Abstract
Renal leiomyosarcoma was diagnosed in a 10-year-old Domestic Shorthair cat with a 3-year history of clinically managed, chronic renal disease. Sudden death was preceded by a brief episode of mental dullness and confusion. At postmortem examination, the gross appearance of the left kidney was suggestive of hydronephrosis, and a nephrolith was present in the contralateral kidney. However, histology revealed an infiltrative, poorly differentiated, spindle cell sarcoma bordering the grossly cavitated area. Neoplastic cells were immunoreactive for vimentin and smooth muscle actin, which led to a diagnosis of renal leiomyosarcoma; neoplastic cells were not immunoreactive for desmin. Leiomyosarcoma arising in the kidney is a rare occurrence in humans and an even rarer occurrence in veterinary medicine with no prior cases being reported in cats in the English literature. The macroscopic appearance of the tumor at postmortem examination was misleadingly suggestive of hydronephrosis as a result of the large cavitation and may be similar to particularly unusual cases of renal leiomyosarcomas in humans that have a cystic or cavitated appearance.
Keywords
Primary renal tumors of domestic animals, including renal carcinoma and nephroblastoma, are uncommon; mesenchymal tumors are even more rare. 1 In cats, the most common tumor affecting the kidneys would be lymphoma followed by renal cell carcinoma (Pressler B. Diagnostic approach to renomegaly. Proceedings of 79th Western Veterinary Conference; March 4–8, 2007; Las Vegas, Nevada). Mesenchymal tumors that have been most often reported in dogs and cats include undifferentiated sarcoma, fibroma, fibrosarcoma, hemangioma, and hemangiosarcoma, with renal hemangiosarcoma being the most commonly reported (Pressler B. Diagnostic approach to renomegaly). 24 Renal leiomyosarcoma is particularly rare, with only 1 report in the English literature of renal leiomyosarcoma in the dog and none in the cat. In human medicine, there are limited reports, as sarcomas constitute ~1% of all renal neoplasms, although leiomyosarcomas account for ~50–60% of those cases.22,26,29 Renal leiomyosarcomas are thought to arise from the renal capsule, the pelvis, or vessels within the kidney. 29 In humans, primary renal leiomyosarcoma is associated with a poor prognosis because an aggressive clinical course with early recurrences is common despite radical nephrectomy. 26
A 10-year-old, spayed female, Domestic Shorthair cat was presented for autopsy after a 3-year history of clinically diagnosed and managed, chronic renal disease. Sudden death was preceded by a brief episode of mental dullness and confusion. On postmortem examination, the left kidney was rounded and larger than the right kidney measuring ~4 cm in diameter. On the cut surface, the renal cortex and medulla were replaced by a single, large cavity containing ~15 mL of clear, slightly viscous, mucinous fluid surrounded by a thin, ~5 mm wide band of white to tan, firm tissue. The left adrenal was adhered to the left kidney. The right kidney was rounded with ~5 mm in diameter nephrolith in the renal pelvis, which was mildly ectatic. Tissues were fixed in 10% buffered formalin and embedded in paraffin; 5-μm sections were stained with hematoxylin and eosin. Histology of the ~5 mm of tissue remaining in the left kidney revealed a moderately cellular, poorly demarcated, infiltrative, unencapsulated neoplasm with cells loosely arranged in streams and occasionally individualized, supported by a fine, fibrovascular stroma. Cells were plump and pleomorphic, ranging from spindled to rounded, with distinct cell borders containing abundant intensely eosinophilic homogeneous cytoplasm and variably eccentric to central, elongated to reniform nuclei with coarsely clumped chromatin and 0–1 variably distinct, central nucleoli. Anisocytosis and anisokaryosis were severe (Figs. 1, 2), and mitoses were 1–2 per high power field. Neoplastic cells invaded the renal capsule. Additionally, there was subcapsular infiltration by large numbers of lymphocytes, which were also present in large numbers along with plasma cells and histiocytes among the neoplastic population. The large cavity observed at postmortem examination was not lined by epithelium, but instead by neoplastic cells inconsistent with a cyst or hydronephrosis. Regarding the right kidney, histology showed severe, multifocal to coalescing, chronic, lymphoplasmacytic tubulointerstitial nephritis and nephrolithiasis with glomerulosclerosis, tubular casts, and peripelvic fibrosis.
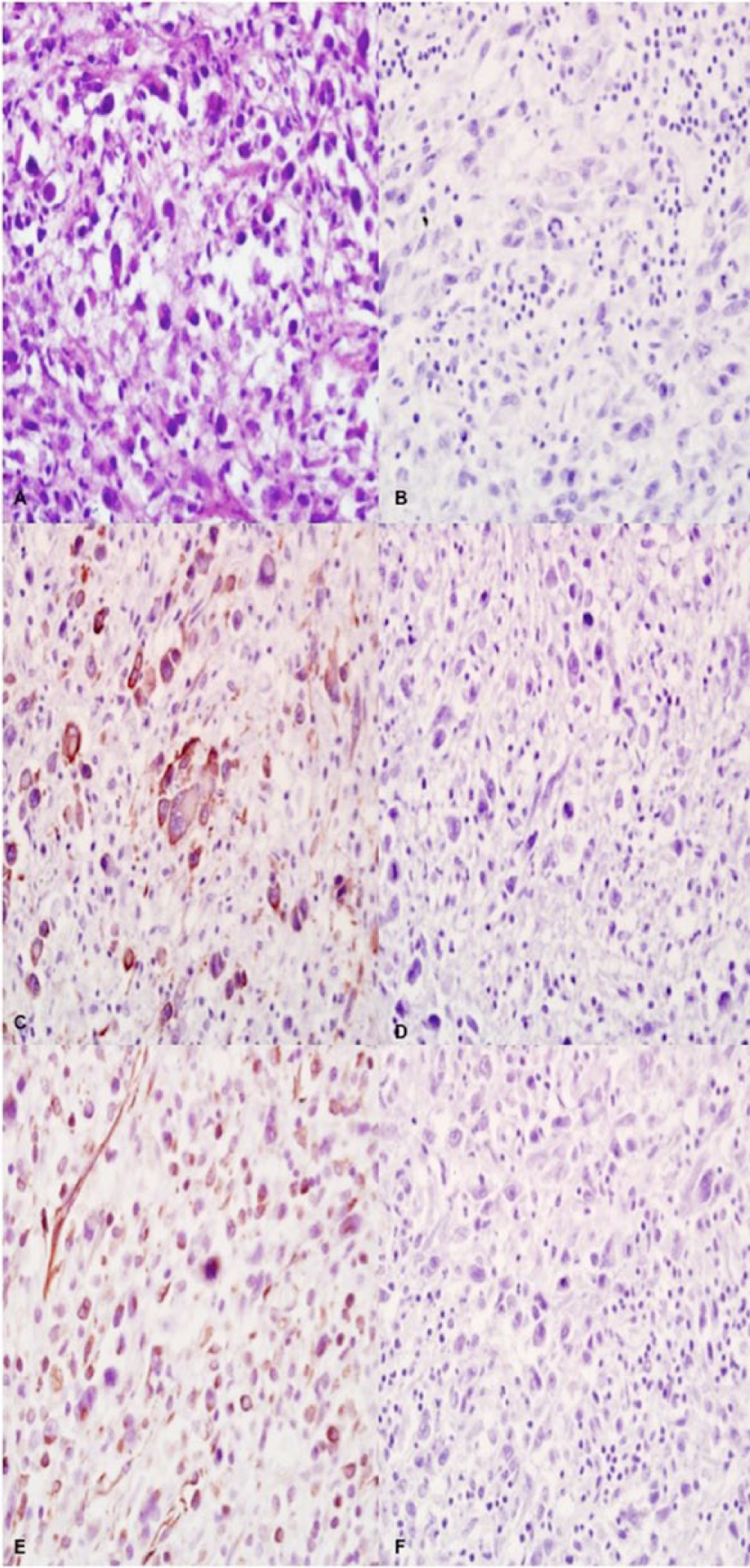
Left kidney; neoplastic cells.
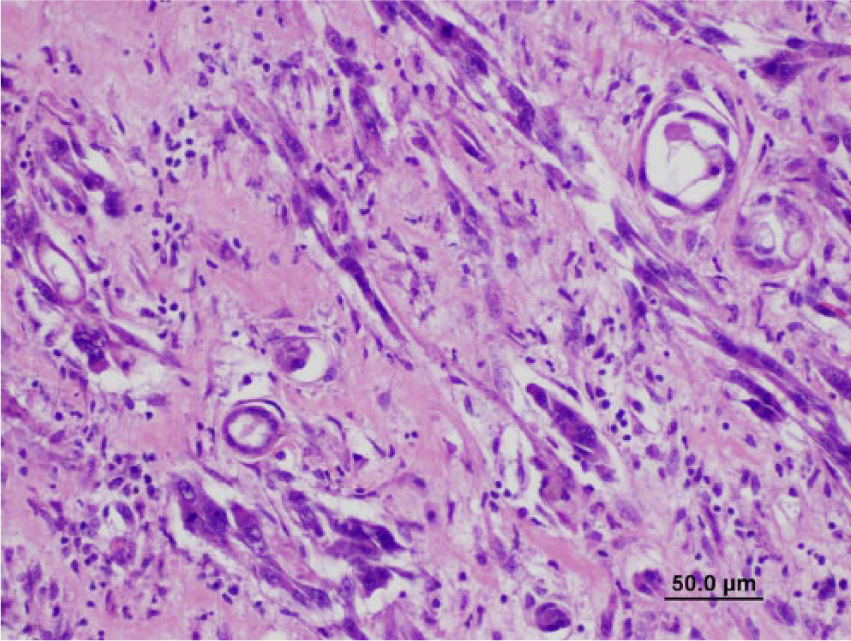
Spindyloid neoplastic cells with severe anisocytosis and anisokaryosis and multinucleation in the left kidney. Hematoxylin and eosin. Bar = 50 μm.
Immunohistochemical staining of formalin-fixed, paraffin-embedded tissue sections from the neoplasm in the left kidney was performed as described by the manufacturer. a Neoplastic cells were not immunoreactive for cluster of differentiation (CD)18 (Fig. 1B), cytokeratin AE1/AE3 (Fig. 1D), and sarcomeric actin (Fig. 1F), ruling out neoplasms of histiocytic, epithelial, and skeletal muscle origin, respectively. Cells were diffusely, strongly immunoreactive for vimentin (Fig. 1E), indicating mesenchymal origin, and also α-smooth muscle actin (Fig. 1C), indicating smooth muscle origin, thus warranting a diagnosis of leiomyosarcoma. Cells were not immuno-reactive for desmin.
Renal neoplasia is generally rare in cats. The most common renal neoplasm in cats is lymphoma; however, the kidneys are more often a site of metastasis rather than a primary site for the development of lymphoma. 11 Regarding primary renal neoplasia of cats, similar to other species, epithelial neoplasms predominate with mesenchymal neoplasms being reportedly exceedingly rare. A challenging feature of this case was that, at postmortem examination, the macroscopic appearance of the left kidney was suggestive of hydro-nephrosis; therefore, neoplasia was not suspected at that time. Renal leiomyosarcomas with a cystic appearance have been reported in the human literature, although very rarely. 6 Additionally, the clinical history of mental dullness and confusion could have suggested a lesion within the brain; however, it is presumed that the clinical presentation was related to uremia as no gross anatomic or histologic lesions were observed in the brain.
Immunohistochemistry was crucial in evaluating the cell-type of origin in this case given the severely pleomorphic histomorphology of the cells. Based on immunohistochemical results, a presumptive diagnosis of leiomyosarcoma was made. It was an unexpected finding that the neoplasm did not show immunoreactivity for desmin, because desmin is often used as a muscle marker. However, there are cases in the literature of leiomyosarcomas in cats lacking immunoreactivity for desmin. 12 There may be differential expression of desmin by smooth muscle depending on anatomical location or specific tissue or origin and, in fact, it has been reported that smooth muscle of blood vessels may not express desmin. 17 Because renal leiomyosarcomas in human cases have been speculated to arise from vessels within the kidney, the lack of immunoreactivity for desmin in this case is not confounding to the diagnosis of renal leimyosarcoma. 29 Another differential diagnosis that was considered in this case was inflammatory myofibroblastic tumor as a result of the presence of a spindloid population of neoplastic cells admixed with intense inflammation consisting of lymphocytes, plasma cells, and histiocytes. There is a paucity of reported cases of inflammatory myofibroblastic tumors in the veterinary literature, with only 1 case in a brush-tailed porcupine and 1 case report of 2 dogs with that specific tumor designation.13,15 However, neoplasms with a similar histologic appearance have been reported in various species in various locations, but frequently are described as “pseudotumors” or “inflammatory pseudotumors” or some other similar term, so their actual incidence may be underestimated.2,3,8,9,19,20,23,28,30 None of these cases in veterinary species involved the kidney. Similar varying terminology has been problematic in the human literature with such varied terms as “inflammatory pseudotumor,” “pseudosarcomatous myofibroblastic or fibromyxoid lesion,” and “plasma cell granuloma” for the same lesion. 14 There are cases in the human literature of inflammatory myofibroblastic tumors affecting a wide variety of tissues including the kidney, but these are rare with occurrence in the lungs of children and young adults being more typical and extrapulmonary cases overall being considered more unusual. Inflammatory myofibroblastic tumors can present a diagnostic challenge for both clinicians and pathologists in both human and veterinary medicine. In human cases, h-caldesmon immunoreactivity has been helpful in differentiating leiomyosarcoma from inflammatory myofibroblastic tumors because it is specific to smooth muscle and not expressed in myofibroblasts. 4 It is uncertain if h-caldesmon could be helpful in feline cases. In the present case, although it is difficult to completely rule out myofibroblastic origin and inflammatory myofibroblastic tumor in particular, it was considered less likely. Neoplastic cells in our case were nearly diffusely immunoreactive for smooth-muscle actin, whereas myofibroblastic tumors often have more patchy immunoreactivity; lack of immunoreactivity for pancytokeratin would also make a myofibroblastic tumor less likely as some reports indicate myofibroblastic tumors often express cytokeratins in addition to vimentin. 5 Additionally, there was a history of chronic renal disease in our case, and chronic inflammation was present in the contralateral kidney as well and not confined to the kidney affected by the neoplasm; the inflammation was therefore thought to be related to the chronic renal disease and further destruction of the kidney by the neoplasm rather than a distinctive feature of the tumor.
Because human cases of renal leiomyosarcoma typically have a relatively short and aggressive clinical course, it is considered unlikely that the renal neoplasm was the cause of chronic renal disease for the 3 years it was clinically noted and managed. The clinical history of chronic renal disease and evidence of nephrolithiasis and lymphoplasmacytic inflammation observed at postmortem examination and on histology, respectively, led to speculation and discussion on the possible role that chronic renal disease and/or chronic inflammation could have had in the development of the tumor. One case report describing the development of renal neoplasia in the face of chronic kidney disease in 2 cats exists in the literature. 10 The case report described 2 cats, which developed renal transitional cell carcinoma ~1 year after the diagnosis of chronic renal disease; in one of these cases, the neoplasm was confined to a single kidney, whereas the other one had bilateral disease. The role that chronic renal disease played in the development of neoplasia in these cases, similar to ours, was unclear, but the authors pointed out that an increased incidence of malignancy including renal tumors has been reported in humans with end-stage renal disease with or without the history of renal transplant. 27 Rudolph Virchow appreciated a tendency of neoplasia to occur at sites of chronic inflammation as early as 1863, and in the last several years, the idea of inflammation as a key event in the development of cancer has continued gaining steam. 16 Evidence is continually building in support of the idea that the generation of cytokines, reactive oxygen and nitrogen species, and growth factors by inflammatory cells in chronic inflammatory conditions may create an environment of continued cellular proliferation and tissue and DNA damage that contributes to the development of cancer. In general, a large number of cancers have been identified in domestic animals and humans alike that seem to be associated with chronic inflammatory conditions, and an extensive comparative review is currently available in the veterinary literature.18,21,25 In cats specifically, there are many tumors with well-described associations with chronic inflammation including posttraumatic ocular sarcomas, vaccine-associated sarcomas, cutaneous squamous cell carcinoma, oral squamous cell carcinoma, and intestinal lymphoma.7,21 Given the apparent propensity of cats to develop sarcomas in instances of chronic inflammation, we speculated on the role of chronic, low-grade inflammation in the development of a primary renal sarcoma in our case. However, it is impossible to draw any definitive conclusions on the role chronic inflammation and/or chronic renal disease could have played in the development of neoplasia in this case considering the fact that chronic renal disease is extremely common in the cat and renal sarcomas are not. It could therefore be more likely that because chronic renal disease and neoplasia in general are more common in geriatric cats, these 2 conditions occurred simultaneously as a matter of coincidence. Furthermore, because chronic inflammation was also observed in the contralateral kidney in which the neoplasm was not present, whether or not inflammation initiated the development of the neoplasm in this case could not be discerned.
This case presented unique challenges in gross and histologic interpretation. Macroscopically, the cavitated appearance of the tumor mimicked severe hydronephrosis, and neoplasia was therefore not a consideration at the time of postmortem examination. Histologically, the pleomorphic appearance of the cells made the cell of origin difficult to determine prior to immunohistochemical evaluation. Challenges of this case highlight the importance of including neoplasia in the differentials for a hydronephrotic kidney and of including leiomyosarcoma as a differential for a primary renal sarcoma in cats.
Footnotes
Acknowledgements
We thank Dr. Brett A. Berryhill at Staring Plaza Veterinary Center in Baton Rouge, Louisiana, for his submission of the case and collaboration on review of the clinical history.
Authors’ contributions
N Fowlkes drafted the manuscript. All authors contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; critically revised the manuscript; gave final approval; and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
The antibody for CD18 was obtained from Dr. Peter Moore, University of California, Davis, California. Antibodies for cytokeratin AE1/AE3, vimentin, sarcomeric actin, α-smooth muscle actin, and desmin were obtained from Dako North America Inc., Capinteria, California.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for color figures was provided by the Department of Pathobiological Sciences, School of Veterinary Medicine, Louisiana State University, Baton Rouge, Louisiana.
