Abstract
Tissue fixation, a central element in histotechnology, is currently performed with chemical compounds potentially harmful for human health and the environment. Therefore, alternative fixatives are being developed, including alcohol-based solutions. We evaluated several ethanol-based mixtures with additives to study fixative penetration rate, tissue volume changes, and morphologic effects in the bovine testis. Fixatives used were Bouin solution, 4% formaldehyde (F4), 70% ethanol (E70), E70 with 1.5% glycerol (E70G), E70 with 5% acetic acid (E70A), E70 with 1.5% glycerol and 5% acetic acid (E70AG), and E70 with 1.5% glycerol, 5% acetic acid, and 1% dimethyl sulfoxide (DMSO; E70AGD). Five-millimeter bovine testicular tissue cubes could be completely penetrated by ethanol-based fixatives and Bouin solution in 2–3 h, whereas F4 required 21 h. Bouin solution produced general tissue shrinkage, whereas the other fixatives (alcohol-based and F4) caused tissue volume expansion. Although Bouin solution is an excellent fixative for testicular tissue, ethanol-based fixatives showed good penetration rates, low tissue shrinkage, and preserved sufficient morphology to allow identification of the stages of the seminiferous epithelium cycle, therefore representing a valid alternative for histotechnology laboratories. Common additives such as acetic acid, glycerol, and DMSO offered marginal benefits for the process of fixation; E70AG showed the best preservation of morphology with excellent nuclear detail, close to that of Bouin solution.
The preservation of biological tissues after death or excision from the body plays a central role in histotechnology. Tissue fixation prevents autolysis in order to preserve cytoarchitecture very close to the in vivo situation. Many approaches for tissue fixation have been undertaken, but aldehydes, and particularly formaldehyde, have long been considered the “gold standard” in histopathology laboratories around the world.6,12 The use of formaldehyde is legislated and is banned in many countries. As a consequence, several formaldehyde substitutes have been developed commercially. However, major concerns related to these substitutes include availability (particularly for laboratories located in developing countries with high import costs), undisclosed formulations that interfere with scientific interpretation of research results, irregular supply, and even sudden exit from the market. 18 Moreover, they introduce issues such as slow tissue penetration, tissue shrinkage, and problems during microtomy.12,18
Fixatives can be classified into 2 main groups: coagulative or denaturing, and cross-linking fixatives (Table 1). Coagulative fixatives disrupt proteins into their native structure, whereas cross-linkers react covalently to form bridges among the amino acid chains of proteins. Apparently, penetration rates are unrelated to the fixative type. However, it is accepted that fixation occurs almost immediately with coagulative fixatives, whereas cross-linkers require many hours for protein cross-link formation (formaldehyde requires 24 h at 25°C for this reaction to occur). 2
Examples of some common fixatives classified according to mechanisms of action and their known penetration speeds.*

Penetration speed expressed as the constant
DMSO (dimethyl sulfoxide) is a molecule carrier and therefore would enhance the penetration of other substances.
Alcohol-based fixatives belong to the coagulant group of fixatives 7 and, as such, tend to produce coagulated proteinaceous materials in histologic preparations 6 (a problem not present with formaldehyde). Additionally, alcohols cause shrinkage, with subsequent distortion of tissue microanatomy. In order to overcome the disadvantages of alcohol-based fixatives, additives can be added to the fixative mixture, as demonstrated with the introduction of Carnoy fixative 16 and with a modified Davidson solution. 9 Furthermore, alcohol-based fixatives allow the performance of immunohistochemistry procedures without antigen-retrieval steps, as well as the recovery of intact nucleic acids from tissue fixed in such mixtures.7,18
We evaluated several ethanol-based mixtures for the fixation of bovine testicular tissue, with respect to penetration rate, tissue volume changes, and morphology. Testes were collected from one ~2-y-old Holstein bull at a commercial slaughterhouse. The bull was fasted and water deprived for at least 12 h before death. Testes were collected early in the morning, within 15 min of death, and were held in an ice box (at ~4°C) until experiments began (total elapsed time of 4 h). The vaginal tunic and epididymis were excised and the testis weight recorded. Volume was estimated by the water displacement method. Testes were grossly normal. The experiments were performed in a temperature-controlled room (~22°C). Fixatives used were 1) Bouin solution; 2) 4% formaldehyde v/v (F4); 3) 70% ethanol (E70); 4) 5% glacial acetic acid in 70% ethanol (E70A); 5) 1.5% glycerol in 70% ethanol (E70G); 6) 5% glacial acetic acid and 1.5% glycerol in 70% ethanol (E70AG); and 7) 5% glacial acetic acid, 1.5% glycerol, and 1% dimethyl sulfoxide (DMSO) in 70% ethanol (E70AGD). In some experiments, 95% ethanol (E95) was used. Bouin solution was freshly prepared (70% saturated solution of picric acid, 25% of 40% formaldehyde, 5% glacial acetic acid).
For the penetration assay, 84 cubes of testicular tissue of ~1 cm3 from 1 bull testis were cut and immediately placed in chilled isotonic saline solution (4°C, 0.9% NaCl). Metal rulers guided the cuts of 1-cm thick slabs along the major axis of the testis; the slabs were further cut into 1-cm wide strips, which were subsequently cut to the final size (~1 cm3). The tissue pieces were cut avoiding the central testicular mediastinum. Twelve random pieces of tissue were simultaneously immersed in each of the 7 fixatives. The containers were glass jars with 120 mL of fixative each, at 1:10 (tissue volume:fixative volume; usual working concentration). 3 A plastic mesh (2-mm diameter glass fiber mesh with polyvinyl chloride coating) was placed on the bottom of each jar to guarantee uniform access of the fixative to all surfaces of the tissue pieces during stationary fixation. Three random pieces of testicular tissue were removed from the fixatives at each of the 4 time points, and each tissue piece was cut in half, to photograph 1 of the 2 newly exposed cut surfaces, from a fixed distance of 10 cm, adjacent to a ruler (smallest unit = mm) in order to register fixative penetration in mm. Once photographed, the pieces of tissue were discarded. The time points for fixation stop and photography were 0.5, 2, 3, and 4.5 h.
Because formaldehyde present in the tissue during fixation is not readily visible to the naked eye, 10 its presence in formalin-fixed tissues was evaluated through the reaction of formaldehyde with Tollens reagent. 15 This reagent was prepared by adding 2–3 drops of 5% (w/w) NaOH solution to 1 mL of 5% (w/w) AgNO3 solution. The brown precipitate formed by the addition of NaOH solution was dissolved by dropwise addition of 2 M NH3 solution and stirring until total dissolution of the precipitate. Tollens reagent was prepared fresh before use. Pieces of tissue were removed from the formalin-containing flask, gently blotted with a paper towel, cut in half, and submerged in Tollens reagent (newly exposed tissue surface facing up) until a dark brown precipitate appeared in the tissue where aldehydes were present (typically 2–5 min). Tissues were then photographed as described with other fixatives.
Penetration was evaluated with Image-J software (http://imagej.nih.gov/ij/). The program was calibrated before measuring the distance from the surface of the tissue to the inner front line of the fixative advance. This depth of penetration was measured at a right angle from the mid-point of each side of the nearly square exposed surface of each tissue piece, therefore allowing 4 penetration measurements per tissue piece, averaged for statistical analysis.
In a separate experiment (from the same bull, contralateral testis), the individual volumes of 3 pieces of tissue (~3 cm3) per fixative were taken fresh, placed in the specific fixative (volumetric ratio of 1:10) and their volume reevaluated after 24 h (total number of pieces = 24; 8 fixatives). Volumes were estimated using the water displacement method; the graduated cylinders were photographed before and after water displacement, from a fixed distance (horizontal axis, 15 cm), and the displacement estimated on digital images with Image-J software.
For histologic processing, testicular tissue pieces (1 × 1 × 0.5 cm, from the testis used for the penetration assay) were immersed in the different fixatives for 24 h (1 per fixative; total number of pieces = 7) and the specimens then transferred into biopsy cassettes. The newly cut surface of the 24-h fixed tissues were placed face down in the cassettes and subsequently toward the bottom of the embedding mold. Samples were dehydrated (two 70% ethanol steps, two 96% ethanol steps, two 100% ethanol steps, 30 min each; 2 xylene steps, 45 min each) and infiltrated with paraffin in a histoprocessor. a Samples were embedded in paraffin blocks and cut 5 μm thick. b Some sections were cut at 20 μm for nuclear diameter evaluation. Slides were deparaffinized, stained with Harris hematoxylin, and mounted. c A testicular tissue cryosection (5 μm) was cut in a cryostat d to be used as a reference for several morphometric parameters. Briefly, fresh material was obtained simultaneously from the same regions of the testis as described for the fixative penetration assay. The testis tissue was immediately glued to the base of a specimen cryostat holder (round, 30 mm) with tissue-embedding medium e and frozen at −5°C. Sections (5 μm) were cut d (–5°C, coldest equipment temperature; room temperature = 22°C.) and were stained similarly to the histologically processed material.
Fixed testicular tissues were analyzed for general morphology and then staged (12 stages of the bovine seminiferous epithelium cycle, acrosomic system 4 ). Stereologic and morphometric parameters were estimated. Volume density (Vd) of testicular compartments—seminiferous tubules and interstitial (intertubular) space—were estimated with a 30-point, 195-μm spaced graticule, digitally superimposed at random positions over testicular tissue images corresponding to the several fixatives (treatments) used, in a total of 20 images per fixative (600 points per fixative). All images were captured with a digital video camera. f Tubule diameters were measured on the digital images by averaging the major and minor axis of randomly chosen near-round cross-section profiles of seminiferous tubules (~55 cross-section profiles per fixative), separated at least 250 μm from each other by using Image-J software. Average nuclear diameters of spermatocytes in pachytene (at stage VII of the seminiferous epithelium cycle) were also taken, by estimating the major and minor axes of randomly chosen cells in 20-μm thick sections, by adjusting the micrometer knob to obtain the highest (true) diameter dimensions. 14 Pachytene spermatocytes have a rounded to slightly ovoid nucleus with discrete chromatin patches representing homologous chromosomes in the process of genetic recombination.
Roundness was evaluated in 8 randomly chosen near-round seminiferous tubule cross-sections per fixative. The roundness (
All statistical analyses were performed using specialized software.
g
Data were tested for normal distribution, and no transformations were required. General statistical analysis of stereologic and morphometric variables was performed using one-way analysis of variance. The level of significance was set at
Best-fit curves for fixative penetration of bovine testicular tissue over time showed a sigmoid shape, the best fitting model being third order (cubic) polynomial functions in all cases (high
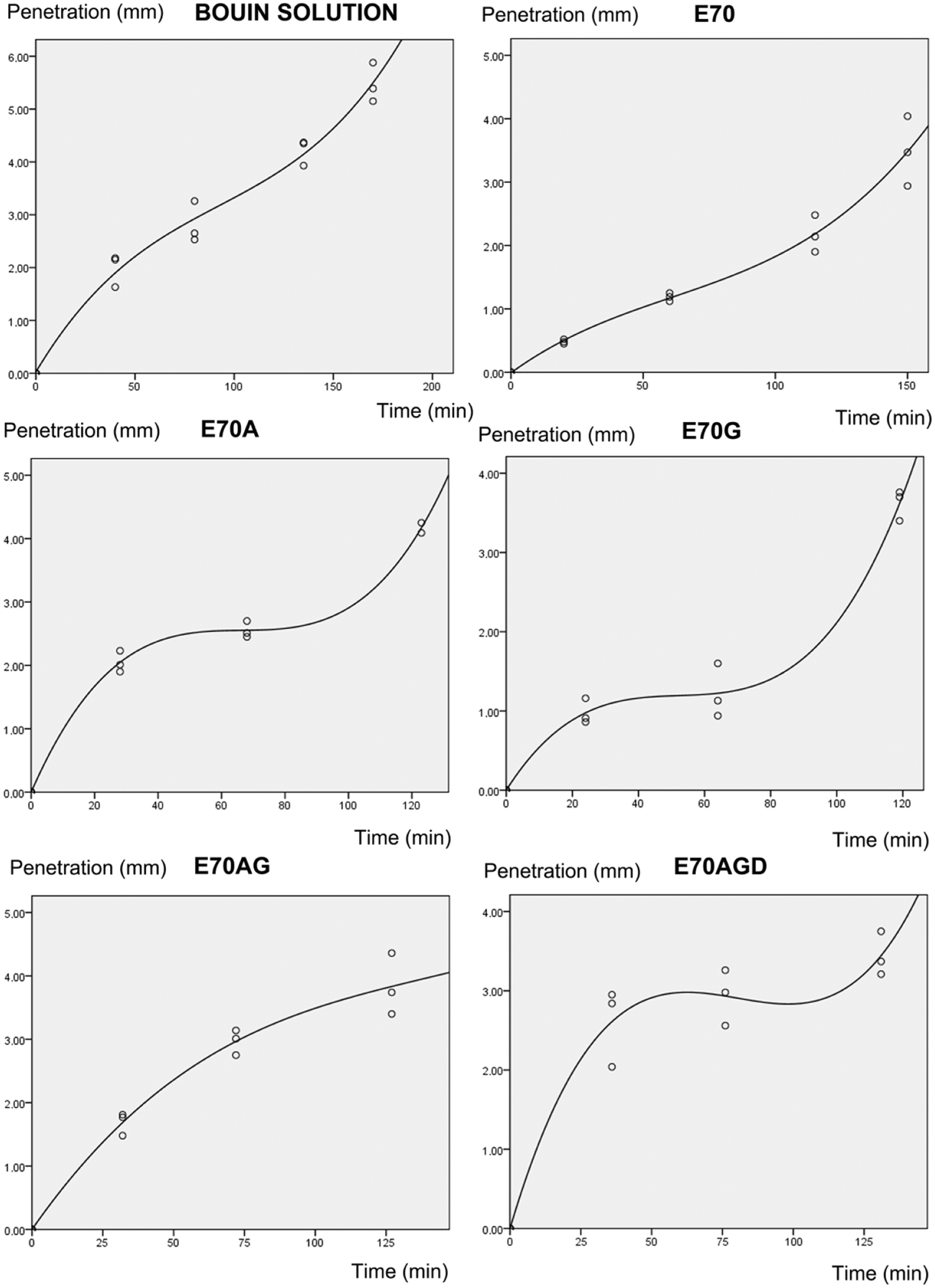
Best-fit curves for fixative penetration in bovine testicular tissue over time. Fixatives used were Bouin solution, 70% ethanol (E70), 5% glacial acetic acid in 70% ethanol (E70A), 1.5% glycerol in 70% ethanol (E70G), 5% glacial acetic acid + 1.5% glycerol, in 70% ethanol (E70AG), and 5% acetic acid + 1.5% glycerol + 1% DMSO, in 70% ethanol (E70AGD). Equations and
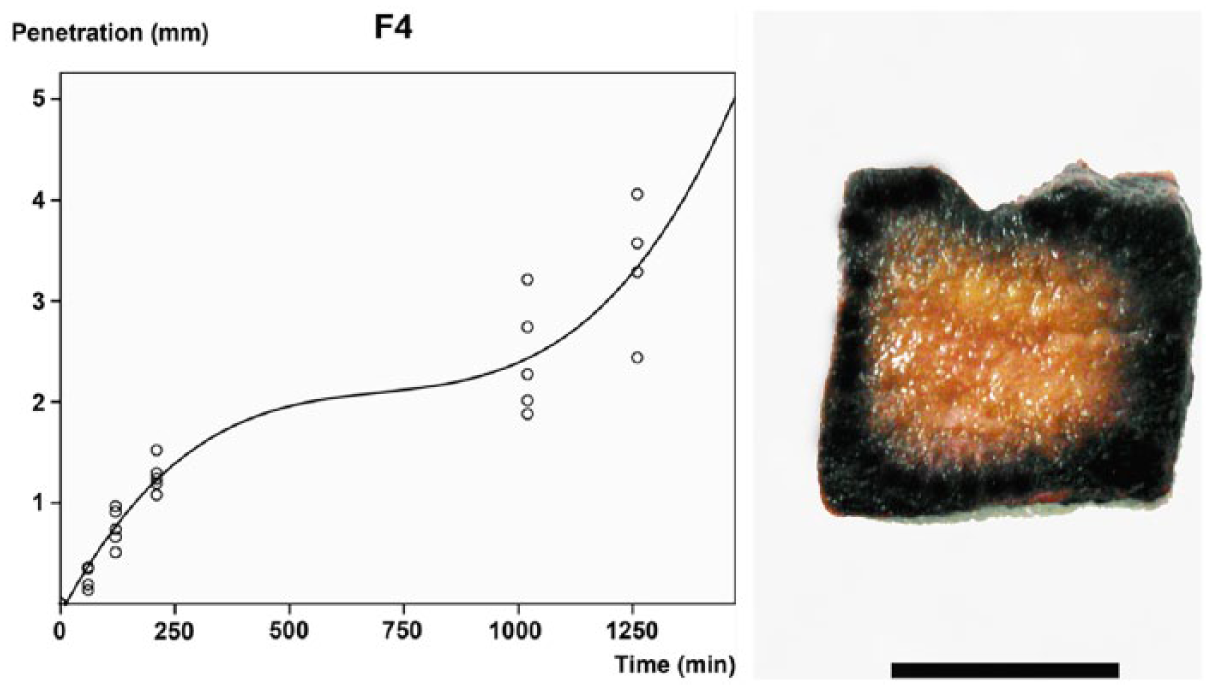
Best-fit curve for 4% formaldehyde (F4) penetration in bovine testicular tissue over time. Equation and
Duration of penetration (time taken for 5-mm thick testicular tissue pieces to be completely penetrated by selected fixatives, as predicted by curve equations).*

DMSO = dimethyl sulfoxide.
All ethanol-based fixatives and Bouin solution showed similar penetration times for 5-mm thick bovine testicular tissue (i.e., complete penetration in 2–3 h). Fastest-penetrating Bouin solution and DMSO-containing fixative (E70AGD) would completely infiltrate that tissue in about the same time (~2 h, 40 min). However, after exactly the first hour of exposure to the fixative (rapid penetration stage of the curve), DMSO-containing fixative (E70AGD), penetrating 3 mm in total, would outperform Bouin solution by 0.6 mm (Bouin solution total penetration in hour 1 = 2.4 mm), representing a 25% higher penetration rate. In general, the first rapid phase of penetration was steeper in the case of acetic acid–containing fixatives, particularly with DMSO-containing fixative (E70AGD). Acetic acid is well known for its rapid penetration into tissues.10,17 DMSO probably promotes the penetration of acetic acid, as DMSO acts as a carrier molecule for many compounds. 8
Formaldehyde solution had a slow penetration rate given that only 3.3 mm of testicular tissue were penetrated after 21 h. The reaction of Tollens reagent to verify formaldehyde presence in bovine testis tissue occurred very fast. The positive reaction with Tollens reagent, after only a few hours of immersion of tissues in F4, confirms that formaldehyde does not react with tissue proteins in this time range, thus remaining chemically free.
The volume of bovine testicular material did not change significantly with the different fixatives used (Fig. 3). Because the testis is a very water-rich tissue, it would be expected that high water content fixatives (e.g., Bouin solution and F4), as opposed to those containing alcohol, would produce less shrinkage. F4 has been known to cause tissue swelling during fixation, generating increments in cell height. 1 The rest of the fixatives, although expected to cause shrinkage (given their alcohol base), instead generated tissue volume expansion. Acetic acid is probably involved in counteracting shrinkage in those fixatives containing it, 11 which leads to the question of why 70% ethanol alone did not produce major shrinkage in our experiments, as would be expected for alcohols (and was actually shown with ethanol 95%, used as an internal control, which produced marked shrinkage, i.e., ~23%). Perhaps the higher presence of water in the 70% ethanol solution resulted in a slight volume expansion, thus counteracting the action of ethanol.
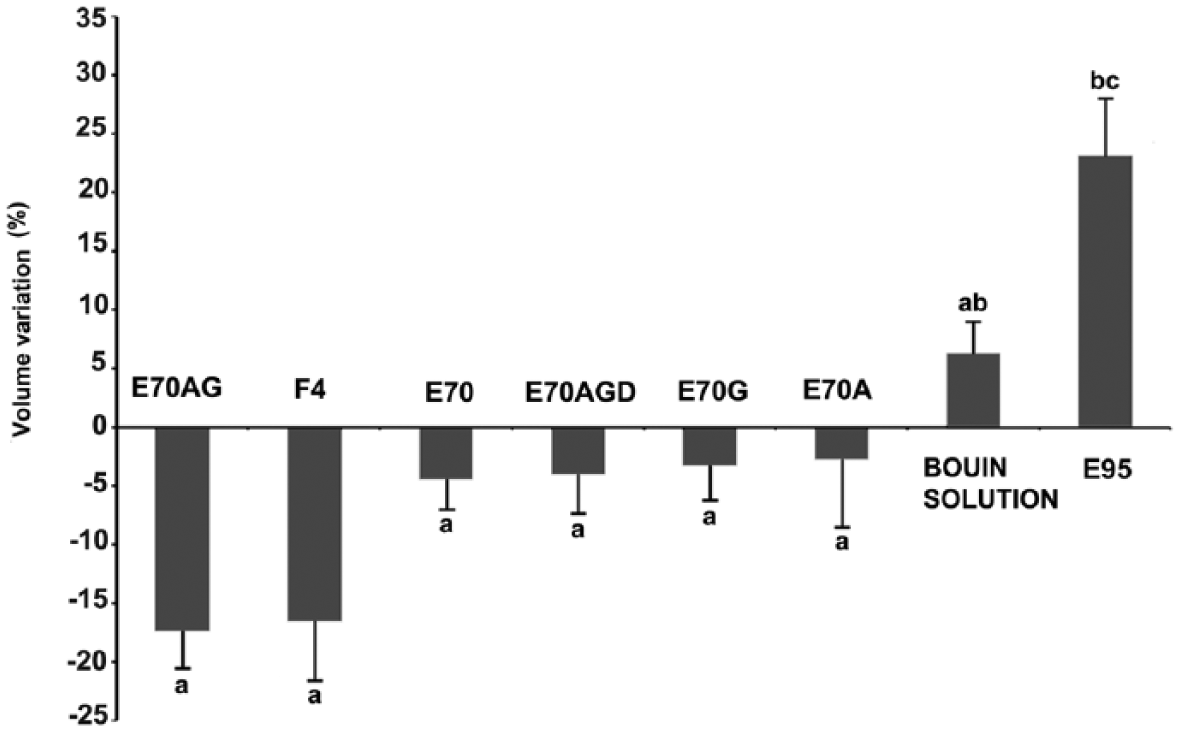
Percentage of bovine testicular tissue volume change after fixation with different fixatives after 24 h using the water displacement method. E70AG = 5% glacial acetic acid + 1.5% glycerol, in 70% ethanol; F4 = 4% formaldehyde; E70 = 70% ethanol; E70AGD = 5% acetic acid + 1.5% glycerol + 1% dimethyl sulfoxide (DMSO), in 70% ethanol; E70G = 1.5% glycerol in 70% ethanol; E70A = 5% glacial acetic acid in 70% ethanol; E95 = 95% ethanol. Different letters (a, b, c) represent significant differences (
Fixative composition was a factor for gross tissue volume changes. In order to see fixative effects at lower scales (tissue and cellular levels), stereologic and morphometric techniques were used. Cryostat (unfixed) sections were assumed as non-changing (control). Bouin solution produced smaller interstitial space Vd than the rest of the fixatives (~47% lower than in cryostat sections,
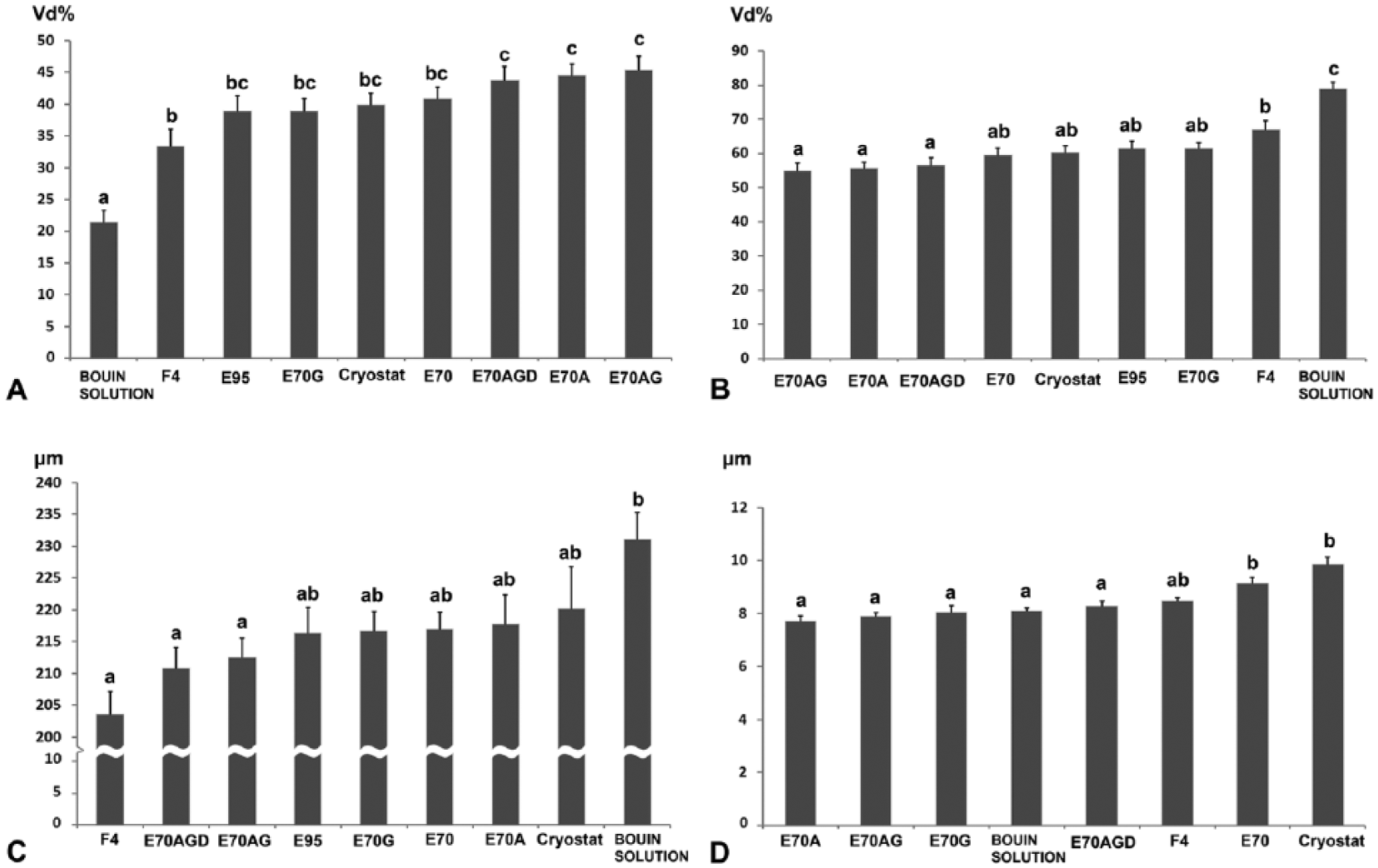
Several end-points of bovine testicular tissue fixation with different fixatives.
At the cellular level, nuclear diameters of pachytene spermatocytes at stage VII of the seminiferous epithelium cycle were significantly higher in cryostat sections (
Acetic acid, which is a component of Bouin fixative, is known to penetrate very fast, probably coagulating proteins encountered on its diffusion front almost instantaneously. As well, acetic acid incorporates water molecules between protein chains and disrupts salt-like bonds between amino and carboxyl groups of proteins. 16 Picric acid probably produces the same effect, but its penetration is slower than that of acetic acid, 11 likely stabilizing different groups of proteins. These combined coagulative effects and the presence of formaldehyde in Bouin solution would then produce an overall effect of tissue expansion affecting both tubular and intertubular spaces.
In the end, results concerning relative volume changes of testicular compartments and cells were very likely also affected by the influence of the histologic processing through alcohol series and xylene. Dehydration during processing probably modulates the patterns of local volume change in the Bouin-fixed tissues, preferentially producing shrinkage of the interstitium, a less cellular compartment. The dehydrating effect of alcohols makes denatured proteins lose water molecules normally located among them. In this way, proteins can better come into contact with each other, allowing for noncovalent attractions between charged amino acids. In our study, a rehydration of 95% ethanol-fixed testicular tissues, when transferred to 70% alcohol (at the start of histologic processing) would be expected. However, because proteins present on these tissues were already coagulated, the hydration would be only transient because the tissues are subjected to histologic processing dehydration. More research is needed to clarify the effects of histologic processing on the subtle details of testicular tissue after fixation. Any amount of cross-linking produced by F4 or Bouin solution would not be expected to reverse during histologic processing, particularly during the first step (70% ethanol). Although Bouin solution produced the best overall morphology, with very round seminiferous tubule cross-sections, good stain affinity, and sharp nuclear detail, alcohol-based fixatives gave comparable testis morphology (Fig. 5). Alcohol-based fixatives produced some cell sloughing toward the lumen of the seminiferous tubules. E70AG gave excellent morphology (close to Bouin solution) and did not show significant nuclear retraction compared to Bouin solution and F4 (
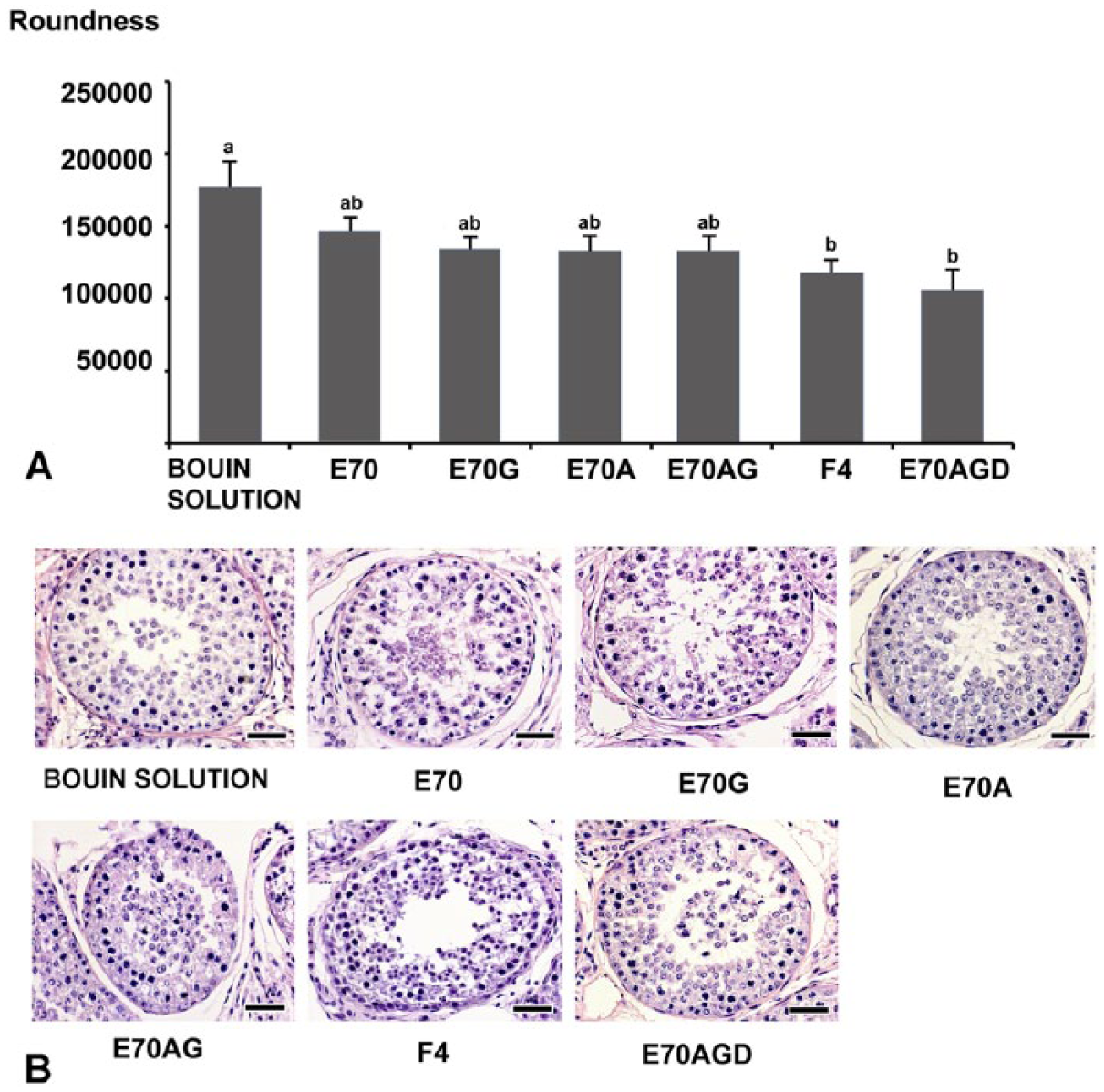
Roundness of seminiferous tubule cross-sections of bovine testis tissue after fixation for 24 h with different fixatives. Roundness is a dimensionless end-point that was evaluated in seminiferous tubule cross-sections. Higher values describe shapes closer to a perfect circle. Roundness (
As a reference for good morphology, the parameter roundness (
Thus, although Bouin solution is an excellent fixative for testicular tissue, particularly suited for research purposes, ethanol-based fixatives represent a very interesting alternative for histotechnology laboratories. They are economical, environmentally safer, and provide fairly good tissue morphologies, not posing the well-known health and safety–related drawbacks of Bouin solution. When including specific additives to the fixative mixtures, such as glacial acetic acid and/or DMSO, they rapidly penetrate tissues, outperforming F4, which in addition requires at least 24 h for cross-linking reactions to occur (after complete penetration), sequesters antigens, and affects nucleic acid integrity. Of all ethanol-based fixatives used, E70AG showed the best morphology with excellent nuclear detail, close to Bouin solution. It will be important to study the long-term stability of paraffin-embedded sections and sample blocks for archival purposes and nucleic acid stability for molecular biologic forensic analysis when using alcohol-based fixatives.
Footnotes
Acknowledgements
We thank the slaughterhouse “Camal Metropolitano,” Quito, Ecuador, for providing the biological materials used in the present work.
Authors’ contributions
NC Cabrera and LA Ramos contributed to conception of the study and critically revised the manuscript. JR Espinoza and P Sandoval contributed to acquisition of data and drafted the manuscript. P Vargas-Jentzsch contributed to conception of the study; contributed to acquisition and analysis of data; and critically revised the manuscript. PM Aponte contributed to conception and design of the study; contributed to analysis and interpretation of data; and drafted the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Histoprocessor, Slee Medical, Mainz, Germany.
b.
Microtome Cut 6062, Slee Medical, Mainz, Germany.
c.
Synthetic Canada balsam, Biopack, Buenos Aires, Argentina.
d.
Cryostat, Slee Medical, Mainz, Germany.
e.
CryoGlue, Slee Medical, Mainz, Germany.
f.
Digital video camera, Tucsen Imaging Technology, Beijing, China.
g.
SPSS Statistics for Windows version 17.0, SPSS, Chicago, IL.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study received financial support from SENESCYT—Project Prometeo and AGROCALIDAD, Ecuador.
