Abstract
Based on microscopic and immunohistochemical characterization, we documented spontaneous proliferative and neoplastic lesions in the thyroid and parathyroid glands of nondomestic felids. Ten animals (4 leopards, 3 tigers, and 3 cougars), all with a previous diagnosis of thyroid neoplasia were identified from the University of Tennessee College of Veterinary Medicine database. The mean age of affected animals was 15.9 y. Twelve neoplasms were identified; 2 animals had 2 concurrent neoplasms. After immunohistochemical characterization using a panel of chromogranin A, thyroglobulin, and calcitonin, 7 of the former thyroid neoplasms were diagnosed as thyroid adenomas, 1 was diagnosed as a thyroid carcinoma, and 4 were diagnosed as parathyroid adenomas. No thyroid medullary neoplasms (C-cell tumors) were diagnosed in the current study. Most of the diagnosed neoplasms were benign (11 of 12), and metastasis was not documented in the single carcinoma. Only 2 animals were suspected to have functional neoplasms (1 thyroid adenoma and 1 parathyroid adenoma), based on associated tissue lesions or serum biochemistry. Other documented lesions in the thyroid and parathyroid glands included thyroid nodular hyperplasia (n = 7), parathyroid hyperplasia associated with chronic renal disease (n = 2), a thyroid abscess, and a branchial cyst. Parathyroid adenomas were more commonly diagnosed than expected in comparison with domestic cats. We demonstrated that an immunohistochemistry panel for thyroglobulin, calcitonin, and chromogranin A can be used to differentiate neoplasms of thyroid from parathyroid origin in nondomestic felids.
Introduction
Thyroid nodular hyperplasia and thyroid follicular adenomas are common in older domestic cats.1,15 Previous case reports document functional and nonfunctional thyroid carcinomas in captive nondomestic felids, including a cougar (Felis concolor) 9 and multiple leopards (Panthera pardus).6,11 In a study at the Philadelphia Zoological Gardens that spanned 55 y, the most commonly documented neoplasms of carnivores were of the endocrine organs. 10 Thyroid adenomas were documented in multiple nondomestic felids, and thyroid carcinomas were documented in a leopard, lion (Panthera leo), puma (Puma concolor), and Sumatran tiger (Panthera tigris). 10 Both the benign and malignant neoplasms were proposed to be secondary to an iodine-deficient diet. 10 In a 2008 study of neoplasia in nondomestic felids at the Knoxville Zoological Gardens that spanned 24 y, neoplasms of the endocrine organs were the second most commonly reported, following neoplasms of the integument. 14 Of these endocrine neoplasms, 80% were associated with the thyroid gland, 14 suggesting that proliferative lesions in nondomestic felids occur with some frequency.
In this retrospective study, we describe microscopic characteristics and immunohistochemical staining patterns of previously diagnosed thyroid neoplasms. The primary goal of this study was to use immunohistochemistry (IHC) to definitively characterize these neoplasms as either thyroid or parathyroid in origin. Additionally, other thyroid lesions and lesions in the associated parathyroid glands are described. The final goal of the study was to compare nondomestic felid thyroid lesions with those seen in domestic cats.
Materials and methods
Autopsy reports on nondomestic felids filed by the Pathology Service at the University of Tennessee Veterinary Medical Center (Knoxville, Tennessee) were reviewed for a diagnosis of thyroid neoplasia or other lesions. Species, sex, age at death, previous and final diagnoses, and cause of death or euthanasia were recorded for each case (Table 1). Information about clinical pathology, when performed, was also obtained from these animals’ medical records for the 2 most recent analyses (Table 1).
Signalment, descriptive diagnosis, relevant clinical chemistry findings, and pertinent cause of death or euthanasia in 10 nondomestic felids with thyroid or parathyroid neoplasia.
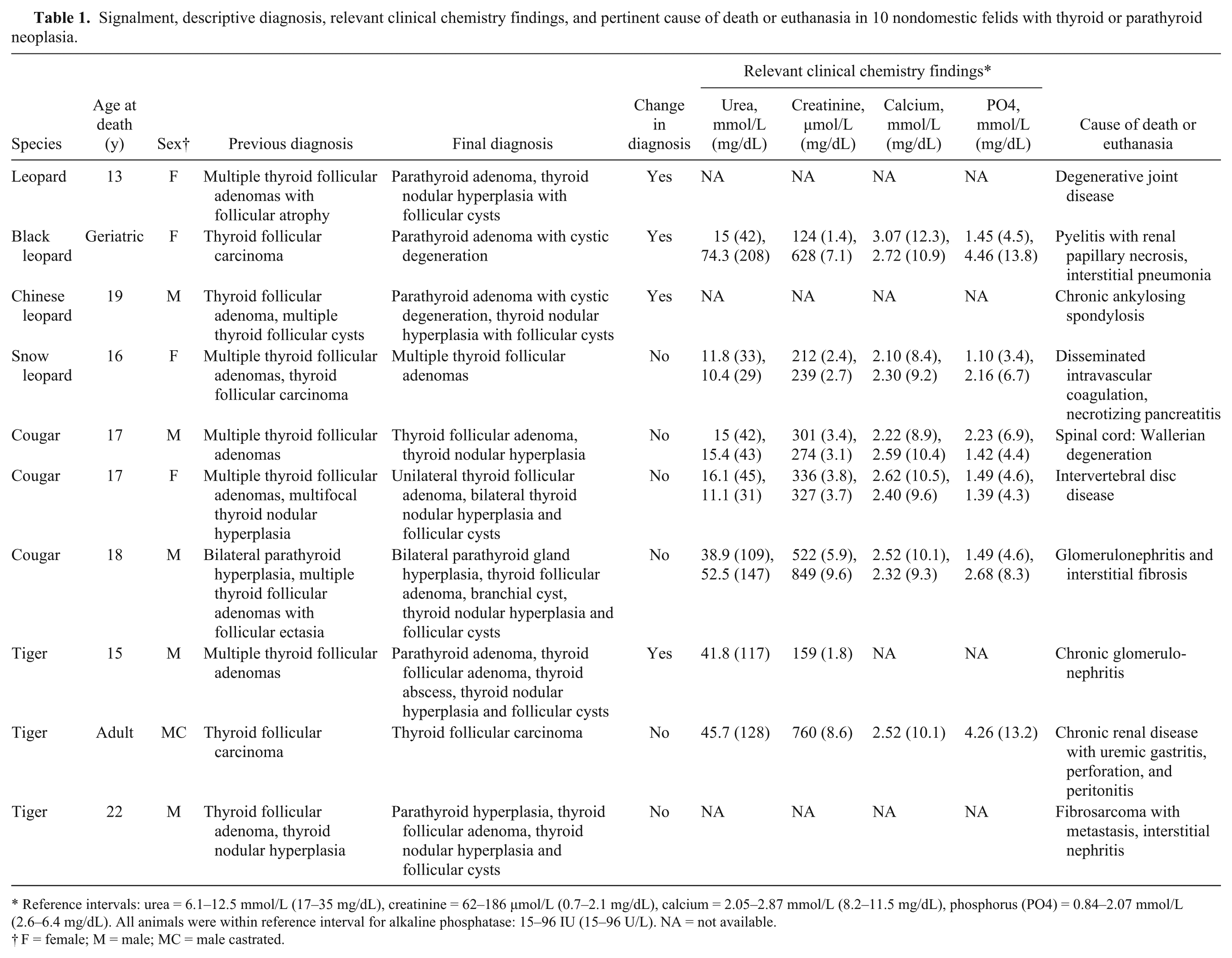
Reference intervals: urea = 6.1–12.5 mmol/L (17–35 mg/dL), creatinine = 62–186 µmol/L (0.7–2.1 mg/dL), calcium = 2.05–2.87 mmol/L (8.2–11.5 mg/dL), phosphorus (PO4) = 0.84–2.07 mmol/L (2.6–6.4 mg/dL). All animals were within reference interval for alkaline phosphatase: 15–96 IU (15–96 U/L). NA = not available.
F = female; M = male; MC = male castrated.
Thyroid and parathyroid tissues were fixed in 10% buffered formalin, sectioned at 5 µm, and stained with hematoxylin and eosin. All stained sections were reviewed by a board-certified pathologist (SJ Newman) and a pathology resident (JP Pope).
Chromogranin A, a thyroglobulin, b and calcitonin c antibodies were optimized using thyroid, parathyroid, and archived adrenal medullary tissue from both nondomestic felids and domestic cats. These same tissues not receiving the primary antibodies in each case served as negative controls. Nonneoplastic thyroid follicular cells, C-cells, and chief cells within the individual cases served as internal positive controls. All thyroid, parathyroid, and adrenal gland tissues were cut at 5 µm, placed on charged slides, air dried, and heated at 60°C for 15 min. All slides were deparaffinized using xylene and then rehydrated through graded ethanols to deionized water. For chromogranin A slides, heat-induced epitope retrieval was done with citrate buffer at pH 6 in a steamer at 95°C for 20 min. Thyroglobulin slides had no antigen retrieval performed. For calcitonin slides, enzyme-induced antigen retrieval d was done at room temperature for 5 min. All slides were rinsed in deionized water and soaked in Tris-buffered saline–Tween (TBST) for 10 min before being placed into an autostainer e at room temperature and rinsed with TBST between steps. A 3% hydrogen peroxide block was applied for 5 min to all slides. A serum-free protein block was applied for 20 min to the chromogranin A slides, for 30 min to the thyroglobulin slides, and for 5 min to the calcitonin slides. Chromogranin A was applied for 90 min at a 1:200 dilution; thyroglobulin was applied for 20 min at a 1:60,000 dilution; and calcitonin was applied for 30 min at a 1:500 dilution. The labeled polymer anti-mouse f for chromogranin A slides and anti-rabbit g for thyroglobulin and calcitonin slides (commercial horseradish peroxidase system) were applied for 30 min, and DAB (3,3′-diaminobenzidine) h was applied as the chromogen for 10 min to all slides. All slides were then rinsed with deionized water, stained for 5 s in hematoxylin, blued in ammonia water, dehydrated through ethanols, cleared with xylene, and coverslipped. Diagnosis was based on criteria outlined in Table 2.
Diagnostic criteria for lesions included in the study: thyroid nodular hyperplasia, thyroid follicular adenoma, thyroid carcinoma, parathyroid hyperplasia, and parathyroid gland adenoma.

Results
Ninety accessions were reviewed, and 12 nondomestic felids were identified with thyroid neoplasia; 2 animals were excluded because tissues were either not available or too autolyzed for inclusion in the study, reducing the number of study animals to 10. Seven of these neoplasms in 5 non-domestic felids were published previously 14 but are further characterized in the current study; 5 additional animals were included from the autopsy reports. The ages of 1 tiger and 1 leopard were unknown. The youngest and oldest cases, with known ages at death, were a 13-y-old leopard and a 22-y-old tiger, respectively. The mean age at death for all nondomestic felids in the current study was 15.9 y.
Twelve neoplasms were originally diagnosed as thyroid neoplasms, with 2 animals having 2 concurrent neoplasms. Following IHC characterization in conjunction with the diagnostic criteria described (Table 2), 7 of these neoplasms were diagnosed as thyroid adenomas (Fig. 1C, 1D), 1 as a thyroid carcinoma (Fig. 1E, 1F), and 4 as parathyroid adenomas (Fig. 1A, 1B). No thyroid medullary neoplasms (C-cell tumors) were identified in the current study. Antibodies for calcitonin and thyroglobulin were able to be optimized for use in nondomestic felids, but an anti–parathyroid hormone (PTH) antibody was unable to be utilized for confirmatory diagnosis of parathyroid neoplasms in our laboratory, despite use of multiple protocols and several commercially available reagents. Most (11 of 12) of the diagnosed neoplasms were benign, and metastasis was not recorded in any reports, including the final autopsy report of the animal with the thyroid carcinoma. Other lesions in these 10 felid thyroid glands and parathyroid glands included thyroid nodular hyperplasia with follicular cysts (n = 7), parathyroid hyperplasia associated with chronic renal disease (n = 2), a thyroid abscess, and a branchial cyst.
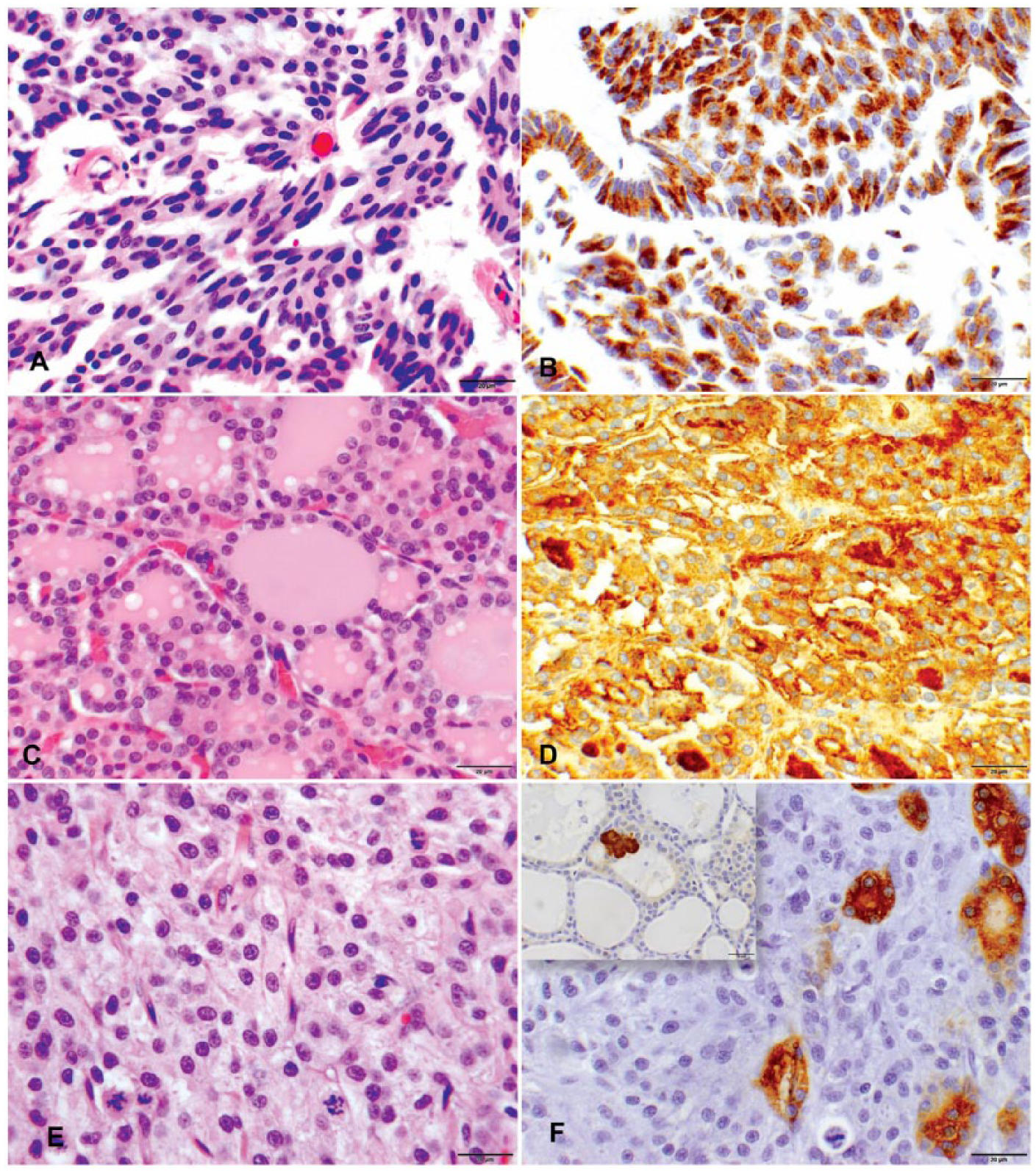
Histopathology and immunohistochemistry of parathyroid and thyroid neoplasms in nondomestic felids. (
Clinical pathology results were available from 2 animals with parathyroid adenomas (Table 1): 1 leopard with a parathyroid adenoma, and 1 tiger with both a parathyroid adenoma and a thyroid adenoma. The leopard had an increased calcium concentration (3.07 mmol/L [12.3 mg/dL]; reference interval = 2.20–2.89 mmol/L [8.8–11.6 mg/dL] 19 ) at 1 sampling, but calcium concentrations decreased to within the normal reference interval in subsequent plasma samples. The tiger had no biochemical changes suggestive of thyroid or parathyroid disease. PTH levels were not available for any of the study animals. Triiodothyronine (T3), thyroxine (T4), and thyroid-stimulating hormone (TSH) levels were not available antemortem for any of the study animals, and plasma biochemical abnormalities that would be consistent with, but not specific for, clinical hyperthyroidism (such as elevated alkaline phosphatase activity) were not observed in any felid for which clinical pathology testing was performed (n = 5; Table 1). In addition, for animals with thyroid neoplasms, body condition varied from emaciated to obese. Iodine levels were not available for any of the study animals. Hyperthyroidism was considered in the snow leopard with a thyroid adenoma because of concurrent hypertrophic cardiomyopathy (a lesion documented in domestic cats with hyperthyroidism) and hyperphosphatemia, without primary renal tubular disease, but was not confirmed.
The causes of death or euthanasia varied and included renal disease (n = 3), renal disease and interstitial pneumonia (n = 1), intervertebral disc disease (n = 2), Wallerian degeneration of the spinal cord (n = 1), degenerative joint disease (n = 1), necrotizing pancreatitis and disseminated intravascular coagulation (n = 1), and metastatic fibrosarcoma (n = 1). Thyroid or parathyroid neoplasms were not considered to contribute to the cause of death or euthanasia in any of the cases. Additional lesions found at autopsy that were potentially related to the thyroid lesions included hypertrophic cardiomyopathy in a snow leopard with bilateral thyroid adenomas, and a pancreatic islet cell tumor in a tiger with a thyroid adenoma and parathyroid gland hyperplasia.
Discussion
Thyroid neoplasms were more common than parathyroid neoplasms in the current study; the parathyroid neoplasms were diagnosed only following IHC characterization. However, parathyroid neoplasms were more commonly diagnosed than expected, compared with domestic cats.3,4,8,13 A leopard with a parathyroid adenoma had a 1-time increased plasma calcium concentration, as well as anorexia, which were interpreted as nonspecific findings potentially associated with a functional neoplasm. Neoplasms in the other 3 animals were suspected to be nonfunctional.
Bilateral parathyroid gland hyperplasia (secondary hyperparathyroidism) associated with chronic renal failure is commonly reported in domestic cats. Although renal failure was not confirmed in our cases, 2 of the felids in the current study had findings supportive of significant renal disease and possible associated secondary hyperparathyroidism, 1 based on laboratory findings of azotemia and 1 with an autopsy finding of interstitial nephritis. 3 Parathyroid gland hyperplasia can also occur in association with hyperthyroidism. 3 The thyroid neoplasms diagnosed in these 2 animals were suspected, but not confirmed, to be nonfunctional.
Domestic cats with thyroid neoplasms are typically geriatric, with a mean age of 12.4 y at diagnosis of adenomas and 15.8 y for diagnosis of carcinomas. 2 The mean age at diagnosis for the nondomestic felids in our study, 15.9 y, was similar to domestic cats. Thyroid glands can develop hyperplastic and neoplastic lesions from both the thyroid follicular cells and the surrounding C-cells (parafollicular cells). Domestic cats most commonly develop either thyroid nodular hyperplasia, with bilateral involvement in up to 70% of cases, or follicular adenomas, both of which can result in hyperthyroidism.1,3,15 Seven nondomestic felids had foci consistent with thyroid nodular hyperplasia, which, to our knowledge, has not been described previously in nondomestic felids. Unlike domestic cats, these hyperplastic lesions were suspected to be nonfunctional, based on available supporting laboratory results, but this suspicion cannot be confirmed without evaluation of T3 or T4 values. Because of the single case of concurrent hypertrophic cardiomyopathy, the possibility of functionality requires further study. All the nondomestic felids in our study were captive, and previous reports have implicated an iodine-deficient diet in the development of thyroid neoplasms in captive exotic cats. 9 Neither blood iodine levels nor dietary iodine levels were measured in our study, so it is unclear if iodine deficiency played a role in thyroid neoplasm development in these exotic cats.
Thyroid follicular adenomas are more common than follicular carcinomas in domestic cats,1,3 and similarly, only 1 thyroid follicular carcinoma was diagnosed in a tiger in the current study. Thyroid follicular carcinomas with metastases have been described in a tiger, a puma, and a leopard, 7 but metastases were not recorded in the autopsy report for the tiger included in our study. In domestic cats, thyroid follicular carcinomas metastasize <50% of the time, most commonly to the regional lymph nodes rather than lungs.1,2,15 No medullary (C-cell) thyroid neoplasms were diagnosed in the study, which is similar to the case in domestic cats, in which these neoplasms are rare. 2
A single tiger had lesions consistent with multiple endocrine neoplasia (MEN) and included bilateral parathyroid gland hyperplasia, a thyroid follicular adenoma, and a pancreatic islet cell carcinoma. In humans, 17 MEN-1 is associated with a mutation in the menin gene that results in lesions in parathyroids and endocrine pancreas with subtypes that can lead to additional lesions in the thyroids, 17 as in this tiger. A MEN-1–like syndrome has been reported in 2 Domestic Shorthair cats, but examination of the menin gene for mutations in those cats was unsuccessful. 17 We were not able to evaluate the tiger in our study for menin gene mutations.
All neoplasms (n = 12) included in our study were originally diagnosed as thyroid neoplasms. Following IHC characterization, 4 were reclassified as parathyroid adenomas (Table 1). IHC has not been used in nondomestic felids to further characterize these neoplasms, but has been used in domestic cats to differentiate tumors arising from thyroid follicular cells (thyroglobulin), C-cells (calcitonin), and chief cells (PTH; Chaitman SJ, et al. Immunohistochemical staining of thyroglobulin in thyroid glands of normal cats, hyperthyroid cats, and cats with euthyroxinemia. Proc 18th Am Coll Vet Internal Med Meeting; 2000 May 25–28; Seattle, WA).2,13 Both medullary thyroid tumors (C-cell tumors) and parathyroid gland tumors can be immunoreactive for chromogranin A and synaptophysin.2,5 The single thyroid carcinoma included in our study had a few neoplastic cells (<5%) that stained positively with thyroglobulin, and poorly differentiated thyroid follicular carcinomas, as in this case, have been reported to be negative for thyroglobulin.12,16
Chromogranin A was used to confirm neoplasms of parathyroid based on its previous use in dogs 5 and its reported conservation between species. 2 IHC staining with chromogranin A was observed to be most intense in leopards and less intense in tigers, suggesting a species difference in immunoreactivity; 1 tiger had poor IHC staining with both thyroglobulin and chromogranin A. Chromogranin A staining was most intense in hyperplastic parathyroid lesions. Staining differences of PTH and chromogranin A in normal, hyperplastic, and neoplastic parathyroid lesions have been documented.5,18,20
Parathyroid neoplasms were more common than expected in this population of nondomestic felids, when compared with domestic cats, but an IHC panel may be required for accurate diagnosis. Most of the thyroid neoplasms and all of the parathyroid neoplasms in these nondomestic felids were benign. Most of the thyroid and parathyroid neoplasms were suspected to be nonfunctional based on the available laboratory results, clinical history, and recorded lesions; however, without evaluation of more specific serum biochemistry parameters, including TSH, T3, T4, and PTH, this suspicion cannot be confirmed. For the nondomestic felid cases in our study, spindle cell morphology of a portion of the neoplastic cells in parathyroid gland tumors that was confirmed by IHC was one of the most discriminating features for differentiation of origin by microscopic examination. Chromogranin A, thyroglobulin, and calcitonin can be used as a panel in nondomestic felids to distinguish parathyroid, thyroid follicular, and thyroid medullary origin of neoplasms.
Footnotes
Acknowledgements
We thank Misty Bailey (paid by the University of Tennessee College of Veterinary Medicine) and Elizabeth Carter for technical editing assistance, Joy Chambers (paid by the University of Tennessee College of Veterinary Medicine) for technical graphic support, and Dr. Deanna Schaefer for SI unit calculations.
Authors’ contributions
JP Pope contributed to design of the study and drafted the manuscript. J Steeil contributed to design of the study. EC Ramsay, D Reel, and SJ Newman contributed to conception and design of the study. All authors contributed to acquisition, analysis, and interpretation of data; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Monoclonal mouse chromogranin A antibody DAK-A3, Dako North America, Carpinteria, CA.
b.
Polyclonal rabbit thyroglobulin antibody, Dako North America, Carpinteria, CA.
c.
Polyclonal rabbit calcitonin antibody, Dako North America, Carpinteria, CA.
d.
Proteinase K, Dako North America, Carpinteria, CA.
e.
Autostainer S3400, Dako North America, Carpinteria, CA.
f.
EnVision + system HRP anti-mouse, Dako North America, Carpinteria, CA.
g.
EnVision + system HRP anti-rabbit, Dako North America, Carpinteria, CA.
h.
Dako North America, Carpinteria, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received financial support from the Companion Animal Fund, Department of Small Animal Clinical Sciences, College of Veterinary Medicine, University of Tennessee.
