Abstract
We describe a hernia of the swim bladder, with a concurrent mycotic granulomatous inflammation, and carcinoma of the swim bladder in a wild mullet (Mugil cephalus) referred for an exophytic dorsal mass. Grossly, the mass was white, soft, and composed of multiple cystic gas-containing chambers connected by a funnel-shaped tissue segment to the coelomic swim bladder. Histologically, the mass was characterized by cysts of variable size, multifocally contiguous with the subepithelial rete mirabile, supported by abundant fibrous tissue. The skin covering the herniated swim bladder was focally ulcerated and replaced by abundant granulation tissue in which multiple scattered granulomas centered on pigmented fungal hyphae were observed. These granulomas were also seen in the remaining coelomic portion of the swim bladder as well as in the spleen, perivisceral pancreas, and peritoneal adipose tissue; the fungus was molecularly identified as Cladosporium spp. Focally, arising from the herniated swim bladder epithelium, an unencapsulated poorly demarcated, moderately cellular neoplasm, composed of islands, lobules, and acini of neoplastic epithelium, was found.
The swim bladder is the most important buoyancy organ in teleosts and helps fish to balance hydrostatic pressure and reduce the energetic cost of swimming. The anatomy, as well as the pathology of this organ, has not been commonly described.7,15 Developmental defects of the swim bladder have been reported in intensively reared larvae and juveniles of Atlantic cod (Gadus morhua) and Atlantic salmon (Salmo salar).12,17 Swim bladder herniation and dilation have been described in pet fish by radiographic and autopsy investigations 6 ; herniation has also been reported in a white mullet (Mugil curema) caught in the Colorado River, Texas, and in a specimen of catla (Catla catla); however, in both cases, the reports were limited to gross description of the lesions.5,10 Epithelial neoplasms of this organ have not been frequently reported; adenomas and adenocarcinomas have been described in Japanese medaka (Oryzias latipes) and guppy (Poecilia reticulata), as well as in juvenile cod.4,17 Virally induced mesenchymal neoplasms of swim bladder, specifically leiomyosarcoma, have been described in Atlantic salmon by several authors.2,3,9
We describe 3 significant concurrent lesions associated with the swim bladder in a wild mullet (Mugil cephalus). These include: 1) a hernia of the swim bladder, 2) a mycotic granulomatous inflammation of herniated tissue and visceral organs, and 3) a concurrent carcinoma arising from the swim bladder herniated epithelium.
A wild mullet, showing a dorsolateral mass adjacent to the dorsal fin, was caught by an angler in the Caldoli River, which flows into Lesina Lake (Puglia, Southern Italy). The fish was brought to the aquaculture facility of Cesenatico, Department of Veterinary Medical Sciences (University of Bologna, Italy). The animal was kept alive in a 2-m3 tank (water temperature 28°C; salinity 3 g/L) for 2 months; during this period, the fish displayed a tendency to float to the surface and difficulty in maintaining its position in the water column. No other clinical signs were evident. No therapeutic interventions were performed. After 2 months, the animal accidentally died jumping out of the tank; a thorough autopsy was performed. Serial transverse sections of the body comprising the mass and the underlying skin, muscle, vertebrae, and kidney were obtained and photographed. Other organs (gills, liver, stomach, intestine, spleen, and gonads) were also sampled. Cytologic smears were obtained from the mass and stained with May-Grünwald/Giemsa. All tissues were formalin-fixed, decalcified when necessary, paraffin-embedded, and processed for routine histology. Two mullets of approximately the same size, obtained from a commercial supplier, were used as control specimens to describe the macroscopic and histologic features of the normal swim bladder, as data on the anatomy of this species are scarce in the literature. 15 One control was cut cranially to the dorsal fin in order to obtain a normal reference of the topography of the swim bladder, and notochord and epaxial and hypaxial muscles; the other was opened to show coelomic organs that were removed to visualize the swim bladder in the lateral view. The swim bladder is located ventrally to the spinal cord and kidney, retroperitoneal in position, and covered ventrally with black peritoneum. Along the dorsal side, there is the oval; anteriorly, the swim bladder bifurcates into 2 cornua. From the dorsal surface of the esophagus arises the pneumatic duct. The gas gland is found on the anteroventral wall where it divides, passing into each cornua of the swim bladder. Two segments (cranial and caudal) of the swim bladder were sampled and processed as described above. Histologically, the normal swim bladder is composed of 2 layers of elastic fibers (external and internal tunica); the gas gland lumen is lined by glandular epithelium covered by a squamous epithelial lining. The rete mirabile is seen in groups of small fan-shaped tufts, as described in the literature for this species. 15
The affected fish was an immature male, 25 cm in length and weighing 150 g; body condition was normal. Grossly, the mass was white, 3 cm in diameter, glistening, and soft. It was formed by multiple cystic gas-containing chambers (herniated swim bladder; Fig. 1A, 1B), which were connected by a funnel-shaped tissue segment to the remaining portion of the organ in the coelomic cavity, which separated the epaxial muscles (hernia orifice). The coelomic portion of the swim bladder was hypoinflated. The vertebrae and kidney were not involved in the pathologic process or displaced by it (Fig. 2). Other organs were grossly unremarkable. Histologically, 50% of the exophytic mass was formed by herniated swim bladder sustained by abundant fibrous stroma, 40% was edematous granulation tissue with multifocal scattered granulomas, and the remaining 10% was composed of focal neoplasms arising from the epithelium of the herniated swim bladder.
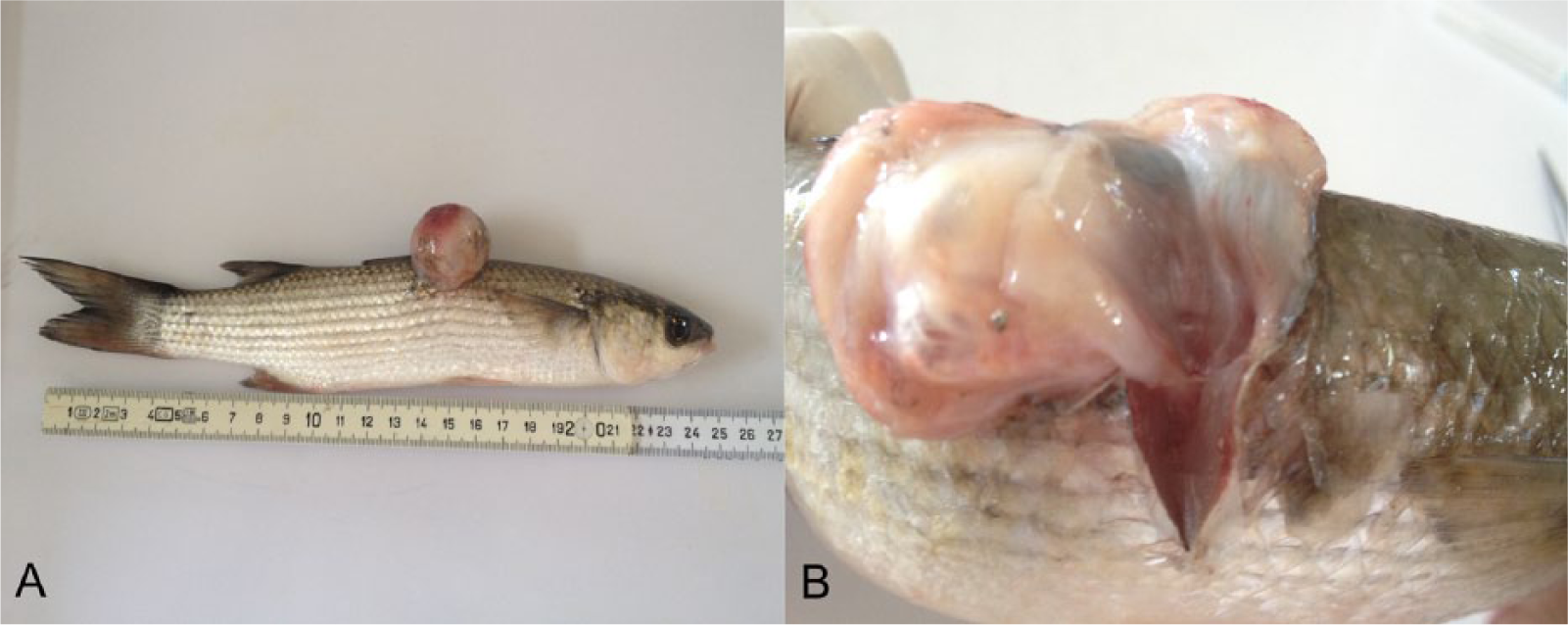
Mugil cephalus.
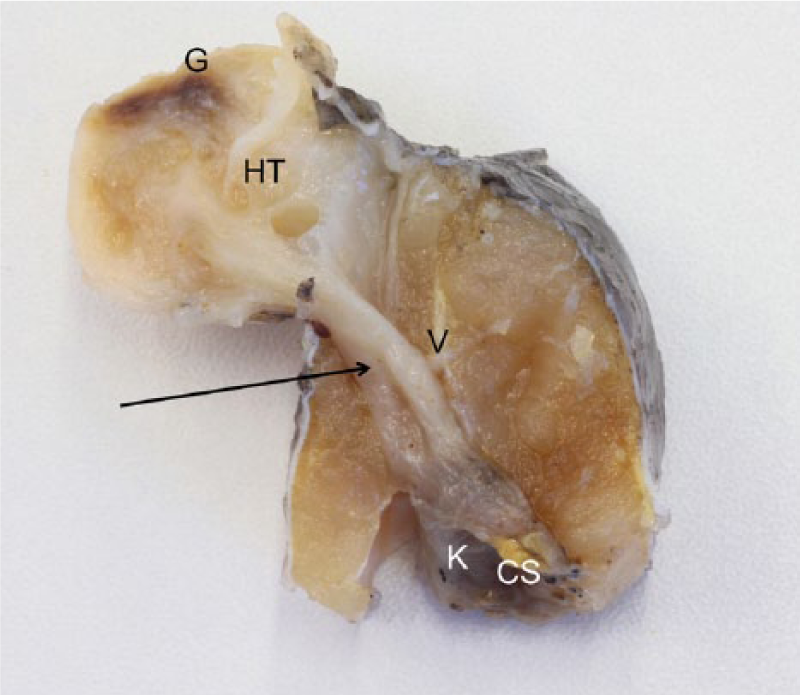
Mugil cephalus. Cranial view of transverse section of a swim bladder hernia (aerocystocele) in a fixed specimen. Herniated tissue (HT) shows cystic spaces and a brown area corresponding to ulcerated skin and granulation tissue (G). This herniated tissue is connected through a funnel-shaped tissue segment (arrow) to the swim bladder of the coelomic cavity (CS; hypoinflated). Vertebral body (V) and kidney (K; covered) are not involved in the pathologic process or displaced by it.
The herniated swim bladder tissue was characterized by round cysts of variable size (0.2–3 mm), lined by a single layer of attenuated epithelium, which was multifocally contiguous with the subepithelial rete mirabile (Fig. 3A). An abundant amount of fibrous tissue (fibroplasia) supported the hernia, dissecting and compressing the epaxial musculature (hernia orifice) causing muscular fibrosis and atrophy. This fibrous tissue was contiguous with the coelomic portion of the swim bladder. The kidney and vertebral bodies were not involved, and they showed a normal topography. The skin, which was elevated by the herniated swim bladder, was focally ulcerated, subjacent to which there was abundant granulation tissue with multifocal scattered granulomas centered on pigmented fungal hyphae. Granulomas had extensive central regions of laminated, acellular debris, rare hyphae, and foci of mineralization, surrounded by thick concentric walls of epithelioid macrophages and variable fibrosis. A vegetal foreign body was also detected.
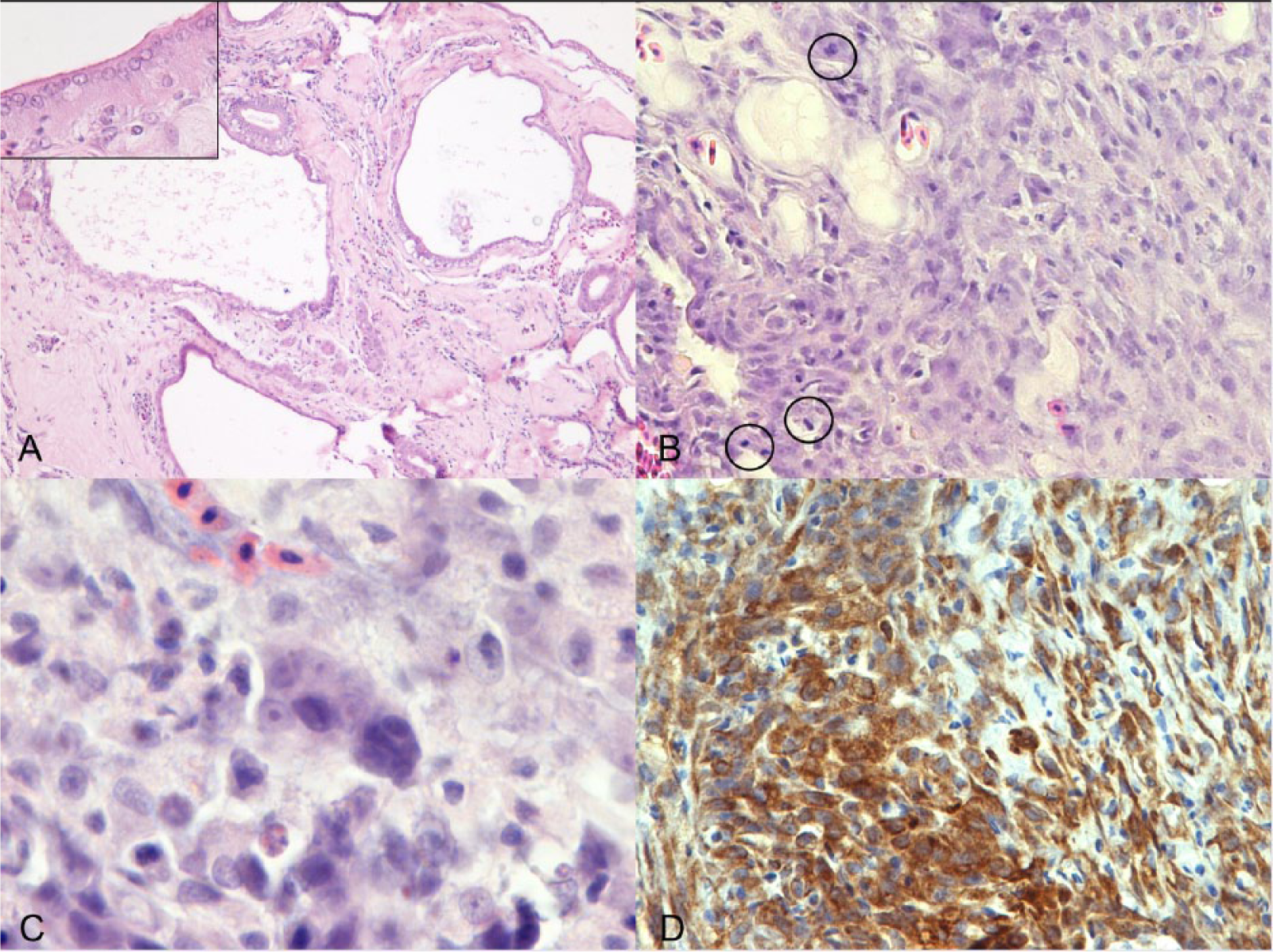
Mugil cephalus.
Focally, arising from the herniated swim bladder epithelium, the fibrous tissue was infiltrated by an unencapsulated, poorly demarcated, moderately cellular neoplasm composed of islands, lobules, and acini of neoplastic epithelial cells bounded by abundant stroma (desmoplasia). Neoplastic cells were 15–30 µm, polygonal, with distinct cytoplasmic borders, intermediate nuclear-to-cytoplasmic ratio, and a moderate amount of eosinophilic cytoplasm. Nuclei were oval, with finely stippled to marginated chromatin and a single, prominent magenta nucleolus. Anisocytosis and anisokaryosis were moderate, and the mitotic rate was up to 3 per high-powered field (Fig. 3B, 3C).
The spleen, perivisceral pancreas, peritoneal adipose tissue, and the remaining coelomic portion of the swim bladder were multifocally expanded by granulomas sharing the same histologic features described above; the intestine displayed mild lymphoplasmacytic inflammation, and the gills showed epithelial hyperplasia with fusion of secondary lamellae with moderate lymphoplasmacytic inflammation. The kidney, liver, stomach, and gonads were histologically within normal limits.
To characterize the pathologic process and etiologic agents involved, histochemistry (Alcian blue pH 2.5–PAS; Grocott and Ziehl–Neelsen), immunohistochemistry (anti-cytokeratin AE1/AE3, a dilution 1:50), and molecular biology assays for fungal agents were carried out. DNA was extracted from 2 stained cytologic smears by using a commercial kit b following the manufacturer’s instructions. Molecular characterization of the fungi was carried out by using primers targeting the nuclear ribosomal internal transcribed spacer (ITS) region as indicated previously. 13 Polymerase chain reaction (PCR) was executed in a final volume of 25 µL. PCR conditions were: buffer c (2.25 mM MgCl2), 200 µM dNTPs, 400 nM primer, and 1.25 U Taq polymerase. d The PCR protocol was as follows: step 1 (1 cycle) 94°C for 2 min, step 2 (repeated 40 times) 94°C for 25 s, 60°C for 25 s, 72°C for 1 min. A PCR product was obtained and then sequenced with an automated sequencer e ; the resulting data were blasted with an online genetic database (http://blast.ncbi.nlm.nih.gov/) giving 99% identity with Cladosporium spp.
From cytologic smears stained with May-Grünwald/Giemsa, the fungal hyphae appeared septate, irregular in width, with non-parallel pigmented walls and form chains (pseudohyphae). By histology, the fungal hyphae inside granulomas stained black with Grocott, and Ziehl–Neelsen did not reveal acid-fast bacteria within the granulomas.
Immunohistochemistry for AE1/AE3 showed a diffuse, cytoplasmic positivity of epithelium lining the swim bladder cysts, neoplastic cells (Fig. 3D), and rete mirabile, and the epithelioid cells forming the granuloma. These findings confirm the epithelial origin of the neoplasia from the glandular epithelium of the gas gland; the positivity of epithelioid cells does not have a diagnostic significance but represents a specific immunoreactivity reported in the literature. 11
On the basis of postmortem and histologic findings, a diagnosis of swim bladder herniation with concurrent carcinoma was proffered. The presence of a multiorgan granulomatous process caused by pigmented fungi (phaeohyphomycoses) consistent with Cladosporium spp. was interpreted as an opportunistic agent as reported in domestic and wild animals, and particularly in fish,1,14 most likely carried by the vegetal foreign body found.
Because of the topographic distribution of granulomatous lesions and their extent, the proposed pathogenesis of the 3 concurrent lesions described was as follows: 1) the herniation of the swim bladder through a putative malformation of epaxial muscles; 2) the ulcerated herniated tissue was damaged by the vegetal foreign body, favoring entry of the hyphae and causing a granulomatous inflammation in the fibrovascular stroma, as well as into the coelomic tissues adjacent to the hernia; and 3) the focal transformation of the swim bladder epithelium into a carcinoma, likely a result of organ displacement, as with human pathology, 16 and/or subsequent prolonged inflammatory stimulation.
In mammals, a hernia (etymologically “rupture”) is defined as an abnormal protrusion of an organ or tissue beyond the cavity in which it is normally situated, through a congenital or acquired defect in its surrounding walls (most often abdominal), with the preservation of natural fibrovascular connections. 8 In the present case, the following components of the hernia are present: the herniated organ (swim bladder) and the hernial defect (the body wall skeletal muscle defect with fibrosing reaction). The swim bladder is displaced through a body wall defect (congenital or acquired). To correctly classify the presented organ displacement, the definition of “aerocystocele” is proposed. This definition literally means “hernia of the swim bladder.” In this case, the displacement could have acted putatively as a causative carcinogenetic event; in fact, the malignant transformation of an organ or tissue not located in the normal site has been reported in humans. 16 Moreover considering the fibrosis embedding the hernial ring, the presented case could be a long-lasting process.
The occurrence of swim bladder developmental disorders in farmed teleosts is reported,12,17 whereas herniation of the swim bladder through the body wall is rare, and only a few case reports exist in the literature.6,10 Only 1 case is recorded in a white mullet (Mugil curema), and strikingly shared the same gross appearance of the case presented. 5 Regarding swim bladder neoplasms, there are few reports of spontaneous occurrence: adenomas and/or adenocarcinomas have been described in juvenile cod, 17 Japanese medaka, and guppy, 4 whereas several authors have reported virally induced leiomyosarcoma in Atlantic salmon.2,3,9
Footnotes
Authors’ contributions
R Sirri and C Bianco contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. G Zuccaro contributed to acquisition of data. ME Turba contributed to analysis and interpretation of data. L Mandrioli contributed to interpretation of data and drafted the manuscript. All authors critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Mouse monoclonal AE1/AE3 antibody, Dako Denmark A/S, Glostrup, Denmark.
b.
DNA tissue kit, Makerey Nagel, Milan, Italy.
c.
GoTaq buffer, Promega Italia srl, Milan, Italy.
d.
GoTaq polymerase, Promega Italia srl, Milan, Italy.
e.
Automated sequencer ABI310, Life Technologies, Milan, Italy.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
