Abstract
Choroid plexus neoplasms are uncommon brain tumors in dogs. Choroid plexus carcinomas often spread diffusely throughout the ventricular system and subarachnoid space and, in aggressive forms, can mimic histologic patterns of other carcinomas, including being embedded in a desmoplastic reaction. Although choroid plexus tumors (CPTs) heterogeneously express pan-cytokeratin, little is known about other markers to identify choroid plexus and their associated tumors. Kir7.1, an inward-rectifier potassium channel, is reported to have high diagnostic utility in human neuropathology to distinguish CPTs from other primary brain tumors and cerebral metastases. To determine Kir7.1 expression in the dog brain, we analyzed the immunoreactivity of Kir7.1 in normal brain, gliomas, ependymomas, CPTs, meningiomas, and carcinomas. In normal brain tissue, the immunostaining was restricted to the choroid plexus where there was robust membrane immunoreactivity along the apical border of the cells with less intense cytoplasmic staining. Similar strong immunoreactivity was detected in 12 of 12 CPTs, whereas 5 of 5 gliomas, 4 of 5 ependymomas, 5 of 5 meningiomas, and 5 of 6 carcinomas had no immunoreactivity. One ependymoma and 1 nasal carcinoma with squamous metaplasia were up to 75% immunopositive, with moderate cytoplasmic and membranous immunoreactivity, but lacking the robust apical immunoreactivity pattern. Analysis for immunoreactivity in a tissue microarray failed to yield any other locations in which immunoreactivity was detected. These results, including the distinctive pattern of immunostaining in CPTs, suggest that Kir7.1 is an excellent marker for CPTs in the dog.
In the dog, choroid plexus tumors (CPTs) account for ~7–10% of the total number of primary brain tumors.14,16 Most of these tumors affect middle-age to older dogs and are most commonly recorded in the fourth ventricle, with lesser involvement of other ventricular locations. 16 Dissemination can occur throughout the ventricular system, with metastasis found in the central canal or within the subarachnoid space of the spinal meninges. In the dog, CPTs are classified as grade I (choroid plexus papilloma), grade II (atypical choroid plexus papilloma), and grade III (choroid plexus carcinoma), with increasing features of malignancy associated with each grade. 16 This grading scheme is similar to the most recent human World Health Organization (WHO) classification of brain tumors; however, whether the underlying pathogenesis of canine CPTs and their human counterpart is similar is not known. 7 Because these tumors can disseminate via the cerebrospinal fluid, they can mimic metastatic carcinomas, and immunohistochemical determination of the cell of origin is useful especially with small surgical biopsies in which the primary source of the tumor is unknown.
With the increasing grade of CPTs, the increased degree of necrosis, atypia, and bizarre cellular growth can make diagnosis of choroid plexus carcinomas challenging. 9 Furthermore, diagnosis of brain biopsies, which may lack overall architecture and histologic pattern, can be difficult. Although CPTs arise from the choroid plexus epithelium, cytokeratin immunoreactivity in canine tumors has been reported to be variable with at least 1 study indicating only a small number of cases immunoreactive to wide-spectrum cytokeratin; however, immunoreactivity of these are either variably or not specific to CPTs.3,5,9 In human samples of CPTs, tissue microarray analysis has identified several candidate genes that are specific to choroid plexus and allow for differentiation from metastatic carcinomas. 2 One of these diagnostic markers is the inward rectifier potassium (K+) channel Kir7.1, also known as inward rectifier potassium channel 13 or potassium inwardly rectifying channel, subfamily J, member 13. 2 Inward rectifier potassium channels are characterized by allowing potassium to flow into the cell, and the inward rectification is mainly due to the blockage of outward current by internal magnesium. 8 Kir7.1 has a low single channel conductance, providing steady background potassium current to establish the membrane potential. 6 In human CPTs, Kir7.1 is consistently expressed in the apical membrane of normal choroid plexus epithelium and neoplastic choroid plexus.2,10 The present report describes the expression of Kir7.1 in canine CPTs and provides a comparison of immunoreactivity in other primary and metastatic brain tumors in the dog.
The pathology database at the Cornell University College of Veterinary Medicine (Ithaca, New York) was searched for cases of choroid plexus neoplasia in the dog from the period of 1990–2015. The selected cases were from dogs of different breeds ranging in age from 3 to 13 years; 12 cases had CPTs. Additionally, 6 cases of metastatic central nervous system tumors, including salivary (1/6), nasal (3/6), pulmonary (1/6), and unknown primary (1/6), as well as 5 cases each of gliomas, meningiomas, and ependymomas were retrieved from the archive. All cases were studied at autopsy, which was performed within 12–24 h after death. Tissues were collected, fixed in 10% neutral-buffered formalin, embedded in paraffin, sectioned at 5 µm, and stained using hematoxylin and eosin. Immunohistochemistry for Kir7.1 a was performed on all cases using the following protocol: formalin-fixed, paraffin-embedded sections were deparaffinized, rehydrated, and subsequently blocked with 3% hydrogen peroxide. Pretreatment involved microwaving for 20 min in 0.01 M citrate buffer followed by 20 min of cooling. All steps were followed by a Tris-buffered saline wash. Prior to application of the primary antibody, all slides were treated with a biotin block b and a protein block c for 10 min. Sections were incubated with anti-human Kir7.1 (goat polyclonal, 1:50, overnight at 4°C). Slides were then incubated with secondary antibody biotinylated horse anti-goat d (1:200, 30 min at room temperature) followed by 30-min incubation at room temperature with a commercial kit. d The slides were developed with DAB chromogen d and counterstained with Mayer hematoxylin. Negative control slides consisted of isotype-matched anti-goat immunoglobulin G d (1:1,000, 30 min at room temperature). Positive control tissue consisted of normal canine brain, which included gray and white matter, lateral ventricle lined by ependyma, and choroid plexus. Additionally, a canine tissue microarray was assayed for immunoreactivity. Tissues included in the canine tissue microarray were liver, kidney, spleen, skeletal muscle, salivary gland, colon, small intestine, stomach, adrenal, thyroid, testis, prostate, lung, skin, lymph node, and aorta.
Briefly, 5 choroid plexus papillomas and 7 choroid plexus carcinomas (CPCs) were identified. These tumors corresponded to WHO grade I and III, respectively, with grade III tumors displaying features that included increased mitotic figures, regions of necrosis, metastatic spread, and disorganized growth pattern. 15 Arborizing trabeculae and cords of neoplastic choroid plexus epithelium supported by cores of fibrovascular tissue were typical in all cases. Two of the CPCs were embedded in a dense desmoplastic response. Normal choroid plexus epithelium displayed intense apical immunoreactivity with lesser cytoplasmic immunoreactivity. Mild nonspecific staining was noted in the arachnoid and pia mater in a minority of the cases. Normal ependyma did not show any immunoreactivity. Immunoreactivity and staining pattern for Kir7.1 was found in 12 of 12 (100%) CPTs in the present case series. Immunoreactivity was most intense at the apical membrane of neoplastic choroid plexus and less so within the cytoplasm; the staining pattern did not vary across tumor grade or location (Table 1; Fig. 1). Immunoreactivity was diffuse throughout all neoplasms. All cases of meningioma and glioma had no immunoreactivity. One of 5 ependymomas displayed cytoplasmic immunoreactivity similar to that seen in the CPTs; however, the pattern was patchy in the neoplasm with large regions of the tumor lacking immunoreactivity. In this single case, the most intense staining pattern was found in the pseudorosettes and lacked apical staining. One of 6 carcinomas (the nasal carcinoma) had mild to moderate cytoplasmic immunoreactivity; however, there was no apical membrane immunoreactivity detected, and the significance of the staining pattern is unclear. No immunoreactivity was present in the canine microarray tissues.
Signalment of cases and immunohistochemical staining pattern of Kir7.1 in the current study.*
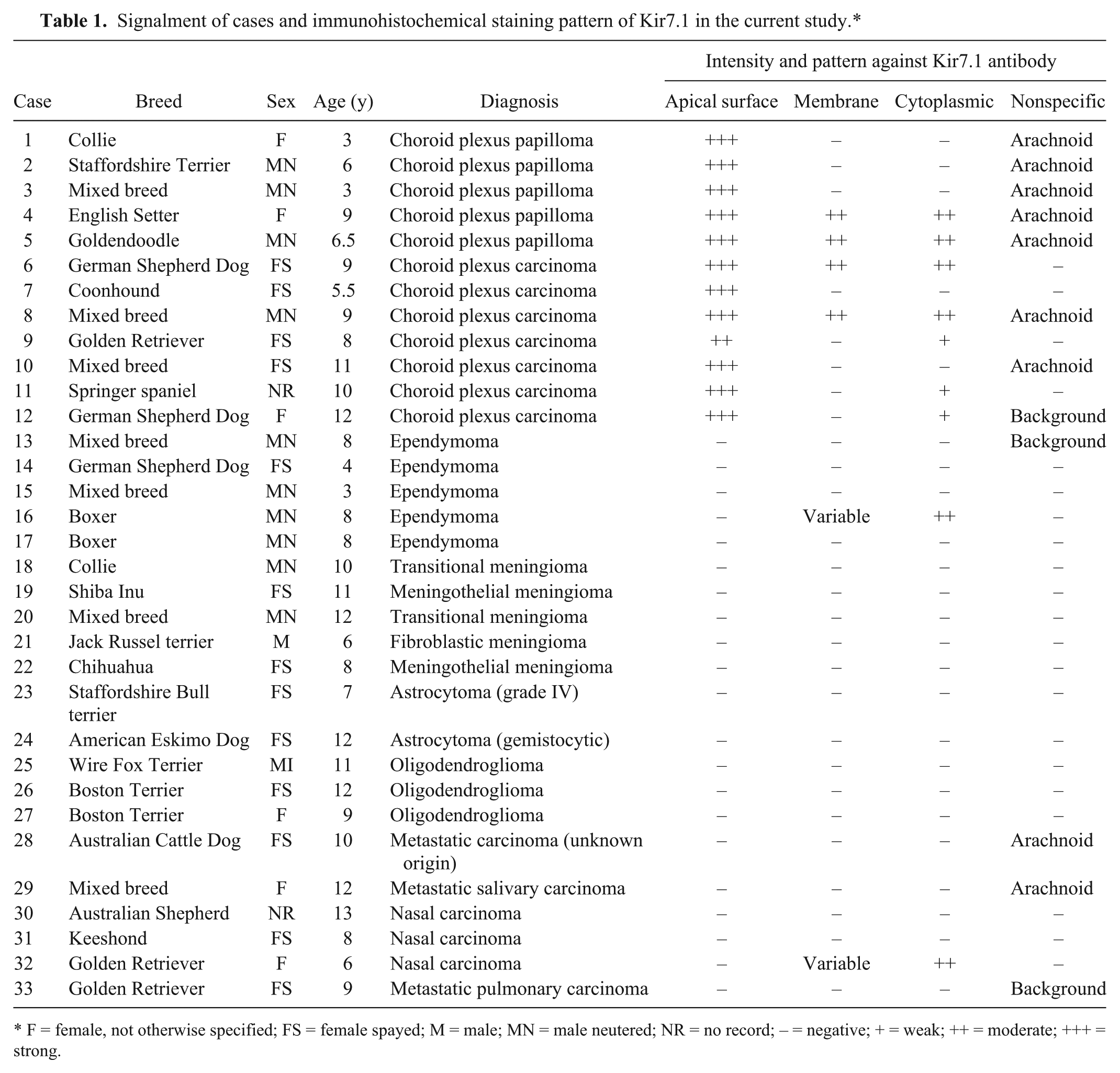
F = female, not otherwise specified; FS = female spayed; M = male; MN = male neutered; NR = no record; – = negative; + = weak; ++ = moderate; +++ = strong.
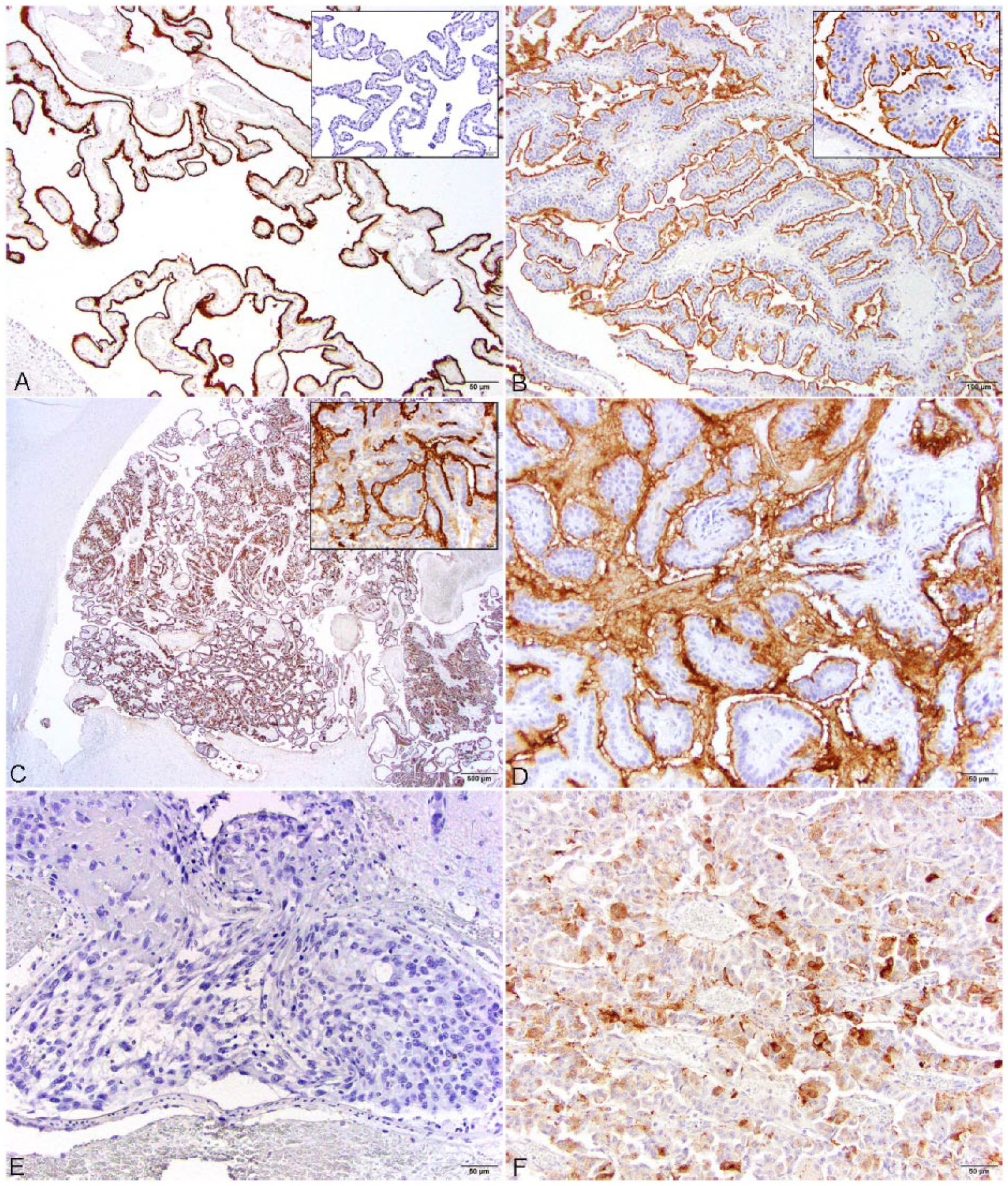
Histologic features and Kir7.1 immunoreactivity in normal and neoplastic canine choroid plexus and other brain neoplasms.
CPTs represent 7–10% of the primary brain tumors in the dog. In dogs, the diagnosis of CPTs has not been difficult if a thorough postmortem examination is performed, unlike humans where differentiating CPTs from metastatic carcinomas in the brain frequently poses a clinical challenge. 9 In the current clinical setting in veterinary medicine, more brain biopsies are being performed, and accurate diagnosis of the neoplasm directly correlates to the outcome of the case. Although CPTs generally have a distinct pattern in the context of location, biopsied specimens may lack the architecture that is required for accurate diagnosis. The staining patterns of a variety of immunohistochemical markers have been evaluated as a marker for CPTs in dogs. In a previous review, pan-cytokeratin and glial fibrillary acidic protein were listed as useful immunohistochemical markers for canine CPTs; however, these are neither specific nor diffuse in staining pattern.1,3–5,9,11,12 Previous investigations revealed inconsistent nuclear and cytoplasmic staining of cell adhesion molecules, E-cadherin, and β-catenin between normal choroid plexus and neoplasm.3,9 Additionally, N-cadherin may be associated with the dissemination of CPC cells. 3 In more broad-scale studies to immunohistochemically characterize canine neuroepithelial tumors, numerous markers including nestin, neurofilament, β-III tubulin, doublecortin (DCX), cytokeratins, ionized calcium-binding adaptor molecule I (IbaI), Ki-67, epithelial growth factor receptor, c-erbB2, pERK1/2, pAkt, p53, cleaved caspase 3, Bcl-2, and Bcl-xL were assessed. 4 Of these markers, all neoplastic cells were positive for Bcl-2 and Bcl-xL. 4 In addition to the previously mentioned markers, an earlier study examined epithelial cell adhesion molecule (EpCAM), epithelial membrane antigen, carcinoembryonic antigen, proliferating cell nuclear antigen, S100, vimentin, and MIB-1, none of which showed consistent immunoreactivity against choroid plexus and derived tumors. 1
In human pathology, multiple studies have been performed to identify specific immunomarkers for CPTs to differentiate against metastatic carcinomas of the brain.2,13 Of the markers that have been explored, Kir7.1 has been demonstrated to be a reliable marker for CPTs. In the present study, the apical membrane of the normal and all neoplastic choroid plexus were consistently strongly immunoreactive with Kir7.1. This indicates that, as in humans, Kir7.1 is an excellent marker for CPT in dogs regardless of grade. Of the 21 non-CPTs, 2 were immunoreactive with Kir7.1, an ependymoma and a nasal carcinoma, both of which showed variable membrane staining and moderate cytoplasmic staining. Both neoplasms lacked strong immunolabeling of the apical membrane, which all CPTs displayed. This should help prevent an erroneous deterring from a diagnosis of CPT. In conclusion, the data presented herein indicate that Kir7.1 is a valuable diagnostic tool for the diagnosis of CPTs in the dog and has a parallel immunoreactivity to that seen in human cases of CPTs.
Footnotes
Acknowledgements
We thank the histopathology technicians at Cornell University College of Veterinary Medicine for assistance with case processing and Jen Patterson for assistance with images. We also thank Drs. Alexander de Lahunta and James Hammond for some of the cases included in the series.
Authors’ contributions
EJ Choi contributed to design of the study, and contributed to acquisition, analysis, and interpretation of data. EA Sloma contributed to conception and design of the study, and contributed to acquisition and analysis of data. AD Miller contributed to conception and design of the study, and contributed to acquisition, analysis, and interpretation of data. All authors drafted the manuscript; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Catalog no. sc-22440, Santa Cruz Biotechnology, Dallas, TX.
b.
Invitrogen/Life Sciences, Grand Island, NY.
c.
Dako North America Inc., Carpinteria, CA.
d.
Vectastain ABC Elite, Vector Laboratories Inc., Burlingame, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
