Abstract
An adult nematode was grossly identified in the meninges of a Malayan sambar (Rusa unicolor equina), with numerous microfilariae associated with encephalitis and vasculitis on histopathology. The nematode was confirmed to be Elaeophora schneideri by sequencing a portion of the 18S rRNA gene. Our report highlights the potential for aberrant migration of E. schneideri in exotic deer species and the use of advanced testing to specifically identify this metazoan parasite, avoiding misidentification of Parelaphostrongylus tenuis.
Elaeophorosis, caused by the filarial nematode Elaeophora schneideri (superfamily Filarioidea, family Onchocercidae), can be a significant health complication for both domestic and wild ruminants. 2 Mule deer (Odocoileus hemionus) and black-tailed deer (Odocoileus hemionus columbianus) are considered to be the natural definitive hosts for E. schneideri.2,9 Transmission of E. schneideri occurs during a bite and acquisition of a blood meal by an infected Tabanus or Hybomitra horsefly (order Diptera, family Tabanidae). Once inside the host, E. schneideri larvae migrate through the circulatory system, where they eventually reach the carotid arteries or cephalic arterial system and mature into adult nematodes.2,11 Infections of atypical hosts, including red deer (Cervus elaphus), elk (Cervus canadensis), moose (Alces alces), Barbary sheep (syn. aoudad; Ammotragus lervia), bighorn sheep (Ovis canadensis), sika deer (Cervus nippon), and domestic sheep and goats, can result in the development of elaeophorosis.1,3–5,14–18,21
Elaeophorosis is generally caused by the adult nematodes lodging within the cephalic arteries, restricting blood flow and leading to ischemic necrosis, blindness, and/or sublingual oral impactions. 2 In previous reports, E. schneideri nematodes have been identified in the internal maxillary arteries, heart, thoracic and abdominal arteries, femoral and digital arteries, and leptomeningeal arteries of atypical hosts. 1 E. schneideri has been identified in sambar deer in Florida previously. 6 However, we describe in detail the subclinical disease caused by E. schneideri in the meninges of a Malayan sambar, including ancillary tests used to definitively differentiate the parasite from the neurotropic parasite Parelaphostrongylus tenuis.
A captive, 12-year-old male Malayan sambar (Rusa unicolor equina) had a 2-week history of progressive neurologic signs, including left ear and facial droop and minor drooling. Additional clinical signs and physical examination findings included ticks in both ears, grade 2 (on a scale of 1–5) lameness of the right rear leg, packing food in the left side of the mouth, corneal ulceration and edema in the left eye, facial swelling, isolating behaviors, and lethargy. Acute trauma from an enclosure mate was the suspected cause of the clinical signs. Given the poor prognosis, the animal was euthanized and submitted for postmortem examination. The animal was in poor body condition. There were multiple skin abrasions on the body, blood in the left external ear canal, and a left temporal bone skull fracture. Between the skull and the brain, dorsolateral to the left cerebrum and just dorsal to the fracture site, a portion of a single nematode was found surrounded by tan and red, firm tissue. Because of the disruption of the meninges as a result of the nearby fracture, the exact layer of the affected meninges was not determined, but the nematode appeared to be outside the dura mater grossly. The portion of nematode recovered was 3 cm long with 1 tapered end. Blood vessels of the head were examined grossly and did not contain nematodes. Histologic sections of the tissue surrounding the adult nematode and adjacent fracture site contained multiple disrupted blood vessels surrounded by focal granulomatous and eosinophilic inflammation as well as a subacute fibrovascular proliferation interpreted as granulation tissue secondary to both the skull fracture and free nematode. Blood vessels contained fibrin thrombi. There was hemorrhage in the surrounding tissues, but the majority of the leptomeninges and brain were histologically normal. Both the gray and white matter of the brain had mild multifocal granulomatous encephalitis and vasculitis with occasional microfilariae (Figs. 1, 2). Microfilariae were not identified in the cerebellum or any other tissue. The carotid arteries and adult nematode were not examined histologically.
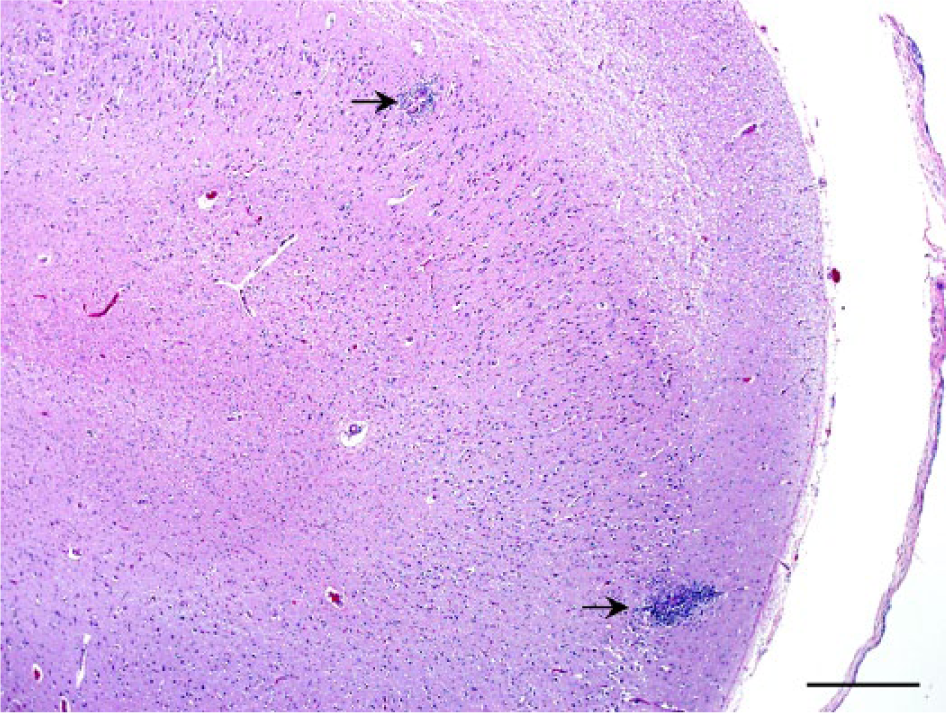
Section of the hippocampus of a Malayan sambar (Rusa unicolor equina). Occasional blood vessels are disrupted by macrophages, lymphocytes, and occasional multinucleate giant cells surrounding rare microfilariae (arrows). Hematoxylin and eosin. Bar = 500 µm.
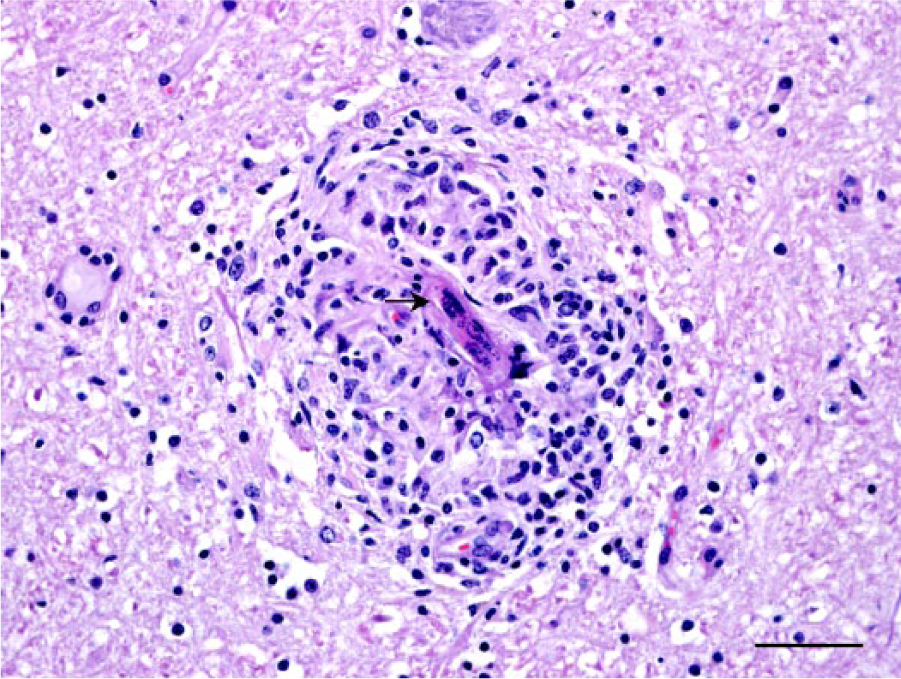
Section of midbrain of a Malayan sambar (Rusa unicolor equina), ventral to the mesencephalic aqueduct. A microfilaria is folded on itself (arrow), disrupting a blood vessel, and is surrounded by moderate lymphohistiocytic inflammation. Hematoxylin and eosin. Bar = 50 µm.
The nematode was examined using a dissection microscope. The size and morphologic characteristics of the nematode, including a coiled appearance and width of ~1 mm, were consistent with E. schneideri. However, given the absence of an entire adult nematode needed to obtain length measurements and examine the tail for a characteristic terminal cone-shaped protuberance, the identity of the nematode could not be confirmed. Thus, molecular testing was used to identify the species of the adult nematode isolated from the sambar.
Given the scarcity of reference sequences from cervid nematode parasites in GenBank, representative E. schneideri, Onchocerca cervicalis, and P. tenuis nematodes were used to aid in identification (Table 1). These reference nematodes were identified based on morphology. A portion of each nematode was excised using a clean razor blade, and the DNA was extracted a according to the manufacturer’s instructions. A DNA extraction negative control was included to ensure that no contamination occurred during the extraction procedure. Polymerase chain reactions (PCR) were performed as previously described and targeted the 18S rRNA gene using the Nematoda-specific primers Nem18SF (5′-CGCGAATRGCTCATTACAACAGC-3′) and Nem18SR (5′-GGGCGGTATCTGATCGCC-3′). 7 Briefly, each reaction was prepared with 12 μL of PCR master mix, b 1 μL of each primer (5 μM), 8 μL of nuclease-free water, and 3 μL of DNA template. A nuclease-free water negative control was included to detect contamination during the PCR procedure. All PCR products were amplified using the following thermocycler conditions: an initial denaturation at 94°C for 5 min; 35 cycles of amplification (94°C for 30 s, 54°C for 30 s, 72°C for 1 min); followed by a final extension at 72°C for 10 min. Portions of the PCR products were examined via electrophoresis on 1% agarose gel. The PCR products were purified a and sequenced at the University of Tennessee (UT Genomic Core, Knoxville, Tennessee). Sequence chromatograms were edited. c The 858-bp consensus sequences were compared against the NCBI nucleotide database and aligned with the 18S sequences from known ungulate nematode parasites from GenBank and our reference nematodes. 19 Neighbor-joining phylogenetic trees were constructed to determine the closest relative of the unknown nematode. Phylogenetic analysis of the 18S rRNA gene revealed a high identity to E. schneideri (Fig. 3). All nematode 18S consensus sequences, including the nematode isolated from this sambar, were deposited in GenBank (Table 1).
18S analysis of several nematodes found in cervids. Reference nematodes were morphologically identified prior to molecular testing. All nematodes were subjected to DNA extraction and polymerase chain reaction, sequenced, and consensus sequences deposited in GenBank.

Isolated from the sambar deer detailed in our study.
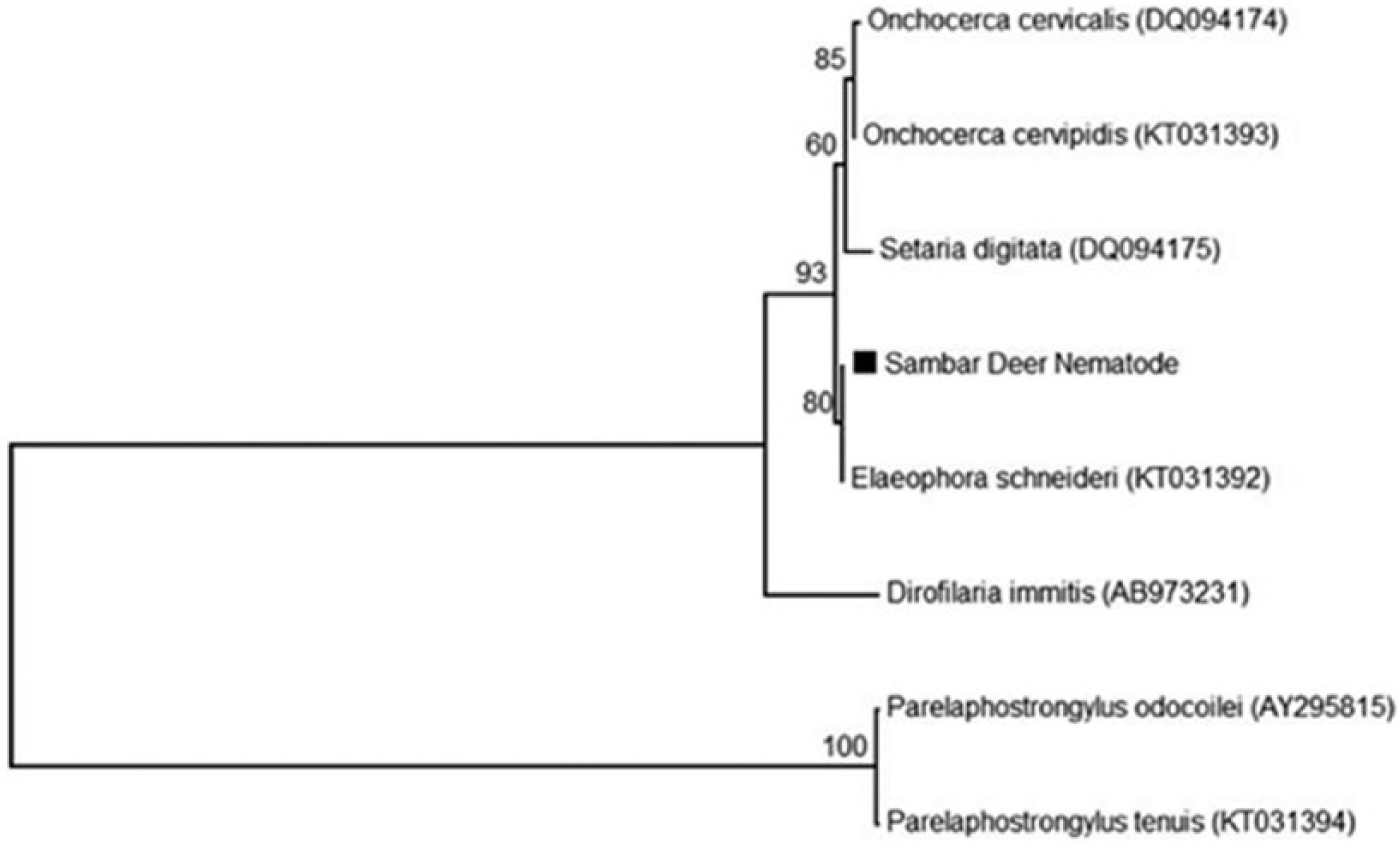
Phylogenetic consensus tree of partial 18S sequences (858 bp) from reference ungulate parasitic nematodes and a nematode from a Malayan sambar (Rusa unicolor equina). Evolutionary relationships were inferred using the neighbor-joining method. The percentages of replicate trees in which the associated taxa clustered in the bootstrap test (1,000 replicates) are shown above the branches. Each reference nematode is labeled with its GenBank accession in parentheses.
The sambar is a large deer species native to southern Asia. 13 Infection with E. schneideri in deer species other than the mule deer and black-tailed deer is related to the tabanid’s nondiscriminatory feeding behavior. 2 Even though deer are considered to be the reservoir and tend to have fewer clinical signs associated with infection compared to elk and moose, 4 there are reports of E. schneideri migrating to vessels other than the carotid, including the internal maxillary arteries, heart, thoracic and abdominal arteries, and femoral and digital arteries.1–3 In our case, the adult nematode was found associated with inflammation and disrupted blood vessels over the cerebrum near the skull fracture site. It is most likely that the trauma from an enclosure mate caused disruption of the vessel containing the nematode and incited the inflammatory reaction. There were no reports of neurologic signs prior to the suspected trauma; however, this animal resided in a large, mixed-species enclosure where prolonged individual observations can be challenging, and mild clinical signs resulting from this nematode infection or the microfilariae might not have been noticed.
Given that Elaeophora sp. are known to live in blood vessels without any clinical signs in some species, it is unknown how long this nematode was in the calvaria and whether it was within a vessel. The pre-patent period is estimated to be 5.5 months in the definitive mule deer host, 11 and although the exact lifespan for E. schneideri is unknown, other filaroid nematodes can survive 2–10 years. 8 There are reports of elaeophorosis affecting other vessels, including within the brain, and even causing large cavitating lesions in the cerebrum as a result of hypoxia; such changes were not seen in this case.
The 18S rDNA sequences and the presence of microfilariae identify this nematode as the filaroid E. schneideri, which is endemic to western North America, including California, where the well-adapted definitive hosts, mule deer and black-tailed deer, reside. Previous studies examining the prevalence of E. schneideri in California found 4–44% of mule deer 10 and 78% of black-tailed deer 20 harbored E. schneideri. Also, nematodes consistent with E. schneideri have been observed in the carotid arteries of native mule deer in southern California examined at the Wildlife Disease Laboratories in San Diego (unpublished observation). This sambar was born at the San Diego Zoo Safari Park and resided there for its entire life. Thus, this animal most likely acquired E. schneideri locally, possibly from the bite of an infected horsefly, which is common in southern California.
Nematodes found in the dorsal meninges may be confused with P. tenuis, which is the most likely differential for a nematode in the brain. However, P. tenuis was considered less likely because this deer lived its entire life in southern California, which is not included in the range of P. tenuis. 12 Our report highlights the importance of looking for E. schneideri in cervids. In addition, it emphasizes the need to perform molecular parasitologic confirmation for partial nematodes recovered from brain or meninges to ensure correct identification and the potential implementation of measures to prevent further transmission to other animals within a collection.
Footnotes
Acknowledgements
We thank Misty Bailey with assistance with manuscript formatting and proofreading.
Authors’ contributions
J Bernard, C Grunenwald, and R Gerhold contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. IH Stalis contributed to design of the study; contributed to analysis and interpretation of data; and drafted the manuscript. M Varney contributed to conception of the study and to acquisition of data. J Zuba contributed to conception of the study and to analysis and interpretation of data. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
DNeasy blood and tissue kit, QIAquick PCR purification kit, Qiagen Inc., Valencia, CA.
b.
Dream Taq PCR master mix, Life Technologies, Carlsbad, CA.
c.
Sequencher 4.1 software, GeneCodes Corp., Ann Arbor, MI.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
