Abstract
Sensitive detection of
The agent thought to cause postweaning multisystemic wasting syndrome (PMWS) is
The development of a molecular technique, TaqMan real-time polymerase chain reaction (PCR), has advanced epidemiological studies of PMWS. 1 The determination of viral loads by TaqMan real-time PCR is dependent on the relationship of the quantification cycle (Cq) to the standard calibration curve. 2 This assay has several intrinsic limitations. First, sensitivity to PCR inhibitors can affect the accuracy of quantification. Additionally, the preparation of a calibration curve is required, which can be a laborious and time-consuming process. 7 Ultimately, quantification relies on the Cq values, which can lead to error amplification and limit its usefulness. 9
Droplet digital PCR (ddPCR) is a novel PCR technology. 11 The assay is an endpoint measurement that enables the quantification of nucleic acids without using a standard curve and without being dependent on the reaction efficiency. Because absolute quantification and enhanced detection sensitivity can be achieved, ddPCR has been used in various research applications, such as in microorganism detection 12 and biomedical research. 3 In our study, a specific and sensitive diagnostic method based on ddPCR for the detection of PCV-2 was established.
We obtained serum samples from 107 pigs that had tested positive by enzyme-linked immunosorbent assay (ELISA) for PCV-2. The pigs were 30–60 days of age from a herd in the southern Sichuan Province. The herd had not been immunized against PCV-2, and some pigs showed signs of PMWS. Some pigs that had died had macroscopic lesions consistent with PMWS infection, including lymphadenopathy and pulmonasry infiltrates. PMWS infection was confirmed by immunohistochemical testing and PCR sequencing. The serum samples were stored at −70°C prior to extraction of total DNA using a commercial kit, a according to the manufacturer’s instructions. Extracts were then stored at −20°C.
Field isolates of
The primers and probe were designed for PCV-2 TaqMan real-time PCR and ddPCR using commercial software b based on the nucleotide sequences of ORF2 (open reading frame 2) that was retrieved from GenBank (KC823059.1) and represented sequences of isolates from China (ZJG1103). The primer and probe sequences were as follows: F1 (5′-GCTGGAGAAGGAAAAATGG-3′); R1 (5′-TTGACAGTATATCCGAAGGT-3′); and probe (FAM-5′-TTCAAC ACCCGCCTCTCCCG-3′-BHQ).
The complete PCV-2 sequence was PCR amplified using the primers F2 (5′-TTTCCGCGGGCTGGCTGAACTTTTGAAAGT-3′) and R2 (5′-AGCCCGCGGAAATTTCTGACAAACGTTACA-3′). The plasmid standard was constructed by inserting the complete sequence of PCV-2 into a commercial vector,
c
according to the manufacturer’s instructions, and was then transformed into
Real-time PCR was carried out using a real-time PCR system. e The procedure was optimized with regard to the concentrations of reagents and the annealing temperature. For each amplification step, the best reaction mixtures consisted of 10 μL of 2× PCR master mix, f 2 μL of template, and the primer and probe at final concentrations of 750 nM and 500 nM, respectively, in a 20-μL final volume. Amplification programs were as follows: 95°C for 30 sec, followed by 40 cycles of 95°C for 5 sec, and 56°C for 60 sec.
The same real-time PCR primers and probe were used in the ddPCR system. g According to the manufacturer’s instructions, the optimized ddPCR reaction mixtures included 10 μL of 2× ddPCR master mix, h 2 μL of template, and the primer and probe at final concentrations of 900 nM and 250 nM, respectively, in a 20-μL final volume. All reaction mixtures were loaded into a disposable plastic cartridge i along with 70 μL of commercial oil, j and then were placed onto a droplet generator g that partitioned each sample into 20,000 water-in-oil, nanoliter-sized droplets. Droplets were transferred into a 96-well plate k for PCR. The cycling conditions were as follows: 95°C for 10 min, followed by 40 cycles of 94°C for 30 sec and 52°C for 60 sec, 1 cycle of 98°C for 10 min, and ending at 12°C. Finally, plates containing the droplets were placed into a droplet reader, g and data were analyzed using commercial software. l
Correlation and regression analyses of standard curves from TaqMan real-time PCR and ddPCR were performed with commercial software. m For ddPCR, Poisson statistics were used to measure the initial template concentration using commercial software. l Kappa statistics were used to determine the agreement between ddPCR and TaqMan real-time PCR. n To avoid generating false-positive results, we monitored for potential contamination throughout the study.
Selection of an optimal annealing temperature is one of the most critical parameters for the specificity of a reaction. Setting an annealing temperature too low can lead to the amplification of nonspecific PCR products, and a temperature that is too high can reduce the yield. Therefore, plasmid (9,830 copies/μL) was annealed at the following temperatures: 61, 60.4, 59.3, 57.3, 54.9, 53, 51.7, and 51°C. Based on the FAM (6-carboxyfluorescein) signals that we displayed as rain plots (Fig. 1), 52°C was selected as the annealing temperature, which resulted in the greatest fluorescence amplitude difference between the positive (gray) and negative (blue) controls.
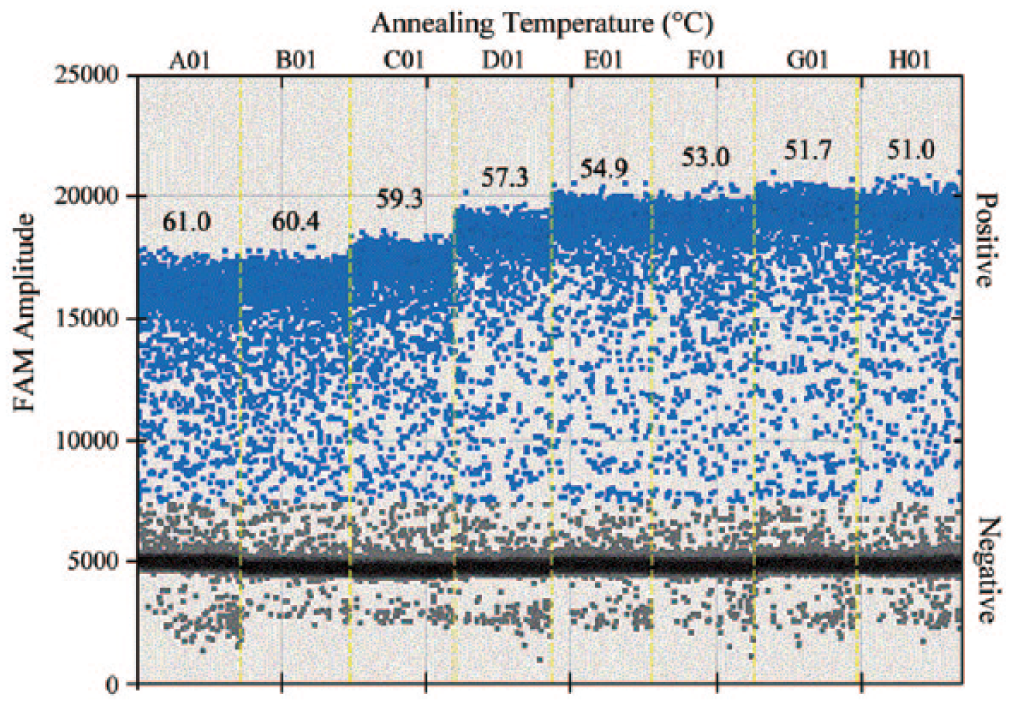
FAM (6-carboxyfluorescein) fluorescence amplitude of different annealing temperatures. The assay was conducted across an annealing temperature gradient: 61.0, 60.4, 59.3, 57.3, 54.9, 53, 51.7, and 51°C.
No FAM fluorescence signal was observed in either the negative control, or in PCV-1, PPV, PRV, CSFV, or PRRSV samples (Supplementary Fig. 1 available at http://vdi.sagepub.com/content/by/supplemental-data). The modified ddPCR developed in the current study was able to detect PCV-2.
Standards with different copy numbers were used to evaluate the robustness and reproducibility of the ddPCR assay. For each sample, samples were tested in triplicate to evaluate intra- and interassay reproducibility. The intra- and interassay coefficient of variation (%) for concentration (copies/μL) ranged from 1.30% to 2.70% and from 0.67% to 2.50%, respectively (Table 1).
Robustness and reproducibility of droplet digital polymerase chain reaction.*
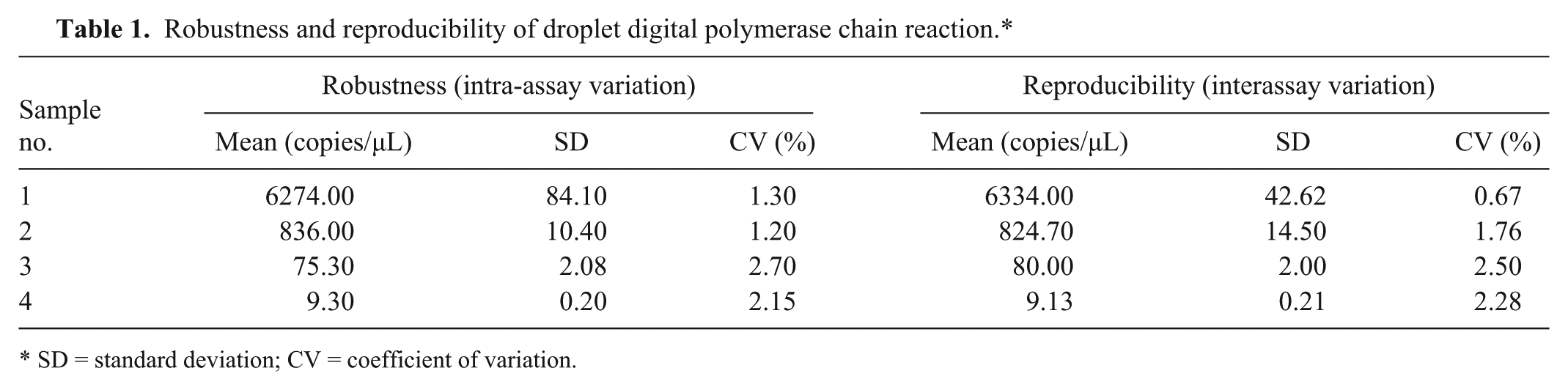
SD = standard deviation; CV = coefficient of variation.
Serially diluted PCV-2 plasmids were prepared in triplicate, and then standard curves of PCV-2 were constructed using ddPCR and TaqMan real-time PCR to compare the limitations, linearity, and efficiency of quantification (Fig. 2; Table 2). We found that ddPCR and TaqMan real-time PCR exhibited good linearity, with
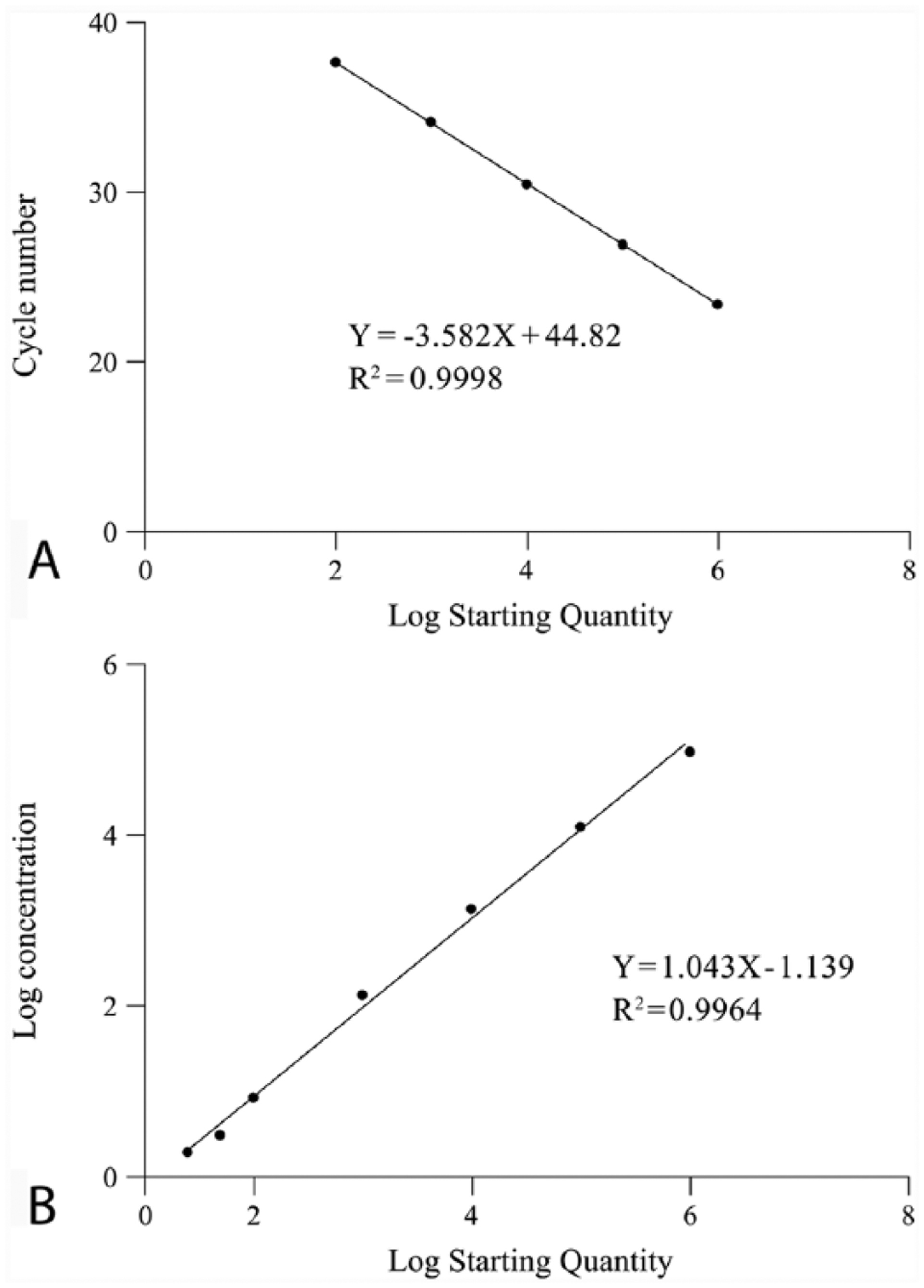
Standard curves of
Comparison of TaqMan real-time polymerase chain reaction (PCR) and droplet digital (dd)PCR using serially diluted
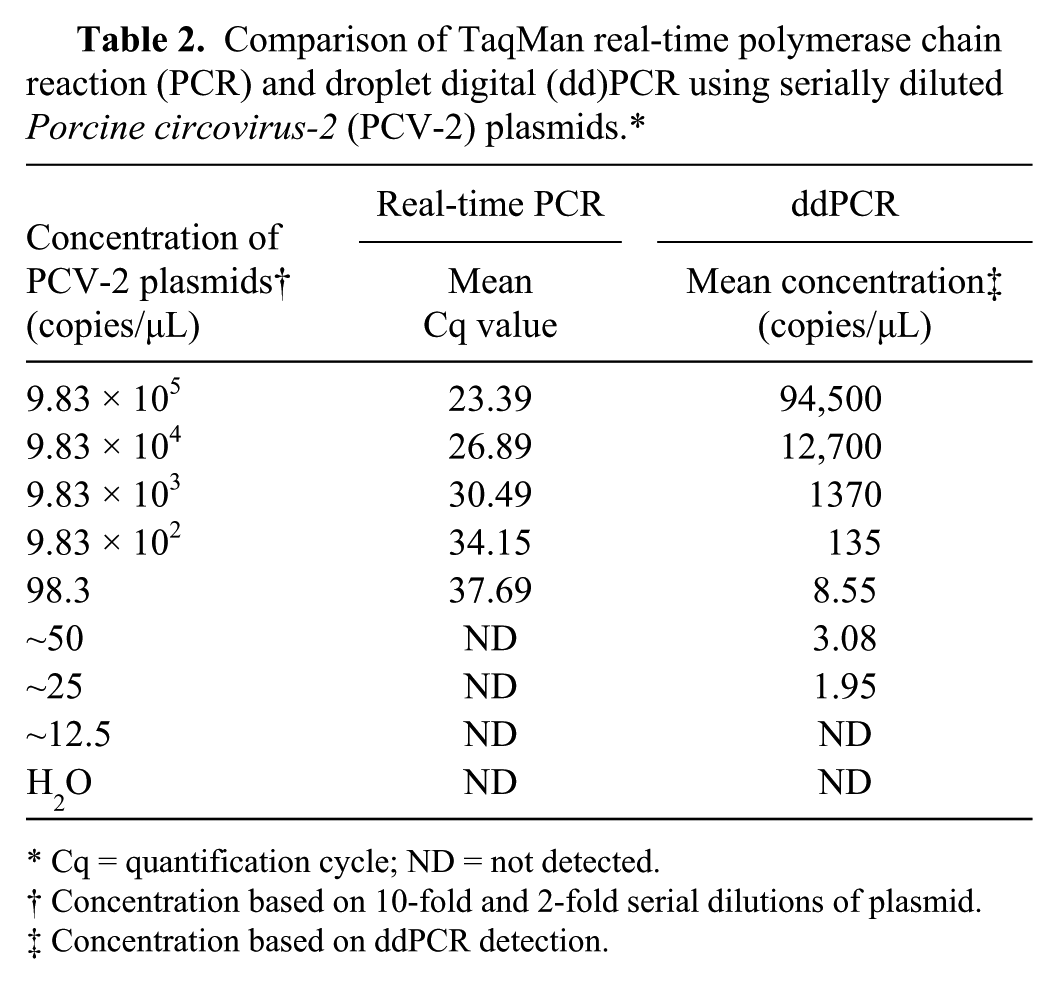
Cq = quantification cycle; ND = not detected.
Concentration based on 10-fold and 2-fold serial dilutions of plasmid.
Concentration based on ddPCR detection.
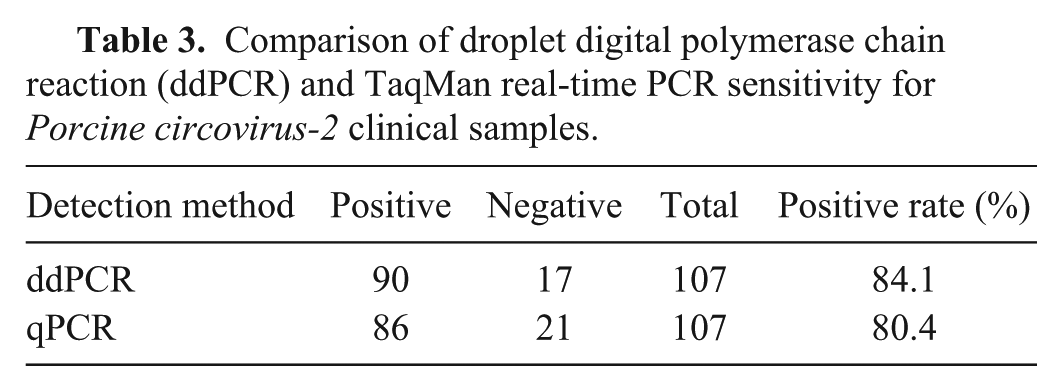
The 107 PCV-2 antibody-positive serum samples were used to compare the detection sensitivities of the ddPCR and the TaqMan real-time PCR (Table 3). The former correctly identified 90 of the samples (84.1%), the latter 86 (80.4%). All of the samples correctly identified by TaqMan real-time PCR were also detected by ddPCR (Supplementary Fig. 2 available at http://vdi.sagepub.com/content/by/supplemental-data). The kappa statistic was 0.87 (standard error: 0.06, with 95% confidence intervals), indicating almost perfect agreement between ddPCR and TaqMan real-time PCR.
Comparison of droplet digital polymerase chain reaction (ddPCR) and TaqMan real-time PCR sensitivity for
The ddPCR, the latest version of digital PCR, 14 partitions each sample into thousands of droplets; PCR amplification is carried out within each droplet. After the reaction endpoint, Poisson statistics are used to measure the initial template concentration by determining the fraction of positive (containing an amplified target) and negative (no amplified target) droplets. 4 Compared with TaqMan real-time PCR, ddPCR has been confirmed to have some of the following advantages: it is more sensitive for low copy number quantification, 6 is better at identifying single nucleotide polymorphisms, 10 is less susceptible to PCR inhibitors, and can detect target DNA in complex environments. 5 All of these features make ddPCR a promising detection technique and a practical method for clinical diagnosis. However, a limitation of ddPCR compared with TaqMan real-time PCR is that target samples should be present at <100,000 copies because high starting concentrations can result in nonlinear results. 8
The ddPCR assay, using a preprocessed mixture, allows for absolute quantification to be achieved. Detection over a wide dynamic range of concentrations by ddPCR allows for the measurement of viral ranges in animals, which can exhibit various levels of infection.
16
In the present study, the linear regression correlation coefficients were measured using either ddPCR or TaqMan real-time PCR showed good
Footnotes
Authors’ note
Shan Zhao and Hua Lin contributed equally to this work.
Authors’ contributions
S Zhao contributed to conception and design of the study, and to acquisition, analysis, and interpretation of data, and drafted manuscript. H Lin contributed to conception and design of the study, and acquisition of data. Q Yan contributed to conception of the study and analysis of data. Y Yan, Y Sun, and J Hu contributed to acquisition of data. S Chen contributed to acquisition and analysis of data. M Yang contributed to acquisition and interpretation of data. Z Chen, and L Xi contributed to analysis of data. Z Hao contributed to analysis and interpretation of data. C Wen contributed to interpretation of data. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
DNeasy tissue kit, Qiagen GmbH, Dusseldorf, Germany.
b.
Oligo Primer Analysis software, Molecular Biology Insights Inc., Colorado Springs, CO.
c.
pMD19-T simple vector, TaKaRa, Dalian, China.
d.
e.
CFX96 Real-Time PCR system, Bio-Rad Laboratories, Hercules, CA.
f.
Premix Ex Taq, TaKaRa, Dalian, China.
g.
QX100 Droplet Digital PCR System, Bio-Rad Laboratories, Hercules, CA.
h.
ddPCR Super Mix for probes, Bio-Rad Laboratories, Hercules, CA.
i.
DG8 cartridges, Bio-Rad Laboratories, Hercules, CA.
j.
Droplet generation oil, Bio-Rad Laboratories, Hercules, CA.
k.
Eppendorf AG, Hamburg, Germany.
l.
QuantaSoft software, Bio-Rad Laboratories, Hercules, CA.
m.
GraphPad Prism, GraphPad Software Inc., La Jolla, CA.
n.
SPSS Statistic 22, IBM Corp., Chicago, IL.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Program for Changjiang Scholars and Innovative Research Team in University, China (IRT0848), and Research Project from the Sichuan Entry-Exit Inspection and Quarantine Bureau (SK201403).
