Abstract
Thyroxine (T4), free T4 (FT4), and thyrotropin (TSH) concentrations were measured in serum from 693 healthy representatives from 7 dog breeds (Alaskan Malamute, Collie, English Setter, Golden Retriever, Keeshond, Samoyed, or Siberian Husky) to determine whether breed-specific reference intervals (RIs) are warranted. Veterinarians reviewed the health history, performed a physical examination, and approved laboratory data for the enrolled dogs. Many purebred dogs had T4 and FT4 concentrations that were at, or below, the lower limits previously determined for non–breed-specific RIs. Mean concentrations of T4, FT4, and TSH varied significantly among breeds. The range of mean concentration of T4 (19.7 nmol/L [1.53 µg/dL] in English Setters to 29.0 nmol/L [2.25 µg/dL] in Keeshonds) and FT4 (12.6 pmol/L [0.98 ng/dL] in English Setters to 20.2 pmol/L [1.57 ng/dL] in Samoyeds) was considerable. Median TSH values ranged from 6.10 mIU/L (0.07 ng/mL; Alaskan Malamute and Golden Retriever) to 17.6 mIU/L (0.26 ng/mL; Collie). Mean T4 and FT4 concentrations were higher in females. Increasing age was associated with decreasing T4 and FT4, and increasing TSH concentration. The substantial ranges across breeds of measures of central tendency (mean, median) for all hormones indicate that breed-specific RIs are warranted. RIs encompassing the central 95% of reference values for all breeds combined, and for individual breeds, were calculated using nonparametric (TSH) and robust (T4, FT4) methods. Use of breed-specific RIs in combination with careful attention to the potential for pre-analytical and analytical variability in test results will improve thyroid function assessment in these breeds.
Introduction
Diagnosis of hypothyroidism in dogs is based on medical history, clinical signs, and the interpretation of laboratory test results. Typically, the concentrations of thyroid and pituitary hormones in the blood are measured and compared with laboratory reference intervals (RIs) derived from populations of dogs deemed to be healthy based on defined criteria that may include health history, physical examination, and biochemical screening. The necessity for healthy reference individuals for the determination of RIs is readily understood. However, additional factors may also affect reference values. For example, few diagnostic laboratories have sufficient data to provide RIs partitioned by breed. This is problematic because several studies have demonstrated that concentrations of thyroid hormones in serum from different breeds of dogs differ significantly.1,7,13,19,32,35 Initially, total thyroxine (T4) concentrations were reported to be greater in “small” dogs when compared with “medium or large” breed dogs. 24 Healthy Greyhounds were shown to have significantly lower serum T4 and free T4 (FT4) concentrations when compared with non-Greyhounds.7,32 Scottish Deerhounds, 31 Alaskan sled dogs,13,19 Sloughis, 18 and Basenjis 30 all have T4 concentrations that are at, or below, a low limit of the previously established reference range for T4. The data illustrate the importance of the lower limit of a RI for the accurate interpretation of laboratory data used to establish thyroid health. Without breed-specific RIs, healthy dogs from these breeds will likely be misdiagnosed as being hypothyroid or equivocal for thyroid health. As a result, these animals may undergo unnecessary additional testing, be treated with thyroid supplementation at a relatively young age, and/or be removed from breeding programs (thereby affecting genetic diversity). In addition, the prevalence of hypothyroidism in these breeds will be overestimated.
The purpose of our study was to measure T4, FT4, and thyrotropin (TSH) concentrations in serum from healthy purebred representatives from 7 common dog breeds and determine whether there were differences in results according to breed. Our objective was to determine whether breed-specific RIs are warranted for use in these breeds.
Material and methods
The work conducted in our study was approved by the Institutional Animal Care and Use Committee of the University of Minnesota (0801A24323). The selection of breeds to include in the study was either purposive (breeds reputed to have a high prevalence of hypothyroidism: Golden Retriever, English Setter, Keeshond, Samoyed) or by breeder interest (breed associations indicating interest in participation: Alaskan Malamute, Collie, Siberian Husky).
Study population
A total of 782 eligible purebred dogs were recruited for the study. Recruitment was conducted by communicating the goals of the study locally (to clients and staff of the University of Minnesota College of Veterinary Medicine), as well as nationally (through strong recruitment efforts by purebred dog organizations). Dog enrollment and direct sample collection occurred at the University of Minnesota Veterinary Medical Center (St. Paul, Minnesota) at study clinics set up by the investigators and breed clubs, or through collaboration with private practice veterinarians. Recruitment, enrollment, and sample collection and analysis occurred over a 5-year period (2005–2010). Owners and veterinarians that participated remotely received an information packet that contained instructions for completing the health questionnaire and consent form, and for preparing their dog for participation in the study (medication and fasting requirements). Written instructions provided to the veterinarians included animal inclusion and exclusion criteria, physical examination requirements, blood and urine collection procedures, and requirements for sample handling and shipping. Data recording forms and a sample submission checklist were provided for completion by the veterinarian prior to shipment to the sample coordinator at the University of Minnesota. On receipt, the sample coordinator verified that the submission packet met the preenrollment requirements of the study before enrolling the dog and forwarding the samples for analysis.
Eligibility for retention in the healthy reference population was based on history, physical examination, complete blood cell count (CBC), urinalysis, and biochemistry profile. Purebred dogs were preferred to be between 1 and 9 years of age, and were required to be nonpregnant, nonlactating, and have no history of receiving medication during the previous 12 weeks, other than heartworm preventive, flea and tick medication, or nutritional supplements. Dog owners completed a health questionnaire related to the health, history, and activity of their dog. Dog owners confirmed that their dogs had been fasted (with access to water) for at least 6 hr and that no nonsteroidal anti-inflammatory drugs, corticosteroids, trimethoprim-potentiated sulfonamides, anticonvulsants, or thyroid replacement therapy had been prescribed or administered in the 12 weeks prior to presentation. Information requested from the questionnaire (available on request from the submitting author) included American Kennel Club registration number, date of vaccination, past and present health history, medication history, exercise tolerance, weight, water consumption, urination, defecation or appetite changes, date of last estrus, pregnancy history, and any skin abnormality (e.g., itchiness, hair loss). Intact, female dogs were estimated to be in diestrus if the day of sample collection was within 2 months of the last known estrus date as recorded in the owner questionnaire. Serum progesterone concentration was not determined. A physical examination was performed by a veterinarian at the preenrollment session, and dogs were excluded from participation if they had physical signs of estrus or lactation, and had any abnormality, including extreme body condition (obese or cachectic). Physically healthy dogs were accepted for additional health screening, which included the review by a clinical pathologist of serum biochemical profiles, a CBC, and urinalysis results. Exclusion criteria included abnormal findings on the CBC or serum biochemical results that could not be accounted for by a transient stress response (evidence of a corticosteroid or epinephrine leukogram and mild hyperglycemia on the chemical profile); urinalysis results compatible with a urinary tract infection (white blood cell count >5 per 10× objective field or bacteria >20 per 10× objective field on sediment examination); hypercholesterolemia 1.09 mmol/L (42 mg/dL) above the RI (within the analytical imprecision of the upper limit of the RI); and CBC or serum biochemical results that suggested compromised sample quality (more than a few smudged or pyknotic cells in blood smears; marked hypoglycemia; enzyme elevations compatible with delayed separation of serum from cellular elements of blood); or marked hemolysis or lipemia. Dogs were also excluded if they were determined to have conclusive laboratory evidence of hypothyroidism defined as the combined presence of thyroglobulin autoantibody (TgAA > 25% of positive control), elevated TSH concentration (>33.5 mIU/L; >0.6 ng/mL), low T4 (<19.3 nmol/L; <1.5 µg/dL), and/or low FT4 (<7.7 pmol/L; <0.6 ng/dL) concentrations.
After screening of the 782 dogs, the healthy reference population (total 693) for each breed included between 96 and 102 privately owned Alaskan Malamute, Collie, English Setter, Golden Retriever, Keeshond, Samoyed, or Siberian Husky dogs. Slightly more than one-third (240, 34.6%) of retained dogs were resident in Minnesota, with the remainder distributed among 35 U.S. states (435 dogs, 62.8%) and Canada (18 dogs, 2.6%).
Sample collection procedures
Owner consent was obtained prior to sample collection, and owners received a copy of all laboratory test results. Urine samples were collected on the day of sampling by free-catch or cystocentesis. Blood samples were collected from the jugular or cephalic vein. Approximately 3 mL were placed in a sterile tube containing ethylenediamine tetra-acetic acid (EDTA) as a preservative for hematological analysis. Another 5 mL were collected in a sterile evacuated plain glass tube with no additive to obtain serum for biochemical and hormone analyses. Blood samples for biochemical and endocrine testing were allowed to clot, and serum was separated from cells by centrifugation within 60 min of collection and refrigerated at 4°C (biochemical tests) or −20°C (hormone tests) until analysis. Biochemical analysis was performed within 24 hr after collection at the University of Minnesota College of Veterinary Medicine Clinical Pathology laboratory, or at a regional clinical pathology service (Antech Diagnostics or IDEXX Laboratories). Serum samples collected for thyroid hormone analysis were frozen at −20°C until analyzed at the University of Minnesota Veterinary Diagnostic Lab within 1 month of sample collection. Not all analyses could be performed for all dogs because of insufficient serum volume. Because of a commercial supply problem that led to national shortages of FT4 reagent, 32 serum samples were sent frozen to the Animal Health Diagnostic Center at Cornell University College of Veterinary Medicine (Ithaca, New York), where thyroid analyses were performed with the same methods used at the University of Minnesota for the other serum samples.
Thyroid hormone and TgAA determination
Serum concentration of T4 was determined by a chemiluminescent enzyme immunoassay b according to the directions of the manufacturer. The intra-assay coefficient of variation (CV) was 20, 10, 15, and 10% at T4 concentrations of 6.4, 12.9, 25.7, and 51.5 nmol/L (0.5, 1.0, 2.0, and 4.0 µg/dL), respectively. The interassay CV was 20, 12, 15, and 10% at T4 concentrations of 6.4, 12.9, 25.7, and 51.5 nmol/L (0.5, 1.0, 2.0, and 4 µg/dL), respectively. The lowest detectable concentration of T4 was 3.2 nmol/L (0.25 µg/dL). To establish the spiking recovery, a 12.9 µmol/L (1 mg/dL) serum pool was prepared, and serial dilutions were made to achieve a range of 3.2–322 nmol/L (0.25–25 µg/dL). The recovery was >90% at all levels. 34
Serum concentration of FT4 was determined by use of equilibrium dialysis c according to the directions of the manufacturer. As determined by the manufacturer, the lowest detectable amount of FT4 was 1.9 pmol/L (0.15 ng/dL), and the intra-assay CV was 10.7% at FT4 level of 19.3 pmol/L (1.5 ng/dL) and 12.3% at FT4 level of 41.2 pmol/L (3.2 ng/dL). The interassay CV was 15% at FT4 concentrations of 16.7 pmol/L and 34.7 pmol/L (1.3 and 2.7 ng/dL), respectively. Concentrations of FT4 measured after serial dilution of 3 human serum samples were linear throughout its calibration range. The intra-assay CV was 8.3% at FT4 concentration of 25.7 pmol/L (2.0 ng/dL) determined in canine serum. The interassay CVs were 8.7% and 6.9% at FT4 concentrations of 23.2 and 78.5 pmol/L (1.8 and 6.1 ng/dL), respectively, for canine serum.
Serum concentration of TSH was determined using a chemiluminescent enzyme immunoassay d according to the manufacturer’s instructions. As reported by the manufacturer, the intra-assay CVs were 5.0, 4.0, 3.1, and 3.8% at TSH concentrations of 14.4, 29.2, 70.4, and 101 mIU/L (0.20, 0.50, 1.6, and 2.6 ng/mL), respectively. The interassay CVs were 6.3, 7.4, and 8.2% at TSH concentrations of 12.0, 18.2, and 107 mIU/L (0.16, 0.27, and 2.8 ng/mL), respectively. The lowest detectable concentration of TSH was 2.8 mIU/L (0.03 ng/mL). Samples that were determined to contain <2.8 mIU/L sensitivity level were recorded as containing 2.7 mIU/L (0.029 ng/mL). Concentration of TSH measured after serial dilution of 3 canine samples was linear (88–102% of expected values after dilution) throughout the calibration range. Three canine serum pools were spiked with 3 solutions of TSH over a range of 35.2–179 mIU/L (0.64–5.6 ng/mL). The recovery ranged from 85% to 104%.
Thyroglobulin autoantibody concentration was determined using an enzyme-linked immunosorbent assay.e,15,16 The intra-assay and interassay CV for 2 canine serum pools (1 positive and 1 negative sample) was <15% and <22%, respectively. The value of TgAA measured after serial dilution of 1 positive canine sample showed good linearity (r2 = 0.95). Results were calculated using the manufacturer’s “preferred method,” which requires the subtraction of nonspecific binding (wells that are not coated with antigen) values from the average values calculated from antigen-coated wells that contained aliquots of canine serum. This result was then divided by the average of 2 wells that contained positive control samples. If the quotient of this calculation was <10% of the positive control value, the sample was negative for TgAA. If the quotient was >25% of the positive control value, the sample was positive for TgAA. Results of 10–25% of the positive control value were categorized as inconclusive.
Statistical analysis
The data obtained from the questionnaires and tests results were entered into a database. f Statistical analysis was performed using a commercially available statistics package. g Descriptive statistics were used to describe distributions of test results and other variables, and exploratory data analysis was performed to identify and evaluate the influence of possible outliers. All reference values (beyond those rejected due to exclusion criteria) were retained because reference individuals were selected from well-defined populations, health was confirmed as reliably as possible, unintended preanalytical and analytical variation was minimized by using study-specific standard operating procedures, and there was no evidence to suggest that individual observations were aberrant. Differences between breeds were tested using 1-way analysis of variance (ANOVA) using the Tukey method for comparison of means. Multiple linear regression models were used to explore the associations between test results (T4, FT4, TSH) and breed, adjusting for potential effect modifiers such as sex, age, TgAA status, and size (height, weight, body mass index). As expected, TSH data were highly skewed; therefore, comparison of TSH concentrations among breeds was performed using the nonparametric Kruskal–Wallis 1-way ANOVA. For multiple linear regression modeling, natural log transformation of TSH data was performed prior to analysis. Breed was forced into all regression models, and a backward elimination approach was used after initially fitting a full model incorporating sex, neuter status, age, TgAA status, and measures of body size (height, weight, body mass index) as covariates.
Reference intervals encompassing the central 95% of reference values (2.5–97.5%) for all breeds combined, and for each of the 7 breeds, were calculated using nonparametric (TSH) and robust methods (T4 and FT4). Guidelines for determining de novo RIs, provided by the Clinical and Laboratory Standards Institute and the American Society for Veterinary Clinical Pathology, recommend that the robust method be used to generate RI when 40–120 reference samples are available. Confidence intervals (CIs; 90%) around the upper and lower reference limits were calculated using the bootstrap method.4,6 Log-transformation was used as needed on data displaying nonnormal distribution prior to CI calculation. All RI determinations were performed using a statistical package. h Two-sided RIs were derived for T4 and FT4, and a 1-sided (upper) interval was derived for TSH concentration.
Results
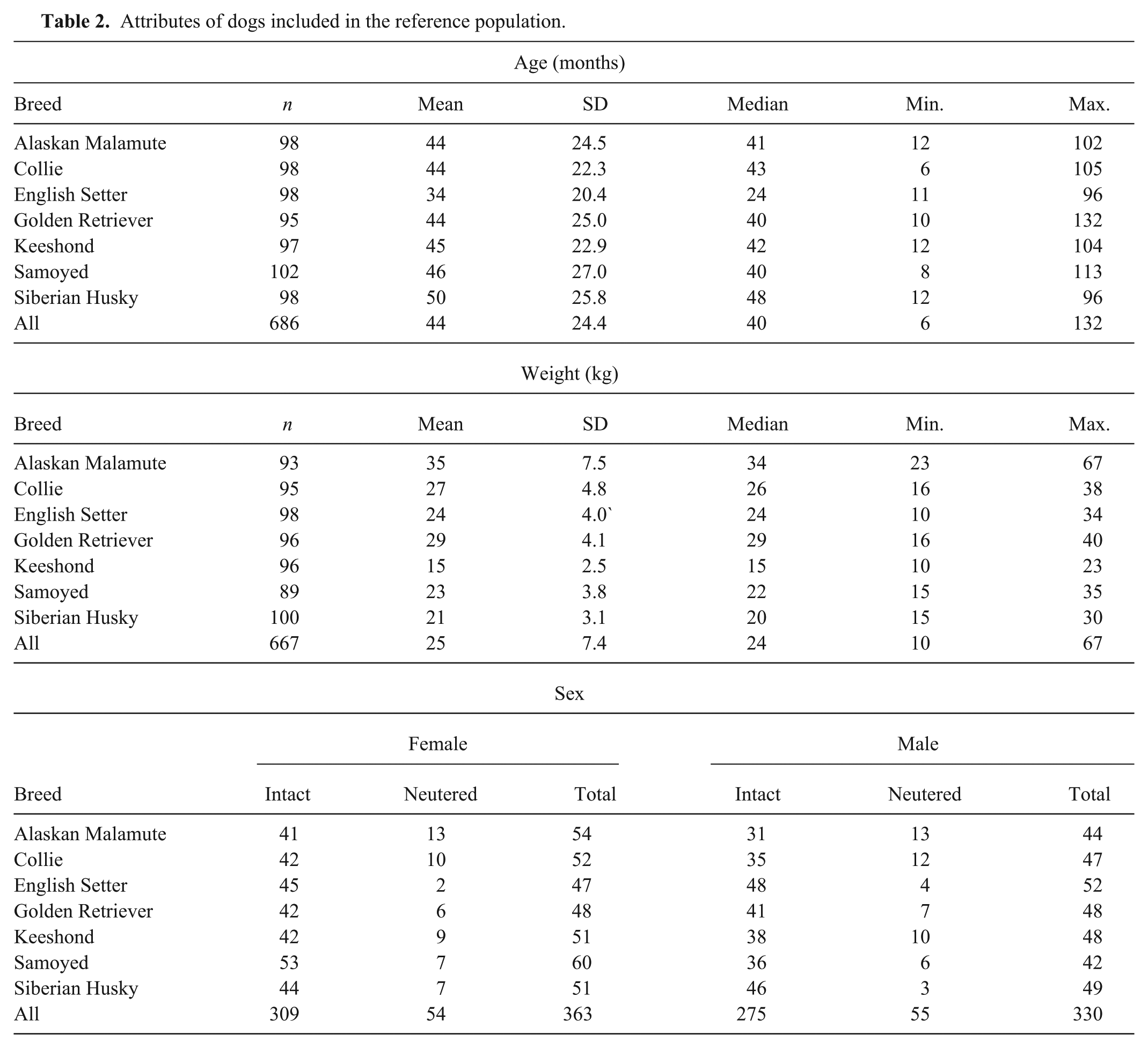
Of the original study population of 782 dogs, 89 (11%) were deemed to be ineligible for the reference population. The most frequent reasons for exclusion (Table 1) were urinary tract abnormalities and elevated cholesterol concentration. The age distributions (median 40 months for all dogs) were similar across breeds, apart from the English Setter population with a median age of 24 months. Six dogs (0.9%) between 6 months and 1 year of age, and 2 dogs >9 years of age (0.3%) were included in the study. Approximately half (48–59%) of the animals for each breed were female, and the majority (84%) were intact rather than neutered animals (Table 2). Partitioning based on neuter status was not performed. An estimated date of last estrus was provided by the owners (questionnaire entry) for 265 of the 309 (86%) intact females. Based on those entries, 76 intact females were estimated to be in diestrus when the date of blood collection was within 2 months of the last estrus date (on average, diestrus in the dog lasts for 60 days after estrus). The date of last estrus was unknown for 44 intact females. Most dogs (87%) were eating dry food, and the others were eating various combinations of dry, moist, and/or raw meat diets. Vaccination history was recorded for 92% of dogs, of which approximately one-fifth (21%) had been vaccinated within 1 and 3 months of sample collection. Ninety percent of the dogs in the study population did not have detectable serum thyroglobulin autoantibodies (Table 3). The English Setter breed had the highest number of animals that tested positive (28/99) or inconclusive (6/99) for the presence of serum TgAA. To evaluate the effect of the TgAA-positive English Setters, the mean, median, and range of the English Setter data were recalculated after exclusion of TgAA-positive English Setters. There was no significant change in the results. All other breeds had ≤5% positive TgAA results, and ≤6% inconclusive results. When tested by 1-way ANOVA, there was no difference in mean T4, FT4, or TSH concentrations (P = 0.94, P = 0.35, P = 0.64, respectively) in serum from animals testing positive, negative, or inconclusive for TgAA. Similarly, the mean T4 (P = 0.80), FT4 (P = 0.15), and TSH (P = 0.16) concentrations did not differ when all positive and inconclusive TgAA results (n = 69) were excluded from the analysis.
Reasons for exclusion of screened dogs by breed.*
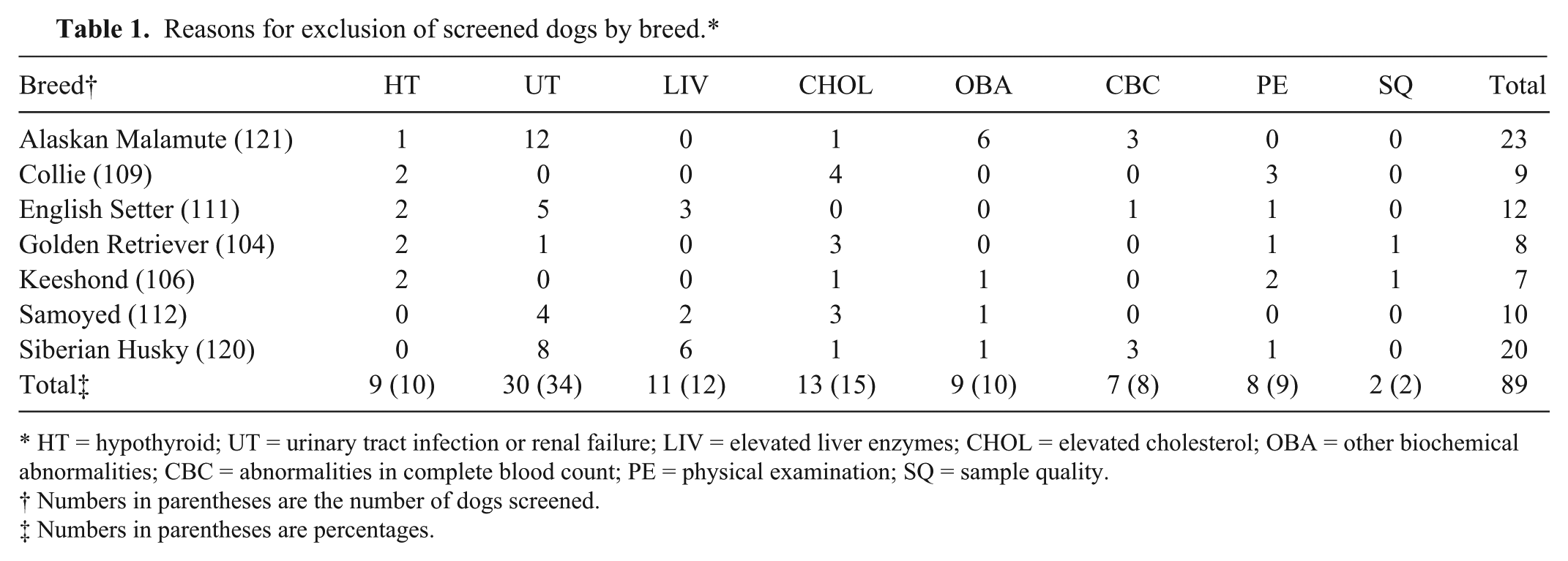
HT = hypothyroid; UT = urinary tract infection or renal failure; LIV = elevated liver enzymes; CHOL = elevated cholesterol; OBA = other biochemical abnormalities; CBC = abnormalities in complete blood count; PE = physical examination; SQ = sample quality.
Numbers in parentheses are the number of dogs screened.
Numbers in parentheses are percentages.
Attributes of dogs included in the reference population.
Thyroglobulin autoantibody results by breed.
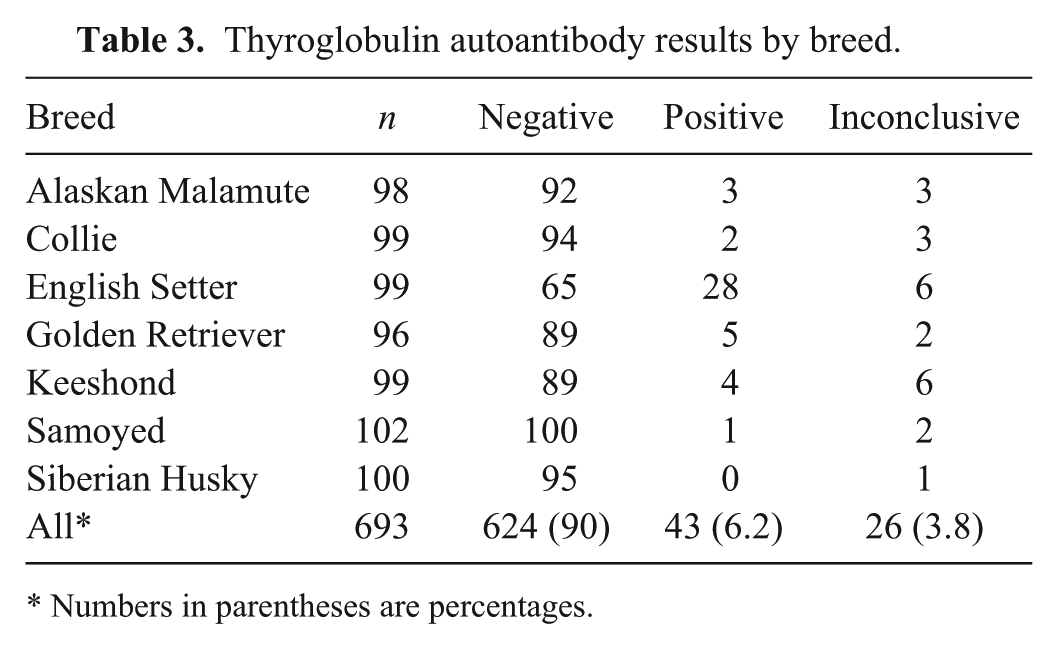
Numbers in parentheses are percentages.
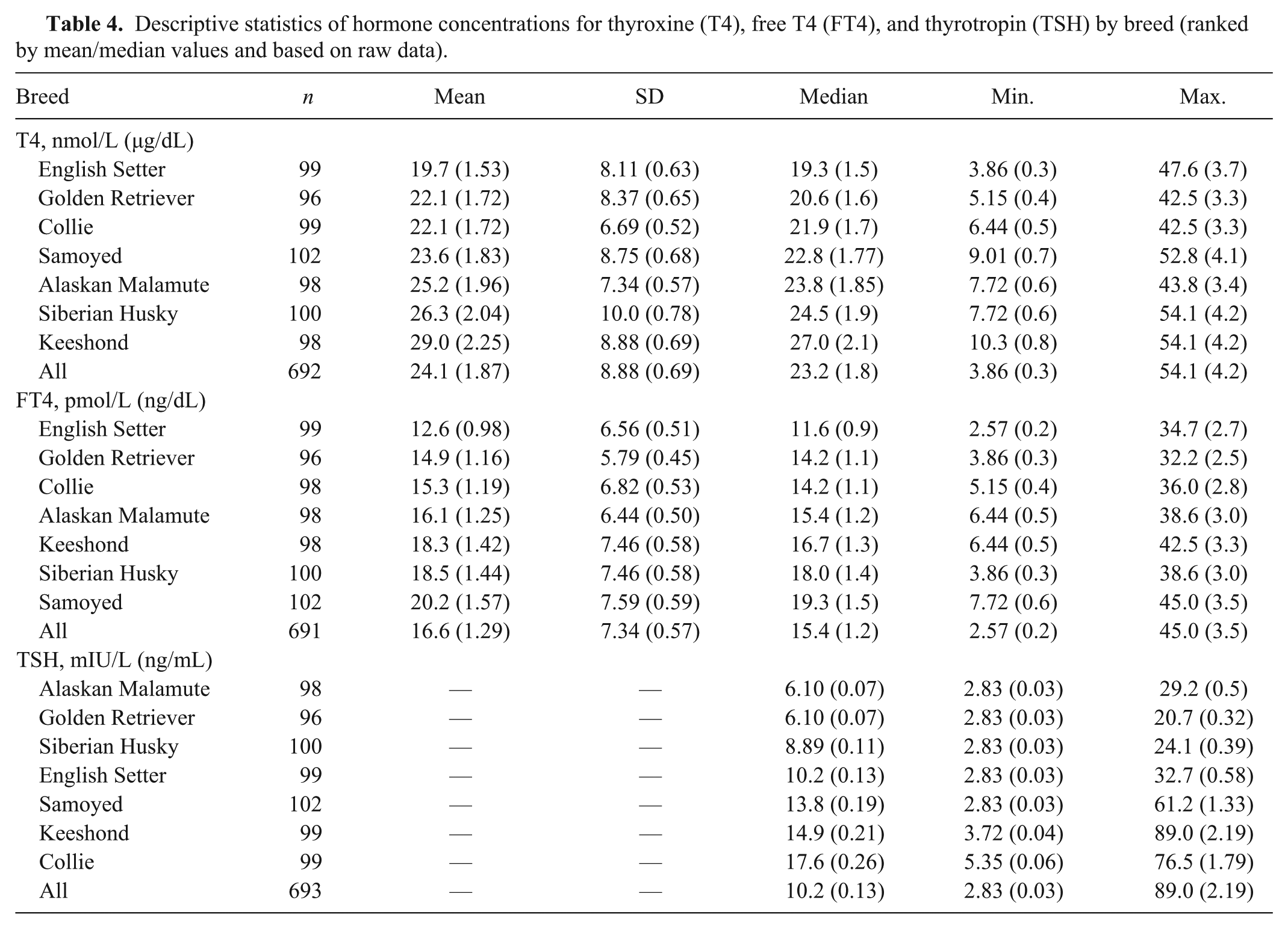
Mean serum concentrations of T4, FT4, and TSH varied significantly among breeds (P < 0.0001), and the magnitude of differences between breeds were substantial (Table 4). Mean concentrations for T4 ranged from 19.7 nmol/L (1.53 µg/dL) in English Setters to 29.0 nmol/L (2.25 µg/dL) in Keeshonds. Based on Tukey pairwise comparison of means, mean T4 concentration in the 3 breeds with the lowest values (English Setter, Golden Retriever, Collie) were significantly lower than in the 2 breeds with the highest values (Keeshond, Siberian Husky), and the breeds with intermediate values (Alaskan Malamute and Samoyed) differed significantly from the breeds with the highest and lowest values. Similarly for FT4, the range among means was considerable (12.6 pmol/L [0.98 ng/dL] in English Setters to 20.2 pmol/L [1.57 ng/dL] in Samoyeds); the breeds with the lowest mean values (English Setter, Golden Retriever, Collie) differed significantly from the 3 breeds (Samoyed, Siberian Husky, Keeshond) with the highest mean values; and the breed with intermediate mean values (Alaskan Malamute) differed significantly from both the breeds with the highest and lowest mean values. The prevalence of TSH >33.5 mIU/L (>0.6 ng/mL) was strongly associated (P < 0.001) with breed, being most common in the Collies, Samoyeds, and Keeshonds (Table 5). The TSH concentration exceeded 33.5 mIU/L (0.6 ng/mL) in 27 (3.9%) samples. Median TSH values by breed ranged from 6.10 mIU/L (0.07 ng/mL; Alaskan Malamute and Golden Retriever) to 17.6 mIU/L (0.26 ng/mL; Collie); the 3 breeds with the highest median TSH values (Collie, Samoyed, Keeshond) differed significantly from all others; breeds with intermediate median values (English Setter, Siberian Husky) differed significantly from the breed with the lowest median value (Golden Retriever), and TSH median value in English Setters was significantly higher than in Alaskan Malamutes. The breeds with the lowest mean T4 and FT4 (Golden Retriever and English Setter) concentration did not have any reference individuals with TSH concentrations >33.5 mIU/L (>0.60 ng/mL).
Descriptive statistics of hormone concentrations for thyroxine (T4), free T4 (FT4), and thyrotropin (TSH) by breed (ranked by mean/median values and based on raw data).
Number of dogs with serum thyrotropin (TSH) concentration >33.5 mIU/L (>0.6 ng/mL), thyroxine (T4) concentration <12.9 nmol/L (<1.0 µg/dL), and free T4 (FT4) concentration <10.9 pmol/L (<0.85 ng/dL) by breed.
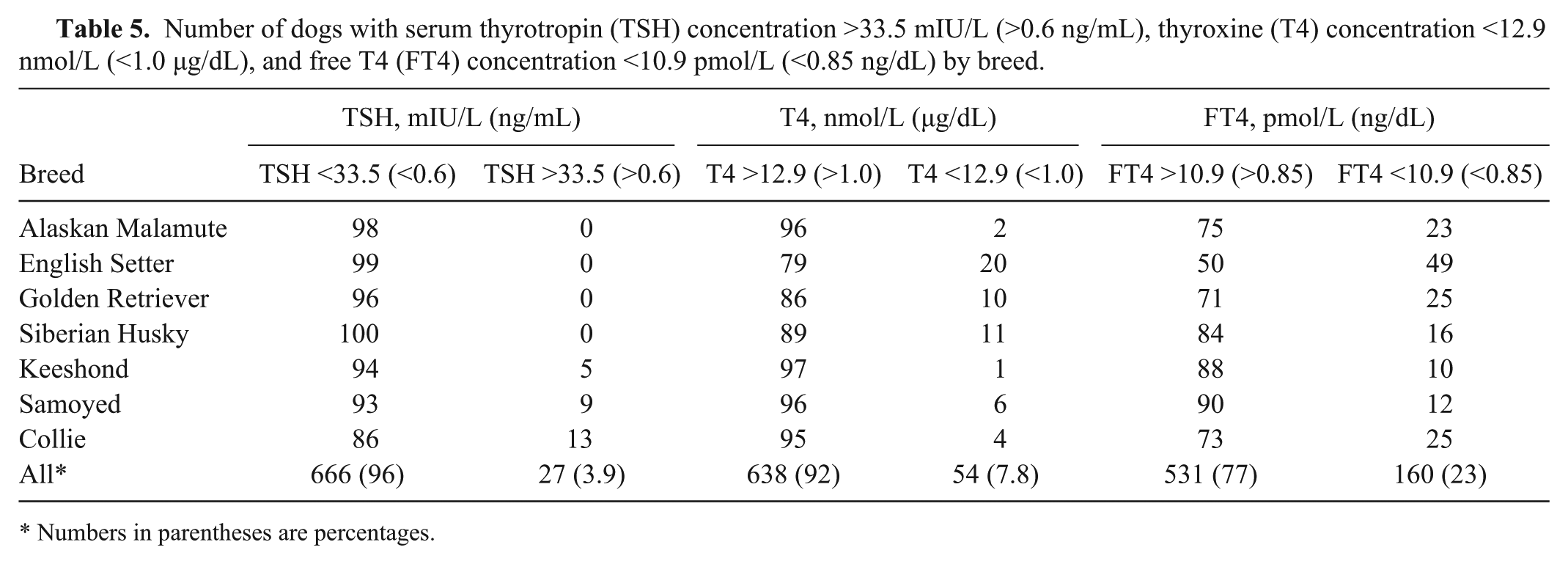
Numbers in parentheses are percentages.
Results of multiple linear regression analysis indicated breed accounted for ~10% of the variance in T4 and FT4, and 30% in TSH (Table 6). Potential effect modifiers or confounding factors that were significant in multivariate models were sex and age. Inclusion of sex in the thyroxine models accounted for a further 4% of variance for both T4 and FT4, but <1% of additional variance of TSH.
Multiple linear regression models including breed (English Setter is the comparison breed), sex (female), and age (months) as predictors of thyroid hormone concentrations.*
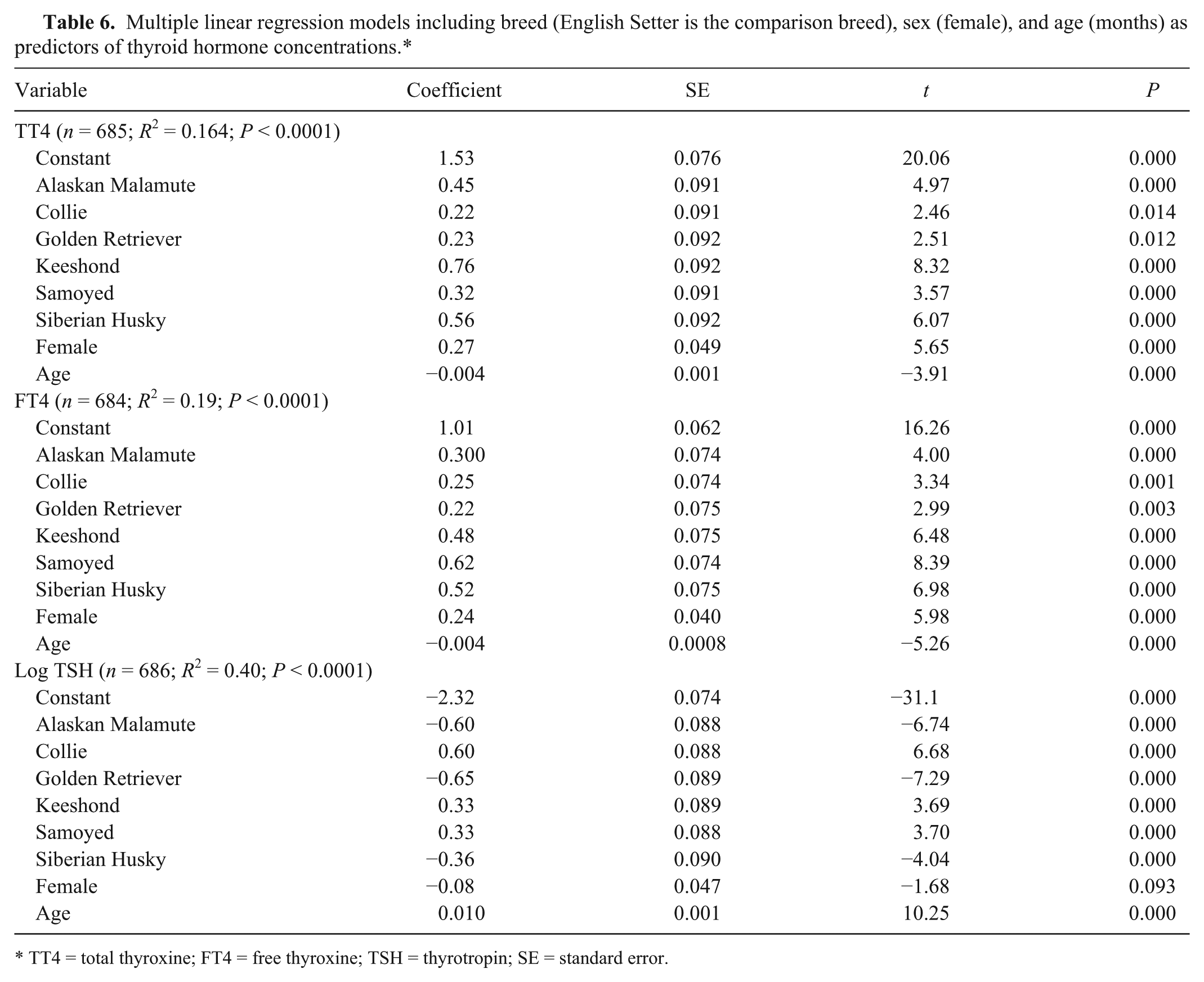
TT4 = total thyroxine; FT4 = free thyroxine; TSH = thyrotropin; SE = standard error.
Mean serum T4 concentration was higher in female dogs (3.47 nmol/L [0.27 µg/dL] higher) when compared with males. When the 76 females in diestrus were removed from the analysis, mean serum T4 concentration remained higher by 2.45 nmol/L (0.19 µg/dL) when compared with mean concentration in the males. Serum concentration of FT4 was 3.09 pmol/L (0.24 ng/dL) higher when females are compared with males and 2.96 pmol/L (0.23 ng/dL) higher when the females estimated to be in diestrus were removed from the analysis. The concentration of TSH did not differ between sexes.
Age was negatively associated with T4 and FT4, but accounted for only ~1% of additional variance. The models indicated that both T4 (0.052 nmol/L [0.004 µg/dL] per month) and FT4 (0.052 pmol/L [0.004 ng/dL] per month) concentration declined with age. In contrast, age was significantly positively correlated with TSH concentration, increasing by 0.75 mIU/L (0.01 ng/mL) each month. Measures of body size (height, weight, body mass index) differed significantly among breeds and between sexes, and were excluded from the final models because of potentially confounding effects.
Groups of purebred reference individuals representing the 7 breed populations ranged from 96 to 102 dogs per breed. The breed-specific RIs (with 90% CI) for T4, FT4, and TSH, derived using nonparametric (TSH) and robust (T4 and FTE) methods, are provided in Table 7. The RIs calculated for the Keeshond breed were most similar to the T4 and FT4 reference ranges previously described for 150 healthy non–breed-specific reference individuals. 21 For all other breeds, RIs were within (but generally lower than) the previously reported upper reference range limit but below the non–breed-specific lower reference range limit. The Collie, Samoyed, and Keeshond breeds had an upper reference limit for TSH concentration that was higher than the previously reported non–breed-specific TSH reference range. The percentage of dogs in each breed that had serum T4 and FT4 concentrations below (T4, FT4) or above (TSH) the previously reported reference range distributions are provided in Table 5.
Lower and upper limits of thyroxine (T4), free T4 (FT4), and thyrotropin (TSH) 95% reference intervals for 7 breeds of dogs, normality of distribution for native values, and statistical method of analysis.*
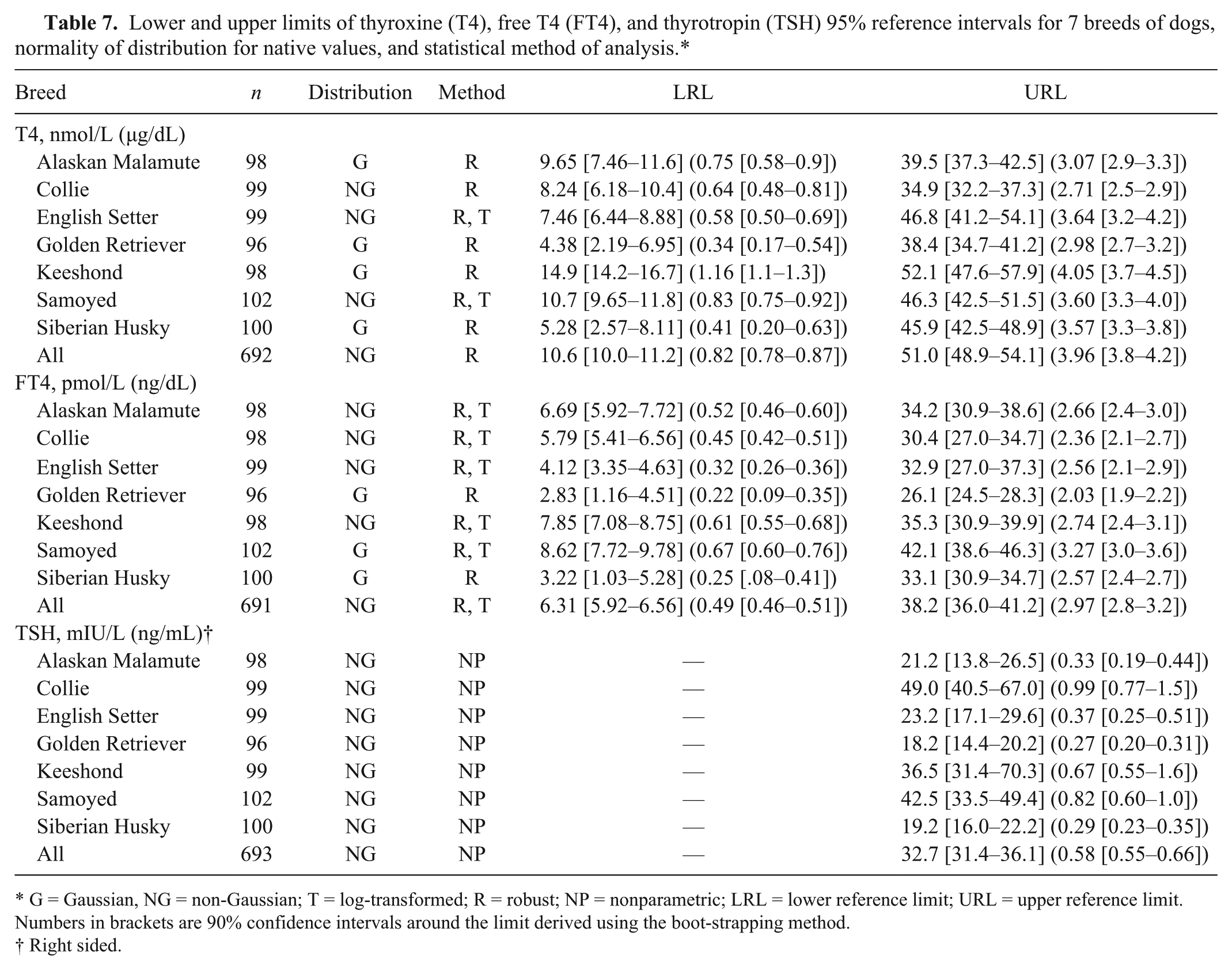
G = Gaussian, NG = non-Gaussian; T = log-transformed; R = robust; NP = nonparametric; LRL = lower reference limit; URL = upper reference limit. Numbers in brackets are 90% confidence intervals around the limit derived using the boot-strapping method.
Right sided.
Discussion
In this study, significant breed-associated variability in serum concentrations of T4, FT4, and TSH was discovered for dogs of 7 common breeds. Collectively, the ANOVA results led us to reject the null hypothesis that the breeds studied can be considered unbiased samples from a single homogeneous canine population. Furthermore, the substantial ranges across breeds of measures of central tendency (mean, median) for all 3 hormones indicate that breed-specific RIs are warranted.
The procedures associated with the selection, preparation, and examination of reference individuals, and the collection, processing, storage, and analysis of samples were performed according to standardized protocols in order to minimize pre-analytical and analytical error. Therefore, the contribution of these factors to the significant differences determined in this study is likely to be minimal.
The median serum T4 (23.2 nmol/L, 1.8 µg/dL, n = 692) and TSH (10.2 mIU/L, 0.13 ng/mL, n = 693) concentrations reported herein for all dogs are comparable with 2 previous reports for T4 (25.7 nmol/L [2.0 µg/dL], 20.6 nmol/L [1.6 µg/dL]) and TSH (13.8 mIU/L [0.19 ng/mL], 8.23 mIU/L [0.10 ng/mL]) from dogs of multiple breeds that were deemed healthy based on history, physical examination, and laboratory testing criteria (n = 150 and n = 62, respectively).21,29 However, the median FT4 concentration (15.4 pmol/L, 1.2 ng/dL) for all dogs is somewhat lower than the value of 21.9 pmol/L (1.7 ng/dL) previously determined in 150 healthy dogs of multiple breed origin. 21
Data reported herein for Golden Retrievers (Table 4) agree with a previous report where the mean ± standard deviation (SD) for hormone concentrations was determined in serum from 36 healthy Golden Retrievers. Serum concentrations of T4, FT4, and TSH were reported to be 25.7 ± 6.44 nmol/L (2.0 ± 0.5 µg/dL), 14.2 ± 5.15 pmol/L (1.1 ± 0.4 ng/dL), and 8.23 ± 8.23 mIU/L (0.1 ± 0.1 ng/mL), respectively, for those dogs. 3
Serum TSH concentration in healthy dogs is usually <33.5 mIU/L (<0.60 ng/mL). 21 However, values above that threshold have been reported, and TSH is secreted episodically, which contributes to inherent variability in measured concentration. 12 In the current study, elevated concentrations of TSH were not equally distributed among breeds. The Collies, Samoyeds, and Keeshonds had the highest percentages (13, 8.8, and 5.1%, respectively) of reference individuals with TSH concentration >33.5 mIU/L (>0.60 ng/mL). Reference individuals from Alaskan Malamute, English Setter, Golden Retriever, and Siberian Husky breeds did not have serum TSH concentrations >33.5 mIU/L (>0.60 ng/mL), suggesting an influence of breed, rather than secretion pattern (Table 5). In a previous study of healthy Whippet dogs, 6 of 51 Whippets had elevated TSH concentration (0–75.2 mIU/L, 0–1.75 ng/mL) compared with lower values (0–28.7 mIU/L, 0–0.49 ng/mL) determined in the control group of multiple breeds. Those differences were not significant. 35
It is unlikely that the higher TSH concentrations observed in some of the breeds (Collie, Samoyed, Keeshond) are the result of a negative feedback effect of low T4 or FT4 concentration because there was no association between a higher than expected TSH value and a lower than expected T4 or FT4 value. Rather, it is possible that the pituitary and/or thyroid set-points differ among individuals and breeds. A study of biological and analytical variation of canine serum TSH concentration in Beagles determined that between-dog variation (43.6%) was much higher than within-dog variation (13.6%) and that analytical variation was 8.8%. It was concluded that population-based RIs are too imprecise to detect small but critical changes in individual dog serum TSH concentration. 11 Some of the uncertainty associated with serum TSH values in the dog (normal concentrations in hypothyroid dogs and elevated values in normal dogs) may be the result of this variation around individual set-points that are not recognized when using population-based decision points. Subject-based RIs may be useful under these conditions, and recommendations for their use for canine analytes have been published.6,8,36
Reference intervals for hormone concentrations were previously reported using the nonparametric method to determine the 5th to 95th percentile range for results from 150 clinically normal animals representing multiple breeds. In that study, the RIs were determined to be 12.9–51.5 nmol/L (1–4 µg/dL) for T4 and 10.9–45.0 pmol/L (0.85–3.5 ng/dL) for FT4. 21 In the current study, the T4 (using the robust method) RI for all dogs is similar to the previous report, while the FT4 RI has lower reference limits (Table 7). The upper limit of the TSH one-sided nonparametric RI for all dogs (Table 7) agree with previous reports of TSH <33.5 mIU/L (<0.60 ng/mL, n = 150) and <35.7 mIU/L (<0.65 ng/mL, n = 62) in healthy dogs representing multiple breeds.21,29
Our study demonstrates that many healthy purebred dogs have serum T4 and FT4 concentrations that are at, or below, the previously established and conventional lower limit of non–breed-specific RIs (Table 5). These findings agree with previous reports where breed specific RIs for the Greyhound,1,7 Saluki, 33 Sloughi, 18 Whippet, 35 Scottish Deerhound, 31 and Basenji 30 have been shown to have lower RI limits than RIs developed from the general dog population. Therefore, we propose that the use of non–breed-specific RIs for some purebred dogs may not be appropriate. Furthermore, it is especially critical to consider the breed-specific variability at the lower limits of the T4 and FT4 RI because clinical decisions are routinely based on these values, rather than on the upper RI limit. In addition, the FT4 and TSH results that are generated for certification programs such as the Orthopedic Foundation for Animals (OFA) thyroid registry (http://www.offa.org/thy_proc.html) may misclassify healthy dogs as hypothyroid or equivocal for thyroid health when non–breed-specific RIs are used to define registry acceptance criteria. In fact, 23% of all of the healthy dogs in the current study had FT4 concentrations below the conventional lower RI limit for the general population of dogs.
The mechanisms for breed-specific differences in these hormones are not known. It is possible that thyroid efficiency and physiological set-points may differ among individuals and among breeds. Regulatory processes such as thyroid hormone receptor function and affinity, or sensitivity at the pituitary level, may contribute to breed differences. Thyroid hormone concentration is affected by thyroid hormone–binding proteins that may also vary by breed. However, in a study of Greyhounds and other breeds, changes in concentration or function of thyroid-binding globulin did not explain breed differences (Shiel E, et al. Qualitative and semi-quantitative assessment of thyroxine binding globulin in the greyhound and other dog breeds. Proc European Congress Vet Intern Med Conf, 2011, Seville, Spain). It is also possible that variable iodine intake through diet or use of supplements such as kelp could contribute to thyroid hormone variability. In the current study, hormone concentrations were not different when supplemented and non-supplemented dogs were compared, and degrees of supplementation were similar across the breed groups.
Serum concentrations of T4 and FT4 were significantly higher in females when compared with males. Previously, thyroid hormone concentrations were shown to be similar when measured in serum from sexually intact males and from females that were in anestrus, proestrus, or lactating. However, serum T4 concentrations were higher during diestrus when compared with other reproductive phases in the dog. 25 In the current study, when the 76 dogs estimated to be in diestrus were removed from the analysis, T4 and FT4 concentrations remained higher in female than in male dogs. The data agree with a report of healthy Saluki dogs wherein T4 and FT4 (but not TSH) concentrations were significantly lower in male dogs when compared with females. Most of the dogs in that study were also intact animals, and information regarding the estrous cycle phase for female participants was not collected. 33 The data suggest that intact females should be evaluated for thyroid function during anestrus. Alternatively, veterinarians should consider the date of last estrus when they are interpreting T4 and FT4 results to account for the potentially higher T4 and FT4 values present during diestrus.
Increasing age was associated with decreasing concentration of T4 and FT4, and increasing concentration of TSH in serum. A progressive decline in serum T4 and FT4 has been previously reported (and recently reviewed) in the dog. 26 Explanations for the effect of age on these hormones may include changes in the thyroid or pituitary responsiveness to feedback regulation or the presence of subclinical thyroiditis or other concurrent illness.
In spite of the careful dog selection, it remains possible that some of the reference individuals in our study were affected by subclinical lymphocytic thyroiditis. Six percent of all dogs in this study were positive for the presence of TgAA. This agrees with other studies that have reported similar percentages (0–5%) of TgAA in the serum of healthy dogs.5,14,16,20,23,33,35 The prevalence of TgAA-positive dogs was much higher (28%) in English Setters when compared with the other 6 breeds (0–5%). This finding agrees with a previous report demonstrating that the English Setter breed has the highest prevalence of thyroglobulin antibody (31% of 585 English Setters tested) out of 140,821 serum samples submitted to a commercial diagnostic laboratory for investigation of thyroid disease. 10 Similar results also appear in the online thyroid statistics maintained by the OFA, which ranks the English Setter breed first for having the highest percentage (26.4% out of a population of 984 English Setters) of dogs classified as having autoimmune thyroiditis (FT4 less than normal range, TSH greater than normal range, TgAA positive). In addition, the English Setter breed has the highest (19.2%) percent equivocal (inconclusive results) classification, and the lowest (54.1%) percentage of dogs classified as normal based on serum concentrations of FT4, TSH, and the absence of TgAA. (OFA thyroid statistics, http://www.offa.org/stats_thy.html, accessed 3/23/15). Lymphocytic thyroiditis is known to be highly heritable, and an influence of breed on age and progression of this disease has been demonstrated.16,17
Dogs that were positive or inconclusive for TgAA (but had normal T4, FT4, and TSH concentrations) were not excluded from the analyses reported in our study because mean T4, FT4, and TSH concentrations were not different from those determined for TgAA-negative dogs, and their removal did not significantly change the results. These results agree with a study of human subjects where it was determined that there was minimal influence of antithyroid antibodies on the TSH reference range. 2 Furthermore, the presence of TgAA in healthy dogs does not mean that hypothyroidism will develop with certainty. For instance, recent rabies vaccination (within 2 weeks of thyroid testing) can result in the temporary (and inconsequential) presence of TgAA in the serum of healthy dogs.27,28 In the current study, animals that were vaccinated within 3 months of enrollment were not more likely to be TgAA positive (P = 0.15) than animals that were vaccinated >3 months prior to participation. In another study, only 20% of 171 TgAA-positive dogs developed signs of hypothyroidism within a 1-year period. 9 The subsequent development of hypothyroidism in the healthy but TgAA-positive or TgAA-inconclusive dogs participating in the current study was not determined because follow-up monitoring of these dogs was not possible.
The presence of TgAA in the serum of 6% of the participants may influence the reliable measurement of T4 in serum because autoantibodies to T4 (T4AA) or T3 (T3AA) may also be present. These autoantibodies can interfere with the immunological measurement method and result in falsely elevated T4 concentration. 17 In our study, T4AA and T3AA were not measured. However, previously, it has been demonstrated that the presence of T4AA and T3AA did not result in falsely elevated thyroid hormone results when the chemiluminescence immunoassay b utilized in the current report was used. 22
Use of the breed-specific RIs reported herein for interpreting data from representatives of these 7 breeds will likely result in more dogs being classified as euthyroid, fewer dogs requiring additional testing and monitoring, and fewer animals being treated for hypothyroidism (especially when based on a single test result) in veterinary practice. On the other hand, the potential to identify and treat subclinical thyroiditis early in its progression may be reduced when comparatively low T4 or FT4, and elevated TSH concentrations (with or without a TgAA-positive result), are viewed as unremarkable. The results of the current study do not resolve this quandary.
It is becoming increasingly clear that T4 and FT4 concentrations at the low limit of RIs are common in many breeds and that the presence of TgAA in the serum of healthy dogs can be spurious, inconsequential, or a potential predictor of future thyroid dysfunction. What remains uncertain is whether these lower levels of thyroid hormone imply any risk for thyroid disease in the future. Longitudinal studies of healthy dogs are needed to determine the predictive value of thyroid indicators (TgAA, T4, FT4, TSH concentrations) for the subsequent development of hypothyroidism. Such studies are especially important for breeds such as the English Setter that have been shown to have a high prevalence of TgAA-positive results, a high prevalence of lymphocytic thyroiditis, and lower RI limits for serum concentrations of T4 and FT4.
In summary, there is breed-specific variability in serum T4 (total and free) and TSH concentrations in the 7 breeds studied, and the use of breed-specific RIs for these animals is warranted. However, laboratory values will always be subject to variability because of the potential for preanalytical, analytical, and postanalytical error. Therefore, the accurate assessment of canine thyroid health will continue to require the essential integration of laboratory data with the full context of the clinical presentation.
Footnotes
Acknowledgements
We thank Sharon Hardy and Anna Sower from the Veterinary Diagnostic Laboratory at the University of Minnesota College of Veterinary Medicine for their excellent technical assistance with this project. We thank the canine health committees of the purebred dog clubs for their considerable assistance with animal recruitment, and sponsorship of sample collection clinics. We thank the dog owners and their veterinarians for their generous participation in this study.
Authors’ contributions
RL Hegstad-Davies and SMF Torres contributed to conception and design of the study, and contributed to acquisition, analysis, and interpretation of data. CA Muñoz-Zanzi contributed to the design of the study, and analysis of data. LC Sharkey contributed to the design of the study and interpretation of data, and PR Davies contributed to analysis and interpretation of data. SC Gresch contributed to analysis of data. RL Hegstad-Davies drafted the manuscript. All authors have critically revised the manuscript; gave final approval; and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
AU400e, Olympus, Center Valley, PA.
b.
T4 kit, Immulite; Diagnostic Products Corp., Los Angeles, CA.
c.
Free T4 by equilibrium dialysis, Nichols Institute Diagnostic, San Clemente, CA.
d.
Canine TSH kit, Immulite; Diagnostic Products Corp., Los Angeles, CA.
e.
Canine TGAA kit, Oxford Laboratories Inc., Oxford, MI.
f.
Access 2010, Microsoft Corp., Redmond, WA.
g.
Statistix version 9.0, Analytical Software, Tallahassee, FL.
h.
MedCalc for Windows version 13.2.2, MedCalc Software, Ostend, Belgium.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the American Kennel Club Canine Health Foundation (Grant 372: Determination of breed-specific reference intervals for assessing thyroid function in several breeds), The Collie Foundation, and the Orthopedic Foundation for Animals.
