Abstract
To investigate the ability of the 35 kDa low-molecular-weight hyaluronan fragment (HA35) to relieve neuropathic and inflammatory pain, including postherpetic neuralgia and shoulder, neck, back and temporomandibular pain. Ten patients with postherpetic neuralgia and 26 patients with shoulder, neck, back or temporomandibular pain were studied and assessed. The 35 kDa hyaluronan fragment (HA35) was prepared by mixing hyaluronidase and 100 mg of high-molecular-weight HA at room temperature for 20 mins. This mixture was locally injected once at the pain point or where the nerve trunk innervated the pain point. Patients scored their pain and comfort on the numerical pain rating scale (NPRS) and the General Comfort Questionnaire (GCQ). After treatment, the NPRS scores and GCQ scores improved. Patients with postherpetic neuralgia and shoulder, neck, back or temporomandibular pain had significantly lower NPRS scores at 30 min to 180 min after injection, especially at 180 min (p < .001). The GCQ scores after treatment for 24 h was significantly greater than that before treatment (p < .01). No adverse reactions occurred. The 35 kDa hyaluronan fragment (HA35) effectively relieved postherpetic neuralgia-induced neuropathic pain and shoulder, neck, back and temporomandibular inflammatory pain.
Keywords
Introduction
Pain can be divided into neuropathic pain and inflammatory pain. 1 Postherpetic neuralgia (PHN) is a typical form of neuropathic pain and is the most common complication of herpes zoster infection. 2 PHN mainly refers to pain that persists 4 weeks or more after clinical healing of the herpes zoster rash area, with an incidence of approximately 9% to 34%. 3 PHN is accompanied by severe pain, which often affects sleep and quality of life 3 and can be difficult to treat clinically. Inflammatory pain occurs mainly in the shoulder, neck, back and temporomandibular joints and is a common condition worldwide,2,4,5 ranking third in overall incidence after tumors and cancers. Regardless of whether the pain is acute or chronic and whether it is inflammatory pain or neuropathic pain, the underlying cause is inflammation or the inflammatory response. 6 There are common points in the treatment of these two kinds of pain 7 : inhibition or suppression of the production of appropriate inflammatory mediators and of neuronal afferent and efferent (motor) transmission. 6 Although currently available pharmacotherapeutics provide symptomatic relief while also affecting the underlying pathophysiology, they usually have various dose-limiting side effects.8,9 Accordingly, a complementary and alternative therapy that works well and has no adverse effects is urgently needed.
Hyaluronan (HA) has been applied for many years as a pharmacological therapy for articular pain relief in clinical practice. 10 The African naked mole rat, whose tissues contain up to 6% HA, is insensitive to acid-stimulated pain. 11 High-molecular-weight (HMW)-HA and its fragments increase the HA content of mouse tissues by gene transfer 12 and alleviate pain through the TRPV1 channel.13,14 The accumulation of HA and its fragments in tissues alleviates the activation of the immune response both in vitro and in vivo, reducing inflammation and the pain caused by the inflammatory response. Topical coapplication of HA with transdermal drug delivery enhancers can attenuate inflammatory and neuropathic pain. 15 HMWHA has poor tissue permeability and skin penetration. In contrast, low-molecular-weight HA (LMW)-HA demonstrated greater tissue permeation. Research by Park showed that LMWHA was faster than HMWHA in a permeation assay and yielded 100-fold greater permeation amounts than those obtained for HMWHA in the first 15 min. 16 A 35 kDa bioactive HA fragment (HA35) was previously isolated from human colostrum.17–19 Specifically sized HA with an average molecular weight of 35 kDa (HA35) isolated from human milk dampens inflammatory responses in murine models of inflammation, demonstrating anti-inflammatory activity similar to that of HA.18,20–23 Our previous in vitro studies revealed that HA35 and HMWHA (1600 kDa) exhibit a high degree of similarity in modulating the cellular activities of immune cells. 24 Therefore, HA35 might be more valuable for medicinal applications than HMWHA (1600 kDa).
In this study, hyaluronidase for injection extracted from bovine testis and sodium hyaluronate for injection were mixed for 20 mins and cleaved into the narrowly distributed HA35 at room temperature. 25 This method mimics the protective effects of HA35 isolated from human colostrum.26,27 Therefore, we used an abdominal fat subcutaneous injection method to examine the analgesic effect of HA35 on human safety and health in patients with PHN and shoulder, neck, back, and temporomandibular pain. This was a single-arm, open-label clinical study in which patients were evaluated before and after treatment.
Methods
Study design and patient groups
This study was approved by a formally constituted review board (Ethics Committee of the Affiliated Hospital of Qingdao University) and registered on ClinicalTris.gov (identifier: NCT05809700). Written informed consent was obtained from all 36 volunteers per the guidelines of the Declaration of Helsinki, as most recently amended in 2013. Patients were enrolled in the study between June 2023 and July 2023.
In this study, 36 patients with PHN and shoulder, neck, back, or temporomandibular pain were studied at the Department of Pain Diagnosis and Treatment of the Affiliated Hospital of Qingdao University. The included patients were aged 18 to 60 years. The clinical diagnosis was in line with the ‘clinical dermatology’ PHN standard and was accompanied by clear herpes zoster pain. The clinical diagnoses included long-term muscle strain; aseptic inflammation caused by shoulder, neck, back or temporomandibular pain; and/or neuropathic pain. The course of PHN ranged from 0–2 months, and the course of shoulder, neck, back and temporomandibular pain ranged from 0 to 8 months. Patients had to be able to make independent judgments and score their pain. Patients who were pregnant and had hepatic or renal failure, neuromuscular disorders, or coagulation disorders were excluded. Other exclusion criteria included immunocompromise, an inability to cooperate with the researchers in this study for various reasons, and participation in other simultaneous clinical or drug trials. No patients who met the exclusion criteria were included according to the above criteria (Figure 1). Flowchart of this study.
A safety study of HA35 showed that a daily dose of 140 mg is safe for healthy individuals. 18 In this clinical study, pain physicians mixed 2000 units of hyaluronidase extracted from bovine testis (SPH No. 1 Biochemical & Pharmaceutical Co., Ltd., H31022111) and 100 mg of HMWHA (Shanghai Haohai Biological Technology Co., Ltd., H20174089) at room temperature for 20 mins in advance. Then, patients with a body weight less than 75 kg were locally injected with 100 mg once at the pain point or where the nerve trunk innervated the pain point, and the pain indicators at different time points before and after treatment were recorded. The patients used the numerical pain rating scale (NPRS) to self-evaluate their pain, and the degree of pain relief was recorded by doctors before and after treatment (described in the ‘Primary outcome measures’ section).28–31 The general comfort questionnaire (GCQ) was used to assess patient comfort (described in the ‘Secondary outcome measures’ section). 32
Primary outcome measures
The primary outcome was the NPRS scores at various treatment time points (30 min, 60 min, 90 min, 120 min, 150 min, and 180 min). The NPRS is a tool that doctors use to help assess patients’ pain. Each patient rated their pain on a scale of 0 to 10, where a score of 0 indicated “no pain” and 10 indicated “the most painful”. More specifically, 0 points indicated no pain, 1–3 points was interpreted as slight and bearable pain, 4–6 points was interpreted as pain that affects sleep but was still tolerable, and 7–10 points meant intense and unbearable pain that affected sleep or other activities.
Secondary outcome measures
The secondary outcome was the GCQ scores 24 h after treatment. The GCQ is a self-rating scale consisting of 30 items in four dimensions: physical, psychological, sociocultural and environmental. Each item is given a score from 1–4, where 1 means “strongly disagree”, 2 means “disagree”, 3 means “agree”, and 4 means “strongly agree”. A total score of less than 60 points indicated low comfort, 60–90 points indicated moderate comfort, and more than 90 points indicated high comfort. 33
Safety was evaluated in a timely manner during the treatment course and at the 24-h follow-up by recording adverse events, including nausea, vomiting, palpitations, dizziness, headache, and long-lasting insomnia.
Statistical analysis
We ran the statistical analyses with SPSS v.24 (IBM Corp., Armonk, NY, USA) in the present study. Continuous data were evaluated for normality and homogeneity and compared between groups using Student’s t test. Normally distributed data are expressed as the mean (SD) and were analyzed by one-way analysis of variance (ANOVA). p < .05 was considered to indicate statistical significance.
Results
Methods for HA35 production and detection
The hyaluronidase extracted from bovine testes and recombinant human hyaluronidase PH20 cleaved macromolecular HA into HA35 after incubation at room temperature for 10–60 mins (Figure 2(A)). The electrophoresis results for HA35 prepared at room temperature for 20 mins are shown in Figure 2(B), lane 7. Molecular weight of the hyaluronidase after enzymatic digestion of the raw HA material at different times. Notes: (A): lane 1: 10 min; lane 2: 20 min; lane 3: 30 min; lane 4: 40 min; lane 5: 50 min; lane 6: 60 min. M1 and M2: the markers for 10 kDa and 50 kDa molecular weight, respectively. (B): Lane 7: 60 min parallel group. The samples were electrophoresed for 1 h.
Patient clinical characteristics
Basic characteristics of participants with neuropathic pain.

Basic characteristics of participants with inflammatory pain.

Analysis of the NPRS scores and improvement in neuropathic pain
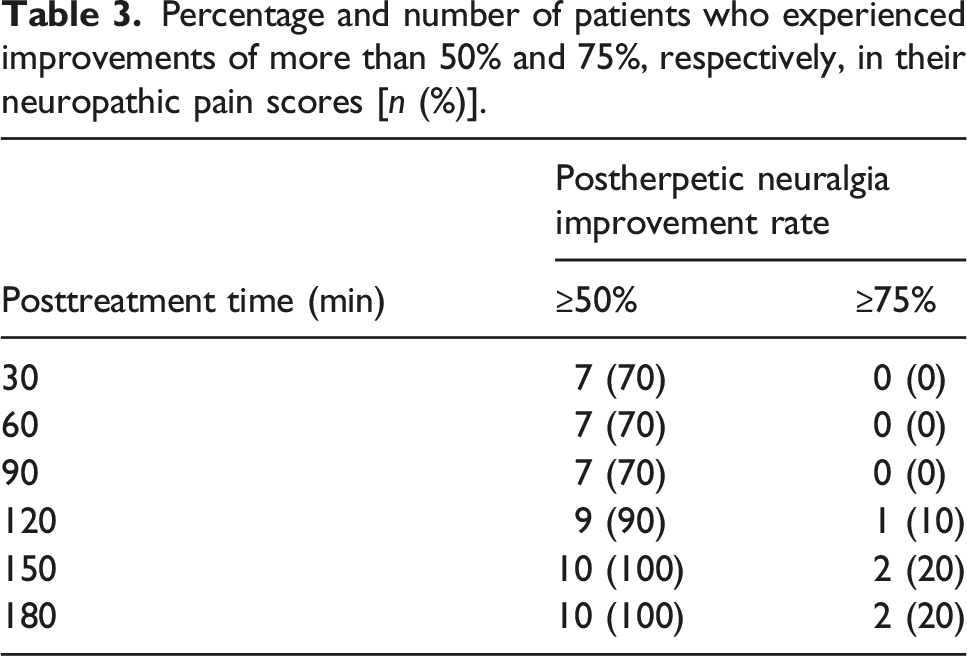
After a local multipoint injection of 100 mg of freshly manufactured HA35 into the pain point area and the nerve trunk innervating the pain point area of the PHN patients, the patients immediately experienced numbness at the pain point, and after 30 mins to 180 mins, the pain scores of all the PHN patients decreased (Figure 3(A)). The patients had a significantly greater reduction in NPRS scores after treatment for 180 mins (p < .001) than after treatment for 30 mins (p < .05). The scores for PHN improvement are shown in Figure 3(B). The average scores increased from 3.3 to 4.9 between 30 mins and 180 mins (p < 0.01). The number of patients with 50% and 75% or greater improvement in PHN scores between 30 mins and 180 mins after treatment are indicated in Table 3. The number and percentage of patients with 50% and 75% or greater improvement in PHN scores increased from 7 (70%) to 10 (100%) and 0 (0%) to 2 (20%), respectively. This finding suggested the success of the treatment administered in this clinical study. NPRS scores and improvement after neuropathic pain treatment. Notes: (A): displays the NPRS scores for patients with PHN before and after treatment with HA35 from 0 to 180 mins. (B): illustrates the degree of improvement in pain scores after treatment from 30 to 180 mins. Percentage and number of patients who experienced improvements of more than 50% and 75%, respectively, in their neuropathic pain scores [n (%)].
Analysis of the NPRS scores and improvement in inflammatory pain
Similarly, 30 mins to 180 mins after the local injection of 100 mg of freshly manufactured HA35 into the pain point areas of patients with shoulder, neck, back or temporomandibular pain, the NPRS scores decreased (Figure 4(A)). As shown in Figure 4(A), the average scores of the shoulder, neck, and back or temporomandibular pain patients increased from 3.6 to 4.5 at 30 mins to 180 mins after treatment (p < 0.01). The shoulder, neck, back or temporomandibular pain scores of patients who achieved at least 50% and at least 75% improvement increased from 23 (88%) to 26 (100%) and from 1 (3%) to 9 (34%), respectively (Table 4 and Figure 4(B)). Improvement in the NPRS scores after inflammatory pain treatment. Notes: (A): displays the NPRS scores for patients with shoulder, neck, back, and temporomandibular pain before and after treatment with HA35 from 0 to 180 mins. (B): illustrates the degree of improvement in pain scores after treatment from 30 to 180 mins. Percentage and number of patients who experienced improvement of more than 50% and 75%, respectively, in inflammation pain scores [n (%)].

Impact of HA injection on general comfort levels
The GCQs for patients with PHN or shoulder, neck, back or temporomandibular pain before and after treatment are shown in Figure 5. When the average satisfaction scores at 0 and 24 h after surgery were compared, statistically significant differences were found in terms of comfort. The average GCQ score of patients with PHN before treatment was 57, and the average score 24 h after treatment was 77 (p < .01). Comfort changed from low comfort to moderate comfort. The average GCQ score of patients with shoulder, neck, back and temporomandibular pain before treatment was 64, and the average score 24 h after treatment was 79 (p < .001). After 24 h of treatment and follow-up, there were no treatment-related adverse events in this study, including nausea, vomiting, palpitations, dizziness, headache, or long-lasting insomnia. This finding showed that the treatment not only relieves pain but also improves patient comfort. Comparison of GCQ scores after treatment for neuropathic and inflammatory pain.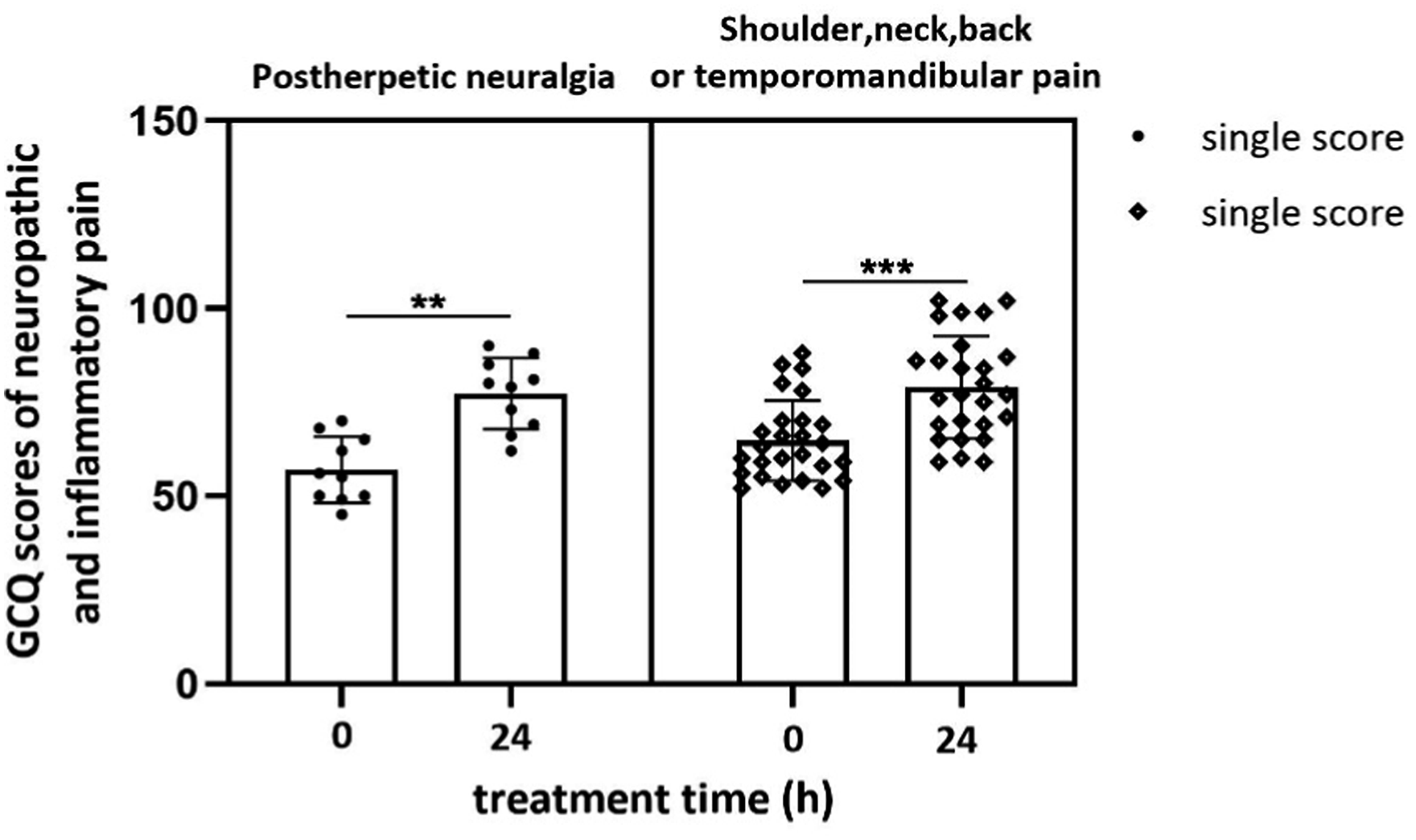
Discussion
Pain is often a symptom of a disease. The pain process usually occurs due to peripheral tissue or nerve injury, disease, or lesions. 34 Pain can be divided into three types based on its pathophysiology: nociceptive pain, inflammatory pain and neuropathic pain. 1 Inflammatory pain and neuropathic pain can occur alone or in combination (mixed pain). 35 PHN is a typical type of neuropathic pain. Shoulder, neck, back or temporomandibular pain generally manifests as a mixture of inflammatory pain and neuropathic pain. For example, back myofascial pain is a typical type of inflammatory pain, and neuropathic pain occurs when myofascial inflammation compresses nerve fibers. 36 However, irrespective of the type of pain, the cause of all the pain is inflammation and the inflammatory response. 6 Treatment methods, including corticosteroids, qutenza, lidocaine and inflammatory mediator blockers or surgical intervention, where appropriate, have inconsistent long-term effects and safety.6,37–39
A new study published in the journal Nature 12 showed that overexpression of the naked mole rat gene nmrHAS2 in transgenic mice significantly increased hyaluronic acid levels in multiple tissues, significantly reduced the risk of aging and DMBA/TPA-induced cancer, and significantly prolonged the average life span and maximum life span of the mice. 12 Zhao et al. assessed 541 patients in a meta-analysis and reported that the NPRS scores of patients in the hyaluronic acid treatment group at 1, 2, 3, and 4 weeks were better than those of patients in the control group (p < .05). 40 These results indicated that HMWHA can reduce neuropathic pain in the rotator cuff and that the short-term analgesic effect is good. Several studies have also shown that topical coapplication of HA with transdermal drug delivery enhancers can attenuate inflammatory and neuropathic pain. 15 Similarly, in the present study, LMWHA reduced the NPRS scores of neuropathic and inflammatory patients within 30 mins, with a shorter analgesic time (Figures 3A and 4(A)). On the one hand, the results suggest that HA35 has an anti-inflammatory function. 25 This finding is consistent with the finding that HA35 reduces hepatic triglyceride levels and the expression of hepatic proinflammatory cytokines and chemokine mRNAs. 41 On the other hand, the rapid analgesic effect of HA35 may be related to its permeability, which is predominantly related to its molecular weight. For example, research in the International Journal of Pharmaceutics showed that HMWHA (>600 kDa) has very limited permeability through the skin and primarily stays on the skin surface, where it can form a thin, protective, hydrating layer. 42 HMWHA particles cannot pass through a filter membrane with a 220 nm pore size or through human tissue before being degraded. In contrast, LMWHA can permeate the stratum corneum, epidermis, and even the deeper dermal layers. Freshly manufactured HA35 can freely pass through filter membranes with a 220 nm pore size, suggesting that the human tissue permeability (with an approximate pore size less than 220 nm) of the nanosized HA35 would be good. LMWHA has been shown to achieve equilibrium permeation of tissue cells within 1 h, whereas HA permeation progresses gradually over 24 h. 16 Cytologically, we confirmed that HA35 has the same function as HA, including cell migration, nitric oxide production and reactive oxygen species (ROS) secretion. 27 These findings suggested that HA35 may have the same effect, given its good injection permeability and ability to overexpress the hyaluronic acid polymer in vivo.
The NPRS is one of the most commonly used pain scales in medicine. During the process of returning patients, we found that HA35 also relieved discomfort. Therefore, a new index, the GCQ scores, was used in the evaluation of neuropathic and inflammatory pain. The GCQ can identify patients’ needs, including pain intensity and anxiety, and help patients feel the effect of treatment. 43 In this study, we used the hyaluronidase extracted from bovine testes and HMWHA to produce the HA35 after incubation at room temperature for 10 mins, 20 mins, 30 mins, 40 mins, 50 mins, or 60 mins (Figure 2(A)). The results showed that the cleavage of HA using hyaluronidase from bovine testis and recombinant human hyaluronidase PH20 is a stable method to obtain the HA35. 24 The key to this study is that hyaluronidase injection and HMWHA injection are already on the market and can be legally used outside their specifications (off-label). In this study, the patients felt numb immediately after being injected with 100 mg of freshly manufactured HA35. The analgesic mechanism of the HA35 may be related to the cellular electrophysiological regulation of neuropathic pain.44,45 Local multipoint injection of 100 mg of HA35 into the pain point area and the nerve trunk innervating the pain point area significantly improved the neuropathic pain score of all patients with PHN (Figure 3(A)) after 3 h. Local injection of 100 mg of HA35 into the pain site also resulted in a significant (Figure 4(A)) improvement in the pain score of all shoulder, neck, back and temporomandibular pain patients after 3 h, meeting and even exceeding the expectations of this clinical study. This study also tracked the GCQ scores of patients with pain 24 h after treatment (Figure 5). The results showed that once HA35 was injected, the patients’ comfort levels changed significantly, and no adverse reactions related to the injection were found, demonstrating its safety. We have also demonstrated that injectable HA35 solution is a promising candidate for lung cancer treatment (Clinical Trials: NCT05852002). Taken together, these findings indicate that HA35 has the potential to become a clinical drug for treating neuropathic and inflammatory pain. Its therapeutic action is likely due to its anti-inflammatory properties.24,25
This study has several limitations. First, no calculation of the sample size needed for this study was performed. Second, the observation time after treating neuropathic pain in patients with PHN was short. Future studies should calculate and justify the sample size and evaluate pain over more than 2 weeks of multiple injection treatments and 3 months of observation to determine the long-term therapeutic effect of HA35. Our patients with neuropathic or inflammatory shoulder, neck, back or temporomandibular pain were treated with only one injection. Because this was a proof-of-concept study, the observation time was relatively short, and only the NPRS was used to assess pain. Future studies should evaluate data after more than 4 weeks of multiple injection treatments and 6 months of posttreatment observation to determine the long-term therapeutic effect. 30 Observation indicators should include behavioral pain assessments, such as the neck, shoulder, back and waist disability index (NDI), which is used to quantitatively evaluate the degree of pain and disability.29,30
Conclusion
1. Local injection of the 35 kDa HA fragment (HA35) into the pain point area and the nerve trunk innervating the pain point area effectively reduced neuropathic pain, including postherpetic neuralgia, within 3 h. 2. Local injection of the 35 kDa HA fragment (HA35) into the pain site effectively reduced inflammatory pain, including shoulder, neck, back and temporomandibular pain, within 3 h.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Qingdao Hui Nuo De Biotechnology Co., Ltd., Hai Shi Hai Nuo Group (24-1-6-ghgg-1-hz).
Ethical statement
Trial registration
Clinicaltrials.gov: Injection Therapy for Neuropathic Pain;NCT05809700. Recruitment Status: Completed. First Posted: April 12, 2023. Last Update Posted: September 26, 2023.
Animal welfare
The present study followed international, national, and/or institutional guidelines for humane animal treatment and complied with relevant legislation.
