Abstract
An ~21-year-old female Barbary sheep (Ammotragus lervia) died spontaneously following a lengthy episode of difficulty in walking. An ~6 × 3 × 3 cm, unilocular cystic growth was found in the cranioventral thorax. The fibrotic cystic wall, lined by a single layer of flattened to cuboidal epithelial cells, was invaginated and partially encircled solid masses of fusiform neoplastic cells with multiple intratumoral cystic structures. The fusiform neoplastic cells were intensely positive for cytokeratin (CK) and partially positive for α–smooth muscle actin and vimentin, but negative for thyroid transcription factor–1 (TTF-1) and neuron-specific enolase (NSE). The intratumoral cysts were lined by CK-positive but TTF-1– negative, NSE-negative, flattened, cuboidal to columnar epithelial cells, suggestive of cystically dilated medullary duct epithelium-derived structures. Based on the location and histopathologic findings of the growth, concurrent spindle-cell thymoma and thymic cysts was diagnosed. We also discuss the correlation between thymic cysts and thymoma and review the literature of thymomas in ovine and wildlife species.
Keywords
Thymomas are neoplasms derived from thymic epithelial cells with various degrees of benign lymphoid proliferation. 9 Although thymomas are considered an uncommon neoplasm, cases have been reported in different animal species.2,9 Thymic cyst is an extremely rare condition, characterized as a unilocular or multilocular cystic structure(s) lined by stratified squamous, flattened to cuboidal epithelium with the concurrent presence of thymic tissue at least focally within the cystic wall11,27; it must be distinguished from cystic degeneration of thymoma, embryonic remnant of ectopic parathyroid gland or thyroid gland, and thymic branchial cleft cyst.11,27 To our knowledge, only 1 feline case of thymic cyst has been reported, but no concurrent thymic cysts and thymoma has been reported in animals. Here, we describe a case of concurrent spindle-cell thymoma and thymic cysts in a captive Barbary sheep (syn. aoudad; Ammotragus lervia), discuss the correlation between thymic cysts and thymoma, and review the literature of thymomas in ovine and wildlife species.
An ~21-year-old female Barbary sheep had suffered from difficulty in walking and had been isolated from other animals for a long period of time. She gradually became emaciated, weak, and reluctant to walk, and died spontaneously. Grossly, an ~6 × 3 × 3 cm, well-demarcated growth was found in the cranial thorax. The growth had a smooth surface with tight adhesion to the ventral aspect of the adjacent right cranial lung lobe (Fig. 1A). The cut sections of the growth showed a clear fluid-filled cystic structure of which the wall was irregular in thickness and consisted of white, yellow-to-pink solid tissues with some soft gelatinous substance (Fig. 1B). The heart displayed marked serous atrophy of fat in the coronary groove. There was generalized depletion of subcutaneous and visceral adipose tissue and moderate atrophy of the skeletal muscle masses. Both carpal joints were severely swollen and contained a moderate amount of clear, straw-colored joint fluid. No changes were found in other organs.
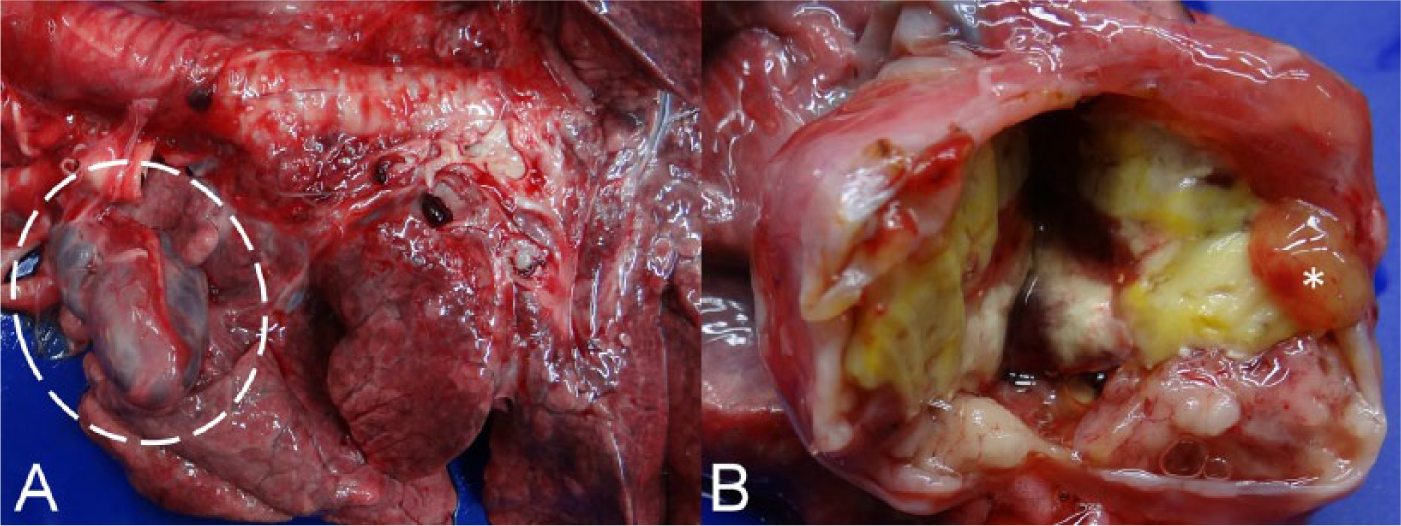
Gross appearance of the cranial thoracic mass from a Barbary sheep (Ammotragus lervia).
Representative tissue samples were collected, fixed in 10% neutral-buffered formalin, processed routinely, sectioned at 5 μm, and stained with hematoxylin and eosin and periodic acid–Schiff (PAS). For immunohistochemical (IHC) staining, deparaffinized tissue sections were heated in retrieval solution, a incubated in 2.5% normal goat serum in Tris-buffered saline (TBS) solution for 30 min at room temperature, and then incubated with each of the monoclonal antibodies against cytokeratin b (CK; diluted 1:400), vimentin c (diluted 1:400), neuron-specific enolase d (NSE; diluted 1:800), and CD3 e (diluted 1:400) at 4°C overnight. The sections were then treated with 3% hydrogen peroxide followed by peroxidase-conjugated secondary antibodies f for 60 min at room temperature. After exposure to 2% diaminobenzidine g for 3 min, the slides were counterstained with hematoxylin for 1 min. In addition, IHC staining for thyroid transcription factor–1 h (TTF-1; diluted 1:400) and α–smooth muscle actin i (α-SMA; diluted 1:100) was performed in an automated system. j All of the internal controls from the normal tissues of this Barbary sheep for the monoclonal antibodies used in the present case were positive, indicative of good quality of IHC staining for the various targeted cell markers.
Microscopically, the cyst wall was composed of layers of fibrous connective tissue and lined by a single layer of flattened-to-cuboidal epithelial cells on the inner surface, which became focally invaginated and partially encircled the masses of fusiform neoplastic cells (Fig. 2A). The fibrotic cyst wall contained small foci of residual thymic tissue, including small mature lymphocytes and epithelial cells with occasional formation of Hassall corpuscles. In addition, scattered small nests of fusiform neoplastic cells were also found in the cyst wall. In the solid masses, the fusiform neoplastic cells were arranged in an interwoven or storiform pattern along with variable infiltration of lymphocytes and eosinophils and formation of multiple variably sized and shaped small intratumoral cysts (Fig. 2B). The fusiform neoplastic cells had a moderate amount of indistinctly bordered, light eosinophilic cytoplasm, and a variably sized, oval, elongated-to-fusiform nucleus with stippled chromatin and inconspicuous nucleolus. No mitotic figures were seen. IHC staining revealed that the fusiform neoplastic cells were strongly positive for CK and displayed multifocal weak-to-moderate positivity for vimentin and α-SMA (Fig. 3A–3C), but were negative for TTF-1 and NSE. Most of the infiltrating lymphocytes were CD3+, consistent with T lymphocytes. The intratumoral cysts were lined by a single layer of flattened, cuboidal-to-columnar epithelial cells with formation of occasional papillary projections; the cysts were filled with PAS-positive or PAS-negative proteinaceous secretion admixed with scattered lymphocytes, macrophages, eosinophils, and cell debris. The epithelial cells of the intratumoral cysts were positive for CK (Fig. 3D), but were negative for TTF-1 and NSE. The soft gelatinous substance seen grossly was composed of a myxoid matrix. Based on the location, cell morphology, and results of IHC staining of the tumor growth, spindle-cell thymoma with multiple intratumoral small cysts was diagnosed. Furthermore, the neoplasm was located inside a large, thymic tissue–contained unilocular cyst, suggestive of a thymic cyst. Thus, concurrent spindle-cell thymoma and thymic cysts were the final diagnoses.
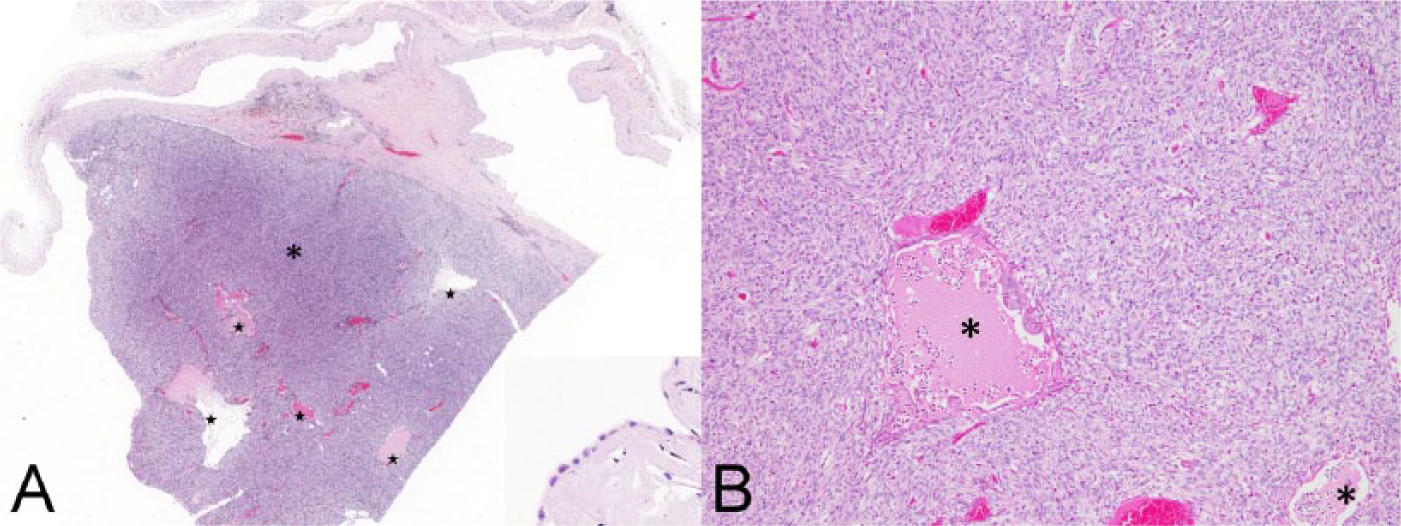
Histologic appearance of the thymic cyst with spindle-cell thymoma from a Barbary sheep (Ammotragus lervia).
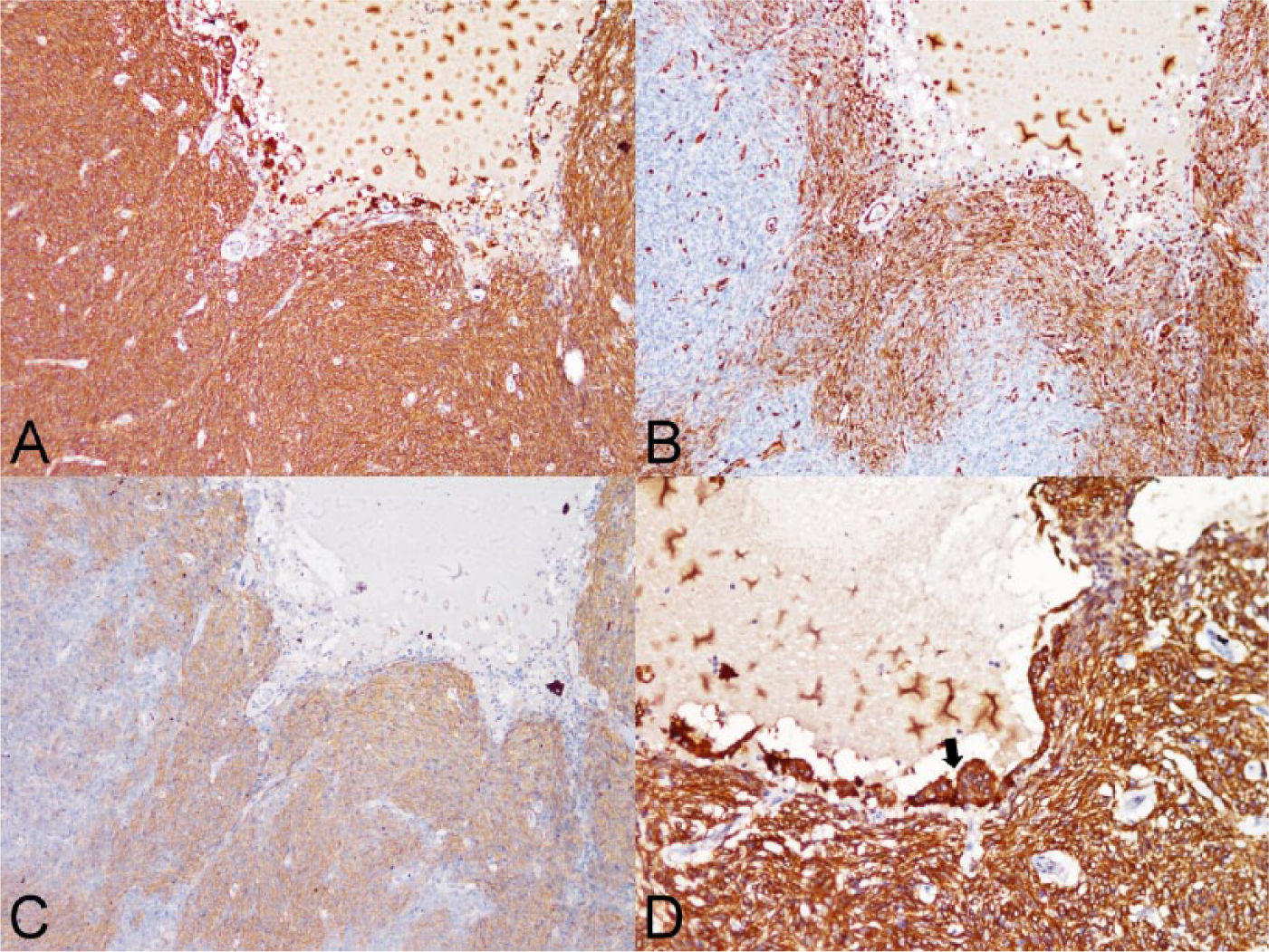
Immunohistochemical staining of the spindle-cell thymoma and intratumoral acquired thymic cysts from a Barbary sheep (Ammotragus lervia).
In animals, thymomas are uncommon but have been reported in the cat, dog, goat, sheep, cattle, horse, rat, rabbit, crab-eating macaque (Macaca fascicularis), African spot-necked otter (Lutra maculicollis), Java sparrow (Padda oryzivora), polar bear (Ursus maritimus), and Siberian tiger (syn. Amur tiger; Panthera tigris altaica).2–8,10,11,13,16–24,27 In animals, thymomas are classified as lymphoid, epithelial, and mixed types on the basis of the predominant cell population of the neoplasm. 9 The epithelial thymoma can be further classified as clear-cell and spindle-cell subtypes based on the histologic features of the neoplastic cells. 9 In the present case, the fusiform neoplastic cells were positive for CK but negative for NSE and TTF-1; therefore, the possibility of a neoplasm derived from thyroid gland or mesenchymal tissue such as nervous tissue could be ruled out. Based on the cell morphology and CK positivity of the neoplastic cells, a diagnosis of spindle-cell thymoma was made.
A summary of the reported thymomas in ovine and wildlife species is presented in Table 1, including the information of age, sex, histologic type, and the status of capsular invasion.2,6,8,10,17–20,22 To our knowledge, there have been 4 thymoma cases reported in female sheep, which were categorized as lymphoid type in 2 cases and epithelial type in the other 2 cases, but there was none of the spindle-cell type (Table 1).18,22 On the other hand, there have been 21 thymoma cases reported in goats (Table 1), and thymomas seem to be more common in dairy goats (Capra hircus) than other animal species.6,8,17,20 In the cases of goat thymoma, lymphoid thymoma was the most common histologic type (Table 1), and only 1 mixed thymoma and 2 spindle-cell thymomas have been reported in goats.6,8,17,20 The spindle-cell thymomas of goats were mainly composed of fusiform epithelial cells arranged in interlacing bundles and whorls with scattered lymphocytes,6,20 similar to our case.
Thymomas reported in ovine and captive wildlife species.*
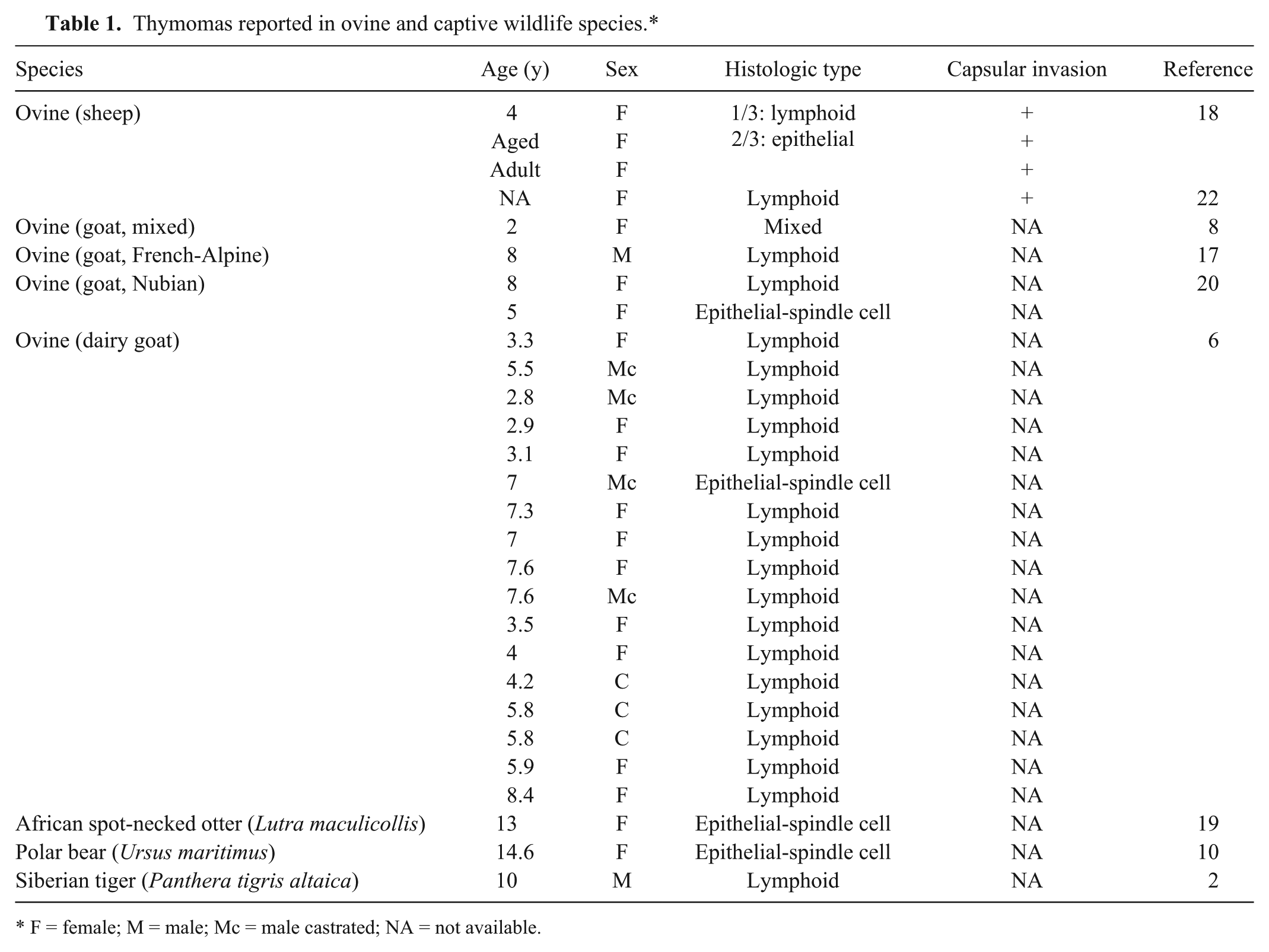
F = female; M = male; Mc = male castrated; NA = not available.
The fusiform neoplastic cells in the present case displayed multifocal weak-to-moderate positivity for α-SMA, and a similar phenomenon has been observed in a previous study of human spindle-cell thymoma. 28 The IHC staining result of vimentin in the present case also displayed a similar multifocal weak-to-moderate positivity. Performing IHC staining for vimentin has only been reported in a few animal thymoma cases.2,8 The thymoma of a Siberian tiger showed poor cross-reactivity for vimentin. 2 The spindle-cell thymoma from a mixed-breed goat was completely negative for vimentin. 8
Human metaplastic thymomas are biphasic and characterized by the concurrent presence of islands of polygonal-to-short fusiform cells and bundles of elongated fusiform cells. 12 The polygonal-to-short fusiform cells were consistently positive for CK, but the elongated fusiform cells had variable IHC features, which might be weak or negative for CK but positive for vimentin and α-SMA. This phenomenon has been thought to be associated with the mesenchymal metaplasia of neoplastic cells, also known as the epithelial–mesenchymal transition (EMT).12,14 EMT is considered to be a malignant progression, which could facilitate the invasion, spread, and metastasis of neoplastic cells. 14 In our case, the fusiform neoplastic cells were monomorphic, but some of them were concurrently positive for CK, vimentin, and α-SMA. Therefore, we speculated that the neoplastic cells might have gone through the process of EMT. Furthermore, although no neoplastic emboli and metastasis were found in the present case, the presence of neoplastic cells in the cyst wall may be suggestive of malignant potential. In the previously reported ovine thymoma cases, capsular invasion without metastasis was a common finding, but no IHC stains were applied.18,22 The correlation between the spindle-cell thymoma and vimentin positivity is still controversial.1,15 Therefore, although some ovine thymoma cases were described as spindle-cell thymomas, the presence of EMT cannot be ascertained solely by cell morphology.
To our knowledge, there have been only 3 reported thymoma cases in captive wildlife species, including a Siberian tiger, a polar bear, and an African spot-necked otter (Table 1).2,10,19 In the case of the African spot-necked otter, no clinical signs were noted, but the Siberian tiger and polar bear cases were accompanied by severe myasthenia gravis, characterized by the presence of progressive muscle wasting, weakness, acute-onset collapse, and elevated serum acetylcholine receptor antibodies.2,10,19 The recorded maximal longevity of captive Barbary sheep was 21.7 years, and the Barbary sheep of the present case was 20 years and 9 months old, which can be considered as an extremely old individual. Although the difficulty in walking, weakness, and moderate atrophy of skeletal muscle masses with bilateral carpal joint lesions were likely aging or arthritic changes, the possibility of thymoma-associated myasthenia gravis cannot be completely excluded.
Cystic degeneration is a frequent finding in thymomas and results from dilation of perivascular space without an epithelial lining.11,26,27 Nevertheless, the intratumoral cysts in the present case were lined by a layer of non-ciliated epithelial cells, which might derive from the embryonic remnants of ectopic parathyroid gland or thyroid gland, from the branchial cyst of the endoderm of the third and fourth pharyngeal pouches, or from the cystic dilation of medullary duct epithelium-derived structures.8,11,25,27 In embryogenesis, the thyroid gland, parathyroid gland, and thymus all originate from the pharyngeal pouch; therefore, the intratumoral cysts of thymic tissue and/or neoplasm could be ectopic thyroid or parathyroid tissues.8,25 The ectopic thyroid and parathyroid tissues usually occur as follicular structures with PAS-positive secretion, and the ectopic epithelial cells of thyroid and parathyroid tissues should be positive for TTF-1 and NSE, respectively. However, the secretion in the intratumoral cysts of our case was inconsistently PAS-positive, and the epithelial cells were negative for TTF-1 and NSE. Hence, the possibility of ectopic thyroid or parathyroid tissues is unlikely. On the other hand, branchial cysts are lined by ciliated columnar epithelial cells,11,27 and thus the possibility of a branchial cyst could also be ruled out in the present case.
The medullary duct epithelium forms tubular structures in the mesenchymal septa of the fetal thymus, which then give rise to Hassall corpuscles. 26 Cystic dilation of the medullary duct epithelium-derived structures, including Hassall corpuscles and vestigial tubular structures, may form epithelium-lined cystic structures with accumulation of proteinaceous secretion, cellular debris, and infiltration of lymphocytes, macrophages, and eosinophils, 26 which are compatible with the intratumoral cysts seen in our case. The cystically dilated medullary duct epithelium-derived structures are also known as acquired thymic cysts.
Thymic cysts are a relatively uncommon condition in humans and extraordinarily rare in animals; to our knowledge, only 1 feline case has been reported in the veterinary literature. 3 The thymic cyst in the feline case was lined by a flattened, non-ciliated epithelium, and the cystic wall was composed of fibrous connective tissue with multifocal remnants of thymic tissue. 3 Generally, thymic cysts are classified as congenital or acquired based on the mechanism of formation. Congenital thymic cysts are associated with remnants of the thymopharyngeal ducts, and characterized as a clear fluid-filled unilocular structure, in which the cystic wall contains thymic tissue and is lined by stratified squamous, flattened-to-cuboidal epithelium without inflammation. 27 In contrast, acquired thymic cysts are associated with various inflammatory and neoplastic conditions; they are generally multilocular and have turbid fluid and thick cystic walls with marked inflammation and fibrosis. 26 In our case, the mass was a large unilocular cyst without marked inflammation, which is compatible with a congenital thymic cyst. In addition, there were multiple epithelium-lined small cystic structures within the thymoma, which were considered acquired thymic cysts originating from the medullary duct epithelium. Although the multiple small cysts within the thymoma could also be part of an invaginated cystic wall from the congenital thymic cyst, the presence of mixed inflammatory cells favors the hypothesis of acquired thymic cysts. Furthermore, the spindle-cell thymoma was encapsulated by the fibrous connective tissue from the invaginated cystic wall of the congenital thymic cyst, suggesting that the thymoma might have originated from the wall of the congenital thymic cyst. It is thus considered that the time and mechanism for the formation of the large thymic cyst and multiple small cysts within the thymoma could be different. If the thymoma indeed arose from a congenital thymic cyst, the multiple small cysts within the thymoma could be the consequence of the development of the thymoma, which further supports the speculation of acquired thymic cysts. Therefore, the possibility of coexisting mixed congenital and acquired thymic cysts should be considered.
Footnotes
Authors’ contributions
WT Li contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; and critically revised the manuscript. HW Chang, CR Jeng, CH Liu, and FI Wang contributed to conception and design of the study, and contributed to analysis and interpretation of data. LJ Chang contributed to conception of the study, and contributed to acquisition and analysis of data. VF Pang contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Trilogy, Cell Marque, Rocklin, CA.
b.
Mouse monoclonal antibody (clone AE1/AE3), Dako Denmark A/S, Glostrup, Denmark.
c.
Mouse monoclonal antibody (clone Vim 3B4), Dako Denmark A/S, Glostrup, Denmark.
d.
Mouse monoclonal antibody (clone BBS/NC/VI-H14), Dako Denmark A/S, Glostrup, Denmark.
e.
Mouse monoclonal antibody (clone F7.2.38), Dako Denmark A/S, Glostrup, Denmark.
f.
Dako Real Envision-HRP (rabbit/mouse), Dako Denmark A/S, Glostrup, Denmark.
g.
Real DAB+ chromogen, Dako Denmark A/S, Glostrup, Denmark.
h.
Mouse monoclonal antibody (clone SPT24), Novocastra Laboratories Ltd., Newcastle upon Tyne, UK.
i.
Mouse monoclonal antibody (clone ASM-1), Novocastra Laboratories Ltd., Newcastle upon Tyne, UK.
j.
BondMax, Leica Microsystems Inc., Bannockburn, IL.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
