Abstract
A 5-year-old male captive corn snake (Pantherophis guttatus) with caudal coelomic swelling was admitted for surgical treatment. Laparotomy revealed a 5 × 4 × 2.5 cm, firm, expansile, irregularly shaped mass arising from the middle portion of the right kidney with a mild lobulated pattern and mottled white-to-tan. Microscopically, the mass was composed of numerous bizarre angulated tubules of polygonal neoplastic cells separated by a scirrhous stroma with remarkable heterophilic infiltrates. The neoplastic cells were nonciliated and mucin secreting, with abundant brightly eosinophilic cytoplasm. There were marked cellular and nuclear atypia, frequent cell individualization, and stromal invasion, indicative of malignant behavior, which was confirmed by metastasis to the left kidney 1.5 months postoperatively. Both neoplastic epithelial cells and mesenchymal cells contributing to the scirrhous stroma had variable immunopositivity for pan-cytokeratin. The neoplasm was considered a renal adenocarcinoma resembling human collecting duct carcinoma.
Renal adenocarcinomas (RAs) are relatively common malignancies in various snake species, although less frequent than soft tissue sarcomas and lymphomas.3–5,8,9,11,13 In a previous retrospective study, colubrids were found to be predisposed to developing RA, especially king snakes. 8 These tumors, which are usually large and solitary, affect only one side of the kidney and have a tubular pattern, little cytologic atypia, and strong scirrhous reaction. Although RAs of snakes often show capsular invasion, metastasis is slow and uncommon.3,8,11 Because of the presumable disruption of renal function, urate tophi are also commonly found in the remaining renal parenchyma.8,11,13 Structurally, the snake nephron contains a poorly developed glomerulus, a long proximal convoluted tubule, a short intermediate segment, a shorter distal tubule, and a collecting duct. The loop of Henle is absent in reptilian nephrons, and mucin secretion can be detected only in the distal tubule and collecting duct.1,2,10
To date, the pathogenesis and classification of human renal tumors have been established not only by their histologic features but also by their immunohistochemical and molecular characteristics correlated with clinical course. 23 Similar information on renal tumors in snakes is limited. The 2004 World Health Organization classification defined several renal cell tumors, such as clear cell renal cell carcinoma, oncocytoma, and collecting duct carcinoma (CDC). Among them, CDC is a rare, highly aggressive, and commonly metastatic tumor derived from the principal cells of the collecting duct of Bellini.15,21 We report herein a RA in a corn snake (Pantherophis guttatus) resembling human CDC. To our knowledge, this is the second case of RA documented in a corn snake.
A 5-year-old male corn snake, housed in the Taipei Zoo, was presented with enlargement of the caudal coelomic cavity and was admitted for laparotomy. At surgery, the caudal coelomic cavity was distended by a 5 × 4 × 2.5 cm mass in the middle of the right kidney. Tumor resection and removal of the adjacent lobules were performed. Grossly, the mass was expansive and firm with a poorly defined margin toward the surrounding compressed renal parenchyma, wherein myriad small white-yellow foci were dispersed, and had a gritty texture on cutting (Fig. 1). On sectioning, the mass was gray-to-tan and multilobulated.
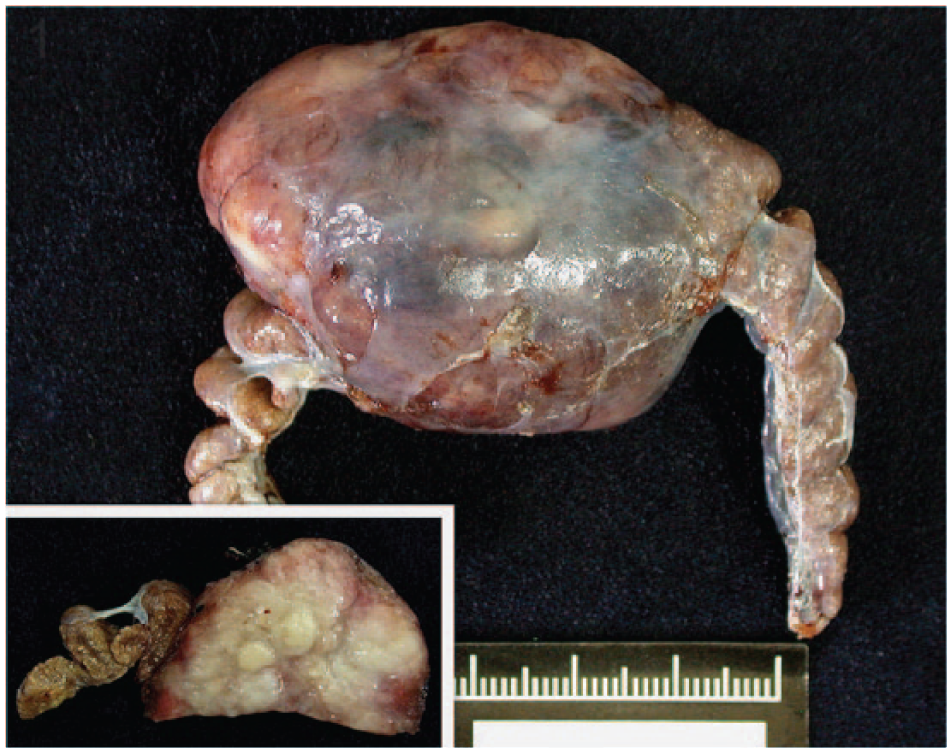
An irregularly shaped, expansile, renal adenocarcinoma in a corn snake, with compression of adjacent kidney lobules. The cut surface is white and lobulated (inset).
Histologically, the tumor was composed of lobules of angulated and irregular tubules (Fig. 2) with a bizarre hobnail appearance (Fig. 3) that were separated by fibrous lamellae of variable thickness, as demonstrated by trichrome staining. The neoplastic cells showed significant anaplasia characterized by marked variation in size and shape, and individual cells were commonly seen infiltrating into the surrounding tissue. The cells had abundant brightly eosinophilic cytoplasm, indistinct cell borders, and pleomorphic vesicular nuclei with conspicuous magenta nucleoli (Figs. 2, 3). No cilia were observed. Both intracytoplasmic and luminal mucin secretion were seen and confirmed by Alcian blue staining (Fig. 4). Mitotic figures were common, estimated at 3–5 per 10 high-power fields, with atypical mitoses (Fig. 3). Scattered throughout the mass was marked infiltration by heterophils (Figs. 2, 3) with sporadic perivascular lymphoplasmacytic aggregates. In the adjacent renal parenchyma were many urate crystal deposits surrounded by multinucleated giant cells and fibrous bands.
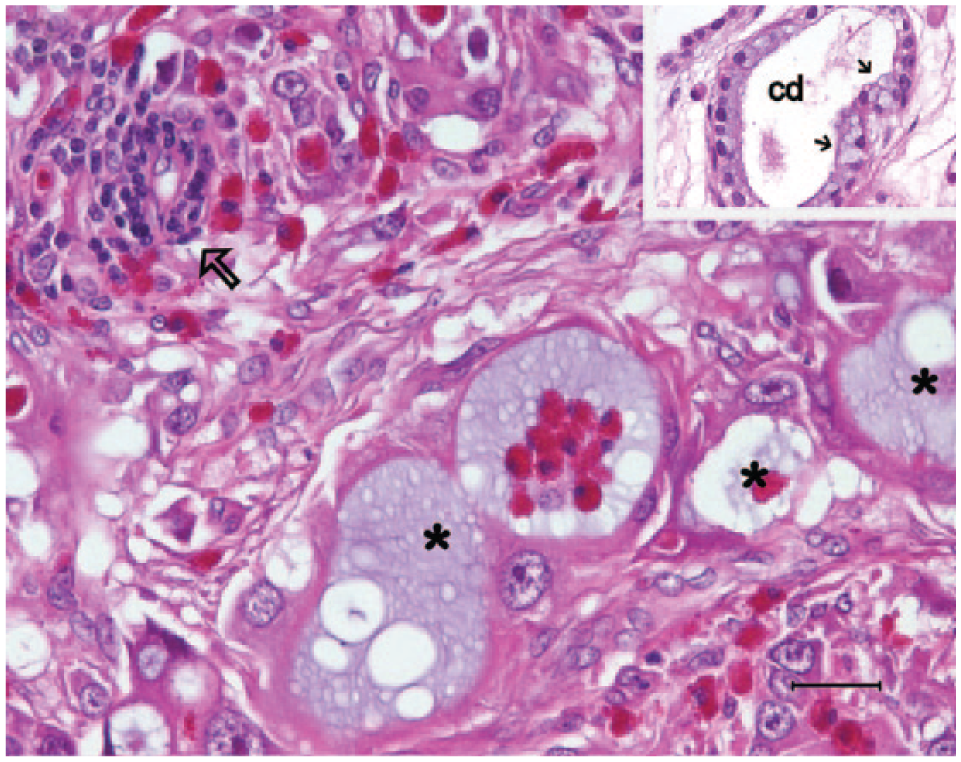
Neoplastic cells forming irregular tubules (asterisks) with many individual cells penetrating and disrupting the renal parenchyma. Marked heterophilic infiltrates are present either within the neoplastic tubules or the desmoplastic stroma. A small lymphocytic aggregate is marked by an arrow. Inset: The residual collecting ducts (cd) characterized by apical mucin secretion (small arrows) are usually dilated; the surrounding stroma is edematous. Hematoxylin and eosin. Bar = 25 μm.
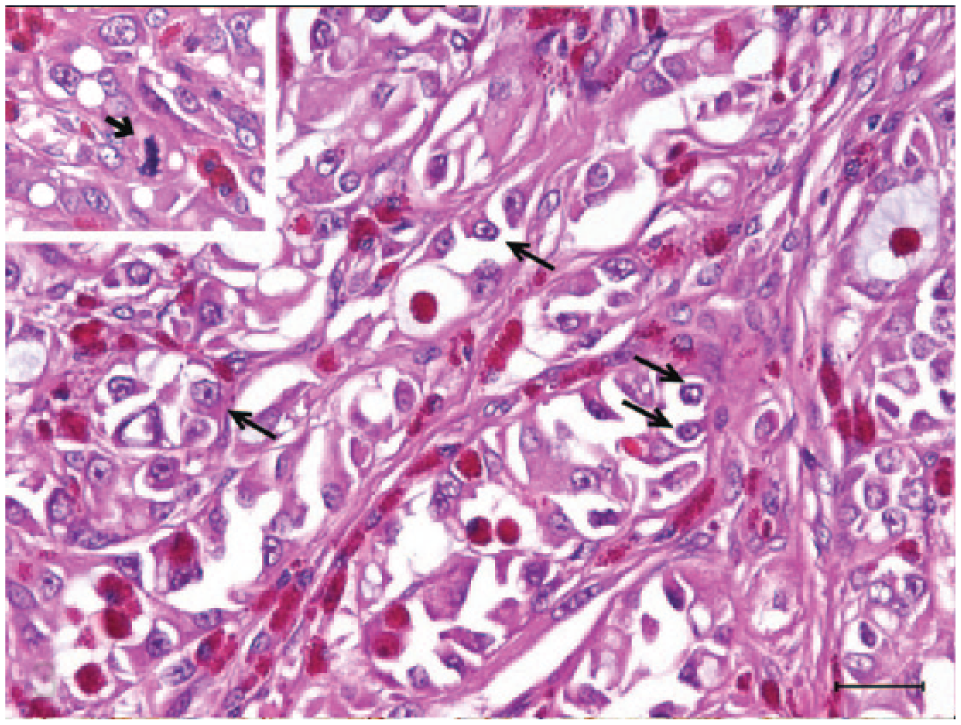
A bizarre hobnail lining frequently occluding tubular lumens. More typical hobnail cells featuring eccentrically located nuclei and sometimes tailed-off cytoplasm are marked by long arrows. Note the presence of both luminal and intracytoplasmic blue mucin. Inset: A dividing cell with an atypical mitotic figure (short arrow). Hematoxylin and eosin. Bar = 25 μm.
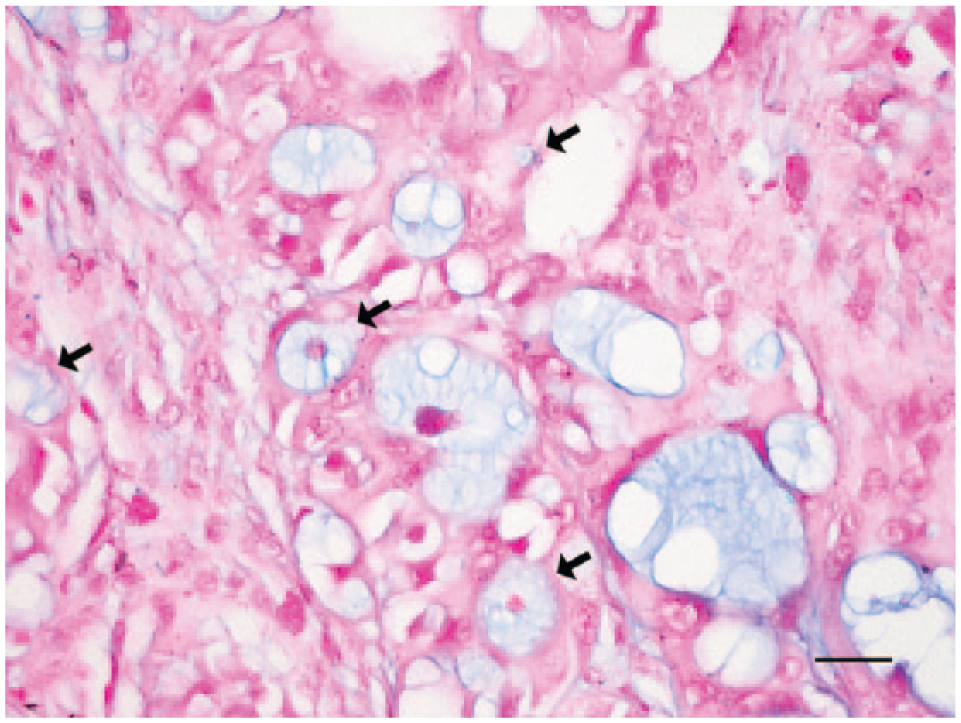
Mucin vacuoles in tumor cells (arrows) and neoplastic tubules. Alcian blue. Bar = 25 μm.
Because the microscopic findings in our case, including mucin secretion and hobnail cells, were reminiscent of human CDC, immunohistochemistry (IHC) was performed on paraffin-embedded tissue sections to confirm the cellular origin. Ideally, neoplastic cells of CDC express markers normally detected in collecting ducts such as pan-cytokeratin (CK), low- and particularly high-molecular-weight cytokeratin (CK-HMW), but lack immunoreactivity to CD10 and villin, which label the brush borders of the proximal renal tubules. Coexpression of vimentin with CK may be seen.19–21 To this end, mouse anti-human CK a and mouse anti-human vimentin a were employed with a non-biotinylated horseradish peroxidase method b using diaminobenzidine as a chromogen substrate. Antibodies of mouse anti-human CK-HMW c and mouse anti-human villin c were used in an automated immunohistochemistry system. d Normal renal tubules and stromal cells in the present case served as internal controls, and mammary gland tumor and small intestine from cats served as external controls. The normal renal tubules of the snake, the epithelial cells of the feline specimens, and the neoplastic cells lining the irregular tubules were successfully labeled by CK, but the latter displayed an uneven distribution and occasional loss of CK expression (Fig. 5). Surprisingly, the spindle-shaped cells of the scirrhous stroma also stained strongly positive for CK (Fig. 5). Antibodies to vimentin, CK-HMW, and villin did not label the snake specimens. The results of IHC and histopathology were suggestive of a RA resembling human CDC.
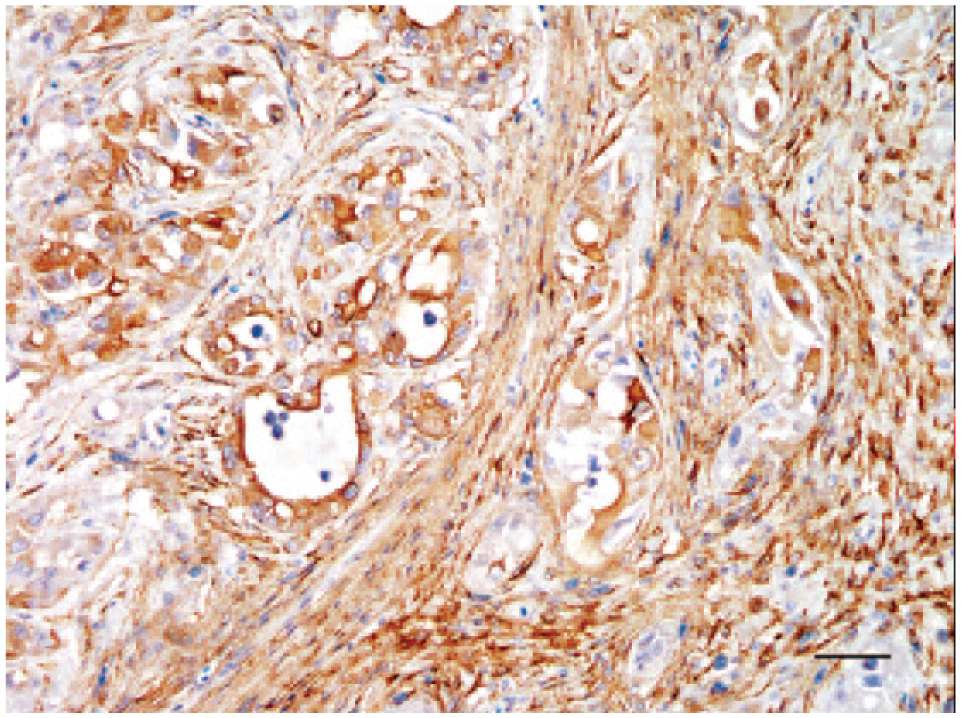
Neoplastic cells exhibiting variable positivity for pan-cytokeratin. Note also consistent immunolabeling in spindle shaped cells of the desmoplastic stroma. Bar = 50 μm.
The snake survived 1.5 months postoperatively, during which time gradual weight loss occurred. At autopsy, the left kidney was swollen and contained multiple randomly distributed 0.5 × 0.5 cm diameter white nodules (Fig. 6). Imprint smears stained with Liu stain (a modified Romanowsky stain) revealed mostly pleomorphic polygonal cells tending to form a sheet, presumably as part of tubular structures. Multinucleation and anisokaryosis were frequently noticed, and mitotic figures were common. Histopathology revealed multifocal infiltrative neoplastic cells and occasionally intravascular bizarre cells morphologically resembling those in the right kidney tumor. Gouty tophi were often seen. The liver was severely affected by multiple necrohemorrhagic inflammation with intralesional bacterial colonies. The cause of death was considered concurrent contralateral renal metastasis with subsequent renal failure and severe hepatitis.
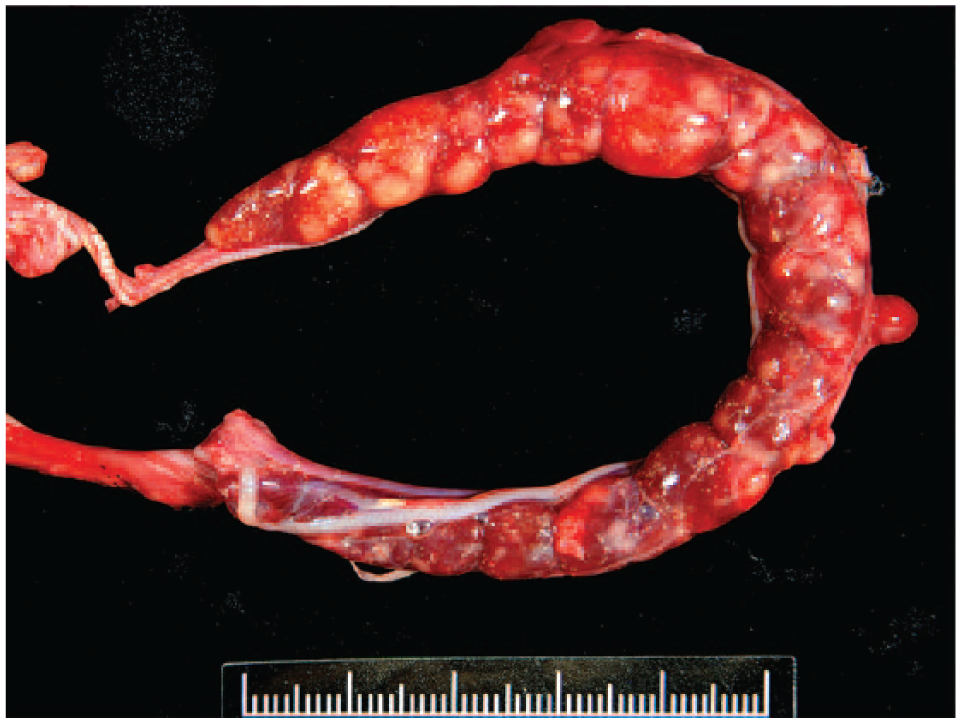
Left kidney is expanded by multiple variably sized white nodules and contains numerous pinpoint spots with a gritty texture of gouty tophi.
Human CDC is an infrequent but aggressive form of renal malignancy with early metastasis and hence unfavorable prognosis. When it is small, CDC can center on the renal medulla near the pelvis, indicating a collecting duct origin. Histologically, CDCs have irregular tubular or tubulopapillary patterns with desmoplasia and pronounced neutrophilic infiltration. The neoplastic cells exhibit significant nuclear atypia. A hobnail growth pattern and mucin production may be seen.14,19,20 Application of specific IHC markers (see above) is often required, and reactivity to Ulex europaeus agglutinin-1, Dolichos biflorus agglutinin, and peanut agglutinin also assists in diagnosis.14,20,21
Our case presented characteristic histologic findings that highly resembled human CDC and are suggestive of a collecting duct origin. The neoplastic cells were extremely anaplastic, with high-grade nuclei and tended to form irregular tubules with occasional hobnailing. Neoplastic tubules were embedded in a desmoplastic stroma with many heterophils. Mucin secretion was evident. Although a rare variant of human clear cell renal cell carcinoma (RCC) of proximal tubule origin can produce extracellular mucin, features in the present case, such as abundant eosinophilic cytoplasm and hobnail growth pattern, clearly distinguished this case from a clear cell RCC. 22 A medullary or central location of the renal tumor is diagnostic for human CDC but may not be relevant to snakes because of different anatomic distribution of collecting ducts. The collecting ducts of snakes begin dorsolaterally in each lobule and move along the lateral margin ventrally to enter the ureter. 10 A lateral location should therefore serve as an indicator of snake CDC. Nonetheless, the massive expansion of the present tumor made this criterion inapplicable.
Immunohistochemically, although only CK successfully cross-reacted with the present snake specimens, there was occasional loss of CK-positivity from the tumor cells lining the tubules, accompanied by acquisition of CK-positivity in spindle cells of the desmoplastic stroma. Commercial anti-human cytokeratin antibodies can cross-react with snake epithelium, but whether they are also able to stain snake mesenchymal cells, at least under a severe inflammatory or neoplastic condition, remains unknown. 16 The loss of CK-positivity may be partially explained by the epithelial–mesenchymal transition, a critical process in which high-grade neoplastic epithelial cells break cell–cell adhesion (which is consistent with the hobnail growth pattern) and favor metastasis characterized by gradual loss of CK-positivity along with increased vimentin expression. 12 Explanations for the CK-positivity in mesenchymal cells may include complex cross-reaction between different species or an induction of unusual expression of surface marker(s) in snake mesenchymal cells by cytokines or growth factors secreted by inflammatory infiltrates. The actual mechanism requires further elucidation. In addition, the CK staining in the present case (Fig. 5) should be technically valid because the negative controls (stroma) in other normal kidney lobules were consistently negative. We thus concluded that the CK-positivity of mesenchymal or spindle cells in the desmoplastic stroma of the neoplasm may have been real. The negative staining for vimentin was compatible with previous reports in snakes.16,18 The application of anti-human vimentin antibody in diagnosing snake tumors remains unstable, and additional investigation on its utility is needed. CK-HMW corresponding to cytokeratins 1, 5, 10, and 14 can normally detect human squamous and ductal epithelia and is suggested to facilitate the diagnosis of CDC. Although CK-HMW could not label the present case, presumably because of the lack of cross-reaction, the result does not rule out the possibility of CDC.14,19,21
Caudal coelomic swelling is a nonspecific but typical presentation in snakes with RA.3,4,13,17 Differential diagnoses for caudal coelomic swelling are impacted or retained eggs in females, abscess, obstipation, or granuloma. Diagnostic imaging such as contrast radiography and ultrasonography may assist in locating lesions; biopsy is recommended for making a definitive diagnosis.4,17 Compared with other reports of RA in snakes, our report describes mucin secretion and hobnail appearance as histologic features. The massive heterophilic infiltrate was also striking, and a similar but milder infiltrate had been recorded in the other RA in a corn snake. 3 Studies in humans have shown that tumor-infiltrating neutrophils could favor tumor growth and metastasis. 7 Being a reptilian counterpart, the role for heterophils in promoting tumor progression remains to be understood.
Viral infection has been proposed to play a role in the development of several snake neoplasms. 6 In our case, a viral insult could not be investigated given the lack of fresh tissues. A genetic problem or exposure to carcinogens is also a concern because the 5-year life span of the present snake was quite short compared with the normal life span of captive corn snakes of 20 years or more. Despite the uncommon occurrence of metastasis in snake RA, contralateral renal metastasis occurred in the present case, and has been documented at variable intervals after surgical resection in other cases.3,4 Accurate classification of snake RA, as well as identification of reliable prognostic indicators, is required to better predict outcomes.
Footnotes
Acknowledgements
We thank Dr. Chih-Chun Wu for her assistance with collection and interpretation of the clinical data.
Authors’ contributions
CF Kao contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. JL Chen, WT Tsao, and AH Lee contributed to acquisition and analysis of data. CH Liu contributed to acquisition and interpretation of data. FI Wang contributed to conception and design of the study; contributed to analysis and interpretation of data; and gave final approval. CF Kao and FI Wang agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Dako North America Inc., Carpinteria, CA.
b.
REAL Envision kit, Dako Denmark A/S, Glostrup, Denmark.
c.
Novocastra Laboratories Ltd., Newcastle, United Kingdom.
d.
BOND-MAX autostainer, Leica Biosystems, Newcastle, United Kingdom.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
