Abstract
Snake fungal disease is an emerging infectious disease caused by the fungus Ophidiomyces ophiodiicola leading to severe dermatitis and facial disfiguration in numerous free-ranging and captive snakes. A free-ranging mud snake (Farancia abacura) from Bulloch County, Georgia, was presented for autopsy because of facial swelling and emaciation. Extensive ulceration of the skin, which was especially severe on the head, and retained shed were noted on external examination. Microscopic examination revealed severe heterophilic dermatitis with intralesional fungal hyphae and arthroconidia consistent with O. ophiodiicola. A skin sample incubated on Sabouraud dextrose agar yielded a white-to-tan powdery fungal culture that was confirmed to be O. ophiodiicola by polymerase chain reaction and sequence analysis. Heavy infestation with adult tapeworms (Ophiotaenia faranciae) was present within the intestine. Various bacterial and fungal species, interpreted to either be secondary invaders or postmortem contaminants, were associated with oral lesions. Although the role of these other organisms in the overall health of this individual is not known, factors such as concurrent infections or immunosuppression should be considered in order to better understand the overall manifestation of snake fungal disease, which remains poorly characterized in its host range and geographic distribution.
Fungal skin infections, or dermatomycoses, have been implicated as significant causes of morbidity and mortality in wildlife. Global amphibian declines have been associated with Batrachochytrium dendrobatidis, the causative agent of chytridiomycosis,19,21 and a novel chytrid fungus, Batrachochytrium salamandrivorans, has caused similar lethal disease in European species of salamanders. 12 White-nose syndrome of bats, caused by Pseudogymnoascus destructans, has killed at least 5 million North American bats and continues to spread throughout the United States and in 1 Canadian province.5,10,13,23
Since 2006, increasing numbers of snakes with fungal skin infections have been reported from the eastern United States. Clinical signs in these snakes include facial swelling, cloudy eyes, improperly shed skin, roughened or thickened scales, dermal or subcutaneous granulomas, and destruction of the venom glands in pit viper species.1,4 This syndrome has been referred to as snake fungal disease (SFD). The fungus Ophidiomyces (formerly Chrysosporium) ophiodiicola has been consistently associated with SFD, although a number of other fungi and bacteria have also been isolated from similar cases.3,4 Although experimental infection establishing O. ophiodiicola as the causative agent of this disease has been demonstrated in captive populations, many questions remain about the manifestation of SFD in free-ranging individuals and various snake species.1,9
A free-ranging, female, 260 g, subadult mud snake (Farancia abacura) with a swollen head and improperly shed skin was found in May 2014, in Bulloch County, Georgia. The snake was brought to the Orianne Society (Athens, Georgia) where it was observed in captivity for several days. The snake was emaciated and appeared weak. No improvement was noted while in captivity so the snake was transferred to the Southeastern Cooperative Wildlife Disease Study (SCWDS; University of Georgia, Athens, Georgia) for euthanasia and autopsy.
On presentation, the snake was lethargic and had a large facial swelling as well as ulceration of scales around the mouth. The snake was anesthetized in an air-tight container with a cotton ball saturated with isoflurane, a and euthanized with an intracardiac injection of pentobarbital sodium and phenytoin sodium followed by decapitation and pithing. b
The snake was in poor nutritional condition with moderate epaxial muscle wasting. Dark gray, caseous material covered the left side of the oral cavity. The cornea of the left eye was diffusely cloudy and ulcerated on the lateral aspect. The skin surrounding the left labial commissure was erythematous and extensively ulcerated (Fig. 1A). Multiple areas of skin were roughened, thickened, and pale (Fig. 1B). An 11-cm-long section of retained shed skin was adhered to the skin of the dorsum, adjacent to a focus of white, thickened scales. Approximately ten 5–10 cm Ophiotaenia faranciae cestodes were in the jejunum; 4 of these cestodes adhered to the mucosa of the intestines and protruded transmurally to the serosal surface. A single 5-cm Ophidascaris sp. nematode was in the stomach. Parasites were identified by morphologic characteristics and published keys.11,17,22
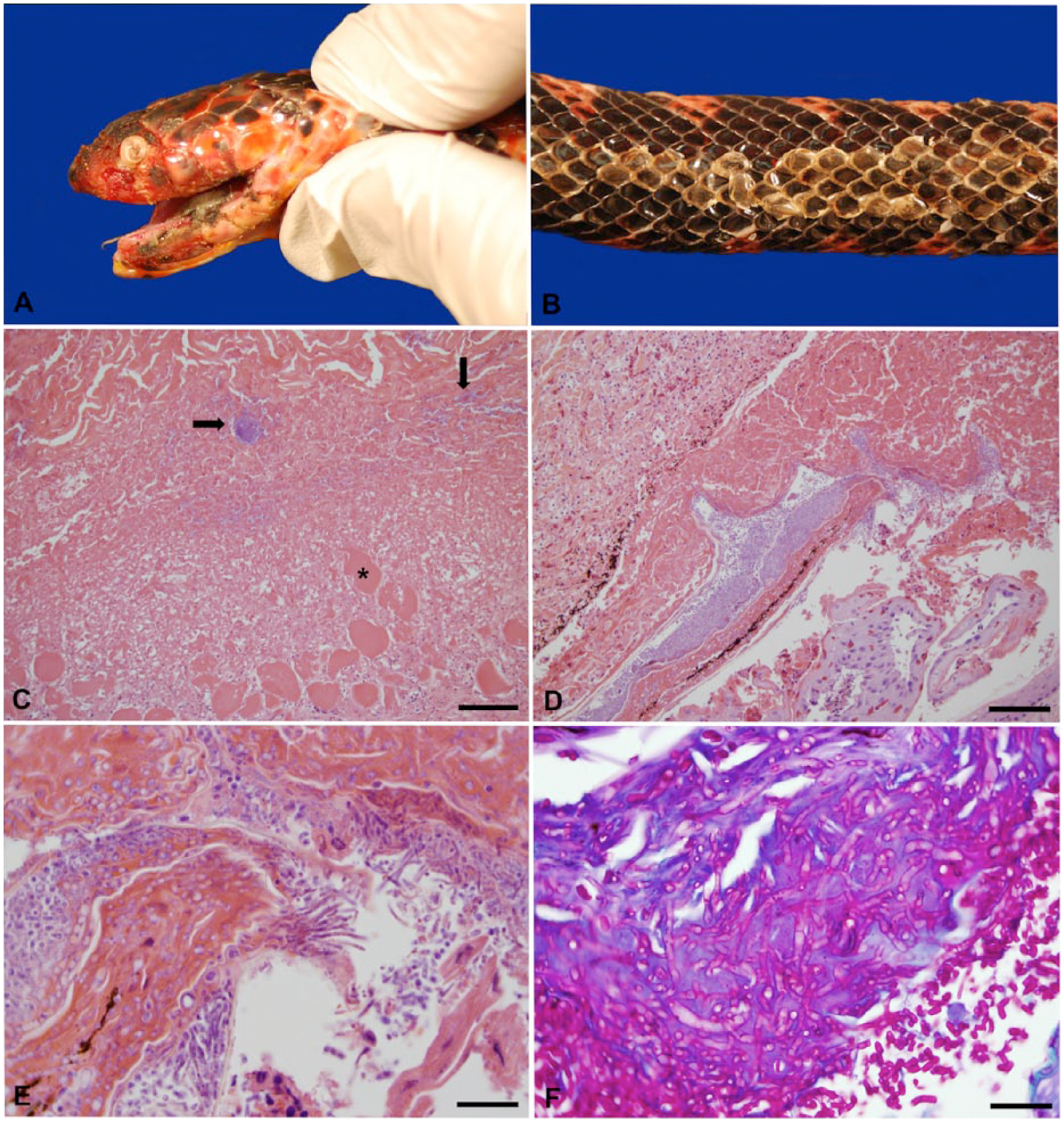
Mud snake (Farancia abacura).
Representative tissues were collected and fixed in 10% neutral-buffered formalin. The head was placed in 10% neutral-buffered formalin and demineralized with acid. Sections of tissues were embedded in paraffin, sectioned at 4–5 µm, stained with hematoxylin and eosin, and examined by light microscopy. Additional sections of skin and head were stained with periodic acid–Schiff (PAS) reaction, Grocott–Gomori methenamine silver (GMS), and Gram stains.
In the oral cavity, necrosis and inflammation extended deeply into underlying tissue and myofibers (Fig. 1C). The surface epithelium often was ulcerated and covered by a serocellular crust with intralesional secondary bacterial colonies (Fig. 1C). The skin from the dorsum was moderately hyperplastic and covered with a serocellular crust composed of fragmented keratin, fibrin, heterophils, and cellular debris. The dermis and underlying subcutis were expanded by fibrous connective tissue and infiltrates of lymphocytes, plasma cells, and macrophages (Fig. 1D). Multiple, ~4–6 µm, undulate, basophilic, pale-staining, septate, fungal hyphae, which occasionally branched at acute angles, and occasional cylindrical to clavate 2 × 4 µm arthroconidia were present in the necrotic debris (Fig. 1E). Fungal hyphae and arthroconidia stained with PAS (Fig. 1F) and GMS. Arthroconidia were most numerous within the hinge between the scales and were also associated with large numbers of mixed coccobacilli. Fungal hyphae occasionally invaded the underlying dermis and were often associated with aggregates of heterophils, lymphocytes, plasma cells, and occasional melanomacrophages. Skin from the ventrum was evaluated histologically, and microscopic changes included infiltration of a smaller number of fungal hyphae that were morphologically similar to those described above, rare arthroconidia, heterophils, and rare superficial colonies of gram-positive coccobacilli. The skin of the head also contained multifocal, thick, serocellular crusts with intralesional fungal hyphae and arthroconidia consistent with those seen on the dorsum, and abundant hyphal invasion of the dermis. Occasional fungal hyphae and large numbers of gram-negative coccobacilli were present along the surface of the spectacle.
The lumens of nearly all examined sections of small intestine were filled with cross-sections of cestodes that occasionally penetrated the tunica muscularis and bulged from the serosa. The cestodes often were encompassed by fibrous connective tissue that was infiltrated by heterophils, lymphocytes, plasma cells, and macrophages. The lamina propria of the intestine was infiltrated by small numbers of lymphocytes and plasma cells.
Multiple samples of skin were plated on fungal culture biplates containing Sabouraud dextrose agar (SDA) and dermatophyte test medium and incubated at 22°C. c After 14 days, a white-to-tan powdery fungal colony was present on the SDA section of a plate containing a sample of dorsal skin. The colony was observed for an additional 14 days. Innumerable rectangular arthroconidia, moderate numbers of parallel aleuroconidia, and undulate hyphae consistent with O. ophiodiicola, were present on cytologic examination of the colony. DNA was extracted from a sample of the cultured fungus using a commercial tissue extraction kit according to the manufacturer’s directions. d A portion of the internal transcribed spacer (ITS) 1 region between the 18S and 5.8S rRNA genes was amplified by polymerase chain reaction (PCR) using primers ChrysoITS-F (5′-TGATCCGAGGTCAACCGGAAGAAA-3′) and ChrysoITS-R (5′-TGGAACCGTGAACGAACTCTGTGA-3′) as described with the following modifications: only 1 µL of DNA template was added to the reaction, and GoTaq e was used as the Taq. 2 The amplified PCR product was electrophoresed in 2% agarose, purified with a commercial gel extraction kit, and submitted to the Georgia Genomics Facility (Athens, GA) for bidirectional sequencing. f The consensus sequence (383 bp) was identical to several strains of O. ophiodiicola (e.g., KF477227, KF477231, KF477233, KF477235). DNA also was extracted from multiple samples of skin and tested by PCR as described above; no O. ophiodiicola was detected.
Samples of skin and oral necrotic tissue also were submitted to the Athens Veterinary Diagnostic Laboratory (AVDL; Athens, GA) for additional fungal isolation. Fungal plates were incubated at 37°C. Heavy mixed fungal growths of a nonpigmented, filamentous fungus and an unidentifiable sterile mycelium were obtained from the skin. Very light growths of Penicillium and an unidentifiable sterile mycelium were isolated from the oral tissue. Samples of skin, oral tissue, liver, and intestines were submitted to the AVDL for aerobic and anaerobic bacterial culture. Heavy mixed bacterial growth of Myroides odoratus, Aeromonas spp., and Stenotrophomonas maltophila was cultured from the oral tissue. Light-to-moderate growth of common environmental contaminants was isolated from the liver and intestine with no detection of Salmonella spp. A sample of intestinal content was sent to the AVDL for electron microscopy negative staining to look for viral particles, and none was detected.
The gross anatomic and microscopic lesions in this snake were highly suggestive of SFD. This diagnosis was confirmed by fungal culture and genetic identification of the isolated fungus as O. ophiodiicola. Although O. ophiodiicola was isolated from fungal culture performed at SCWDS, it was not detected from samples submitted to the AVDL. The latter plates were incubated at 37°C, suggesting that temperature may be an important factor for the isolation of O. ophiodiicola, as isolation was only successful in samples incubated at 22°C at SCWDS. Pathogens adapted for a reptilian host may exhibit inhibited growth at higher temperatures, and diagnostic laboratories should alter their protocols to optimize their chances of isolating such organisms. Additionally, PCR results on DNA directly extracted from tissue samples of the affected lesions were not positive for O. ophiodiicola. Direct testing of tissues has been successful in other instances, and the reasons for the lack of detection in this case are unknown, but it may be a result of the limited sensitivity of the current assay. 2 At the time of diagnosis, a combination of assays (i.e., histopathology, fungal culture, and PCR) was pursued in order to provide maximum diagnostic information because a case definition has not yet been published. Although highly sensitive PCR assays have been published recently, potentially minimizing the need for fungal cultures, histopathology is necessary to differentiate true disease from exposure in suspected cases of SFD.2,6 The role of the serositis caused by a heavy load O. faranciae infection in this individual is unclear, although it could indicate an overall state of poor health.
O. ophiodiicola was first described in 2009 after being isolated from a granuloma on a captive black rat snake (Elaphe obsoleta obsoleta) from Georgia. 16 Frequently associated with cases of SFD, the fungus has been shown to induce lesions consistent with SFD in experimental infections of captive and wild-caught snakes.1,9 The fungus has also been detected in tissue samples going back to 1985 from captive snakes in the United States, Europe, and Australia, which previously had been diagnosed with dermatomycoses caused by other fungal species. 20 Many had been diagnosed with Chrysosporium anamorph Nannizziopsis vriesii (CANV), a related fungal pathogen that has been an emerging cause of dermatomycoses in captive reptiles.14,15 A 2013 publication described analyses of morphologic and molecular characterization of CANV isolates and revealed that these species belong to 3 related lineages, of which only Ophidiomyces has been documented in snakes. 20 Fungal species often are considered to be opportunistic pathogens, but CANV has been documented as an obligate pathogen of reptiles, with Koch postulates fulfilled in at least one experimental trial. 9 The fungi responsible for chytridiomycosis and white-nose syndrome show evidence of adaptation as obligate pathogens of vertebrate hosts and reduced saprophytic activity as compared to their taxonomic relatives.18,19 Nevertheless, disease caused by CANV in captive reptile populations is associated most often with stress or poor husbandry. 14 Although these factors also may be implicated in incidents of SFD in captive snakes, factors including limited genetic variability and cold or wet environmental conditions have been proposed as factors potentially associated with the development of SFD in free-ranging snakes.4,7 Lesions in experimentally infected snakes were relatively mild, with many snakes surviving to the experiments’ endpoints, but conditions experienced by wild snakes may allow for the manifestation of more severe disease.1,9
Snake fungal disease has been diagnosed primarily in individual snakes, but population-level effects have been proposed for isolated, vulnerable populations, including large declines in a population of New England timber rattlesnakes (Crotalus horridus) and endangered eastern massasauga rattlesnakes (Sistrurus catenatus).1,3,7 Geographic and species variability in disease susceptibility have yet to be explored. To our knowledge, our report represents the southernmost detection of SFD in a wild snake at the time of the diagnosis.
Additional unexplored biological factors such as coinfections and immunosuppression may be relevant in the development of SFD. Generally, adult cestodes are not considered pathogenic for their hosts, but in the snake described herein, a heavy parasitic load was associated with serositis. This finding merits attention and supports the need for a more comprehensive investigation of SFD, including the role of any comorbidity. Although no viral particles were detected from electron microscopic examination of feces, other concurrent viral infections or other potential causes of immunosuppression could not be ruled out.
Mud snakes are nonvenomous, highly aquatic snakes found throughout wetlands of the southeastern United States, ranging as far north as the Mississippi River basin of southern Illinois. Although believed to be relatively common throughout their habitat, they are secretive and rarely seen. Mud snakes are not a protected species, but may be vulnerable to wetland degradation. 8 SFD has not been documented previously in this species and further surveillance, as well as population monitoring, is necessary to determine its significance to this species. Further studies are warranted to investigate the geographic and species range of SFD as well as the pathophysiology of the disease and the potential significance to free-ranging snake populations.
Footnotes
Acknowledgements
We thank Hannah Stanford, Carol Maddox, Jeffrey Lorch, the histology laboratory at the University of Georgia, College of Veterinary Medicine, and the federal and state agency members of Southeastern Cooperative Wildlife Disease Study for their assistance with this case.
Authors’ contributions
LA Last contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. H Fenton and MJ Yabsley contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and critically revised the manuscript. JG McGuire and M Moore contributed to acquisition, analysis, and interpretation of data, and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
IsoFlo, Abbot Laboratories, Chicago, IL.
b.
Beuthanasia-d Special, Merck Animal Health, Summit, NJ.
c.
Derm-Duet II, Hardy Diagnostics, Santa Maria, CA.
d.
DNeasy blood & tissue kit, Qiagen Inc., Germantown, MD.
e.
GoTaq Flexi, Promega Corp., Madison, WI.
f.
QIAquick gel extraction kit, Qiagen Inc., Germantown, MD.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding was provided by the sponsorship of the Southeastern Cooperative Wildlife Disease Study by the fish and wildlife agencies of Alabama, Arkansas, Florida, Georgia, Kentucky, Kansas, Louisiana, Maryland, Mississippi, Missouri, North Carolina, Ohio, Oklahoma, Pennsylvania, South Carolina, Tennessee, Virginia, and West Virginia, USA. Support from the states to SCWDS was provided in part by the Federal Aid to Wildlife Restoration Act (50 Stat. 917).
