Abstract
Obesity and endogenous hyperadrenocorticism (HAC) are common clinical conditions in veterinary practice, and both conditions have clinical and laboratory similarities, such as weight gain and dyslipidemia. The objective of the present study was to characterize and compare the lipid profiles and plasma lipoprotein fractions in healthy dogs (
Keywords
Introduction
Hyperlipidemia is characterized by hypercholesterolemia and/or hypertriacylglycerolemia, which are frequently associated with several metabolic complications in primates as well as pets. 8,23 In the dog, hyperlipidemia is generally associated with ingestion of lipid-rich diets or diseases such as nephrotic syndrome, hypothyroidism, diabetes mellitus, hyperadrenocorticism, and obesity. Primary hyperlipidemia also occurs but is infrequent. 8,24 Obesity is commonly observed and is generally associated with a decreased life span and decreased quality of life in pets. The causes and social factors contributing to obesity include genetic and sexual characteristics, physical inactivity, excessive food ingestion, and excessive caloric intake. 14,15 Hyperadrenocorticism (HAC) also may occur spontaneously with weight gain, and its high prevalence in dogs may be associated with several morbid systemic effects related to chronic hypercortisolism. 9,22 Although a frequent phenomenon, obesity becomes clinically relevant when the excessive fat accumulation is associated with the development of hyperlipidemia and, consequently, hyperlipoproteinemia.
Lipoproteins (LPs) transport lipids in lymphatic and blood circulation. They are classified by their lipid and protein composition as follows: chylomicron, very low-density lipoprotein (VLDL), low-density lipoprotein (LDL), and high-density lipoprotein (HDL). Chylomicron transports the lipids of exogenous origin, whereas the other LPs transport endogenous lipids. 3–5 There are several methods to separate lipoproteins depending on their size, form, electric charge, and density.
Hyperlipidemia can be caused by a quantitative increase in circulating LPs or by a higher lipid concentration in the several LP classes. These modifications of the LP profile can result in a greater tendency to develop hepatic lipidosis, pancreatitis, seizures, and ophthalmopathy in dogs. 28 In addition, dyslipoproteinemias may be a manifestation of metabolic syndrome, which is associated with the early development of atherosclerosis and diabetes mellitus in humans and dogs. 6,12,18 The present study endeavored to characterize and compare the plasma LP fractions in obese dogs and dogs with HAC with those of healthy dogs.
Plasma concentrations of total cholesterol (TC), triacylglycerol (TAG), and percentage distribution (fast protein liquid chromatography) of TC and TAG in the lipoprotein (LP) fractions in dogs that are healthy (control), obese, or with hyperadrenocorticism (HAC).*
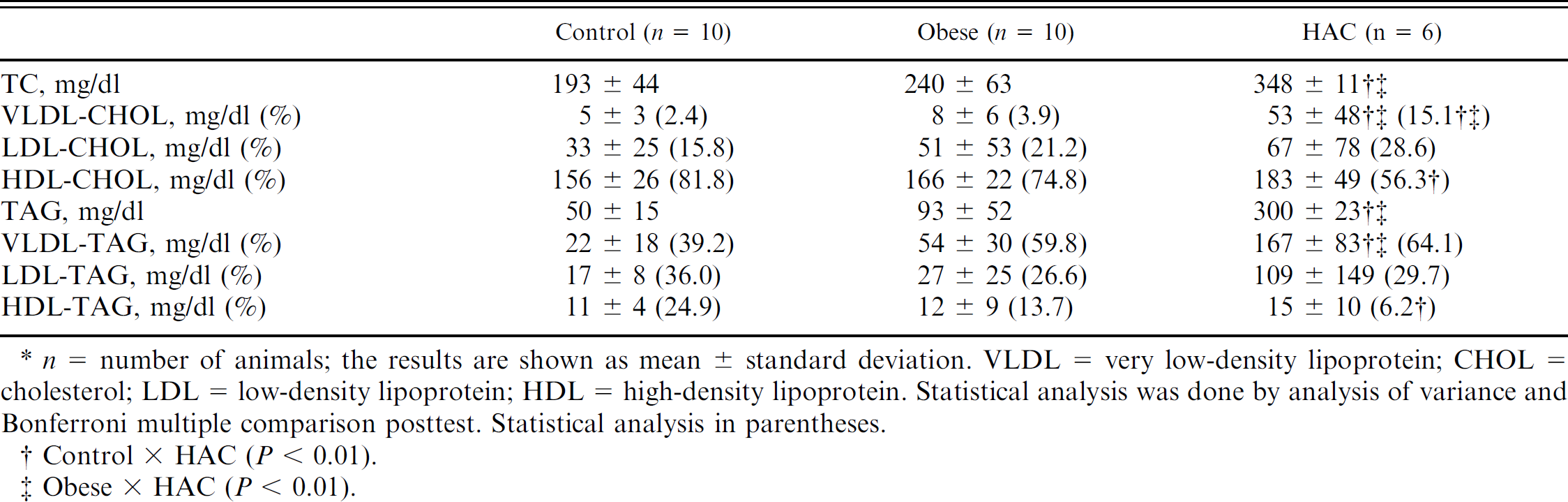
Control × HAC (
Obese × HAC (
Material and methods
Animals
A total of 26 dogs were studied. Ten animals were selected for the control group, 10 for the obese group, and 6 for the HAC group at Hospital Veterinário Anhembi Morumbi (HOVET-AM; Sao Paulo, Brazil). All 26 dogs were client owned, and owner-informed consent was obtained prior to the beginning of the study. The 3 study groups contained dogs of both sexes, several breeds, and various ages. The control group included healthy dogs of several breeds, an age range of 2–10 years, and both sexes (5 males, 5 females). The obese group included dogs of several breeds, an age range of 2–12 years, and both sexes (4 males, 6 females). The HAC group included dogs that ranged from 6 to 12 years in age and included both sexes (1 male, 5 females). The identification of obese dogs was determined by body condition score and measurement of body fat content (%BFC > 20%) that was performed at the Veterinary Clinic of HOVET-AM. The body condition score was determined by the following formula:
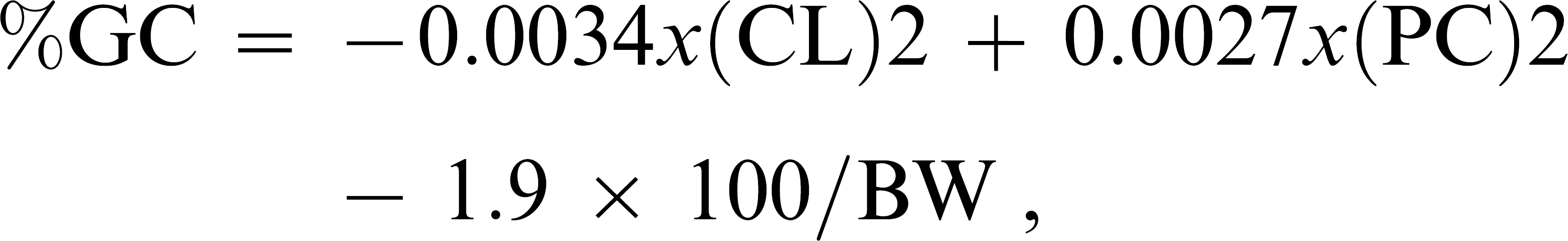
where CL indicates the distance from the calcaneus to the cruciate ligament; PC indicates the pelvic circumference; and BW indicates the body weight. 7 Furthermore, obese dogs lacked clinical signs of endocrinopathy or the administration of medications that could result in weight gain. The diagnosis of HAC was determined by the presence of clinical signs and routine laboratory results (renal and liver function, serum glucose, total cholesterol [TC], triacylglycerols [TAG], albumin, and total protein) related to hypercortisolism and laboratory testing of adrenal function (baseline cortisol value and normal dexamethasone suppression test determined 8 hr after administration of 0.01 mg dexamethasone/kg intravenous). The protocol included animals with a post–dexamethasone plasma cortisol concentration >1.5 μg/dl. All tests were performed at the Laboratory of Clinical Pathology of HOVET-AM.
Lipoprotein sampling and analysis
The plasma LPs were separated through a fast protein liquid chromatography (FPLC) system on an HR 10/30 Superose 6 column. a Two hundred microliters of plasma was applied to the column under a constant flow of 0.5 ml/min of Tris buffer (pH 7.0; 10 mmol of Tris, 150 mmol of NaCl, 1 mmol of ethylenediamine tetra-acetic acid, and 0.03% NaN3). Sixty fractions of 0.5 ml were collected automatically. The TC and TAG concentrations in the VLDL, LDL, and HDL fractions were determined enzymatically using an automated biochemical analyzer. b The procedures above were performed at the Lipids Laboratory (Faculty of Medicine, University of Sao Paulo, Brazil).
Statistical analysis
The data obtained in the present study are expressed as the mean ± standard deviation. The significant differences among the group means were determined by analysis of variance and the Bonferroni multiple comparison posttest. The level of significance tested was
Results
The lipid concentrations and percentages of distribution among the LP fractions are presented in Table 1 and Figure 1. The control animals had a mean plasma cholesterol (CHOL) concentration of 193 ± 44 mg/dl. The percentage distribution among the various LP fractions was 2.4% of VLDL, 15.8% of LDL, and 81.8% of HDL. Plasma TAG concentration was 50 ± 15 mg/dl, and the percentage distribution of the TAG-LP was 39.2% VLDL, 36.0% LDL, and 24.9% HDL. The lipid profile of this group was characteristic of healthy dogs (Fig. 1). 19,20
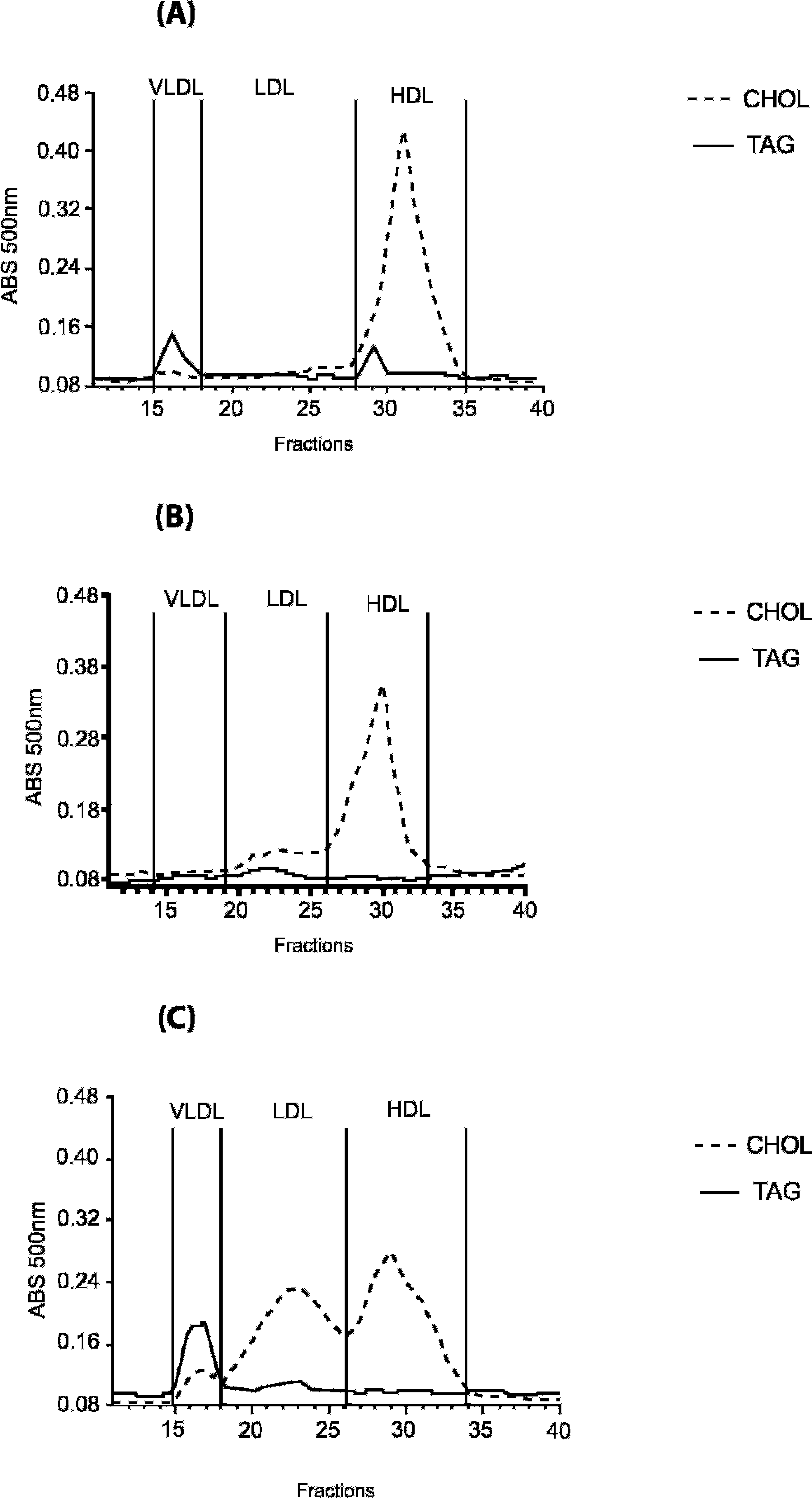
Representative profile of the percentage distribution of total cholesterol and triacylglycerol (TAG) in the lipoprotein fractions determined chromatographically. Fast protein liquid chromatography in control dog (
Plasma TC was significantly higher in the HAC group when compared with the obese and control groups (Table 1). Plasma TC of the obese group was not significantly different from the control group. Within the LP fractions, no differences were observed in the LDL-CHOL among the 3 groups. In dogs with HAC, hypercholesterolemia resulted from a greater percentage of VLDL compared with both obese and healthy groups (Table 1; Fig. 1). On the other hand, HDL-CHOL was lower than that of the control group.
Hypertriacylglycerolemia was greater in the HAC group as compared with the obese and control groups (Table 1), but the percentage of TAG distribution was not different among the 3 groups. When the absolute concentrations of CHOL and TAG in LP fractions were compared, statistical differences in VLDL-TAG were found, but no differences were observed in HDL-TAG and HDL-CHOL. The LP profile of the obese group was similar to that of dogs with HAC, despite a proportionally lower TAG percentage in the same LP classes (Fig. 1). In contrast, the concentration of HDL-TAG was lower in the HAC group (Table 1; Fig. 1).
Discussion
The long-term effects of hyperlipidemia in dogs have not been extensively studied. Dyslipidemia may be associated with neurological disorders, ophthalmopathy, pancreatitis, hepatic lipidosis, and insulin resistance. 4,8 The association between hyperlipidemia, increased visceral fat accumulation, arterial hypertension, and insulin resistance are characteristic of metabolic syndrome, which increases the risk of developing diabetes mellitus and atherosclerosis. 27 The present study describes the lipid profile of healthy dogs and dogs that have spontaneous obesity or HAC.
There are several procedures to fractionate the LPs 11,21 ; however, FPLC allows LP isolation from small plasma volumes (∼200 μl) using frozen plasma samples and reducing apolipoprotein displacement due to the ultracentrifugation method.
Furthermore, the TC concentrations of the LP fractions correlate significantly with the TC content of LPs obtained by ultracentrifugation. 13,17 In dogs, HDL is the main form of CHOL transport in plasma, which represents a different HDL composition as compared with human HDL. 2,3 In most studies, fractionation of the plasma LP has been performed by ultracentrifugation using different density ranges than that established for humans. 19
The lipid profile of the control group was characteristic of healthy dogs. 19,20 These individuals had low concentrations of VLDL-CHOL and LDL-CHOL and a high concentration of HDL-CHOL. 2,5 The higher concentration of HDL-CHOL observed in animals such as dogs and cats decreases the risk of developing atherosclerotic disease. This LP profile is attributed to the absence of CHOL ester transfer protein, which transfers CHOL ester from HDL to VLDL and TAG in the opposite way.
Based on the FPLC analysis, the obese group of dogs did not present alterations in LPs when compared with the control dogs. However, it has been shown that obese dogs can present higher mean values of TAG, VLDL-TAG, and HDL-TAG and lower mean values of HDL-CHOL when compared with healthy dogs. This LP profile may be characteristic of dogs with insulin resistance. 2 This dyslipidemic profile, associated with excessive body fat, may predispose dogs to metabolic syndrome, as has been demonstrated in humans. 10,23,26 It is known that insulin resistance stimulates the hormone-sensitive lipase activity, which increases free fatty acid (FFA) concentration. 2 An increase in FFA concentration also has an important role in insulin sensitivity. The FFAs from visceral fat reach the liver through the portal-hepatic vein, where they impair insulin action on hepatocytes. 1 Perhaps, in the current study, the lipid profile of the obese group of dogs was not different from that of the control group because of the small sample size and because the manifestations of the metabolic syndrome are heterogeneous in obesity.
In contrast, the HAC group had higher cholesterolemia and increased the VLDL-CHOL fraction when compared with the control and obese groups. The concentration of HDL-CHOL was significantly lower in the HAC group when compared with the control group. Also, the absolute amount of VLDL-TAG was significantly higher when compared with the normal dogs. There are very few studies on alterations of the LP profiles of dogs with HAC, although hypercholesterolemia and hypertriacylglycerolemia are common, especially in association with pancreatitis and hepatic lipidosis. 9,22 It is known that the administration of glucocorticoids increases plasma TAG concentration by increasing the hepatic synthesis and secretion of VLDL. In animals with visceral obesity, as well as HAC, insulin resistance results in a lower LP lipase (LPL) activity. The higher plasma concentration of FFA due to increased TAG hydrolysis in adipocytes induces the formation of LPL-FFA complexes. This in turn reduces LPL activity as the FFA competes with the TAG for the active site of the enzyme. 25 Consequently, there is a significant decrease in the plasma VLDL clearance rate. The hypertriacylglycerolemia observed in the HAC group was due to increased absolute amount of VLDL-TAG (mg/dl) as compared with control and obese groups (167 ± 83, 22 ± 18, 54 ± 30, respectively). However, because of wide variations in the percentage of VLDL-TAG among the 3 groups, statistical differences were not observed.
In addition, a higher plasma level of TAG is associated with a reduction in HDL, as this LP fraction can originate from surface components that are released from the TAG-rich LP (chylomicrons and VLDL) by LPL activity. 27 A previous study using ultracentrifugation and electrophoresis of LPs demonstrated an increase in the LDL-CHOL of 14 animals with HAC. 4 However, there was no significant difference in the LDL concentrations of dogs with insulin resistance and control dogs, as observed in the present study. 2
HAC promotes the onset of metabolic syndrome, 6 which explains the change of an antiatherogenic profile, characteristic of healthy dogs, 19,20 to an atherogenic one that is prone to the development of hepatic lipidosis, pancreatitis, diabetes, neuropathies, and ophtalmopathies. 28 increased CHOL concentrations in the VLDL and LDL fractions observed in the HAC group also favor the development of atherosclerosis, 6,12,16 although a previous study in dogs has shown that diabetes mellitus and hypothyroidism have a greater propensity to develop atherogenic lesions than HAC. 12
In conclusion, dogs with HAC differ significantly from healthy and obese dogs with regard to the metabolism of CHOL, TAG, and composition of VLDL and HDL fractions. The diagnosis of such lipid profiles may suggest an increased risk of developing hepatic lipidosis, pancreatitis, neuropathy, ophthalmopathy, diabetes, and atherosclerosis.
Footnotes
a.
FPLC™, Superose™ (HR 10/30); Pharmacia, Uppsala, Sweden.
b.
Cobas Mira® automated biochemical analyzer, F. Hoffmann-La Roche, Basilea, Switzerland.
c.
GraphPad Prism software 2.01, GraphPad Software Inc., San Diego, CA.
