Abstract
Iron-restricted erythropoiesis can occur as a result of an absolute deficiency of iron stores, inflammation-mediated iron sequestration, or functional iron deficiency (in which release of stored iron is slower than the iron uptake by erythroid precursors during intense erythropoiesis). Reticulocyte indices are used to identify iron-restricted erythropoiesis, with the reticulocyte hemoglobin content (CHr) being the most commonly used index in human patients. Dogs with immune-mediated hemolytic anemia (IMHA) may have iron-restricted erythropoiesis caused by inflammation-mediated iron sequestration and/or functional iron deficiency, which could contribute to anemia severity and blunt the regenerative response in some dogs. To investigate this possibility, reticulocyte indices were examined retrospectively in 14 dogs (2–15 years of age; 9 spayed females, 1 intact female, 4 neutered males) with IMHA, and no clinical evidence of blood loss was found to suggest absolute iron deficiency. Five dogs (34%) had CHr below the preestablished lower reference limit (24.5 pg), and hematocrit was significantly lower in these dogs (p = 0.042, nonpaired t-test). Our results suggest that some dogs with IMHA may have iron-restricted erythropoiesis as a result of functional iron deficiency, inflammation-mediated iron sequestration, or (less likely) absolute iron deficiency. Further study is warranted to evaluate if dogs with IMHA may benefit from parenteral iron therapy.
Iron-restricted erythropoiesis occurs when there is too little iron for normal erythrocyte production, and may be caused by an absolute iron deficiency, iron sequestration, or functional iron deficiency (Table 1). 5 Functional iron deficiency has been mostly described in humans with an intense erythropoietic response, where iron is not mobilized rapidly enough from storage sites to keep up with the increased demand from erythroid precursors. This relative decrease in iron availability is thought to be a limiting factor for erythropoiesis, resulting in insufficient regenerative responses in affected people. Functional iron deficiency most frequently occurs in humans given exogenous recombinant erythropoietin (rHuEPO)5,16,17 and is reported to be a common reason for suboptimal erythropoietic response to rHuEPO in human patients with chronic renal failure.3,17 Iron-restricted erythropoiesis, particularly absolute iron deficiency, can lead to the production of microcytic (decreased size) and hypochromic (decreased hemoglobin content) red blood cells, eventually leading to anemia, decreased mean red blood cell volume (MCV), and decreased mean red blood cell hemoglobin concentration (MCHC). 10
Types of iron-restricted erythropoiesis, as classified in humans. 5

Reticulocyte indices are values related to the size and hemoglobin content of reticulocytes; these values are provided by some automated hematology analyzers, such as the ADVIA 120 and 2120. a Reticulocytes circulate in the blood of dogs for ~1–2 days before maturation. 11 Therefore, reticulocyte indices allow for earlier detection of microcytosis and hypochromasia during the development of iron-restricted erythropoiesis than do measures of MCV and MCHC, as the latter indices predominantly reflect cell size and hemoglobin content of mature erythrocytes, which can be in circulation for up to several months.2,4 These hematology analyzers report 18 reticulocyte indices, with reticulocyte hemoglobin content (CHr) being the index most commonly used to identify human patients with iron-restricted erythropoiesis, who may benefit from intravenous iron therapy.3,17 Changes in reticulocyte indices, including decreased CHr, are reported to occur in humans and dogs with absolute iron deficiency anemia, in humans and dogs with iron sequestration caused by anemia associated with inflammatory disease, and in dogs with portosystemic shunting.2,4,6,8,14,15,17
Most dogs with regenerative immune-mediated hemolytic anemia (IMHA) have intense erythropoietic responses, but absolute reticulocyte counts indicate that some dogs with IMHA have unexpectedly weak regeneration. 7 It is possible that regeneration in some dogs with IMHA may be reduced because of functional iron deficiency (analogous to rHuEPO-treated human kidney disease patients) and/or inflammation-mediated iron sequestration. To date, no studies have used reticulocyte indices as markers of iron-restricted erythropoiesis in dogs with IMHA, to our knowledge. The aim of this study was to determine if dogs with IMHA have decreases in CHr that would support underlying iron-restricted erythropoiesis.
Reticulocyte indices were retrospectively collected from the medical records of 14 adult dogs diagnosed with IMHA after being admitted to Cornell University Hospital for Animals (CUHA; Ithaca, New York) for evaluation of anemia. The dogs ranged in age from 2 to 15 years (median: 8 years) and included 9 spayed females, 1 intact female, and 4 neutered males. Breeds included 4 cocker spaniels, 3 mixed-breed dogs, and 1 each of 7 other breeds. Thirteen dogs were diagnosed with presumptive primary (idiopathic) IMHA based on history, physical examination, and clinical pathologic test results. A recently vaccinated dog was interpreted to have IMHA secondary to vaccination. Testing for infectious diseases was done in 10 dogs, all of which lacked antibodies against Ehrlichia canis, Ehrlichia ewingii, Borrelia burgdorferi, Anaplasma phagocytophilum, and Anaplasma platys according to a commercial test. b Blood from 5 of these dogs also tested negative by PCR for Babesia canis. One dog had been diagnosed with lymphoma 2 years previously and was in remission (and not being treated), with no evidence of recurrence after evaluation, so was presumed to have primary IMHA. It is possible that the latter dog and the 4 dogs that were not tested for infectious diseases had secondary IMHA. 12
As part of routine clinical evaluation, blood samples were collected by the attending veterinarian or staff into a tube containing ethylenediamine tetra-acetic acid. The samples were analyzed using an automated hematology analyzer a within 6 h of collection to avoid artifacts of cell swelling from sample aging. 1 If multiple complete blood cell count (CBC) results were available for a patient, the CBC performed at the time of initial evaluation at CUHA was used for the study.
Inclusion criteria included a regenerative anemia (absolute reticulocyte count above the upper reference limit of 95 × 109/L established in the clinical pathology laboratory at Cornell University), and either a positive Coombs test or microscopic red blood cell agglutination with saline dilution. Patients were excluded if they had evidence of conditions other than IMHA that could stimulate or inhibit reticulocyte production, affect red blood cell size, or predispose the dog to iron sequestration. Such conditions included portosystemic shunting, breed-associated microcytosis, renal disease, red blood cell oxidant injury (eccentrocytes or Heinz bodies), generalized bone marrow disease (e.g., neoplasia effacing the bone marrow, myelofibrosis, marrow hypoplasia, or treatment with drugs that could suppress the bone marrow), or inflammation from a condition independent of IMHA (e.g., abscess, dermatitis, or pancreatitis). The increased reticulocyte counts in the dogs of our study support that underlying generalized bone marrow disease was unlikely. An inflammatory leukogram was not an exclusion criterion because most dogs with IMHA have an inflammatory leukogram that may be at least in part caused by tissue necrosis secondary to thromboembolic disease or anemia-associated hypoxia. 9 Dogs were also excluded if they had conditions that could predispose to absolute iron deficiency, including clinically detectable external blood loss or marked thrombocytopenia (platelet count <50 × 109/L). Serum iron was measured in each dog by a standard ferrozine colorimetric method, and was decreased in 2 out of 14 dogs. This finding in itself is not specific for absolute iron deficiency because the degree of decrease in serum iron in these two dogs (59 and 89 µg/dL, reference interval: 98–220 µg/dL) has been reported in dogs with anemia of inflammatory disease. 15 Unfortunately, because additional testing for absolute iron deficiency (bone marrow iron stores, serum ferritin measurement, or response to iron supplementation) was not available in these dogs, it cannot be ruled out that these 2 dogs could have had subclinical absolute iron deficiency.
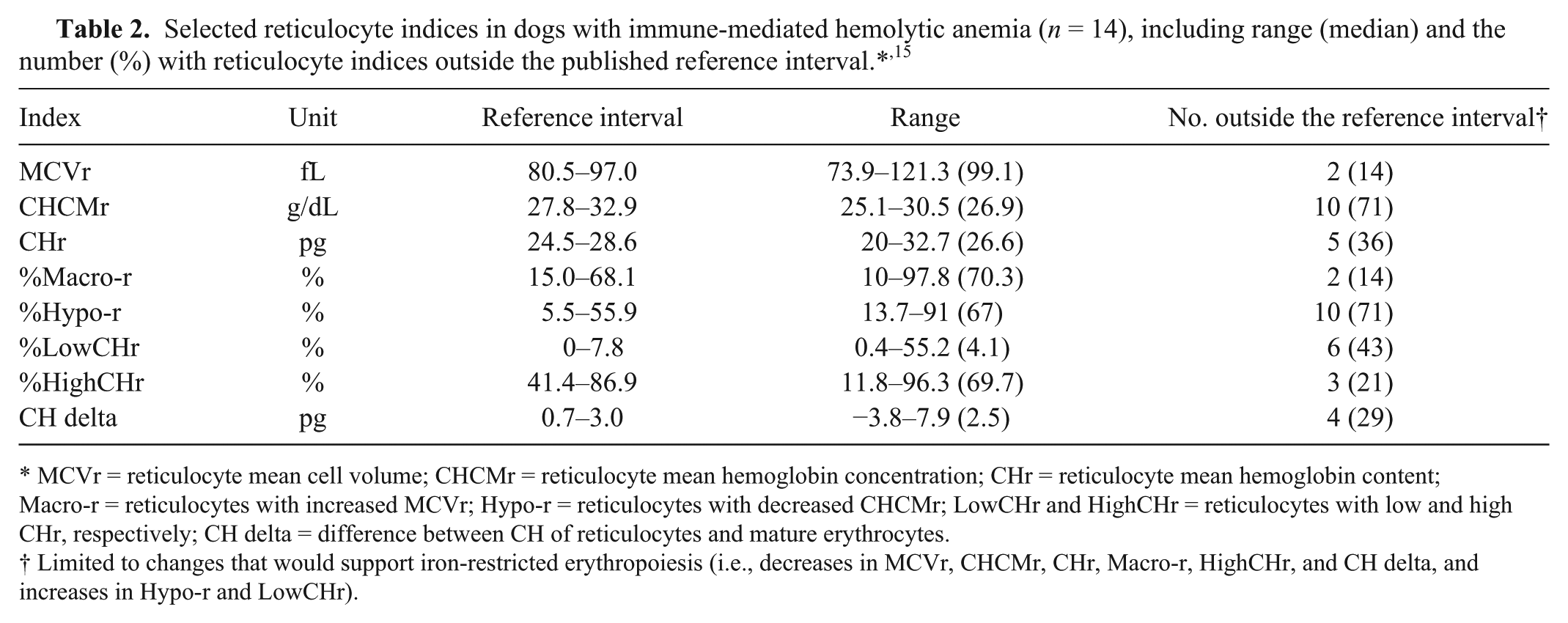
Dogs with IMHA had variable results for the reticulocyte indices that have been reported to be of most interest in dogs with iron deficiency anemia,4,14,15 with the majority of dogs having some values that were above or below established reference intervals (Table 2). 15 The CHr was below the lower reference limit (24.5 pg) in 5 dogs (Fig. 1), including one of the dogs with decreased serum iron. The median absolute reticulocyte count and hematocrit were lower in dogs with CHr <24.5 pg (Fig. 2), which was significant for hematocrit (p = 0.042) but not for the absolute reticulocyte count (p = 0.146, nonpaired t-test). Indeed, in 5 of the 7 dogs with severe anemia (hematocrit <15%), CHr was below the published lower reference limit. The lack of statistical significance in the trend toward lower reticulocyte count in the dogs with CHr <24.5 pg may be a result of the small sample size (i.e., a type II statistical error), or may indicate that the more severe anemia in those dogs is caused by a mechanism other than a blunted regenerative response (e.g., a more severe hemolytic process). Evaluation of a larger sample size is needed to further evaluate the relationship between CHr and reticulocyte count.
MCVr = reticulocyte mean cell volume; CHCMr = reticulocyte mean hemoglobin concentration; CHr = reticulocyte mean hemoglobin content; Macro-r = reticulocytes with increased MCVr; Hypo-r = reticulocytes with decreased CHCMr; LowCHr and HighCHr = reticulocytes with low and high CHr, respectively; CH delta = difference between CH of reticulocytes and mature erythrocytes.
Limited to changes that would support iron-restricted erythropoiesis (i.e., decreases in MCVr, CHCMr, CHr, Macro-r, HighCHr, and CH delta, and increases in Hypo-r and LowCHr).
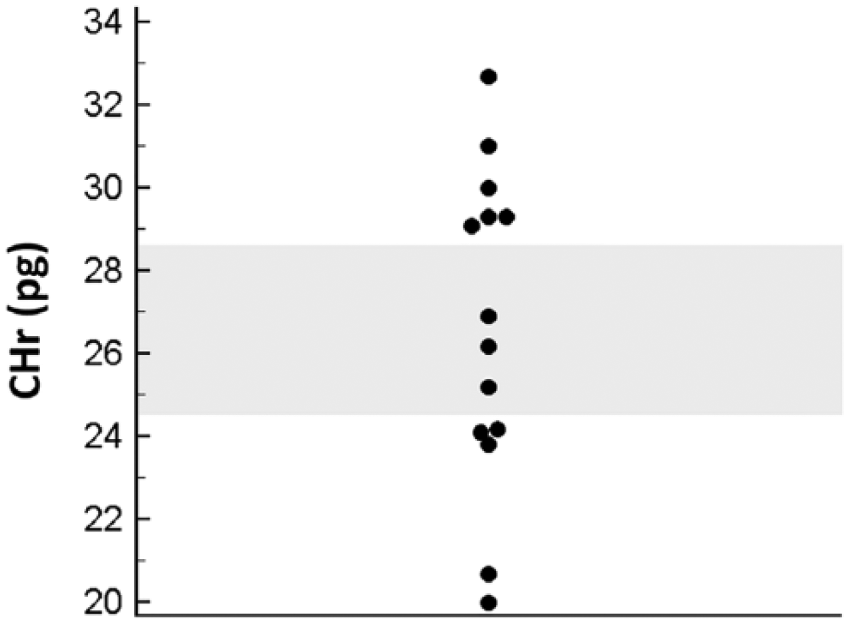
Dot plot of reticulocyte mean hemoglobin content (CHr) in dogs with immune-mediated hemolytic anemia (n = 14). The shadowed region indicates the previously established reference interval, and values below this interval are compatible with iron-restricted erythropoiesis. 15
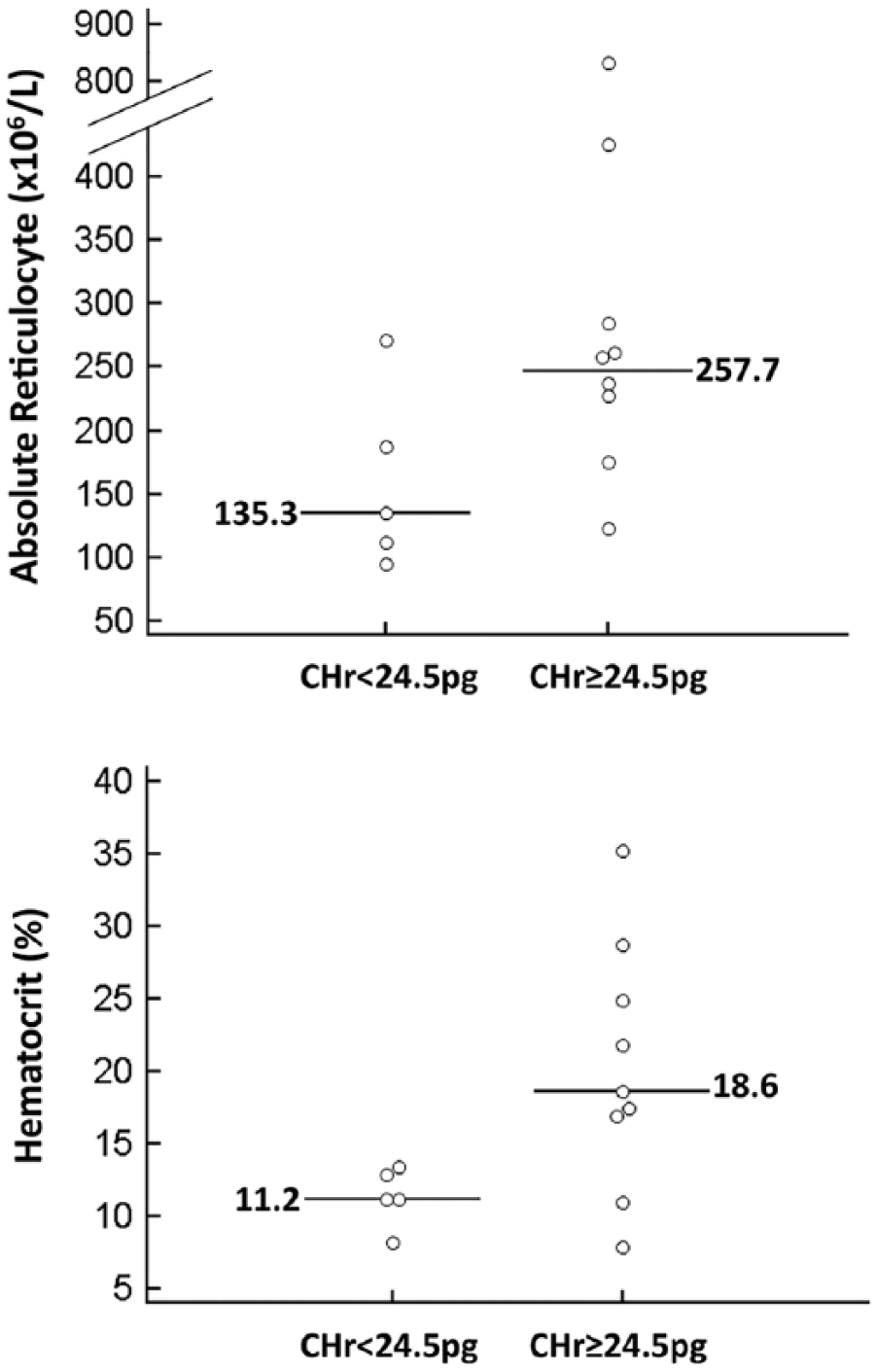
Dot plots of absolute reticulocyte count and hematocrit in dogs with immune-mediated hemolytic anemia grouped on the basis of the lower reference limit of reticulocyte mean hemoglobin content (CHr; i.e., <24.5 pg, n = 5; ≥24.5 pg, n = 9). Horizontal lines indicate median values. Hematocrit was significantly lower in dogs with decreased CHr (p = 0.042). The median reticulocyte count was lower in dogs with decreased CHr but the difference was not statistically significant (p = 0.146, nonpaired t-test).
Small red blood cell agglutinates can sometimes be counted as single large cells by automated hematology analyzers, leading to artifactual increases in MCV and MCHC. 13 Although there is no published data on the effect of agglutination on reticulocyte indices in dogs, it is possible that reticulocyte indices may be affected by agglutination. To test for this possibility, mean reticulocyte index results for dogs with (n = 6) and without (n = 8) agglutination were compared. No difference between the results for these groups was seen (p values ranging from 0.287 to 0.996, nonpaired t-test). Therefore agglutination is unlikely to be affecting the results for the reticulocyte indices. Definitively distinguishing between the 3 types of iron-restricted erythropoiesis described in Table 1 was not possible in this retrospective study, and any type of iron-restricted erythropoiesis could cause CHr values to be below the established lower limit of the reference interval (24.5 pg). 15 Absolute iron deficiency was considered less likely given the lack of detectable external bleeding in the dogs and normal serum iron in 12 out of 14 dogs, but as stated above, this cannot be ruled out. The increased severity of anemia in the dogs with decreased CHr could also be explained if these dogs had either functional iron deficiency or iron sequestration from inflammation. Unfortunately, distinguishing functional iron deficiency from iron sequestration can be difficult, and was not reliably possible in this retrospective study.
Our study reports that, based on decreased CHr, some dogs with IMHA may have iron-restricted erythropoiesis caused by either functional iron deficiency from intense erythropoiesis or iron sequestration caused by inflammation. It should be noted that a single time point, the initial blood sample taken at the time of admission, was used to determine the likelihood of functional iron deficiency in this study group. It is possible that the low CHr was transient and self-limiting (i.e., would correct with recovery or treatment of IMHA without iron supplementation). One would expect the low CHr to persist on subsequent hemograms if these animals truly had functional iron deficiency unless iron supplementation was instituted. Unfortunately, repeat hemograms were not performed on all dogs, and several received transfusions after admission, which could have affected subsequent CHr results. Study limitations include the small sample size and the inability to definitively distinguish between functional iron deficiency, iron sequestration, and subclinical absolute iron deficiency. Even so, our results indicate that a larger prospective study of dogs with IMHA is warranted to determine the frequency of decreased CHr in a larger group, to evaluate if a decreased CHr is associated with a blunted erythropoietic response (lower reticulocyte counts), and to assess the benefit of parenteral iron administration for improving the reticulocyte response in dogs with decreased CHr.
Footnotes
Authors’ contributions
DMW Schaefer contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; and drafted the manuscript. T Stokol contributed to analysis and interpretation of data, and critically revised the manuscript. Both authors gave final approval and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
ADVIA 2120 hematology analyzer, Siemens, Erlangen, Germany.
b.
4Dx SNAP test, IDEXX Laboratories, Westbrook, ME.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.