Abstract
Hypoglycin A (2-amino-3-(2-methylidenecyclopropyl)propanoic acid) is the plant toxin shown to cause atypical myopathy in horses. It is converted in vivo to methylenecyclopropyl acetic acid, which is transformed to a coenzyme A ester that subsequently blocks beta oxidation of fatty acids. Methylenecyclopropyl acetic acid is also conjugated with carnitine and glycine. Acute atypical myopathy may be diagnosed by quantifying the conjugates of methylenecyclopropyl acetic acid plus a selection of acyl conjugates in urine and serum. We describe a new mass spectrometric method for sample volumes of <0.5 mL. Samples were extracted with methanol containing 5 different internal standards. Extracts were analyzed by ultra–high-performance liquid chromatography–tandem mass spectrometry focusing on 11 metabolites. The total preparation time for a series of 20 samples was 100 min. Instrument run time was 14 min per sample. For the quantification of carnitine and glycine conjugates of methylenecyclopropyl acetic acid in urine, the coefficients of variation for intraday quantification were 2.9% and 3.0%, respectively. The respective values for interday were 9.3% and 8.0%. Methylenecyclopropyl acetyl carnitine was detected as high as 1.18 µmol/L in serum (median: 0.46 µmol/L) and 1.98 mmol/mol creatinine in urine (median: 0.79 mmol/mol creatinine) of diseased horses, while the glycine derivative accumulated up to 1.97 mmol/mol creatinine in urine but was undetectable in most serum samples. In serum samples from horses with atypical myopathy, the intraday coefficients of variation for C4–C8 carnitines and glycines were ≤4.5%. Measured concentrations exceeded those in healthy horses by ~10 to 1,400 times.
Keywords
Introduction
Single cases as well as clusters of atypical myopathy in grazing horses, also called seasonal pasture myopathy, have been observed repeatedly in Europe and in North America. Such cases are now ascribed to intoxication by hypoglycin A (2-amino-3-(2-methylidenecyclopropyl)propanoic acid, HGA). 10 This nonproteogenic amino acid has been detected in the seeds of Acer negundo (box elder tree) 10 and was quantified by gas chromatography–mass spectrometry (GC-MS) after methyl chloroformate derivatization. 10 Concentrations of 3–160 µg/seed were found. 10 Freeze-dried seeds of Acer pseudoplatanus (sycamore maple tree) have been found to contain up to 9,120 µg/g/seed. 9 Solid-phase extraction plus dansyl chloride derivatization has been described as a useful tool in the quantification of HGA. 2 The toxin was earlier identified in fruits of the ackee tree 3 (Blighia sapida), a member of the Sapindaceae family, causing severe food poisoning in humans especially in children eating unripe fruits.
High levels of the conjugated metabolite of the toxin, methylenecyclopropyl acetic acid (MCPA), have been identified in the plasma of horses after ingestion of Acer seeds, causing myopathy.10,14 In vivo MCPA is transformed to a coenzyme A (CoA) ester and, in this form, it binds to multiple acyl-CoA dehydrogenases,4,6 –8 without further metabolization. Enzymes loaded with MCPA-CoA are blocked and no longer available for degradation of fatty acids in the mitochondrial ß-oxidation pathway. Consequently, intracellular fat accumulates while energy production via fatty acid oxidation is interrupted. Excess acyl-CoA compounds are then transformed to acyl carnitines, which are found in elevated concentrations in serum and urine. The often lethal disease that results is also described as acquired multiple acyl-CoA dehydrogenase deficiency (MADD). Previous studies10,14 successfully used ultra–high-performance liquid chromatography (UPLC)–tandem mass spectrometry (MS/MS) to determine the carnitine ester of MCPA in serum or plasma and urine after converting it into a butyl ester.
In vivo acyl-CoA compounds are not only conjugated with carnitine but also with glycine, as described previously. 1 Conjugation to glycine is an important detoxification mechanism of the organism because glycine conjugates are readily excreted into the urine. Analyzing urine or plasma samples for acyl glycines, including MCPA glycine, should therefore also be a way to prove HGA intoxication. Previous studies11,12,15 described qualitatively positive results for butyryl, isovaleryl, and hexanoyl glycines in the urine of poisoned horses. One study 10 extracted glycine conjugates by liquid partition chromatography and transferred them, as in a previous study, 15 to trimethylsilyl derivatives. Analysis was done by GC-MS. These authors 15 were also able to quantify MCPA glycine in this way, and found 0.79–3.97 mmol/mol creatinine in 6 of 7 poisoned horses. MCPA glycine was not detected in 1 poisoned horse and 5 normal horses. Unmetabolized HGA has also been detected in serum samples of horses suffering from atypical myopathy. In a previous study, 2 the toxin was measured after solid-phase extraction and dansyl derivatization by UPLC and high-resolution MS/MS. Up to 446.9 µg/L was found.
The methods published so far offer specific laboratory diagnosis of HGA intoxication. However, it is cumbersome to use several different methods for extraction, derivatization, and quantification for different parameters and test materials. Our aim was to facilitate the diagnosis of acquired MADD caused by HGA intoxication by developing a method allowing the determination of the typical metabolites in a simple single analytical procedure suitable for serum as well as for urine samples. We used methanolic extraction for serum and urine, and measured carnitine and glycine conjugates of MCPA and fatty acids after a butylation step in a single UPLC–MS/MS run. We applied the method to samples from diseased horses and a number of healthy horses for comparison.
Material and methods
Reagents
For internal standards, the deuterated chemicals d3-octanoyl, d7-butyryl, and d9-isovaleryl carnitines, and d3-valeryl and d3-hexanoyl glycines were used. a Methanol, acetonitrile, formic acid, and deionized water were commercial products of the highest quality made for liquid chromatography and mass spectrometry. b Butanolic HCl (3 N) and trifluoroacetic acid were available at highest analytical quality. c Creatinine measurement was performed by a manual assay of the Jaffe kinetic method. d
Internal standard preparation
Stock solutions of internal standards were prepared in methanol and stored at −20°C. To prepare the extraction solution spiked with internal standards, these were further diluted with methanol to give final concentrations of 1.27 nmol/L for d3-octanoyl carnitine, 2.53 nmol/L for d7-butyryl carnitine, and 1.34 nmol/L for d9-isovaleryl carnitine. Final concentrations for d3-valeryl glycine and d3-hexanoyl glycine were 17.8 nmol/L and 16.2 nmol/L, respectively.
Sample preparation
Serum (10 µL) or urine (20 µL, diluted with water to give a creatinine concentration of 7.5 mmol/L) were extracted with 300 µL of internal standard solution, vortexed, and then centrifuged for 10 min at RCF 17,000. Two hundred microliters of the clear supernatant were transferred to a microtiter plate and dried under a gentle stream of nitrogen. The residue was treated with 50 µL of 3 N butanolic HCl for 15 min at 65°C, dried at 65°C, and was resolved in 200 µL of methanol:water (80:20 vol/vol). This solution was further diluted 1:5 with water.
Ultra–high-performance liquid chromatography–tandem mass spectrometry
A commercial system e consisting of a gradient pump, integrated solvent degasser, temperature-controlled autosampler, column oven, and triple quadrupole mass spectrometer was used. In the autosampler, the microtiter plates were cooled down to 10°C. Five microliters of the sample solution were injected onto a 1.7 µm, 2.1 mm × 50 mm column. e The chromatography was performed at a flow rate of 450 µL/min using a gradient between solvent A (water containing 0.1% formic acid and 0.01% trifluoroacetic acid) and solvent B (acetonitrile containing the same modifiers as solvent A). The gradient started with 20% B; raised to 27% B within 1.92 min and held to 3.87 min; raised to 99% within 4.13 min and held for 2 min. The gradient returned to the initial conditions within 0.20 min.
The MS/MS system was equipped with an electrospray ionization (ESI) interface. The nebulizing gas was nitrogen; argon was used for collision at a pressure of 4.2 × 10−3 mbar. The source temperature was set at 150°C. Capillary voltage was 0.5 kV. The acyl glycines and acyl carnitines were detected in ESI-positive mode (ESI+) by multiple-reaction monitoring (MRM) mode. Collision energy was 12/20 eV; cone voltage was 16/28 V for acyl glycines and acyl carnitine, respectively. The analyzed transitions [m/z] were as follows: valeryl glycine: 216.1 → 132.0; d3-valeryl glycine: 219.1 → 132.0; MCPA glycine: 226.1 → 132.0; hexanoyl glycine: 230.2 → 132.0; d3-hexanoyl glycine: 233.2 → 132.0; isobutyryl/butyryl carnitine: 288.2 → 85.0; d7-butyryl carnitine: 295.3 → 85.0; isovaleryl/valeryl carnitine: 302.2 → 85.0; d9-isovaleryl carnitine: 311.3 → 85.0; MCPA carnitine: 312.2 → 85.0; hexanoyl carnitine: 316.2 → 85.0; octanoyl carnitine: 344.2 → 85.0; d3-octanoyl carnitine: 347.3 → 85.0; decenoyl carnitine: 370.2 → 85.0. UPLC-MS/MS results were calculated using commercial software.e,f Concentrations of MCPA carnitine were deduced by comparing peak height to d3-octanoyl carnitine, while the reference standard for MCPA glycine was d3-hexanoyl glycine.
Samples
We analyzed serum samples from 18 horses presenting with acute signs of severe myopathy and clinically diagnosed to have atypical myopathy. Clinical signs included muscular weakness, recumbency, and myoglobinuria of varying degrees. Creatine kinase activity was significantly increased due to acute rhabdomyolysis. Serum samples were drawn during clinical illness. There were 2 additional horses (values not included in tables) examined during recovery from atypical myopathy. Fresh serum samples from 14 horses with neither signs of muscular diseases nor metabolic derangements were used as controls. Blood was taken as part of a routine screening or in the diagnostic workup of cases unrelated to atypical myopathy. Urine samples from 11 horses presenting with severe signs of atypical myopathy were taken by catheterization antemortem. Urine samples, taken as free-catch urine, were collected from 5 healthy adult horses as controls. Serum samples were sent at ambient conditions to the laboratory. Urine samples were sent cooled for analysis.
Results
Preparation including all dilution and extraction steps for 20 samples plus 2 controls was completed within 100 min by a single technician. UPLC-MS/MS run time was ~14 min per sample.
Recovery
Method validation included recovery experiments for all relevant acyl conjugates by analyzing spiked urine and serum samples from healthy horses, with 6 replicates each. Recovery in the range of 89–107% was found. For acyl carnitines, this is comparable to results obtained by a previous study. 5 Because of the lack of original substance, we were not able to quantify recovery of MCPA conjugates.
Precision data
For determination of precision, frozen urine and serum samples were used. For intra- and interday analysis, preparation was done separately starting from the original material for each measurement. Preparation and measurement of samples for intraday analysis was done on a single day. Interday analyses were done on 8 consecutive days. For MCPA carnitine, coefficients of variation (CVs) for intraday testing were 3.9% for a serum sample and 2.9% for a urine sample taken from a diseased horse. The respective values for interday analysis were 9.3% and 8.0%. As MCPA glycine was present in concentrations near the detection limit in only a few serum samples, the CV was only determined for a urine sample taken from a diseased horse. The intraday CV was 3.0%, and the interday value 10.9%. For C4–C8 glycines and carnitines, intraday CVs were ≤4.5% for serum and urine from horses with atypical myopathy, except for hexanoyl carnitine in urine (13.4%). Interday CVs were 4.9–17.5%.
The UPLC-MS/MS separation of MCPA carnitine and MCPA glycine in urine from an affected horse is shown in Figures 1 and 2, respectively. Using a method described previously, 13 no matrix effect on quantitative results of the acyl compounds measured was observed during the time window given by the chromatographic separation of these substances. Because of the lack of original substance, we were not able to prove the absence of a matrix effect on the quantification of the carnitine and glycine derivatives of MCPA. However, with respect to retention time and the magnitude of the CV, it seems reasonable to expect there was no effect of coeluting matrix compounds on MS/MS signals.
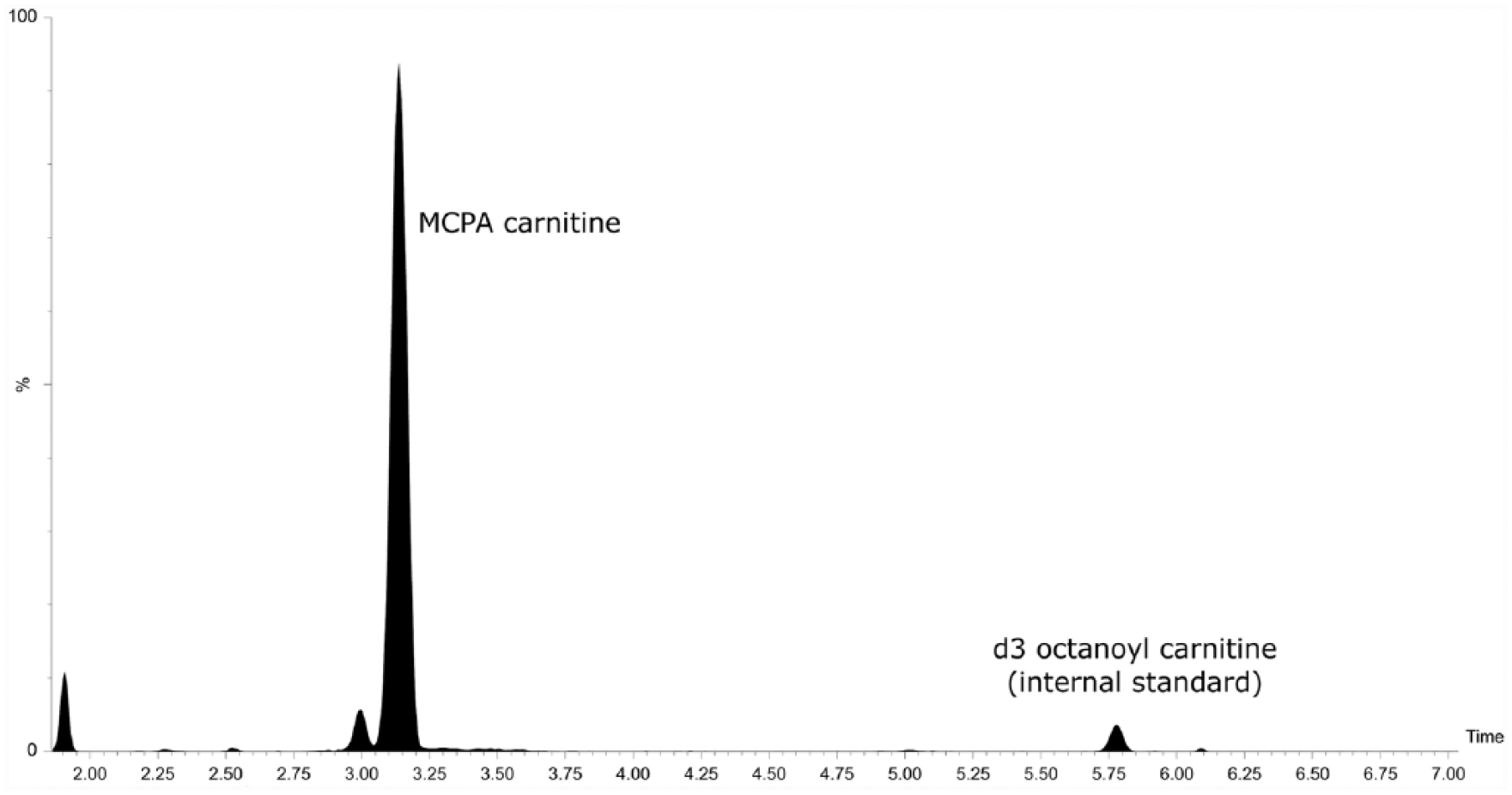
Ultra–high-performance liquid chromatography–tandem mass spectrometry separation of methylenecyclopropyl acetic acid carnitine in a urine sample collected from a diseased horse.
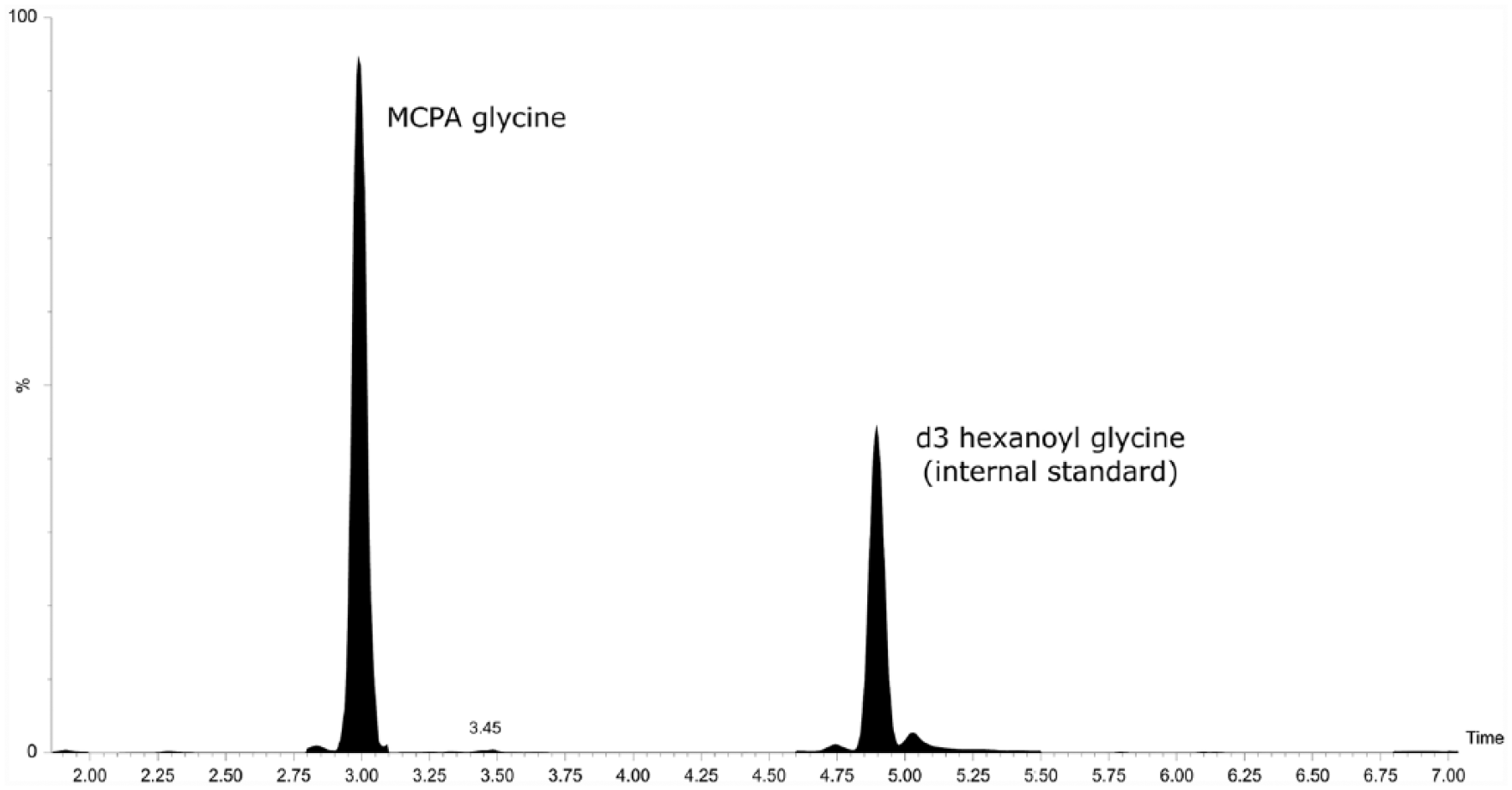
Ultra–high-performance liquid chromatography–tandem mass spectrometry separation of methylenecyclopropyl acetic acid glycine in a urine sample collected from a diseased horse.
Samples
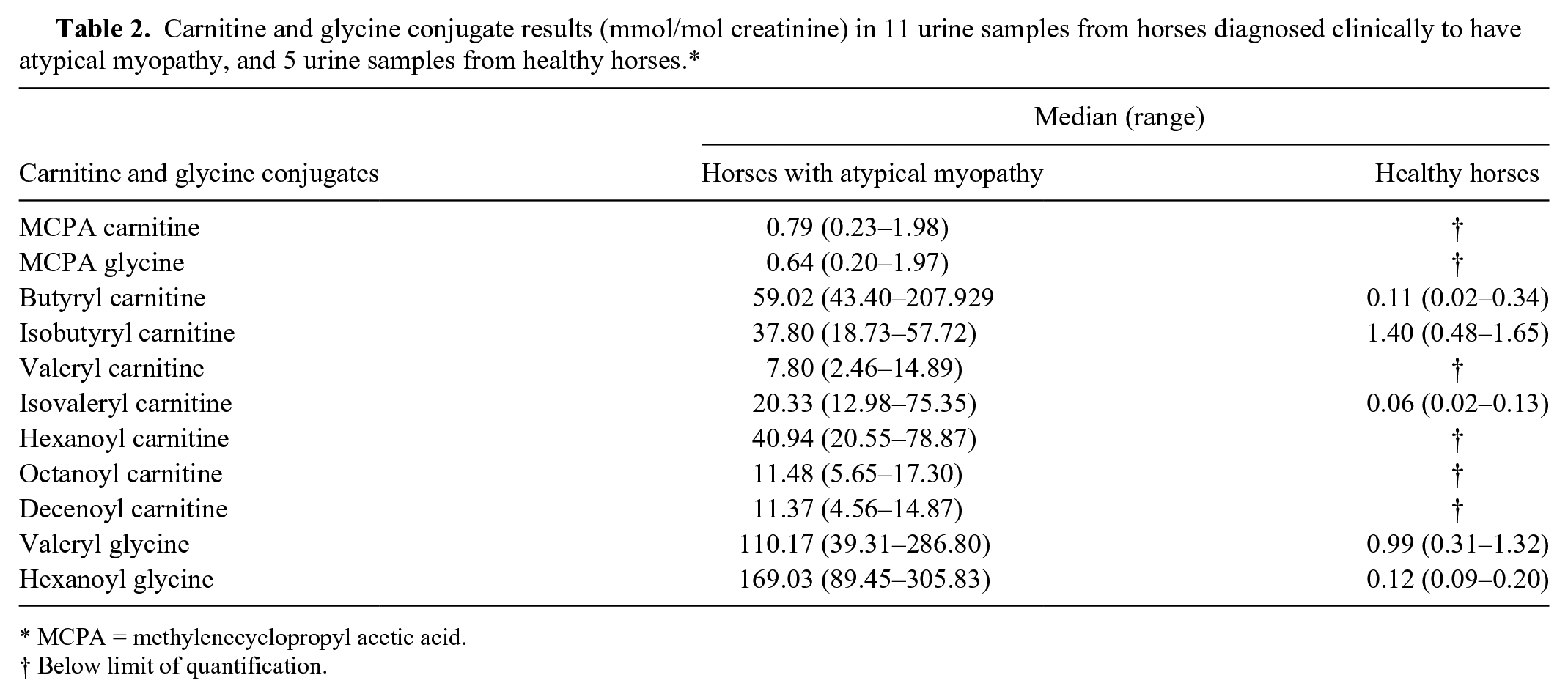
Results obtained in diseased horses are summarized in Tables 1 and 2. MCPA carnitine was found in the serum of all severely diseased horses but MCPA glycine was only found at very low concentrations (<0.04 µmol/L) in serum of 8 out of 18 horses. In urine, however, MCPA glycine was present in all samples obtained from diseased animals. There was no fixed relationship between concentrations of the carnitine and glycine conjugates of MCPA in the urine in individual horses. The factor varied between 0.5 and 2.2.
Carnitine and glycine conjugate results (µmol/L) in 18 serum samples from horses diagnosed clinically to have atypical myopathy, and 14 serum samples from healthy horses.*

MCPA = methylenecyclopropyl acetic acid.
Below limit of quantification.
Median not calculated.
Carnitine and glycine conjugate results (mmol/mol creatinine) in 11 urine samples from horses diagnosed clinically to have atypical myopathy, and 5 urine samples from healthy horses.*
MCPA = methylenecyclopropyl acetic acid.
Below limit of quantification.
Concentrations of medium-chain acyl conjugates were 9–80 times higher in serum, and 27–1,435 times higher in urine samples, compared to controls. On average, the concentration of serum hexanoyl glycine amounted to only 16.4% of that of the carnitine ester; however, glycine conjugate excretion in urine exceeded that of the carnitine conjugate by a factor of 4.3. The serum level of valeryl glycine was 2.9 times higher than that of the carnitine derivative. The respective factor for urine was 18. In 8 diseased horses from which both serum and urine samples were collected simultaneously, the concentrations of the acyl conjugates in each sample were compared. There was no direct correlation between serum and urine concentrations.
In the first of the 2 additional horses that were examined during recovery from atypical myopathy, the serum showed a very low concentration of only 0.03 µmol/L MCPA carnitine, but the concentrations of medium-chain acyl carnitines were still 5–25 times higher than in unaffected horses. Urine was not available from this animal. In the second horse, MCPA carnitine was not detected in serum or urine, but MCPA glycine was detected in urine at a low concentration (0.06 mmol/mol creatinine). Urine levels of glycine and carnitine conjugates of fatty acids were up to 20 times above normal. Concentrations of acyl carnitines in serum, however, were found in the upper normal range only.
Discussion
Until the cause of atypical myopathy or seasonal pasture myopathy in horses was known, diagnosis after the sudden onset of generalized progressive myopathy in grazing horses relied on the detection of some typical biochemical abnormalities. These included markedly increased activities of muscle enzymes and the storage of neutral lipids in type 1 muscle fibers. These are still typical findings but they are not sufficient for the confirmation of HGA poisoning. The major biochemical effect of HGA ingestion, as described previously, 15 is the accumulation of a broad spectrum of acyl carnitines and glycines in plasma and the excretion of increased amounts of acyl carnitines and glycines in urine similar to results obtained in humans with an inborn error of metabolism called multiple acyl-CoA dehydrogenase deficiency. Therefore, quantification of acyl carnitines and acyl glycines in serum and urine has to be undertaken to confirm a diagnosis of HGA intoxication.
Previous studies11,14,15 reported increased levels of most of the up to 35 carnitines tested in plasma or serum. With the serum samples available in our laboratory, we received similar results by parent ion scans of m/z 85 fragments in a spectrum of 32 carnitines (not shown). A full scan does not need more than 2 min, and this is to a large extent independent of the number of substances quantified. However, for the diagnosis of acquired multiple acyl-CoA dehydrogenase deficiency as a consequence of HGA poisoning, it is only necessary to measure a selection of these acyl carnitine and glycine conjugates. In order to be more specific and to increase sensitivity, it is better to reduce the number of metabolites measured and instead to combine MS/MS to chromatographic separation, especially as MS/MS does not allow differentiation of isomers. Specific diagnosis, however, is only achieved by showing the presence of HGA itself or of HGA metabolites in urine and serum or plasma. As mentioned above, methods used for confirmation of HGA intoxication have been expanded by an analysis for HGA in serum.
Methods available, to date, have been based on a variety of procedures for extraction, derivatization, and instrumental quantification. We describe a simple and rapid method that allows quantification of glycine and carnitine derivatives, including MCPA conjugates, in the same run, that is applicable to serum as well as to urine samples.
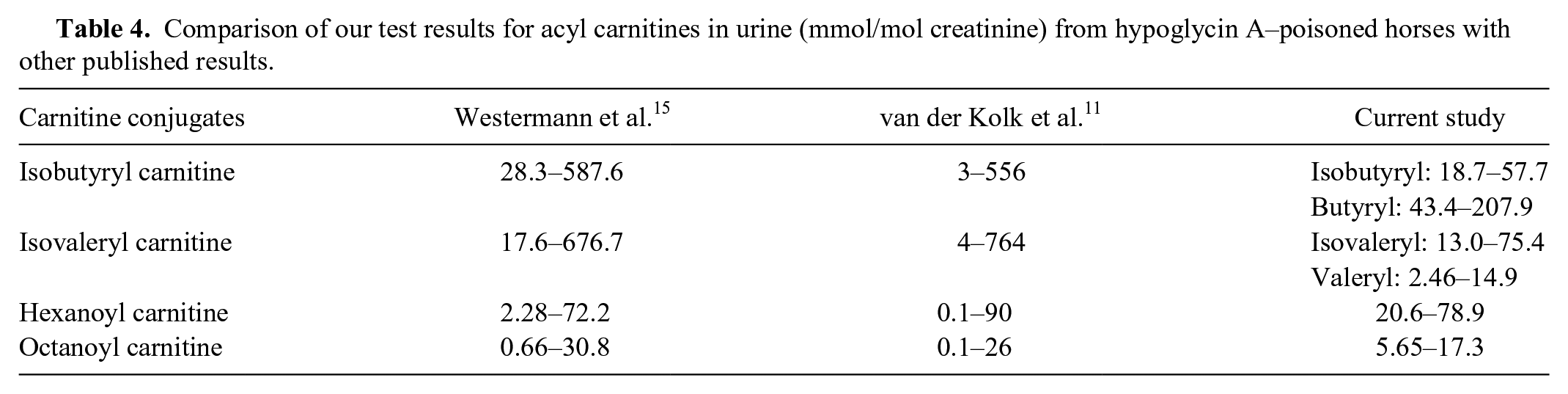
Results for acyl carnitines in serum or plasma obtained with our method correspond well to those previously published by other groups (see Table 3). The concentrations of MCPA carnitine in 3 different laboratories were in the same order of magnitude but were somewhat higher in our patients. As the quantification had been done using deuterized octanoyl carnitine as an internal standard by all groups, differences are not a result of variations in methods but may represent the wide scale of possible in vivo levels. Measurement of urine excretion of acyl carnitines by previous studies12,15 yielded results very similar to ours as shown in Table 4.
Comparison of our test results for acyl and methylenecyclopropyl acetic acid (MCPA) carnitine conjugates in serum (µmol/L) from hypoglycin A–poisoned horses with other published results.*

Dash (—) = not done.
Comparison of our test results for acyl carnitines in urine (mmol/mol creatinine) from hypoglycin A–poisoned horses with other published results.
To our knowledge, there are no other published results for acyl glycines in serum, so comparison to reported results is not possible. Glycine conjugates in urine were quantified by a previous study. 10 For hexanoyl glycine, the researchers 10 found 196 ± 101.4 mmol/mol creatinine, which corresponds well to our results (89.5–305.8 mmol/mol creatinine). Also values obtained for MCPA glycine were within the same range (previous study 0.79–3.97, 10 our results 0.2–1.97 mmol/mol creatinine).
In contrast to other published methods, sample preparation as described in our study does not require any sophisticated chromatographic preanalytic separation, nor is any time-consuming derivatization method necessary. We used a simple butylation process suitable to increase signals sufficiently. One single procedure not lasting longer than 100 min for a batch of 20 samples was applied for serum or urine samples. Measuring MCPA conjugates plus acyl carnitines and glycines in a single run provides a spectrum of detailed results within only 14 min per sample. This is much faster than GC-MS analyses, which take up to 2 hr per sample.
The butylation method provides greater opportunity to study preclinical, subclinical, mild, moderate, and nonfatal cases of HGA poisoning and may help in the development of new therapies. It also provides the opportunity to develop a better understanding of the accumulation and excretion of conjugates of fatty acids and MCPA in the course of HGA poisoning. Considering the rather large concentrations of MCPA conjugates excreted into urine, renal excretion of nonmetabolized MCPA conjugates seems to play an important role in detoxification.
Footnotes
Acknowledgements
We thank Professor Gerald Fritz Schusser (Large Animal Clinic for Internal Medicine, Faculty of Veterinary Medicine, University of Leipzig, Germany) and his team for providing data of their diseased horses.
Authors’ contributions
J Sander, JMV Cavalleri, M Terhardt, M Bochnia, A Zeyner, A Zuraw, S Sander, M Peter, and N Janzen contributed to conception and design of the study, and critically revised the manuscript. All authors contributed to acquisition, analysis, and interpretation of data, and gave final approval. J Sander drafted the manuscript. J Sander and N Janzen agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
H. ten Brink, Academisch Medisch Centrum (AMC), Amsterdam, The Netherlands.
b.
Biosolve BV, Valkenswaard, The Netherlands.
d.
Randox Laboratories Ltd, Crumlin, United Kingdom.
e.
Xevo UPLC-MS/MS system, Acquity UPLC BEH C18 column, TargetLynx 4.1; Waters, Eschborn, Germany.
f.
MS Excel 2010, Microsoft Corp., Redmond, WA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
