Abstract
An outbreak of enteric listeriosis in steers that were fed spoiled silage is reported. The outbreak started 2 days after ~200 animals in a single paddock were given a supplement of spoiled silage. Forty animals (20%) were affected, and 13 (6.5%) died over a period of 10 days. Affected animals were recumbent, depressed, and had diarrhea with mucus and fibrin. Gross and microscopic findings in 3 animals that were subjected to autopsy included excess peritoneal fluid, congestion and edema of abomasum, suppurative enteritis and colitis, and suppurative mesenteric lymphadenitis. Two strains of Listeria monocytogenes were isolated, one of serotype 1/2c from the gallbladder and one of serotype 1/2b from the spoiled silage. Listeria monocytogenes was detected in the mesenteric lymph nodes and intestinal wall of 1 animal by immunohistochemistry (IHC). Clinical history and signs, gross and microscopic findings, bacterial isolation, and IHC results confirmed a diagnosis of enteric listeriosis. The source of infection was likely the spoiled silage.
The genus Listeria currently includes 6 species: Listeria monocytogenes, Listeria ivanovii, Listeria innocua, Listeria seeligeri, Listeria welshimeri, and Listeria grayi. 18 Members of the genus Listeria are gram positive, and some species cause listeriosis, a disease of worldwide distribution. The bacteria are widespread in nature, and infection frequently occurs through consumption of contaminated feed. 11 While Listeria spp. have been isolated from mammals and birds of many species, listeriosis most commonly affects ruminants. 17 Only L. monocytogenes and L. ivanovii are considered to be pathogenic for ruminants. 6 Importantly, L. monocytogenes is responsible for the majority of infections in ruminants, 26 and is also associated with several diseases, including sepsis, meningitis, encephalitis, and abortion, 1 in both humans and nonruminant animals. 16 Local inflammatory lesions in the skin, eye, heart, bone, and peritoneum have also been reported in humans. 12 An enteric form of listeriosis has been described, albeit uncommonly, in humans, sheep, and cattle.2,9,10 The single published report of enteric listeriosis in cattle 9 provided no immunohistochemistry (IHC) results or typing information on the strain involved.
Listeria monocytogenes can be subdivided into 13 serotypes with a wide diversity of individual strains within 4 lineages. Because there is significant variation in virulence and pathogenicity between strains, the determination of the serotypes involved is useful for the epidemiological investigation of listeriosis outbreaks.15,19 The objective of the study is to describe an outbreak of enteric listeriosis in steers and the serotype of the L. monocytogenes strains possibly involved.
The herd involved in this case consisted of 200, ~1-year-old, ~250 kg, Hereford and Angus steers in a paddock of mixed pasture (brome, ryegrass, and white clover) on a beef ranch in Buenos Aires Province, Argentina. This ranch also had a feedlot for steers and occasionally the leftover feed (including silage) from the feedlot yard was fed to the grazing steers. Two days after providing leftover putrid, moldy silage from the feedlot to the grazing steers, 40 showed clinical abnormalities, including depression, recumbency with inability to rise, hyperthermia (41–42°C), ruminal atony, lethargy, and mucofibrinous diarrhea, with a fatal outcome 4–7 days after the onset of clinical disease in 13 animals. The cumulative incidence was 20% (40/200), and the mortality was 6.5% (13/200). An ethylenediamine tetra-acetic acid blood sample collected from 1 sick animal was used for biochemical determinations (urea, alanine transaminase [ALT], and aspartate transaminase [AST]). Serum from the same animal was inoculated onto monolayers of Madin–Darby bovine kidney (MDBK) cells. After 4 blind passages, the cultures were tested by an indirect fluorescent antibody test (IFAT) with commercially available polyclonal conjugates a for Bovine viral diarrhea virus 1 and 2 (BVDV) and Bovine herpesvirus 1 (BHV-1). Postmortem examinations were performed on 3 steers found dead, but samples for laboratory testing were collected only from one of these carcasses. Bile, feces, and intestinal content were collected for bacterial culture. Bile was inoculated onto Columbia agar base with 7% defibrinated bovine blood (CBA) and MacConkey agar. Both media were aerobically incubated at 37°C for 7 days. Bile was also inoculated onto CBA and incubated at 37°C under a 10% CO2 atmosphere for 7 days. Bile, feces, and intestinal contents were inoculated into 40 mL of tetrathionate broth base with the addition of 0.8 mL of iodine solution, 0.4 mL of 0.1% (w/v) brilliant green, and 0.4 mL of 0.1% (w/v) novobiocin. The tetrathionate broth was incubated aerobically at 37°C with subcultures onto xylose-lysine-desoxycholate agar (XLD) after 24, 72 and 120 hr of incubation.
A sample of the silage given to the sick steers was inoculated into a commercial selective enrichment broth for Listeria spp. b with the addition of specific selective supplement 23 at the rate of 10 g per 200 mL of broth. The medium was incubated at 4°C and was subcultured weekly for a 4-week period onto an agar version of the selective broth with the same supplement and CBA, which were aerobically incubated at 37°C for 24–48 hr. Listeria-like colonies were isolated and characterized. 20 Samples of silage were also processed for the detection of mycotoxins by thin layer chromatography. 22
Samples of heart, lung, liver, gallbladder, small and large intestine, mesenteric lymph nodes, spleen, and kidney were fixed by immersion in neutral buffered (pH 7.2) 10% formalin for 48 hr, routinely embedded in paraffin, and then stained with hematoxylin and eosin. Sections of mesenteric lymph nodes and intestine were processed for IHC for the presence of Listeria spp., 4 BVDV, bovine coronavirus (BCV), and BHV-1 according to the Standard Operating Protocols of the California Animal Health and Food Safety Laboratories (University of California at Davis). Briefly, 4-μm thick sections were mounted on histoslides c and stained on a capillary gap system. d A commercial avidin–biotin complex immunoperoxidase kit was used. e Positive controls included formalin-fixed tissue of: 1) brain of a sheep from which L. monocytogenes had been isolated in pure culture, 2) colon of a steer in which BVDV had been detected by polymerase chain reaction (PCR) assay, 3) colon of a calf in which BCV had been detected by IFAT, and 4) lung of a calf in which BHV-1 had been detected by PCR. Negative controls consisted of the positive control tissues in which the primary antibody was replaced by normal serum of the species used to raise the primary antibody. Selected sections of intestine and mesenteric lymph nodes were also Gram stained. Subtyping of the isolated L. monocytogenes strains was performed by multiplex PCR using 4 primer pairs that were specific for L. monocytogenes in addition to 1 primer pair that was specific for Listeria spp., as previously described. 8
The most striking gross finding in the 3 autopsied animals was up to 1 L of translucent yellow fluid in the abdominal cavity and severe congestion of the entire digestive tract. In the abomasum, moderate congestion and edema of mucosal folds was noted. In addition, the small and large intestine had transmural thickening. Small and large intestinal content was semiliquid, yellow-green with presence of multiple fibrin strands. Multifocally, in the small and large intestine, there were fibrin casts loosely adhered to the mucosa. Moderate hepatomegaly and splenomegaly was observed in the 3 animals.
Significant microscopic lesions were present in the small and large intestine, mesenteric lymph nodes, and liver of the 1 animal examined histologically. The small intestinal lesions consisted of moderate generalized mucosal congestion, multifocal mucosal necrosis, diffuse villus blunting, and multifocal to diffuse infiltration of viable and degenerate neutrophils and fewer macrophages, lymphocytes, and plasma cells in the lamina propria, muscularis mucosa, and superficial submucosa (Fig. 1). The neutrophilic infiltration was more severe in the muscularis mucosa than in the other layers of the intestine. Multifocally, the small and large intestinal crypts were dilated and lined by attenuated epithelium that was frequently necrotic; neutrophils and sloughed necrotic epithelial cells were in the lumen of the crypts. Clusters of small, intralesional, gram-positive rods were seen within the mucosa, muscularis mucosae, and submucosa (Fig. 1).
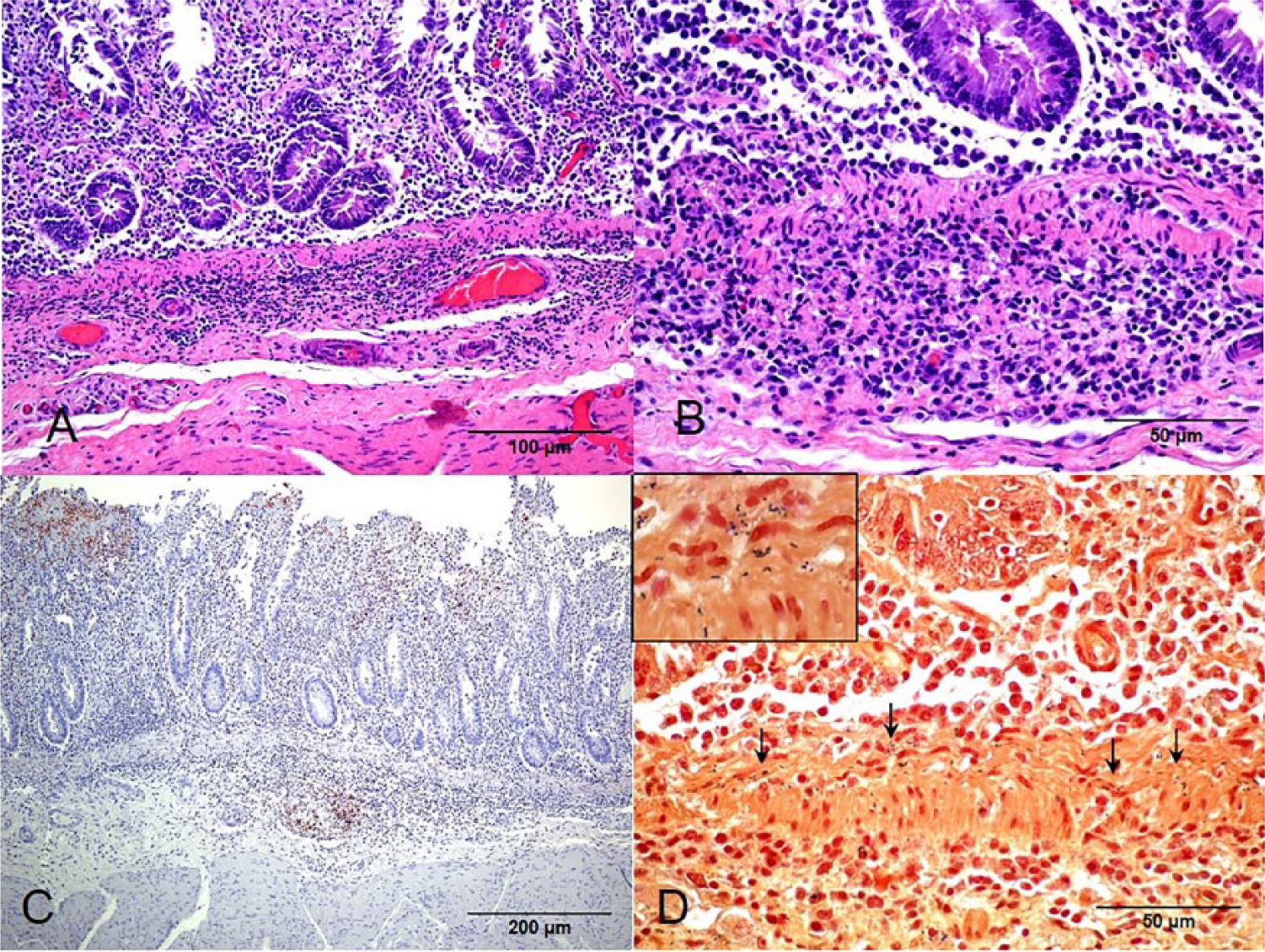
Calf, intestine.
Mesenteric lymph nodes had extensive multifocal to coalescing areas of neutrophilic infiltration, fibrin exudation, and necrosis that affected large areas of the cortex, subcapsular sinuses, and capsule (Fig. 2). Intralesional clusters of gram-positive small rods similar to those seen in the intestine were also present in the affected lymph nodes.
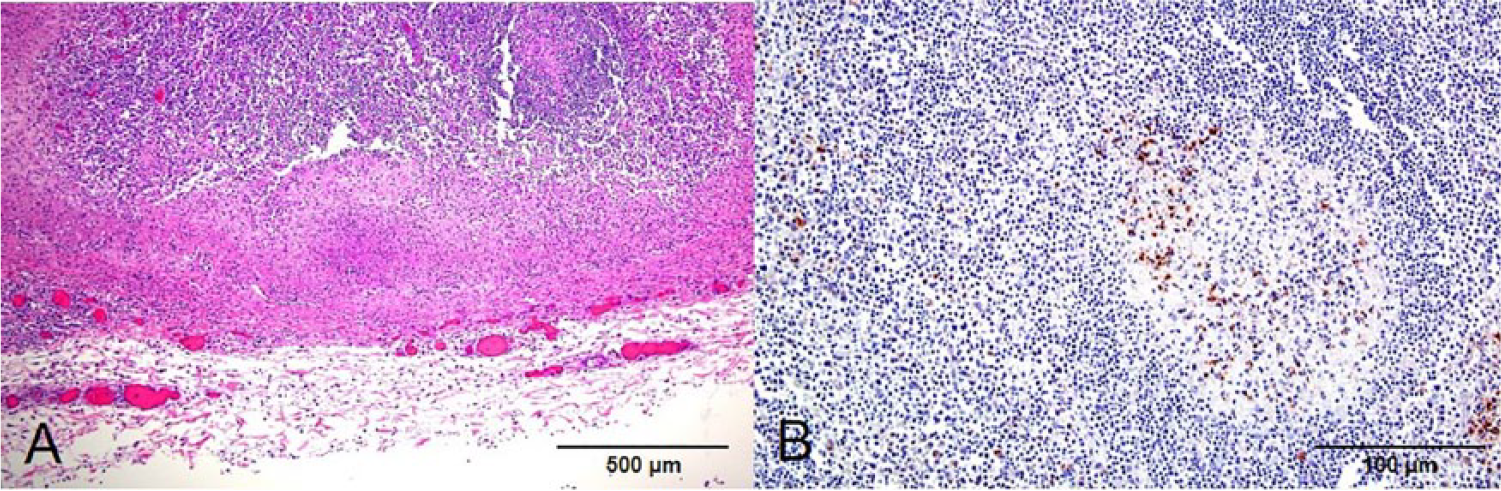
Calf, mesenteric lymph node.
The liver had diffuse congestion and hydropic degeneration of hepatocytes, and multifocal, random, and discrete areas of coagulation necrosis. Multifocal, moderate periportal infiltration of macrophages and lymphocytes, and fewer neutrophils, were seen.
Beta hemolytic colonies developed on CBA, and gray-green colonies with a brown-black halo were seen on the selective agar inoculated with bile and silage. Gram-positive coccobacilli were seen in smears of those colonies, and conventional biochemical tests 23 identified these isolates as L. monocytogenes. Intestinal contents, bile, and feces were negative for Salmonella sp. and Yersinia sp. Viral culture of serum was negative for BHV-1 and BVDV.
Examination by IHC for Listeria spp. was positive for the small intestine (Fig. 1) and mesenteric lymph nodes (Fig. 2). Positive immunolabeling in the form of small rods and granular material was detected in areas of inflammation in the lamina propria, muscularis mucosa, and superficial submucosa of the small intestine. The positive reaction was most intense in the muscularis mucosa. In the mesenteric lymph nodes, the positive reaction was observed in the periphery of areas of necrosis, including the cytoplasm of phagocytes and intercellular space. The IHC examinations for BVDV, BHV-1, and BCV were negative, both in small intestine and in mesenteric lymph nodes samples. No mycotoxins were detected in silage. The levels of ALT, AST, and urea in serum samples were within normal ranges.
Two isolates of L. monocytogenes were obtained, one of serotype 1/2c from the gallbladder and one of serotype 1/2b from the spoiled silage. Culture for Salmonella sp. and Yersinia sp. yielded negative results. A diagnosis of enteric listeriosis was made based on clinical history, and clinical, gross, and microscopic findings, coupled with results of IHC and bacteriological culture. The epidemiological findings and the isolation of Listeria spp. from silage in the present case are highly suggestive that the silage was the source of infection.
The enteric form of listeriosis has been reported in humans, 2 cattle, 10 and sheep, 9 although it is a rare occurrence. Only 1 published report of the condition in cattle 10 exists; that report does not describe typing of the L. monocytogenes isolate or the immunohistochemical features of affected tissues.
The gross and microscopic findings in this case are very similar to those previously described in enteric listeriosis of sheep 9 and cattle 10 in which the intestinal lesions were particularly prominent in the muscularis mucosa. In our steer, immunoreactivity for Listeria spp. was very strong in this layer of the intestine. The presence of microorganisms by electron microscopy within the smooth muscle cells of the muscularis mucosa has been previously described. 9 The reason for the predilection of Listeria spp. for the smooth muscle fibers of the muscularis mucosa is unknown. Some authors have suggested that infection of the muscularis mucosa might occur via direct spread of Listeria spp. from lesions deep in the lamina propria or by infection of the smooth muscle branches in the lamina propria and thence to the muscularis mucosa.9,18
Listeria spp. are mainly found in the soil, forage, water, mud, silage, and foods. 5 Animal outbreaks are primarily associated with poor-quality silage in which the pH is >5, providing ideal conditions for the growth of Listeria spp. 7 In sheep, enteric listeriosis has occurred within 2 days after feeding animals poor-quality silage. 9 A similar incubation period was observed in the current outbreak. This contrasts with the classical encephalitic form of listeriosis in which the incubation period after experimental inoculation into tooth pulp was at least 3 weeks.3,6 This difference in the duration of the incubation period between the enteric and the encephalitic form of listeriosis may be caused by the rapid uptake of large numbers of Listeria spp. present in intestinal contents, while in the encephalitic form the microorganism needs time to ascend the cranial nerves to the central nervous system and multiply in brainstem, which is a very slow process.
The outcome of Listeria spp. intestinal infection depends on the dose ingested, the virulence of the strain involved, 24 and the age of the animals. 27 Molecular epidemiological evidence for such differences in virulence has been presented,13,25 although limited information is available about specific genetic traits that might be responsible for these virulence differences in cattle.
Both L. monocytogenes strains isolated in our case (1/2b from lineage I, and 1/2c from lineage II) have been associated with disease in domestic animals6,19 and are considered primary pathogens for humans. 14 In ruminants, serotype 1/2b (lineage I) strains are usually responsible for the encephalitic form. 5 Conversely, serotype 1/2c (lineage II) strains are commonly associated with the encephalitic and septicemic form of the disease and are usually found in foodstuffs and also are well adapted to diverse environments.19,21 The information in our study should be useful for understanding the pathogenesis of enteric listeriosis. Even though the strain of L. monocytogenes isolated from the gallbladder (1/2c) was different from the strain recovered from silage (1/2b), it is likely that the strain recovered from the gallbladder also came from the silage. Only a small part of the silage was tested, and other areas of the silage could easily have had strain 1/2c.
Footnotes
Acknowledgements
We thank biochemist Raquel Callejo of the Special Bacteriology Service of INEI ANLIS “Dr. Malbran”, who made possible the confirmation and subtyping of the strains, and technicians Tania Lischinsky and Malena Rosana who processed samples for histological and bacteriological study. We also thank Dr. Franciso A. Uzal for his contributions and critical revision of manuscript.
Authors’ contributions
JA García and JF Micheloud contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; and critically revised the manuscript. CM Campero contributed to conception and design of the study; contributed to interpretation of data; drafted the manuscript; critically revised the manuscript; and gave final approval. EL Morrell contributed to conception of the study, and contributed to acquisition and interpretation of data. ER Odriozola contributed to conception of the study; contributed to interpretation of data; critically revised the manuscript; and gave final approval. AR Moreira contributed to conception and design of the study; contributed to acquisition and analysis of data; drafted the manuscript; critically revised the manuscript; and gave final approval. All authors agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
American Bio-Research, Sevierville, TN.
b.
Merck KGaA, Darmstadt, Germany.
c.
Probe-On, Fisher Scientific, Pittsburgh, PA.
d.
Micro-Probe Staining System, Fisher Scientific, Pittsburgh, PA.
e.
Vectastain ABC Elite Rabbit IgG, Vector Laboratories, Burlingame, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
