Abstract
The diagnosis of malignant melanoma can be difficult because these tumors can be amelanotic and may contain diverse variants and divergent differentiations, of which the signet-ring cell subtype is very rare and has only been described in humans, dogs, cats, and a hamster. We describe herein histopathologic and immunohistochemical approaches taken to diagnose a case of signet-ring cell malignant melanoma with myofibroblastic differentiation in a cat. A tumor within the abdominal skin of a 2-year-old cat was composed of signet-ring cells and irregularly interwoven streams of spindle cells. Both neoplastic cell types were periodic-acid–Schiff, Fontana, and Sudan black B negative. Signet-ring cells strongly expressed vimentin and S100 protein. Spindle cells strongly expressed vimentin and smooth muscle actin; some cells expressed S100, moderately neuron-specific enolase, and others variably actin and desmin. A few round cells expressed melan A, and a few plump spindle cells expressed melan A and PNL2, confirming the diagnosis of amelanotic signet-ring cell malignant melanoma with myofibroblastic differentiation in a cat. Differential diagnoses were excluded, including signet-ring cell forms of adenocarcinomas, lymphomas, liposarcomas, leiomyosarcomas, squamous cell carcinomas, basal cell carcinomas, and adnexal tumors.
Keywords
Cutaneous melanoma is uncommon in cats, representing ~0.5% of feline skin tumors, without any sex or breed predilections. 24 According to the World Health Organization’s International Histological Classification of Tumors of Domestic Animals (WHO DA), cell morphology of melanomas is quite variable even within a single tumor and can be composed of spindle cells, epithelioid, polygonal, and/or round cells. 12 Additionally, especially in malignant melanomas, a fusiform, whorled, or nested appearance and an uncommon balloon-cell variant can occur. 12 However, description of the signet-ring cell variant, as it occurs in humans, has so far only been recognized in 21 cats, 3 dogs, and 1 Chinese hamster, and is not yet mentioned in the WHO DA.7,8,12,14,15,17,22,23,25 In addition to the above variants, divergent differentiations within a single malignant melanoma have been reported, such as epithelial, neuroendocrine, ganglionic and ganglioneuroblastic, osteocartilaginous, rhabdomyosarcomatous, smooth muscle, Schwannian and perineurial, as well as fibroblastic or myofibroblastic.3,4 To our knowledge, signet-ring cell melanoma with myofibroblastic differentiation, as proposed in our case, has not been reported in cats. Potential mixture with other cell types (e.g., myofibroblastic cells) can also occur, representing proliferation of reactive stromal myofibroblasts within the tumor microenvironment, and making discrimination from tumor cell differentiation difficult. 3
Demonstration of immunohistochemical (IHC) and ultrastructural features of both melanocytic and myofibroblastic features within the same cell population can be used to differentiate true divergent tumor cell differentiation, as we propose in this case, from reactive stroma. 3 As the melanoma in our case is composed of 2 main components, signet-ring and spindle or myofibroblastic cells, differential diagnoses must be considered and excluded for both components. Signet-ring morphology can also occur in adenocarcinomas, lymphomas, liposarcomas, leiomyosarcomas, squamous cell carcinomas, basal cell carcinomas, and adnexal tumors.4,5,7,15,22,23 Granular cell tumors, malignant triton tumors, extrarenal malignant rhabdoid tumors, pleomorphic rhabdomyosarcomas, undifferentiated sarcomas, and plasmacytomas can also exhibit similar morphology to round signet-ring cells (plasmacytoma, granular cell tumor), or spindle cells (undifferentiated sarcoma), or both cell types (malignant triton tumor, extrarenal malignant rhabdoid tumor, pleomorphic rhabdomyosarcoma).4–7,10,11,15,16,20,22,23
Comparable or overlapping IHC expression patterns make diagnosis of melanoma even more difficult. Markers typically used and often expressed by melanomas are vimentin, S100 protein, and the more melanoma-specific markers HMB45 (only in human melanomas), tyrosinase or tyrosinase-related protein 1 and 2 (TRP-1, TRP-2), melan A, and newer melanoma markers to yet unidentified antigens such as PNL2 and KBA62.2,18,19,21 Other tumors such as granular cell tumors are also positive for S100, frequently express neuron-specific enolase (NSE), and show focal melan A positivity in rare cases. Malignant rhabdoid tumors are vimentin positive and have inconsistently been reported to express S100, NSE, and occasionally melan A.6,10,11 Malignant triton tumors are described as vimentin and S100 positive. 20 Signet-ring cell melanomas in humans are commonly associated with metastatic or recurrent lesions.5,7,17,22 Regardless of the melanoma variant, the prognosis for feline cutaneous malignant melanomas is poor. 25 Herein we report a case of cutaneous amelanotic signet-ring cell malignant melanoma with previously undescribed interspersed myofibroblastic differentiation in a young cat, highlighting histopathologic and IHC features as well as diagnostic challenges.
A 2-year-old female spayed gray-and-black–striped European Shorthair cat was presented with a fast-growing, ~3.2 × 2 × 1 cm, beige, nonulcerated, indolent neoplasm in the abdominal skin near the third right mammary complex. It was first recognized by the owner 3 weeks prior to veterinary consultation. Regional lymph nodes were not enlarged at that time, and the mass was surgically excised by nodulectomy. Two months prior to this, another similarly located mass had been surgically removed by a different veterinarian, but was not further investigated. Three and a half months after the second (recurring) tumor had been removed, the cat was euthanized because of rapidly deteriorating general condition and after another mass was located in the mammary region. Radiographs of the lung revealed radiopaque nodular lesions suspicious for metastasis. According to the owner’s wish, further biopsies and autopsy were not performed.
The tumor was fixed in 10% nonbuffered formalin, embedded in paraffin, and stained with hematoxylin and eosin (HE), periodic acid–Schiff (PAS), Sudan black B, Fontana, Ziehl–Neelsen, and phosphotungstic acid hematoxylin (PTAH). Primary antibodies, antigen retrieval, blocking sera, secondary antibodies, and detection systems were used for IHC (Table 1). Tissues expressing these markers served as positive controls. For negative controls, the primary antibody was omitted, or sections were incubated with a monoclonal antibody against chicken lymphocytes or with rabbit immunoglobulin fraction a instead of monoclonal or polyclonal antibodies, respectively. Sections were deparaffinized in xylene substitute b and rehydrated using a descending alcohol series. Blocking of endogenous peroxidase activity was carried out by incubation in methanol with 0.5% hydrogen peroxide. Primary antibodies were incubated at 4°C overnight. Antibody binding was visualized using 3,3′-diaminobenzidine (DAB) c as chromogen, and sections were counterstained with Papanicolaou solution. d Slides were dehydrated in an ascending alcohol series and mounted with a xylene-based solution.
Antibodies and detection systems used for immunohistochemistry.
Actin (mouse mAb, clone HHF-35, MO 635), CD3 (cluster of differentiation 3; rabbit pAb, A452), Desmin (mouse mAb, M760), GFAP (glial fibrillary acidic protein; rabbit pAb, Z0334), Lysozyme (rabbit pAb, A099), MAC 387 (mouse mAb, clone MAC 387, MO747), MHC II (major histocompatibility complex II; mouse mAb, clone TAL.1B5, M746), melan A (mouse mAb, clone 103, M7916), MUM 1 (multiple myeloma oncogene 1; mouse mAb, clone Mum 1p, M7259), NF (neurofilament; (mouse mAb, clone 2F11, M762), S100 protein (rabbit pAb, Z0311), SMA (smooth muscle actin; mouse mAb, clone 1A4, M0851), Synaptophysin (mouse mAb, clone SY38, MO776), Vimentin (mouse mAb, clone V9, M0725), Rabbit PAP (peroxidase–anti-peroxidase; Z113); Dako Deutschland GmbH, Hamburg, Germany.
CD45R (rat mAb, clone B220, CL8990AP), Cedarlane Corp., Burlington, Ontario, Canada.
Chromogranin A (rabbit pAb, BM5082), Acris Antibodies GmbH, Herford, Germany.
Cytokeratin (mouse mAb, clone Ab1 AE1/AE3, MS-343-P), NSE (neuron-specific enolase; mouse mAb, 18-0196), Thermo Fisher Scientific GmbH, Dreieich, Germany.
Immunoglobulin G (rabbit pAb, 304-005-003), Mouse PAP (peroxidase–anti-peroxidase; ¦ (223-005-025); Dianova GmbH, Hamburg, Germany.
Laminin (rabbit pAb, L-9393), Sigma-Aldrich, St. Louis, Missouri.
PNL2 (melanoma antigen; mouse mAb, sc-59306), Santa Cruz Biotechnology Inc., Santa Cruz, California.
Myogenin (mouse mAb, ab1835), Abcam, Cambridge, UK.
Nerve growth factor receptor gp75 (mouse mAb, clone 7F10, NCL-NGFR), Leica Biosystems GmbH, Wetzlar, Germany.
Avidin–biotin complex, Vector Laboratories, Burlingame, California.
ImmPRESS reagent kit (MP7402), Linaris GmbH, Dossenheim, Germany.
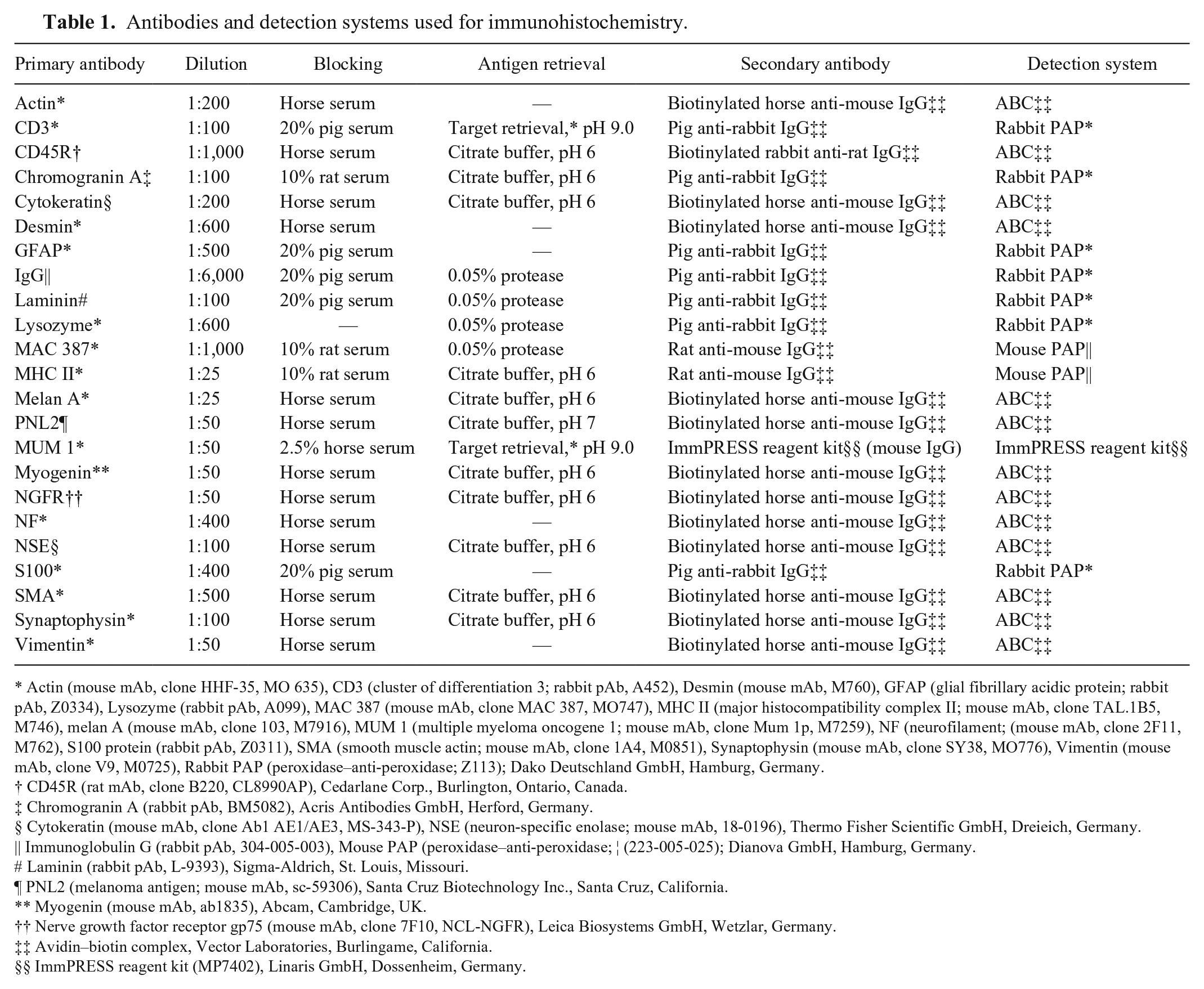
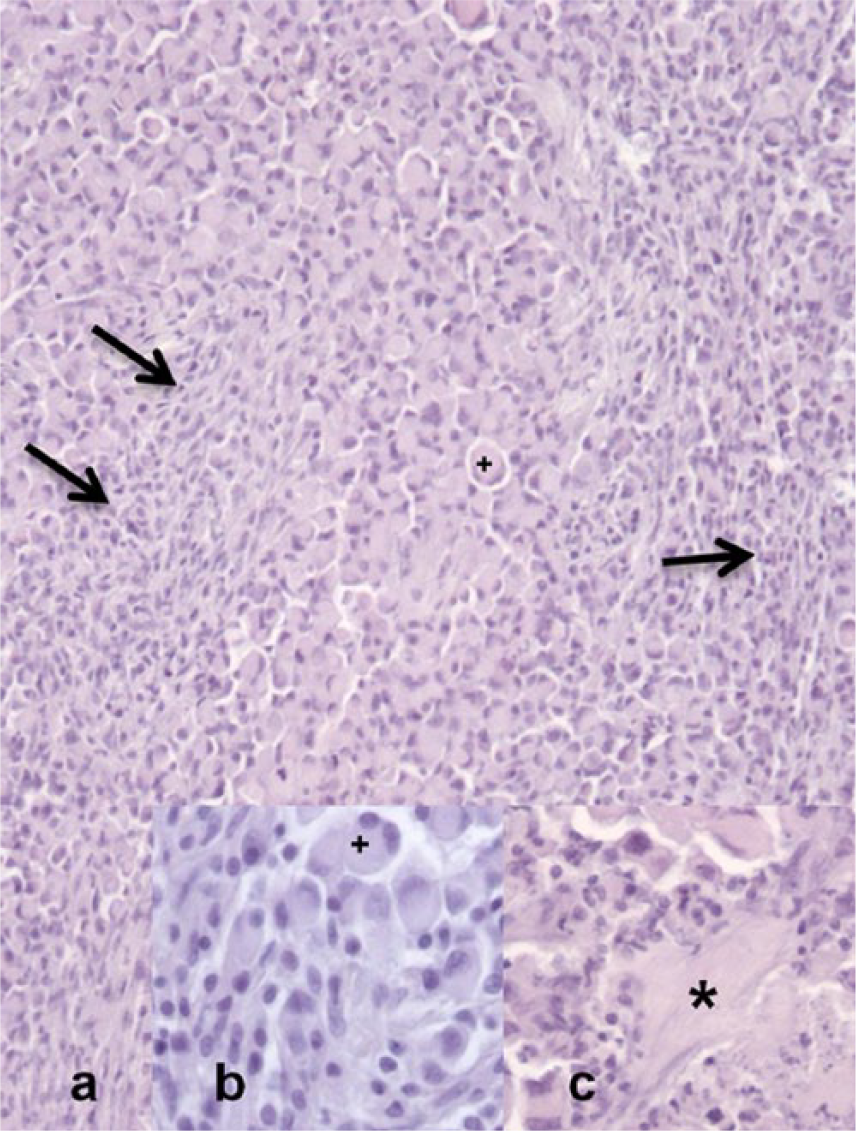
The 3.2 cm × 2 cm × 1 cm tumor was located in the abdominal skin, extended close to the epithelial border, and surrounded hair follicles and a cutaneous nerve. The mass was densely cellular, moderately demarcated, nodular, unencapsulated, with infiltrative growth. Cells were arranged in closely packed sheets of round to pleomorphic, 20–40 µm large cells with distinct cell borders, abundant pale eosinophilic, homogeneous cytoplasm, 1–3 c-shaped, eccentrically displaced nuclei with coarsely stippled chromatin, and 1–2 basophilic indistinct nucleoli (signet-ring cells; Figs. 1a, 1b, 2). Additionally, there were prominent irregularly interlacing streams of spindyloid, ~20 µm × 8 µm large cells with indistinct cell borders, moderate amounts of eosinophilic, homogeneous cytoplasm, an elongated to spindled, centrally placed nucleus with finely stippled chromatin, and 1 basophilic indistinct nucleolus (Fig. 1a, 1b). The tumor exhibited scant fibrous stroma and multiple multinucleated cells throughout the signet-ring cell population (Fig. 1a, 1b). Subtle junctional activity (Supplemental Fig. 1, available online at http://vdi.sagepub.com/content/by/supplemental-data) was evident, and there was prominent anisocytosis and anisokaryosis with 1–3 mitoses per high power field in both the signet-ring and spindle cell population (Fig. 1a, 1b). There were multiple areas of necrosis (Fig. 1c), and vascular invasion was recognized multifocally along the tumor margins (Fig. 2). Within the tumor, multiple accumulations of mononuclear inflammatory cells including lymphocytes, fewer plasma cells, and macrophages were present.
Signet-ring cell malignant melanoma in the abdominal skin of a cat.
Signet-ring cell malignant melanoma in the abdominal skin of a cat. Vascular invasion of periodic acid–Schiff (PAS)-negative signet-ring neoplastic cells. PAS. 400×.
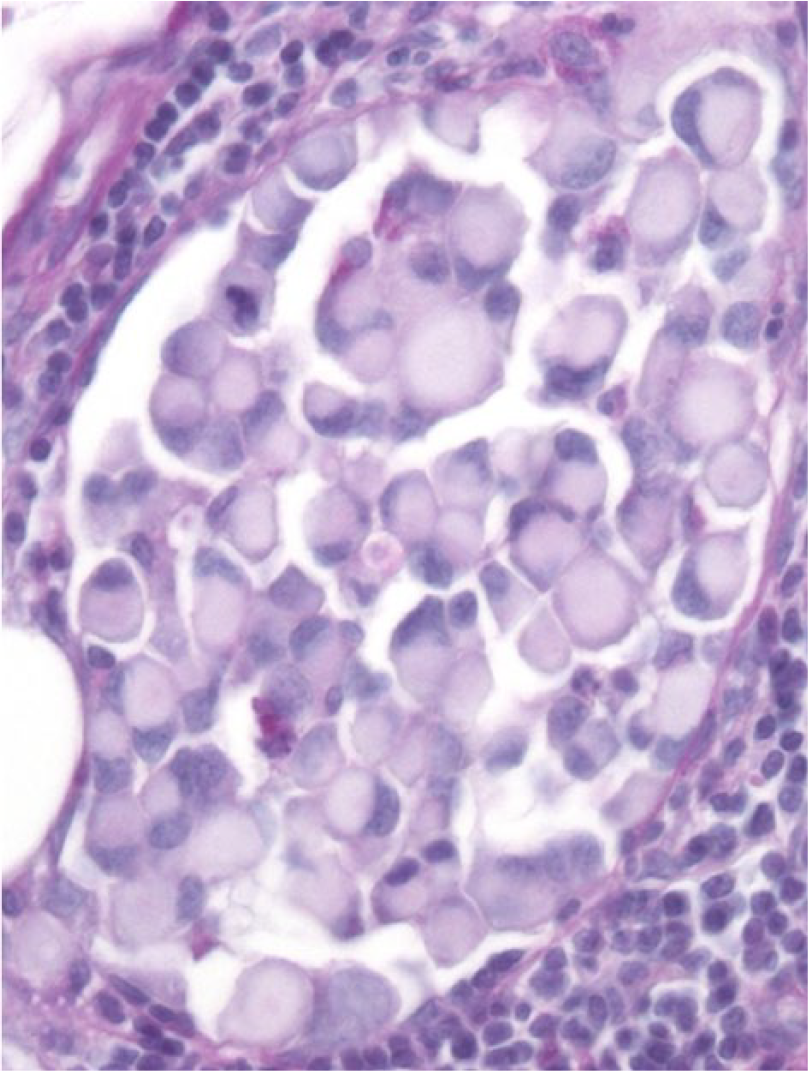
Both types of neoplastic cells stained negatively with PAS (Fig. 2), Ziehl–Neelsen, Sudan black B, and Fontana. The major cell population of signet-ring cells strongly expressed vimentin (Fig. 3c) and S100 (Supplemental Fig. 2, available online at http://vdi.sagepub.com/content/by/supplemental-data) and did not express any of the other markers except for a few cells displaying NSE and even fewer displaying melan A (Fig. 3a). The spindle cells strongly expressed vimentin and smooth muscle actin (SMA; Fig. 4), some of these cells moderately expressed S100 (Supplemental Fig. 2) and NSE, and others were variably positive for actin and desmin. A few plump spindle cells also expressed melan A (Supplemental Fig. 3, available online at http://vdi.sagepub.com/content/by/supplemental-data) and melanoma antigen (PNL2; Fig. 3b). Some interspersed cells were positive for major histocompatibility complex class II (MHC II), macrophage (MAC) 387, myeloma oncogene 1 (Mum 1), lysozyme, immunoglobulin (Ig)G, NSE, and nerve growth factor receptor (NGFR). The nerve within the tumor was labeled with neurofilament (NF) and NGFR. Accumulations of mononuclear inflammatory cells consisted of a mixed population of cluster of differentiation (CD)3- (T cells), CD45R- (B cells), MHC II–, MAC 387–, and lysozyme-positive cells (macrophages), and IgG- and Mum 1–positive cells (plasma cells; Table 2).
Signet-ring cell malignant melanoma in the abdominal skin of a cat.
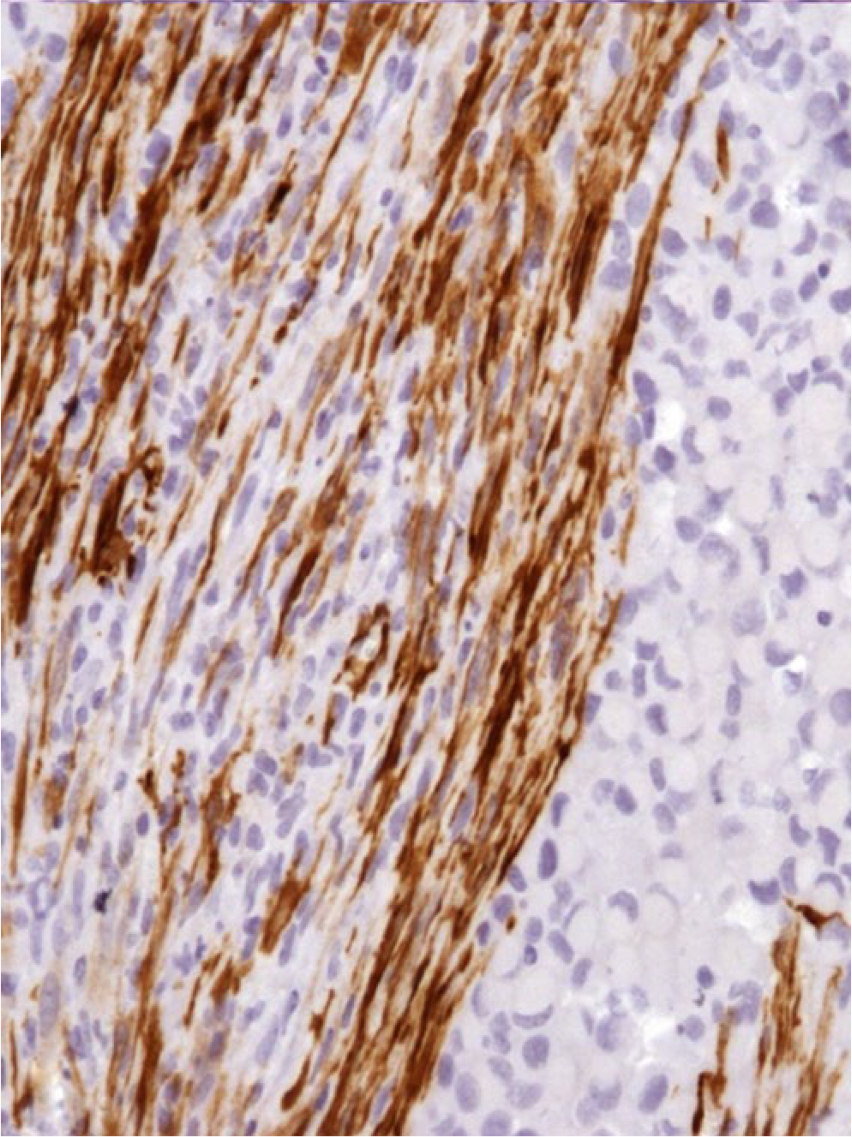
Signet-ring cell malignant melanoma in the abdominal skin of a cat. Interspersed spindle cells with myofibroblastic differentiation. Immunohistochemistry for smooth muscle actin, counterstained with Papanicolaou solution. 400×.
Antibodies and immunohistochemical staining results.*
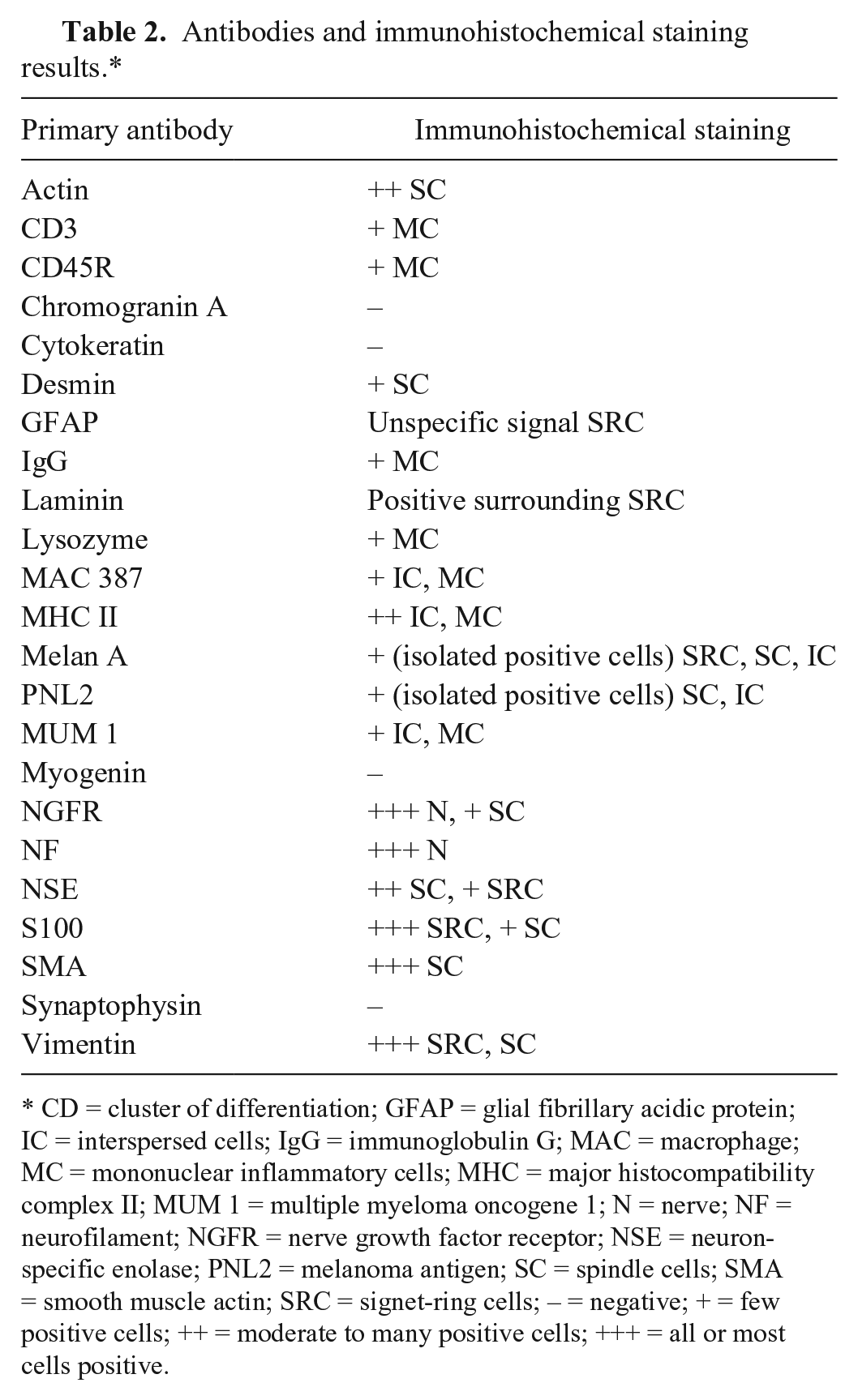
CD = cluster of differentiation; GFAP = glial fibrillary acidic protein; IC = interspersed cells; IgG = immunoglobulin G; MAC = macrophage; MC = mononuclear inflammatory cells; MHC = major histocompatibility complex II; MUM 1 = multiple myeloma oncogene 1; N = nerve; NF = neurofilament; NGFR = nerve growth factor receptor; NSE = neuron-specific enolase; PNL2 = melanoma antigen; SC = spindle cells; SMA = smooth muscle actin; SRC = signet-ring cells; – = negative; + = few positive cells; ++ = moderate to many positive cells; +++ = all or most cells positive.
Immunohistochemical results together with the unusual tumor cell morphology are most consistent with amelanotic signet-ring cell malignant melanoma with interspersed myofibroblastic differentiation. To our knowledge, this variant has not been reported in cats or other animals. 25 It should be emphasized that the affected cat in this case was only 2 years of age, indicating that even very young cats can develop signet-ring cell malignant melanoma. In humans, myofibroblastic differentiation has been reported exclusively in spindle-cell desmoplastic malignant melanomas. 3 According to the WHO DA, melanoma can appear as many different morphologic variants of which the balloon-cell variant is most uncommon; mixed-cell types within a single tumor mass also occur. 12 Mixtures of signet-ring and epithelioid-type cells have been reported in dogs and cats.8,25 Divergent differentiations as reported in human malignant melanomas such as fibroblastic or myofibroblastic, Schwannian and perineurial, smooth muscle, rhabdomyosarcomatous, osteocartilaginous, ganglionic and ganglioneuroblastic, neuroendocrine, and epithelial are not clearly distinguished as such in the WHO DA.3,12
In our case, myofibroblastic differentiation as opposed to proliferation of reactive stromal myofibroblasts was supported by histologic features such as anisokaryosis and anisocytosis of these cells, the appearance of very plump spindle cells suggesting they may represent intermediate cells between signet-ring and spindle cells, and the overall irregularity in which spindle cells interweave between signet-ring cells that is not typical for stromal cells (Fig. 1a, 1b). Myofibroblastic differentiation was also supported by IHC results showing strong expression of SMA in the interwoven spindle cell population (Fig. 4) of which some cells also expressed S100 (Supplemental Fig. 2) and therefore exhibited both myofibroblastic and melanocytic features. 3 Only a few spindle cells expressed desmin using a monoclonal antibody, supporting myofibroblastic differentiation. 3 In previous studies, there was no SMA expression in any of the feline and canine cases of signet-ring cell melanoma, and nonspecific desmin expression was observed in 13 of 21 feline cases using a polyclonal antibody.8,25 Some controversy still remains in distinguishing myofibroblastic differentiation from proliferation of reactive stromal myofibroblasts, and electron microscopy, which was not performed in this case, can be of help by ultrastructurally demonstrating melanocytic and myofibroblastic features within the same cell. 3
The signet-ring cell variant was first described in humans and later in animals, where 21 of 57 feline cutaneous malignant melanomas were identified as signet-ring cell type, of which none exhibited junctional activity, which was subtly present in our case (Supplemental Fig. 1).23,25 In addition to this report, signet-ring cell melanomas have been described only rarely in the skin of dogs and cats.8,17,25 Electron microscopy revealed that the signet-ring cell morphology is caused by numerous aggregates of vimentin-positive intermediate filaments.1,23,25 Comparable to previous canine and feline studies, the presented tumor was amelanotic, confirmed by negative Fontana staining, which made the diagnosis of melanoma even more difficult and strengthened the need for expression pattern analysis of the tumor cells.8,25 All feline and canine signet-ring cell melanomas reported to date were positive for vimentin, S100, and NSE; however, melan A was not tested or only present in 1 canine case.8,25 Signet-ring cells of our tumor also constantly expressed vimentin (Fig. 3c) and S100 (Supplemental Fig. 2); a few of them also expressed NSE and very rarely melan A (Fig. 3a). Spindle cells were also strongly immunopositive for vimentin and SMA (Fig. 4), but some were also positive for S100 (Supplemental Fig. 2) and/or NSE. A few plump spindle cells also expressed PNL2 (Fig. 3b), which is a sensitive melanoma marker especially in conjunction with other typical melanoma markers in humans and other animals such as dogs, horses, and pigs.2,13,18,19,21 NSE expression is commonly described in melanomas of humans and animals and may be the result of neuroectodermal origin of melanocytes.4,8,25 We excluded neuronal or neuroendocrine differentiation in this case, because none of the tumor cell populations expressed other neuroendocrine markers such as NF, synaptophysin, or chromogranin. There was only very faint GFAP labeling, and only very few spindle cells expressed NGFR, which can be accounted for by normal nerve structures in preexisting dermal stroma.
The poor melan A and PNL2 expression might indicate dedifferentiation as the tumor most likely represents a recurrence. In contrast, human signet-ring cell melanomas are mostly positive for melanoma-specific antibody HMB45 and/or melan A. 15 A study on expression of PNL2 in different canine melanomas revealed that 75% of amelanotic melanomas were positive for PNL2; however, signet-ring cell melanomas were not included. 19 To our knowledge, there are no specific studies testing PNL2 expression in human or animal signet-ring cell melanomas. Our case suggests that this melanoma variant does not widely express PNL2, but further studies are needed to validate this hypothesis. The melan A and PNL2 labeling of only a few cells (Fig. 3a, 3b; Supplemental Fig. 3) and amelanotic appearance demanded a broad range of IHC markers to exclude signet-ring cell forms of adenocarcinomas, lymphomas, liposarcomas, leiomyosarcomas, squamous cell carcinomas, basal cell carcinomas, and adnexal tumors.5,7,15,22,23
Other tumors were excluded based on similar histologic appearance and overlapping expression patterns, for example, granular cell tumors (may resemble signet-ring cells, express S100 and frequently NSE, and, in rare cases, melan A), malignant triton tumors (may have similar mixed cell populations, and express vimentin and S100), extrarenal malignant rhabdoid tumors (may have similar mixed cell populations, express vimentin, and have inconsistently been reported to be S100, NSE, and melan A positive), pleomorphic rhabdomyosarcomas, plasmacytomas (may resemble signet-ring cells), and undifferentiated sarcomas (may also appear spindled and pleomorphic, and express vimentin).6,10,11,15,16,20 Rhabdomyoblastic differentiation or lipid accumulations were ruled out by lack of cross striations using PTAH staining or by Sudan black B staining, respectively. Granular cell tumors, which are PAS positive and can also express vimentin and S100, were excluded by negative PAS staining (Fig. 2). Lack of PAS staining has previously been described in human and canine signet-ring cell melanomas.8,15,22
Prognosis for malignant melanoma in cats is independent of melanoma variant or cell type and considered poor because of recurrence and regional metastasis in up to 50% of cases. Survival times after removal of the tumor until death or euthanasia ranged from 0.5 to 18 months in a study employing clinical follow-up data of 45 cats with different melanoma variants, including the signet-ring cell variant.12,24,25 Because the present case represented a recurrence, showed vascular invasion (Fig. 2), and potential lung metastasis, distant metastasis seems likely. Previous studies indicated lymphatic invasion in 6 of 21 feline signet-ring cell melanomas. 25
Initiation of up to 65% of cutaneous melanomas in humans and most melanomas in Angora goats is thought to be caused by ultraviolet A and ultraviolet B radiation, but not much is known concerning other animal species. 24 This cause is not very likely in our case, because the tumor was not located in a sun-exposed location and because of the dark hair color of the cat. However, because breed and familial clustering in domestic animals suggest that genetic susceptibility may play a critical role, even young animals, as in our case, might be affected.9,24 With the exception of gray horses, malignant transformation of benign lesions is very uncommon in animals, and most melanomas are believed to arise de novo.9,24 To our knowledge, there is no described melanoma predisposition for cats with a certain hair or skin color.
Footnotes
Acknowledgements
We thank the referring veterinarian Dr. Petra Leitenstorfer for providing information on clinical and follow-up data.
Authors’ contributions
M Hirz contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. C Herden contributed to conception and design of the study; contributed to analysis and interpretation of data; and critically revised the manuscript. All authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Dako Deutschland GmbH, Hamburg, Germany.
b.
Roti-Histol, Carl Roth GmbH & Co KG, Karlsruhe, Germany.
c.
Sigma-Aldrich, St. Louis, MO.
d.
Merck KGaA, Darmstadt, Germany.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
