Abstract
This study describes the prevalence and distribution of 42 cases of skull base neoplasia in dogs between 2000 and 2014. The average age of affected individuals was 9.5 years, and there was no sex or breed predisposition. The most common skull base neoplasms were meningioma (25 cases) and pituitary adenoma (9 cases). Less common tumors included craniopharyngioma (2 cases), nerve sheath tumor (2 cases), and 1 case each of pituitary carcinoma, meningeal oligodendrogliomatosis, presumed nasal or sinonasal carcinoma, and multilobular tumor of bone. All neoplasms caused some degree of compression of adjacent structures. The distribution of the tumors was greatest in the sellar region (n = 18), followed by the paranasal region (n = 12), caudal cranial fossa (n = 10), central cranial fossa (n = 1), and rostral cranial fossa (n = 1).
The skull base is the anatomic area that composes the floor of the cranial cavity. It is in close contact with the ventral surface of the brain and meninges, as well as the cribriform plate, olfactory bulb, optic chiasm, pituitary gland, and most cranial nerves. 31 The complex anatomy and tissue heterogeneity of the skull base gives rise to a wide variety of neoplasms that are typically difficult to access by the neurosurgeon in human medicine. 11 Therefore, benign or malignant skull base neoplasms typically hold a guarded or a poor prognosis. 31 Given the challenges imposed by these lesions, most of the medical research in this field is concentrated on the development of minimally invasive and precise surgical approaches that aim to replace traditional and frequently life-threatening surgical procedures and on the improvement of potential therapeutic plans that increase postsurgical survival rates and quality of life. 13 In fact, the field of skull base surgery is a medical specialty in human medicine, with multiple organizations and research groups devoted to improve diagnosis, management, and treatment of skull base lesions. Such organizations often have a team of neurosurgeons, neurologists, radiologists, otorhinolaryngologists, plastic surgeons, maxillofacial surgeons, medical oncologists, and radiation oncologists available to manage affected patients.12,13 As a direct result of these efforts, important information has surfaced on the prevalence, location, biology, and therapy of skull base tumors.1,5,8,11–13,29,31,39 In contrast, similar studies have not been conducted in veterinary medicine, and while there have been a number of reports describing primary and secondary intracranial neoplasia in dogs, none has addressed skull base neoplasia as a distinct clinicopathological entity.7,16,19,20,33,34
This article describes the prevalence, anatomic location, and pathologic features of 42 skull base neoplasms in dogs submitted to the diagnostic service of the Athens Veterinary Diagnostic Laboratory at the University of Georgia, College of Veterinary Medicine between January 2000 and December 2014. Cases were retrieved from the web-based laboratory information system using the following key words: canine, intracranial, skull base, pituitary gland, and neoplasia. Selected autopsy and biopsy reports were reviewed, and all neoplasms were assigned a specific anatomic location on the skull base according to an adaptation of similar studies in human medicine, 31 including 1) paranasal region; 2) rostral cranial fossa; 3) central cranial fossa; 4) caudal cranial fossa; and 5) sellar region (Fig. 1). Histology slides were reviewed and, when necessary, neoplasms were classified according to the World Health Organization Classification of Tumors of the Central Nervous System15,18 (Table 1).
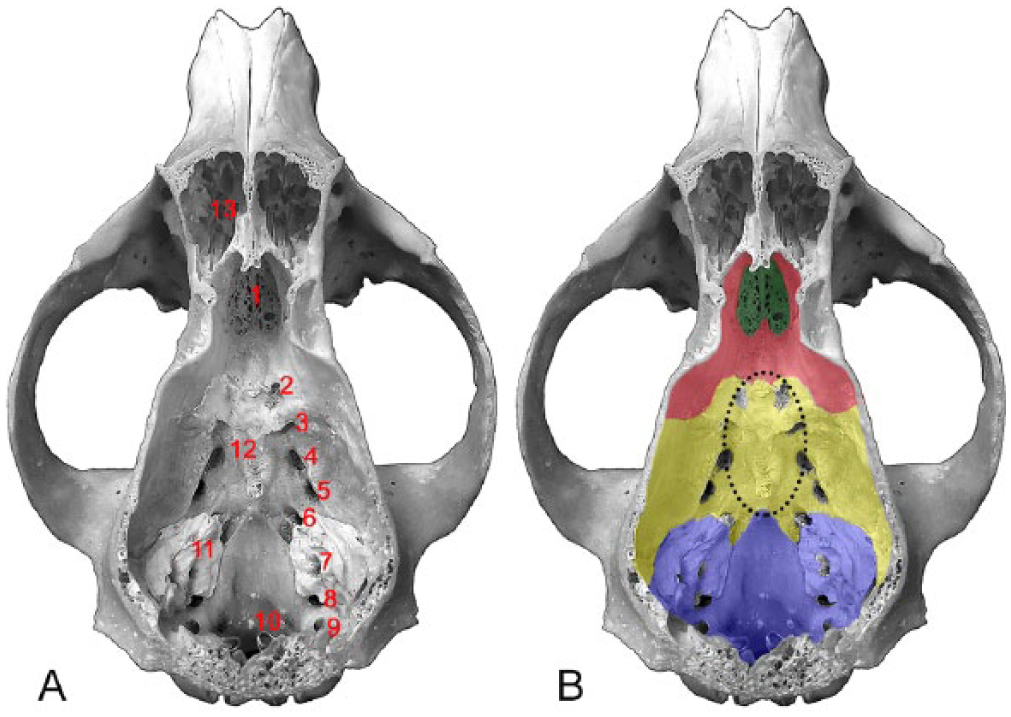
Dorsal view of a canine skull with calvaria removed showing the landmarks of the skull base (A) and its anatomic subdivisions (B).
Anatomic location and diagnosis of skull base neoplasms in 42 dogs.

Numbers in parentheses are the total number of cases.
The anatomic location and final diagnosis of the 42 neoplasms is shown in Figure 1 and summarized in Table 1. All neoplasms caused some degree of compression of adjacent structures. Meningiomas were well demarcated (13 cases) or poorly demarcated (12 cases), 0.5–4 cm in diameter, irregular, white or pale tan and less often gray to red, firm or soft masses or plaques that occasionally (5 cases) infiltrated adjacent structures, such as brain, cribriform plate, nasal sinuses or turbinates, and pituitary gland (Fig. 2). Subtypes of infiltrating meningiomas consisted of transitional (2 cases), meningothelial (1 case), fibrous (1 case), and atypical (1 case). The majority of meningiomas was meningothelial and diagnosed based on histology (Fig. 3), with only 3 cases requiring immunohistochemistry (IHC) for E-cadherin and vimentin (Fig. 4). Pituitary adenomas were all well-demarcated and soft, pale white-to-tan or dark-red nodules that ranged from 1 to 3.5 cm in diameter. The pituitary carcinoma caused overall swelling and gray discoloration of the pituitary gland and infiltrated the optic chiasm, frontal lobes, and midbrain. All pituitary adenomas and the pituitary carcinoma were diagnosed based on histology (Fig. 5). Craniopharyngiomas appeared as poorly demarcated, 1.5 cm in diameter, tan, soft masses that completely effaced the pituitary gland. There was infiltration of the underlying bone in 1 case. The diagnosis was confirmed based on the location of the masses, associated with histology (Fig. 6), positive immunostaining for pancytokeratin AE1/AE3 (Fig. 7), and negative immunostaining for α-fetoprotein. Nerve sheath tumors were 1.5–2 cm in diameter, white, firm masses that obliterated the trigeminal nerves. A presumptive diagnosis was made based on histology (Fig. 8) and confirmed by positive immunostaining for S100 protein and neurofilament (Fig. 9). The case of meningeal oligodendrogliomatosis was characterized by extensive meningeal thickening along the ventral brainstem (Fig. 10), with pituitary gland compression and atrophy. Neoplastic cells were morphologically consistent with oligodendrocytes (Fig. 11) and were immunopositive for Olig2 (Fig. 12). The nasal or sinonasal carcinoma (Fig. 13) was a 3 cm in diameter, white soft mass that effaced the olfactory bulb and infiltrated the frontal lobes. The diagnosis was based on the location, histology, positive immunostaining for pancytokeratin AE1/AE3 (Fig. 14), and negative immunostaining for neurofilament, neuron-specific enolase, synaptophysin, chromogranin A, and S100 protein. The multilobular tumor of bone arose from the lower portions of the frontal bones and appeared as a poorly demarcated, hard, multinodular, pale white mass that infiltrated the optic chiasm, cribriform plate, and frontal sinuses. The diagnosis was based on the typical histologic features that consisted of a poorly demarcated neoplasm arranged in multiple lobules composed of central areas of cartilage and bone surrounded by plump mesenchymal cells that blend into the adjacent supporting fibrous connective tissue. 32
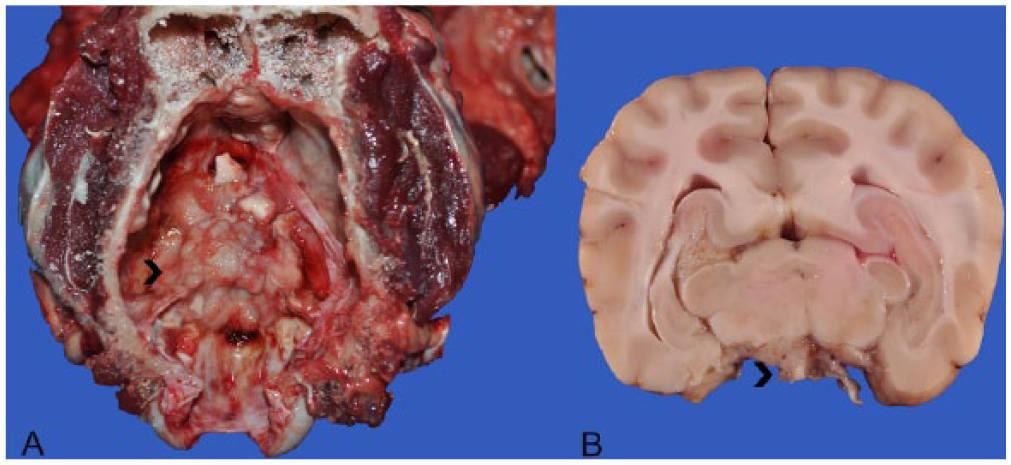
Skull, dog 14.
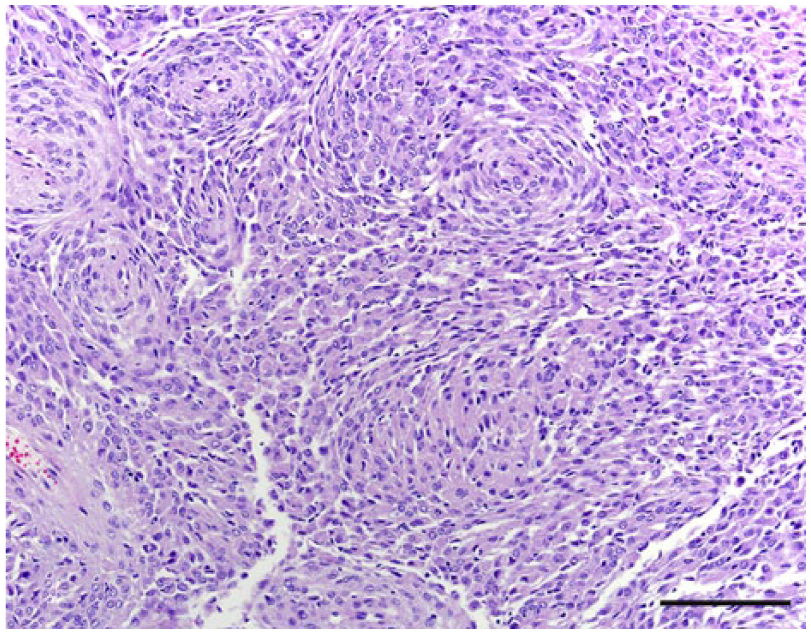
Meningothelial meningioma, dog 24. Slightly elongated neoplastic cells are arranged in solid lobules and bundles. Hematoxylin and eosin. Bar = 100 µm.
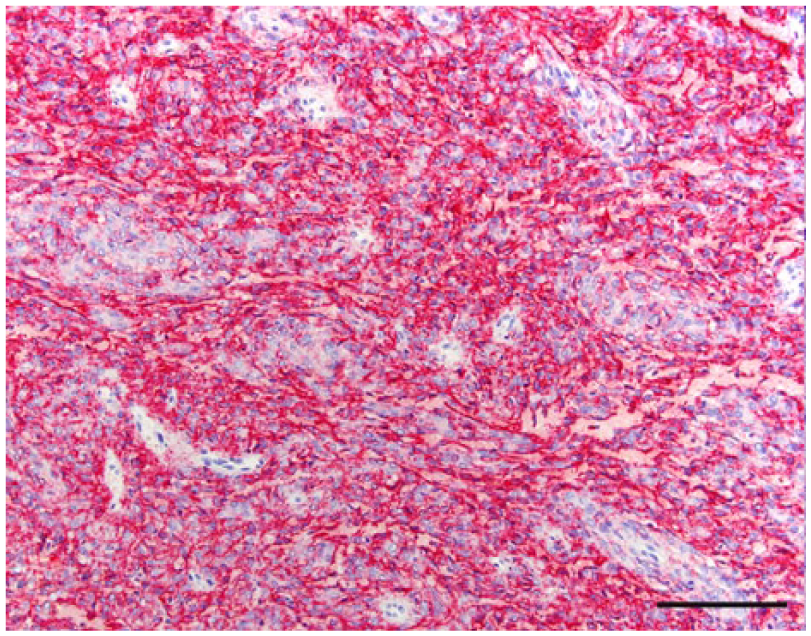
Meningothelial meningioma, dog 24. Neoplastic cells with strong membrane immunostaining for E-cadherin. Bar = 100 µm.
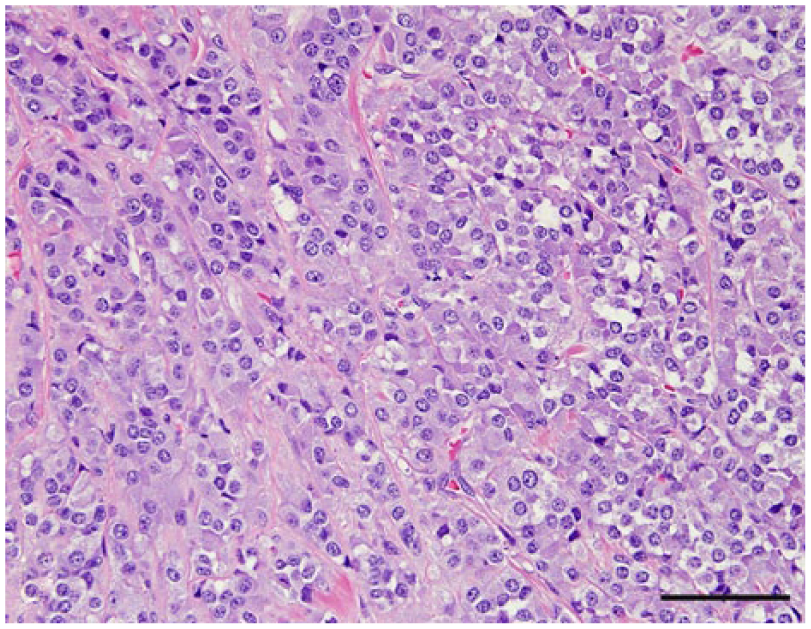
Pituitary adenoma, dog 26. Polygonal neoplastic cells are arranged in small packets. Hematoxylin and eosin. Bar = 50 µm.
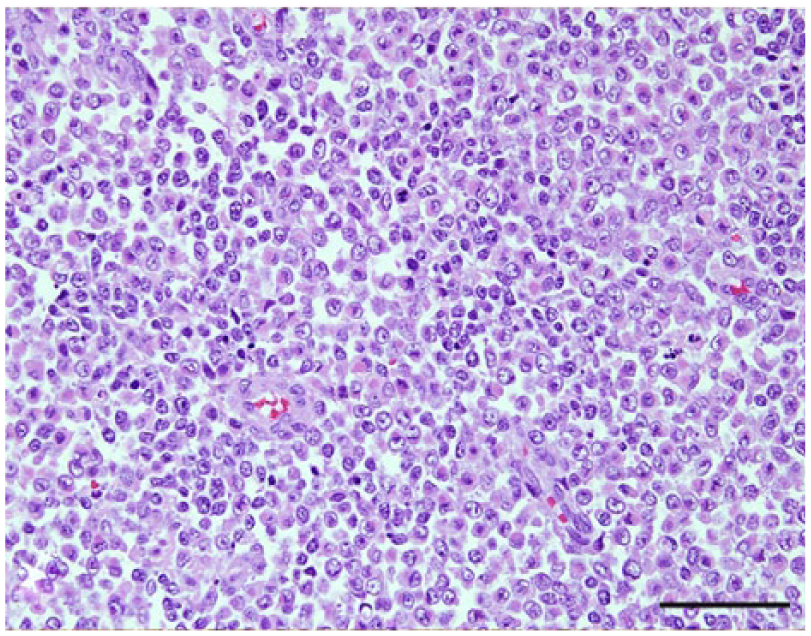
Craniopharyngioma, dog 37. Sheets of round to polygonal neoplastic cells are characteristic of this neoplasm. Rare acini were also present (not shown). Hematoxylin and eosin. Bar = 50 µm.
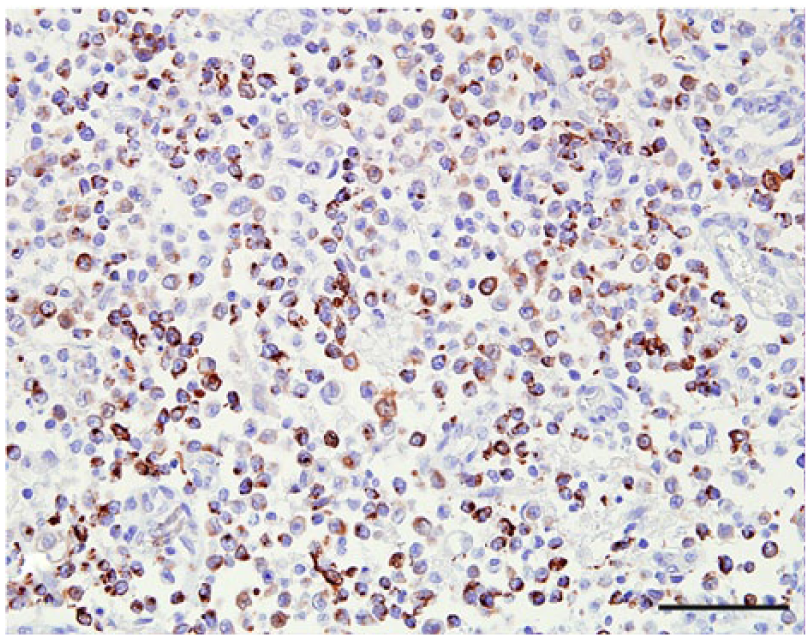
Craniopharyngioma, dog 37. Some neoplastic cells are immunopositive for pancytokeratin AE1/AE3 and negative for α-fetoprotein (not shown). Bar = 50 µm.
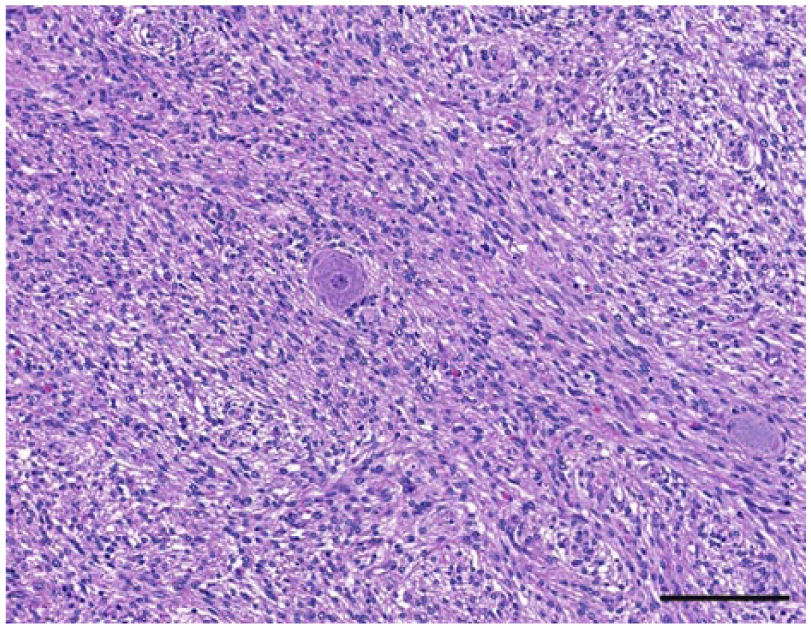
Nerve sheath tumor, dog 39. Spindle neoplastic cells arranged in the characteristic bundles surrounding a ganglionic neuron (center). Hematoxylin and eosin. Bar = 100 µm.
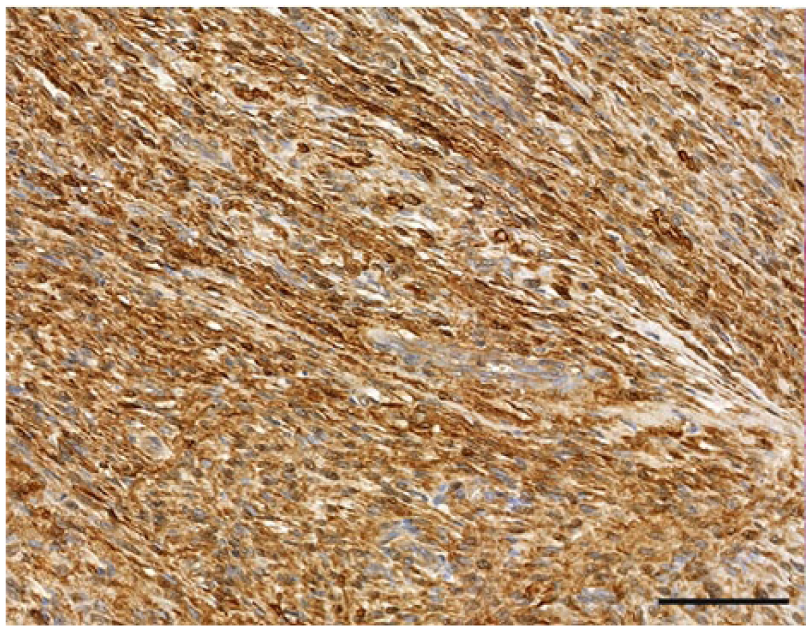
Nerve sheath tumor, dog 39. Neoplastic cells with strong cytoplasmic immunostaining for S100. Bar = 50 µm.
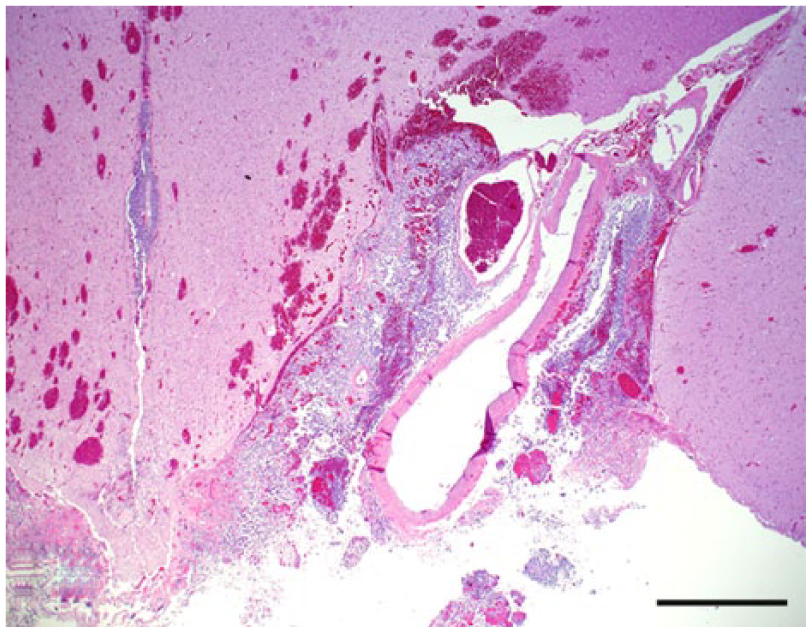
Leptomeningeal oligodendrogliomatosis, dog 40. Neoplastic cells expand the perivascular spaces of the ventral thalamus. Hematoxylin and eosin. Bar = 1 mm.
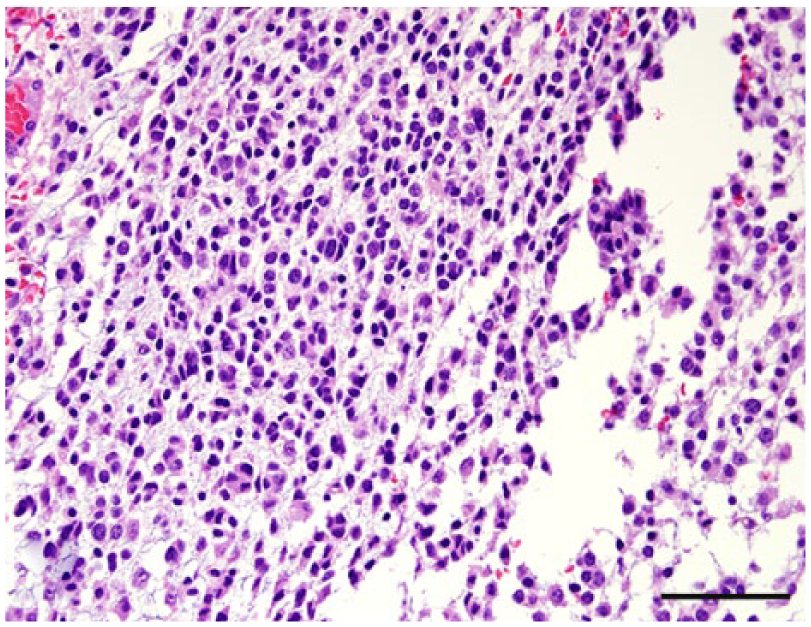
Leptomeningeal oligodendrogliomatosis, dog 40. Neoplastic cells are morphologically consistent with oligodendrocytes. Hematoxylin and eosin. Bar = 50 µm.
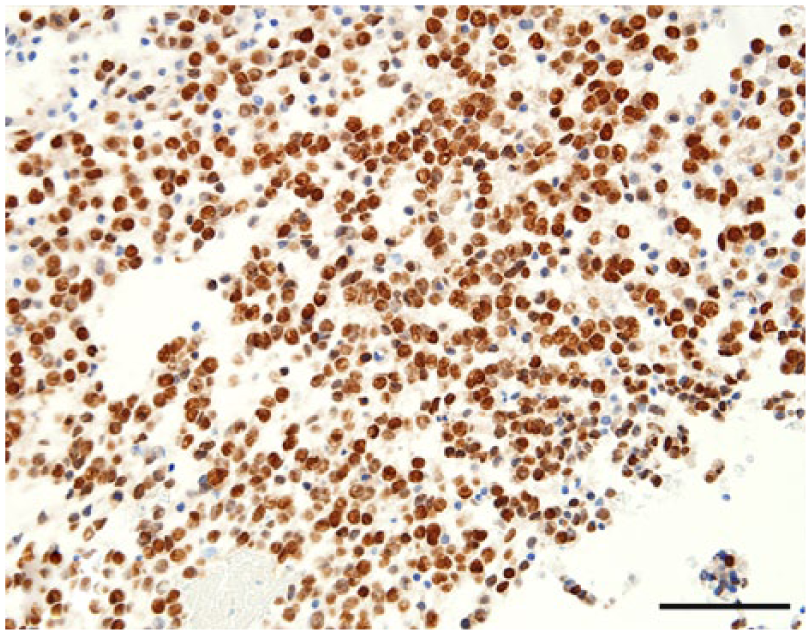
Neoplastic oligodendrocytes are diffusely immunopositive for Olig2. Bar = 50 µm.
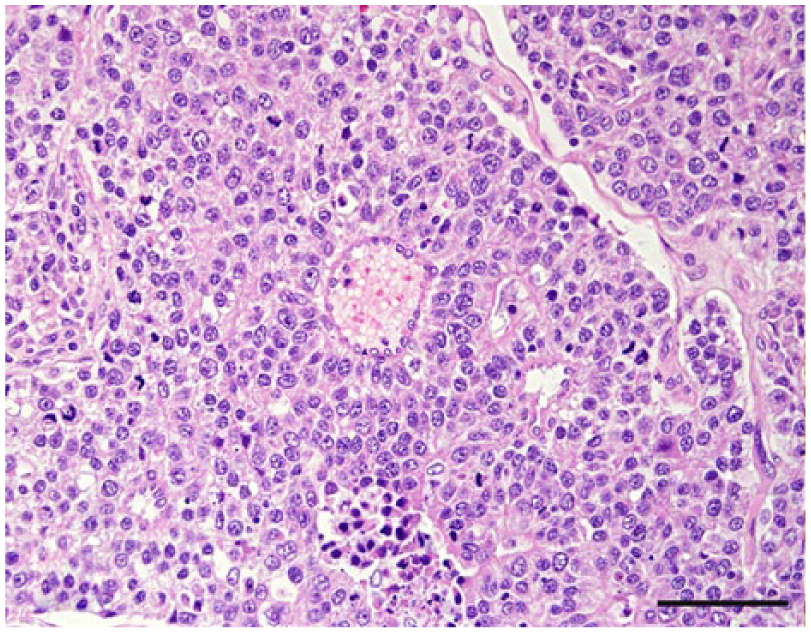
Nasal or sinonasal carcinoma, dog 41. Neoplastic cells palisade around a capillary (center), forming a typical pseudorosette. Hematoxylin and eosin. Bar = 50 µm.
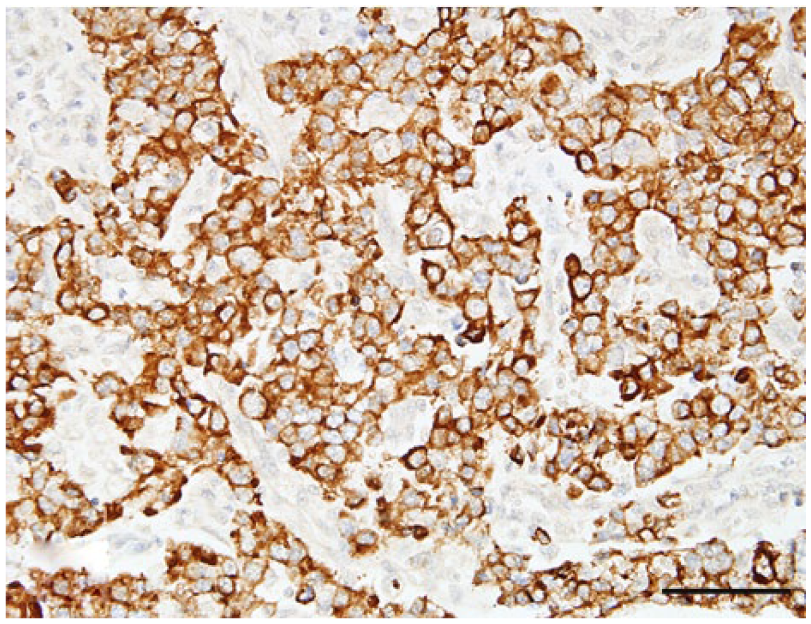
Nasal or sinonasal carcinoma, dog 41. Neoplastic cells with immunopositivity for pancytokeratin AE1/AE3. Hematoxylin and eosin. Bar = 50 µm.
The neuroanatomic location of lesions during a neurologic evaluation is crucial and typically points to a specific group of differential diagnoses that will dictate the upcoming medical procedures to be adopted in a particular case. The advances in diagnostic imaging technologies in veterinary neurology have allowed clinicians to better characterize canine and feline brain tumors and offer better care for patients.9,30,36 While the prevalence and location of skull base neoplasms has been extensively documented in the human medical literature, information is scarce in the veterinary literature. Multiple case series have described central nervous system (CNS) neoplasms in dogs, but no studies, to the author’s knowledge, have reported the aspects such as prevalence and anatomic location of canine skull base neoplasms as a clinical entity that could be compared with its human counterpart.22,25,33–35
The differential diagnoses and prognosis of skull base neoplasms in children differs from those in the adult population. 38 The average age of the canine population in the current study was 9.5 years, and no sex (21 females and 21 males affected) or breed predisposition was noted. Most of the diagnosed neoplasms (29 cases) were primary nervous system tumors (25 meningiomas, 2 nerve sheath tumors, and 1 meningeal oligodendrogliomatosis), followed by 12 pituitary or sellar tumors (9 pituitary adenomas, 2 craniopharyngiomas, and 1 pituitary carcinoma), 1 nasal or sinonasal carcinoma, and 1 multilobular tumor of bone. These results are similar to what has been reported in the adult human population, where the most common skull base neoplasms are meningiomas, pituitary adenomas, vestibular nerve sheath tumors, and craniopharyngiomas, followed by chordomas, chondrosarcomas, chemodectomas, and olfactory neuroblastomas. 27
Canine meningiomas occur more frequently in the brain and less frequently in the spinal cord. 22 Common locations in the brain include the frontal or basal telencephalic areas and less often the sellar area,25,34,35 cerebellopontine angle,10,14,35 retrobulbar spaces,25,26,40 and middle ear. 24 In the current study, meningiomas were the most common skull base neoplasia and occurred more frequently in the paranasal region (11 cases), followed by the caudal cranial fossa (8 cases), sellar region (5 cases), and central cranial fossa (1 case). These results are not surprising given the fact that meningiomas are the most common CNS neoplasms in dogs and cats. 22 All meningiomas in the current report were firmly attached to the adjacent tissues, which would have made surgical excision difficult. 36 No relationship was observed regarding different subtypes of meningioma or their grading and their location on the skull base, and although IHC for vimentin, cluster of differentiation (CD)34, and E-cadherin is useful for diagnostic confirmation, only 3 meningiomas in this study required IHC for a final diagnosis. 28
Most of the pituitary adenomas and the pituitary carcinoma in this study were reported as nodular lesions that completely effaced the pituitary gland. Consequently, these neoplasms were likely >1 cm diameter and would be classified as pituitary macrotumors. Although this classification may be controversial, most clinicians consider pituitary tumors <1 cm in diameter as microtumors (as observed in only 1 case in the current study) and tumors >1 cm in diameter as macrotumors. 21 In the majority of dogs (6 cases) affected by pituitary adenomas and the 1 dog with pituitary carcinoma, clinical signs consistent with hyperadrenocorticism suggested that these tumors were functional. 21 The remaining 3 pituitary adenomas were considered as incidental autopsy findings.
Craniopharyngiomas are sellar neoplasms that originate from the remnants of Rathke’s pouch, and should be differentiated from suprasellar germ cell tumors based on their immunopositivity for cytokeratin and negative immunoreactivity for α-fetoprotein.6,23 The 2 nerve sheath tumors in this study affected the trigeminal nerves. Both dogs had marked atrophy of the ipsilateral masseter and temporal muscles, a common finding in such cases. 2 Diffuse leptomeningeal oligodendrogliomatosis is an uncommon presentation of oligodendroglioma characterized by widespread invasion of the subarachnoid spaces without evidence of a primary mass. The diffuse immunopositivity of neoplastic cells for Olig2 and the absence of a primary cerebral or spinal mass confirmed the diagnosis in this case.4,17 Nasal or sinonasal carcinomas are malignant neoplasms that arise from the nasal cavity or frontal sinuses and can occasionally invade through the cribriform plate and rostral telencephalon. 33 The nasal or sinonasal carcinoma in this study was diagnosed based on its location, histology, and IHC. Because examination of the nasal cavity and frontal sinuses was not conducted during autopsy, the exact origin of the neoplasm could not be confirmed. Differential diagnoses included olfactory neuroblastoma and neuroendocrine carcinoma, but both were ruled out based on the immunostaining profile. 3 Multilobular tumor of bone is a slow-growing, locally invasive neoplasm that typically affects the canine skull. Although most of these tumors are benign, they often cause death or euthanasia of the patient because of their location or undergo malignant transformation, with the potential to invade adjacent tissues or metastasize. 37
Footnotes
Acknowledgements
The author thanks Dr. Elizabeth Howerth (Department of Pathology, College of Veterinary Medicine, The University of Georgia; CVM-UGA) for critical revision of the manuscript; Dr. Steven D. Holladay (Department of Veterinary Biosciences and Diagnostic Imaging, CVM-UGA) for providing the canine skull (![]() ) for photography; and Patricia Rowe (Histology Laboratory, CVM-UGA) for the outstanding support with immunohistochemistry.
) for photography; and Patricia Rowe (Histology Laboratory, CVM-UGA) for the outstanding support with immunohistochemistry.
Author contributions
DR Rissi contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; and agrees to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
