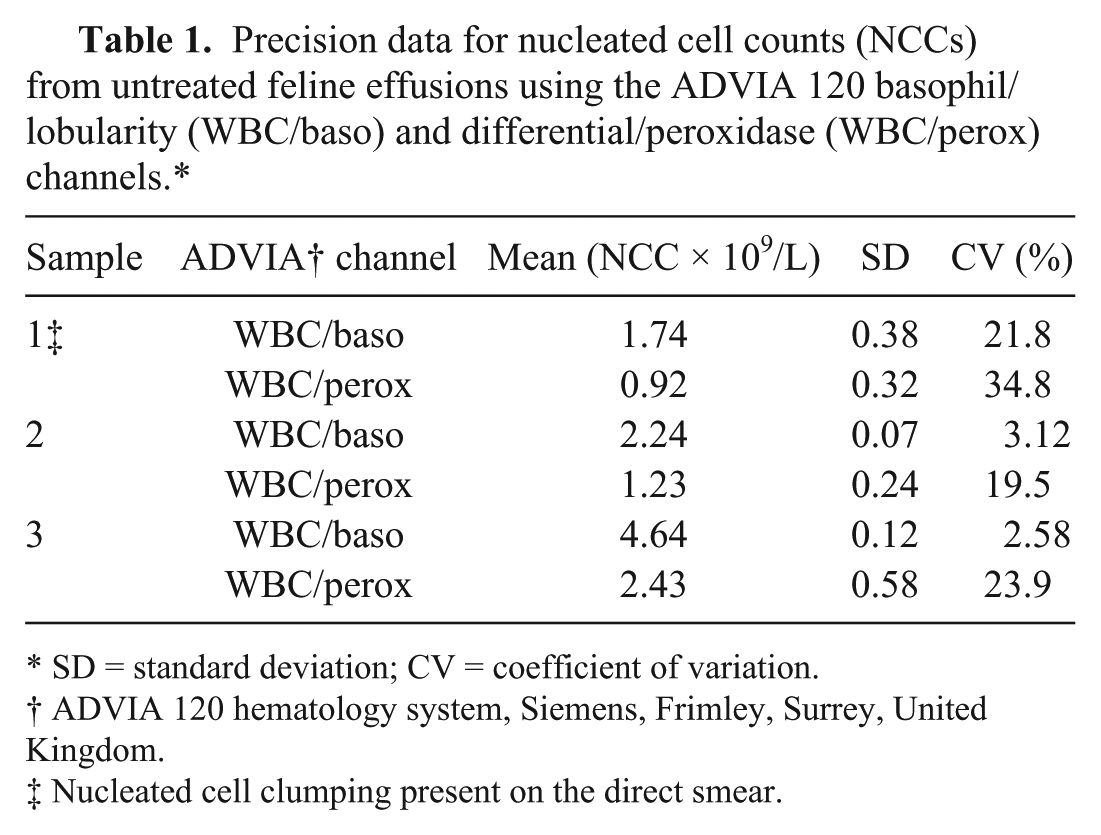
SD = standard deviation; CV = coefficient of variation.
ADVIA 120 hematology system, Siemens, Frimley, Surrey, United Kingdom.
Nucleated cell clumping present on the direct smear.
In total, 25 feline body cavity effusions were submitted to the diagnostic service during the time of the study (13 peritoneal, 12 pleural), and counts were obtained as noted above (Table 2). Smears made from untreated samples were assessed for white cell clumping, and this phenomenon was noted in 13 of 25 samples (9 peritoneal, 4 pleural; Table 2). Smears prepared from hyaluronidase-treated samples showed improved white cell distribution with no cell clumping. This allowed easier assessment of cellularity (taking into account the dilution factor) by microscopy.
Total nucleated cell counts and fluid protein measurements using manual cell counting and the ADVIA 120 basophil/lobularity (WBC/baso) and differential/peroxidase (WBC/perox) channels.*
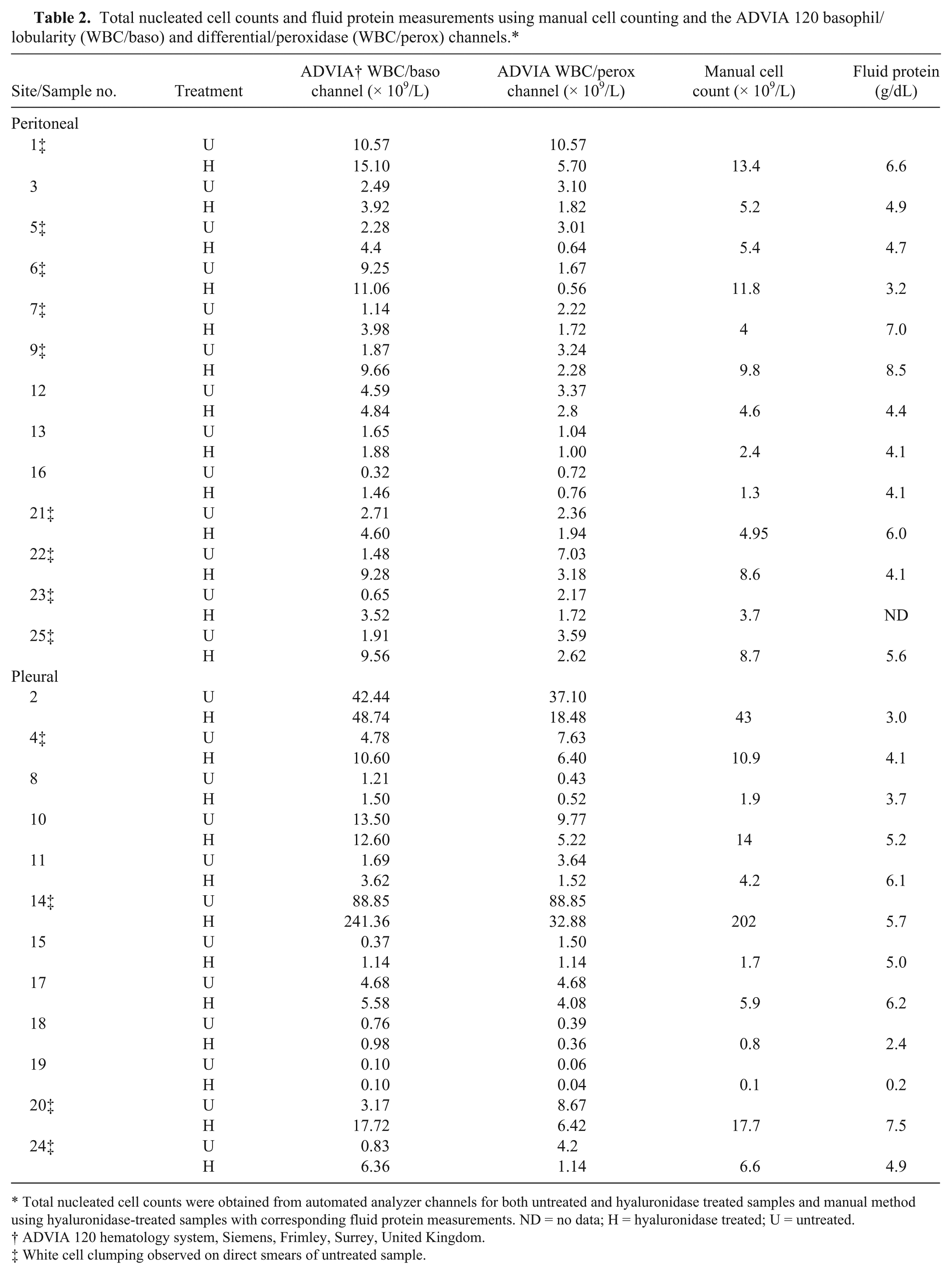
Total nucleated cell counts were obtained from automated analyzer channels for both untreated and hyaluronidase treated samples and manual method using hyaluronidase-treated samples with corresponding fluid protein measurements. ND = no data; H = hyaluronidase treated; U = untreated.
ADVIA 120 hematology system, Siemens, Frimley, Surrey, United Kingdom.
White cell clumping observed on direct smears of untreated sample.
NCCs from untreated samples obtained from both the WBC/baso channel and WBC/perox channel of the ADVIA 120 showed moderate correlation with NCCs obtained from treated samples counted by the manual method (r = 0.68 and r = 0.84, respectively). These results are similar to a previous study of body cavity effusions from different animal species, which found only moderate correlation (r = 0.73) between cells counts obtained from the ADVIA 120 WBC/baso channel and the standard hemocytometer method in feline peritoneal effusions. 6 The Bland–Altman agreement charts showed that both the WBC/baso and WBC/perox channels underestimated the NCCs, and this error was greater in samples where cell clumping was observed. Furthermore, the presence of cell clumping was more common in samples with higher cell counts (Fig. 1A, 1B).
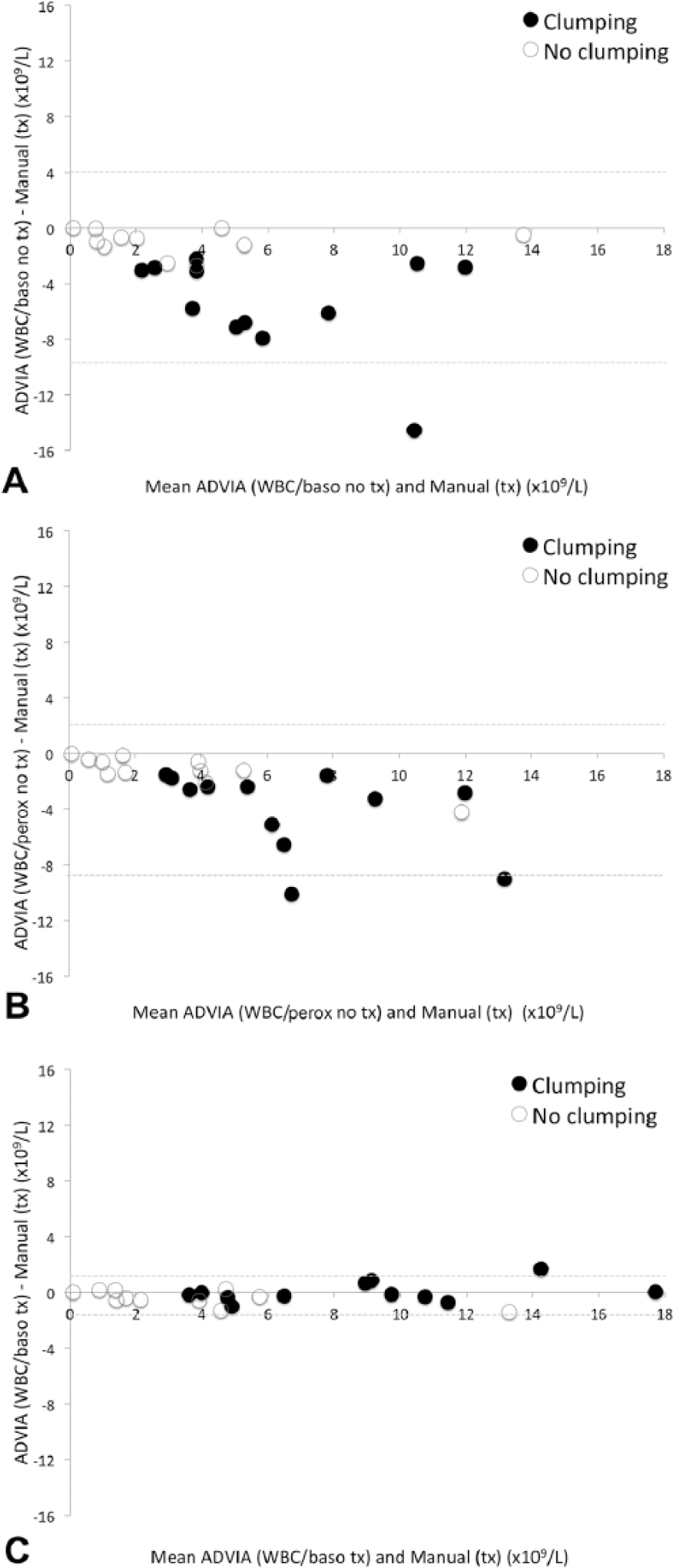
Bland–Altman plots of nucleated cell counts (NCCs) obtained from feline body cavity effusions using the hemocytometer and the ADVIA 120 basophil/lobularity (WBC/baso) and differential/peroxidase (WBC/perox) channels.
After hyaluronidase treatment, greater agreement between the WBC/baso channel and the standard hemocytometer method was shown in both clumping and nonclumping samples treated with hyaluronidase (Fig. 1C); however, the magnitude of improvement was significantly greater in the clumped samples (Student t-test, p < 0.01). The most reliable NCC obtained from the automated analyzer in the treated samples was obtained from the WBC/baso channel given that results showed good correlation with the manual cells counts (Pearson correlation coefficient of r = 0.99). As greater agreement was seen in both clumping and nonclumping samples, it could be argued that treating all feline effusions with hyaluronidase prior to analysis would be beneficial. However, as the cellularity of samples with no clumping tended to be low and the increase in NCC post-treatment small, hyaluronidase incubation of samples with no clumping is unlikely to lead to significant changes in the classification or interpretation of results. Clumped samples had statistically significant greater fluid protein levels than nonclumped samples (Student t-test, p = 0.024); however, fluid protein was not predictive of the need for enzyme treatment because there was a large overlap between the groups, as many non-clumped samples had high fluid protein (Table 2). Enzymatic treatment should therefore be limited to samples where clumping is noted on examination of a direct smear of the effusion.
In contrast to the WBC/baso channel, the NCCs from the WBC/perox channel decreased after treatment, and this resulted in the WBC/perox channel counts being markedly low posttreatment compared with results from the WBC/baso channel, the manual cell count, and visual assessment on a direct smear (Table 2). The reason for the decrease in the WBC/perox channel NCCs posttreatment is unclear, particularly as smear assessment demonstrated that white cell clumping in the samples had resolved after treatment with hyaluronidase. Analysis of the WBC/perox scatter plot shows more events in the “noise” region in treated samples; we hypothesize that enzyme treatment causes increased fragility of the cells, which then disintegrate in the WBC/perox channel.
The results of our study are in contrast to previous recommendations 5 that demonstrated high diagnostic accuracy of the total NCC (ΔTNC, the ratio of the WBC/perox and WBC/baso counts) for diagnosis of feline infectious peritonitis in feline effusions. The previous study hypothesized that this disparity in NCC between the WBC/perox and WBC/baso channels is a result of the acidic WBC/baso reagent causing precipitation of proteins with subsequent entrapment of nucleated cells and low NCCs. Consequently, they advise use of the WBC/perox channel for NCCs in feline effusions. However, the samples in that previous study were not pretreated with hyaluronidase. We demonstrate (Fig. 1B) that, although the WBC/perox NCC is likely to be more accurate in untreated samples, if cell clumping is present then the NCC values are likely to be underestimated by automated analyzers.
Dilution of the samples with an equal volume of saline did not improve the accuracy of the analyzer cell counts (data not shown). This observation along with the success of the hyaluronidase would support the hypothesis that substances such as hyaluronan contribute to the highly viscous nature of some of these effusions.
Pretreatment with hyaluronidase improved the NCCs obtained from the WBC/baso channel in all samples in the study. Greatest improvement was seen in samples where nucleated cell clumping was observed on a direct smear, whereas samples with no clumping observed had only small improvements in NCC. Fluid protein was significantly higher in clumped samples but was not a good predictor for the requirement of enzyme treatment; therefore, the decision for enzyme treatment should be based on the presence of nucleated cell clumping. The WBC/perox channel NCCs were consistently decreased by treatment; therefore, this channel was not reliable for determining accurate NCC after enzyme treatment. Pretreatment with hyaluronidase produced more even distribution of white cells on a direct smear allowing easier assessment of cellularity by microscopy.
Footnotes
Acknowledgements
We thank Ronnie Barron, laboratory manager at Veterinary Diagnostic Services, for useful discussion regarding techniques and during the writing of the manuscript.
Authors’ contributions
SM Lee contributed to design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. K Williamson contributed to conception of the study; contributed to acquisition and analysis of data; and critically revised the manuscript. W Weir contributed to design of the study; contributed to analysis and interpretation of data; drafted the manuscript; and critically revised the manuscript. L Hulme-Moir contributed to conception and design of the study; contributed to interpretation of data; drafted the manuscript; and critically revised the manuscript. H Haining contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
ADVIA 120 hematology system, Siemens, Frimley, Surrey, United Kingdom.
b.
Bovine testicular hyaluronidase, Sigma-Aldrich, Irvine, Ayrshire, United Kingdom.
c.
Olympus AU640, Beckman Coulter, High Wycombe, Buckinghamshire, United Kingdom.
d.
Excel 2010, Microsoft, Redmond, WA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the University of Glasgow Veterinary School student summer vacation scholarship program.
