Abstract
Chronic inflammatory demyelinating polyradiculoneuropathy occurred in an 11-year-old Labrador Retriever dog. Spinal cord compression resulted from massive radiculitis with prominent cholesterol granulomas. Cholesterol deposition and associated granuloma formation is unique in chronic inflammatory demyelinating polyradiculoneuropathy, in both its human and canine expressions.
Chronic inflammatory demyelinating polyradiculoneuropathy (CIDP) is a progressive or relapsing disorder of the peripheral nervous system.
1
In humans, CIDP is characterized clinically by progressive paralysis, sensory loss, albuminocytologic dissociation in the cerebrospinal fluid (CSF), as well as demyelination, remyelination, and inflammation in peripheral nerve and spinal nerve roots.
8
A similar entity with acute and chronic forms has been reported in dogs.2,14 The chronic presentation normally is manifested by demyelination and remyelination with onion bulb formation associated with prominent mononuclear inflammation.
14
Certainly some aspects of this lesion are manifested in other canine radiculoneuropathies, usually in association with wider involvement of peripheral nerves. Other canine radiculoneuropathies include canine ganglioradiculoneuritis (sensory neuropathy),3,17,19 infections such as those caused by
An 11-year-old female spayed Labrador Retriever dog was presented to the Veterinary Teaching Hospital at the Virginia-Maryland Regional College of Veterinary Medicine (Blacksburg, Virginia) for left rear foot self-mutilation. The owner reported a previous history of diabetes mellitus. Clinical evaluation revealed inappetence and bilateral cataracts. Neuropathic effects detected in the limbs were restricted to sensory deficits (analgesia) in the left rear leg distal to the stifle. Biochemical evaluation and complete blood cell count revealed hyperglycemia >67,900 μmol/L (>700 mg/dL), regenerative anemia, elevated alkaline phosphatase (473 U/L, reference interval [RI]: 8–70 U/L), and hyperglobulinemia (47 g/L, RI: 21–38 g/L; 4.7 g/dL, RI: 2.1–3.8 g/dL), hypoalbuminemia (23 g/L, RI: 28–37 g/L; 2.3 g/dL, RI: 2.8–3.7 g/dL), normal serum cholesterol (7.23 mmol/L, RI: 3.34-8.60 mmol/L; 279 mg/dL, RI: 129–332 mg/dL), regenerative anemia (RBC 2.96 x 1012/L, RI: 5.93–8.59 × 1012/L), and leukocytosis (29.2 × 109/L, RI: 5.01–15.55 × 109/L). No Coombs test was done. The patient was suspected to have immune-mediated hemolytic anemia based on the presence of moderate spherocytosis on cytological evaluation of a blood smear and autoagglutination present at blood typing prior to transfusion. Neurological examination, including computed tomography (CT), demonstrated a lesion compressing the lumbar spinal cord (Fig. 1). Specifically, CT scan with intravenous and intrathecal contrast demonstrated loss of epidural fat and presence of contrast in the subarachnoid space from L2 to L5. CSF analysis demonstrated an elevated protein concentration of 2.97 g/L (RI: <0.50 g/L; 297.3 mg/dL, <50 mg/dL) and albuminocytologic dissociation. The owners elected euthanasia given the poor prognosis associated with loss of sensation and medullary compression observed during the CT scan.
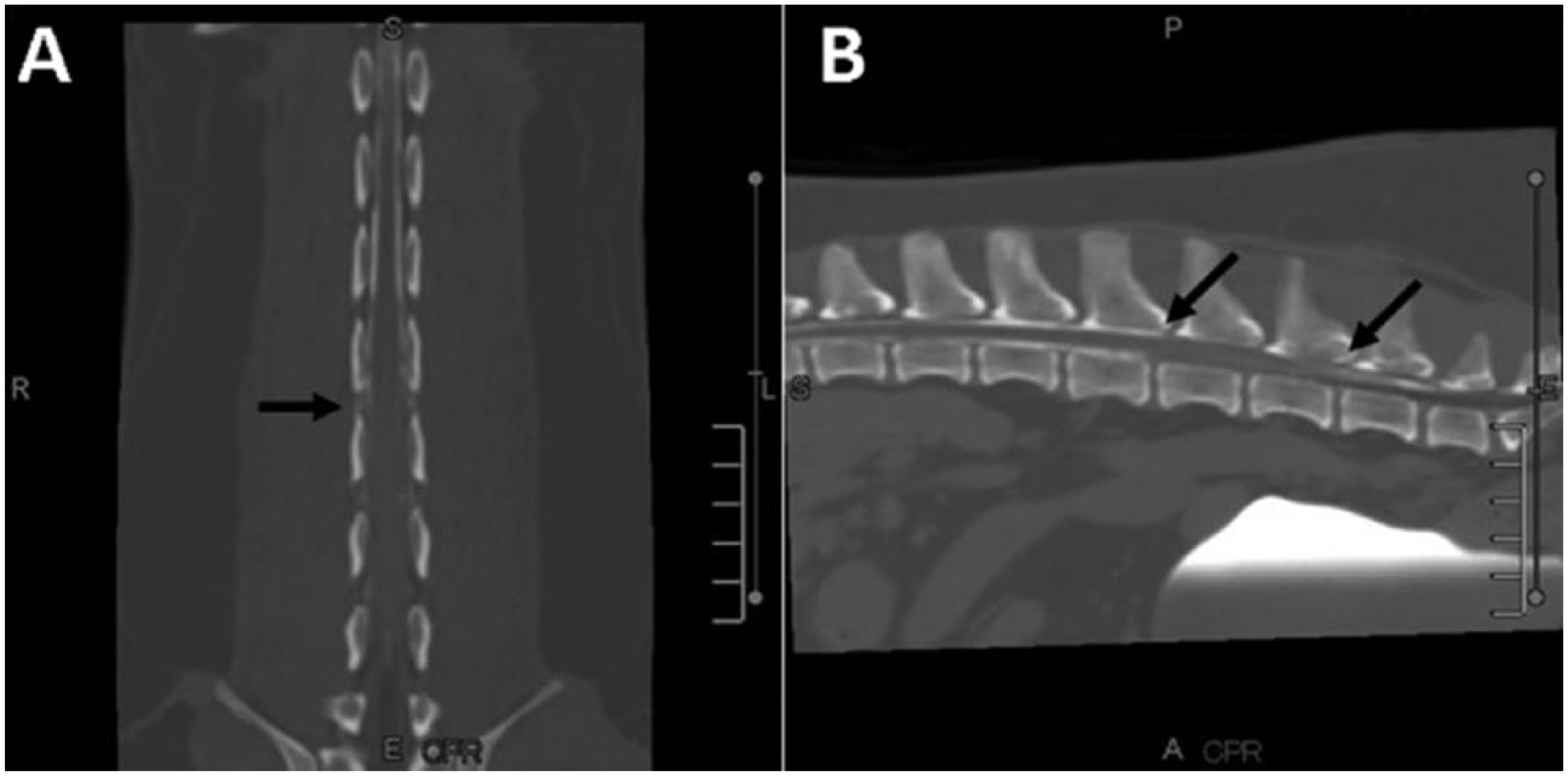
Thoracolumbar, spinal cord computerized tomography (CT) scan. Dorsoventral (
At autopsy, major pathological findings included bilateral gray-tan firm masses in the lumbar subarachnoid space, which incorporated the spinal nerve roots and compressed the spinal cord (Fig. 2). Other findings included loss of the fourth and fifth digits of the left hind limb related to self-mutilation, enlargement of the associated popliteal lymph node, nodular hyperplasia of the liver and pancreas (confirmed by histology), and moderate valvular endocardiosis of the mitral valve.
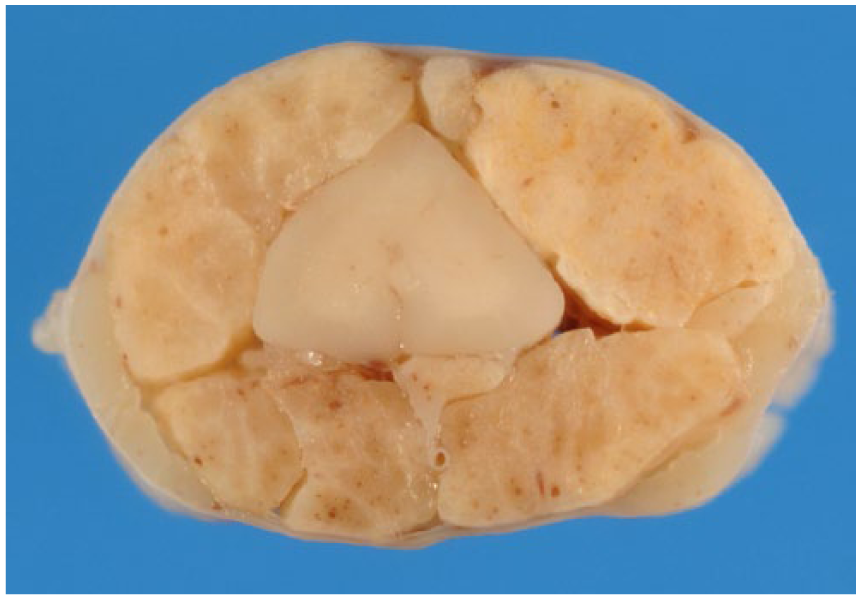
Gross specimen of spinal cord cross-section at the lumbar region. Enlarged chronically inflamed nerve roots surround the spinal cord and compress the cord dorsolaterally.
Tissues were fixed in 10% neutral buffered formalin, dehydrated, impregnated with, and embedded in paraffin, sectioned at 5 µm, and mounted on glass slides. Sections of spinal cord and nerve roots were stained using hematoxylin and eosin, Holmes silver nitrate, Luxol fast blue–periodic acid-Schiff–hematoxylin, and Ziehl–Neelsen acid-fast procedures. In addition, immunostaining using primary antisera against cluster of differentiation (CD)3 (monoclonal, a 1:50), CD79 (monoclonal, a 1:100), CD18 (monoclonal, a 1:50), CD68 (monoclonal, a 1:100), pan-neurofilament (monoclonal, a 1:100), and myelin basic protein (monoclonal, a 1:800) was performed. Nerve root samples, postfixed in buffered 2.5% glutaraldehyde and buffered 1% osmium tetroxide, were dehydrated, embedded in epoxy resin, sectioned at 1-µm thickness, and stained with toluidine blue.
Microscopically, both dorsal and ventral spinal nerve roots in the subarachnoid space from affected cord levels were expanded by prominent chronic inflammation, including extensive cholesterol-bearing granulomas (Fig. 3A, 3B). These lesions contained prominent cholesterol deposits, manifest as acicular clefts, associated multinucleated giant cells, and endoneurial fibrosis; the lesions largely replaced the normal nerve tissue of affected roots (Fig. 3A–C). No acid-fast microorganisms were noted in the lesions. The exudate was characterized by CD18+ and CD68+ macrophages, occasional perivascular clusters of CD3+ lymphocytes, and plasma cells (Fig. 3C, 3D). Both neurofilament and myelin basic protein staining of nerve fibers was diminished in affected nerve roots, but better preservation of neurofilament immunoreactivity relative to myelin basic protein (Fig. 3E, 3F) implies loss of myelin on a considerable proportion of the axons. A few nerve roots were relatively spared (Fig. 3B). Microscopic examination of more distal peripheral nerves was not conducted. Although there was some compression of the spinal cord by the inflamed nerve roots (Fig. 2), no significant intramedullary white matter fiber degeneration or neuronal cell body lesions were detected.
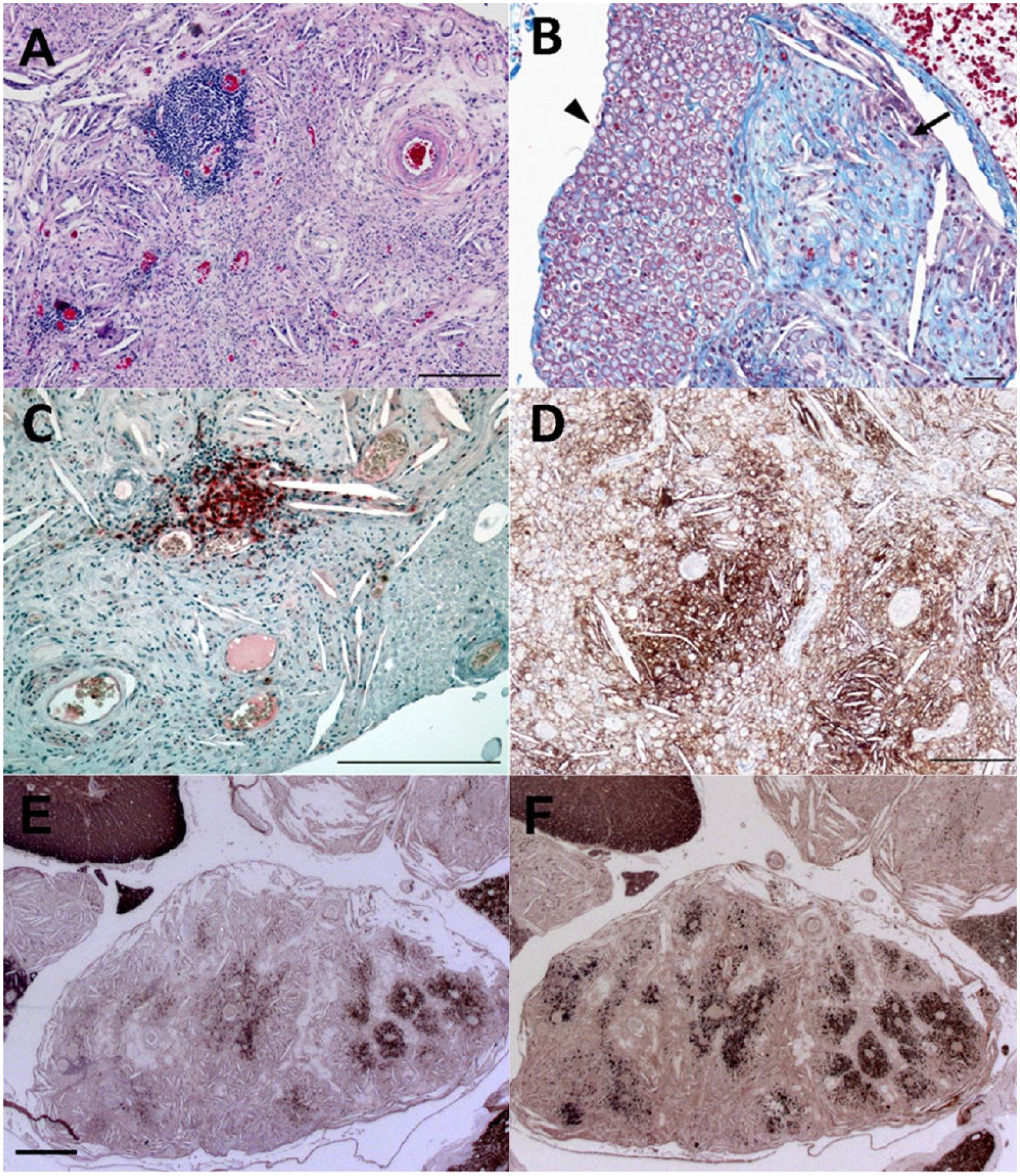
Histological features of the granulomatous inflammation of the polyradiculoneuropathy (all images are of ventral roots).
Examination of 1-µm thick sections of epoxy resin–embedded nerve roots confirmed the above findings. There was significant loss of myelinated fibers in affected roots, with associated endoneurial fibrosis (Fig. 4A). In addition, some actively demyelinating fibers and occasional degenerating myelinated fibers were present (Fig. 4A, 4B). Several clusters of small myelinated fibers consistent with regeneration were noted (Fig. 4A). There were numbers of small thinly myelinated fibers in affected roots, likely representing remyelination (Fig. 4C).
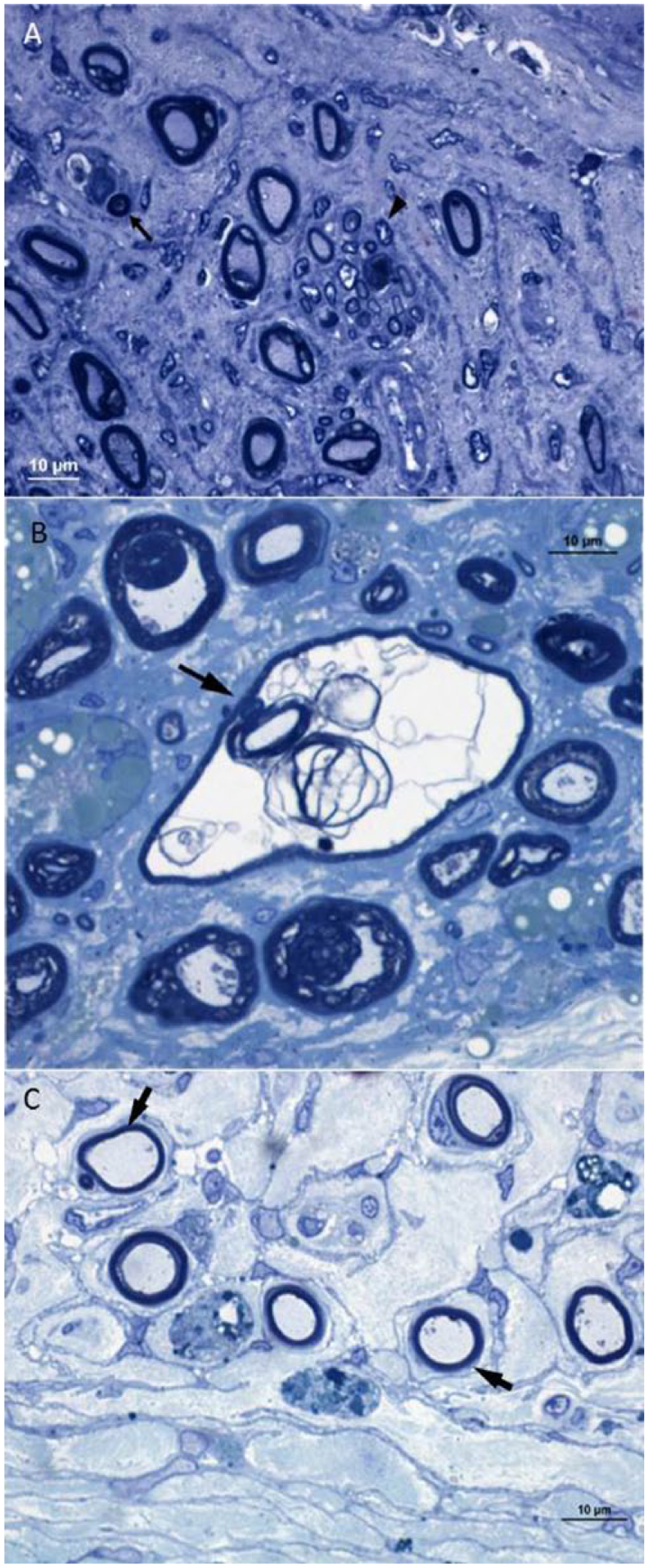
The diagnostic criteria for CIDP in humans require the presence of demyelination and remyelination. 18 In our case, the granulomatous inflammation was sufficiently severe to partially obscure some of these characteristics; however, these diagnostic features were observed histologically and under electron microscopy, corroborating the diagnosis of CIDP-like disease in this dog. Lesions considered supportive of the diagnosis of CIDP, such as perivascular mononuclear infiltration, presence of macrophages, and onion bulb formations, 18 were observed histologically and confirmed by specific immunostaining and electron microscopy.
The pathogenesis of CIDP has not been fully elucidated, but current data supports the theory that it is an immune-mediated disorder, driven by cell-mediated and humoral responses against peripheral nerve antigens. 11 In our case, the presence of hyperglobulinemia may be part of this underlying pathogenesis. Another important feature observed in cases of CIDP in humans is the presence of elevated protein concentration in the CSF. 8 Whether this is related to hypergammaglobulinemia is unclear; however, few reports in human medicine correlated these 2 findings. 11 Although the veterinary literature is scarce, in a case report series where 12 dogs and 8 cats with this condition were evaluated, only 1 dog and 1 cat showed elevated CSF protein. 2 In our case, CSF protein concentration was significantly elevated along with marked hyperglobulinemia, 2 features well described in the human condition, but rarely seen in veterinary medicine.
In human medicine, CIDP has been associated with many systemic disorders such as diabetes, hypoproteinemia, thyroid dysfunction, and neoplasia.11,18 In our case, there was a history of diabetes. Although diabetic neuropathy in humans may have a variety of clinical presentations, the major lesion is considered to be demyelination or loss of myelinated fibers caused by ischemia.9,16 There are some forms of human diabetic neuropathy with associated inflammation, but the pattern of that lesion differs from what was seen in our case. 16 In canine diabetic neuropathy, the major lesion is axonal atrophy of myelinated and unmyelinated fibers, demyelination, and, to a lesser degree, intra-axonal accumulation of glycogen.6,17 It has been postulated that, in diabetic lumbosacral radiculoplexus neuropathy in humans, there could be severe vascular damage in the nerve roots with cholesterol crystals derived from extravasated blood. 7 Somewhat similar appearances of crystalline deposits in the dermis of the foot have been reported in diabetic patients and are thought to be associated with damage to arterioles. 15 Because these neuropathologic descriptions of diabetic neuropathy differ from our case, we feel that this metabolic disorder did not significantly contribute to its lesions.
As noted above, we postulate that a unique aspect of the present case is the presence of prominent cholesterol granulomas in chronically inflamed lumbar spinal nerve roots. The association of such granulomas with chronic neuritis or inflammatory radiculoneuropathy is rare, although this lesion has been encountered in the sural nerve in a study of experimental allergic neuritis in guinea pigs. 10 Because myelin is rich in cholesterol, we suggest that its deposition in these lesions is related to the destruction of myelinated nerve fibers secondary to chronic inflammation 12 as seen in the optic nerve after experimental damage. 13 Although the lesions in the affected nerve roots appeared chronic, there was some evidence of ongoing demyelination and axonal degeneration, which was likely the basis of the fiber loss. In addition, some remyelination and fiber regeneration were present. While more distal peripheral nerves were not examined, there was clinical evidence of involvement in such regions, as indicated by self-mutilation of the foot.
Footnotes
Authors’ contributions
DP Sponenberg and T Pancotto contributed to acquisition of data. RHM King contributed to acquisition and interpretation of data. All authors critically revised the manuscript. P Piñeyro gave final approval. P Piñeyro and BS Jortner contributed to conception and design of the study; contributed to acquisition, analysis, and interpretation of data; drafted the manuscript; and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Dako North America Inc., Carpinteria, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
