Abstract
The purpose of our study was to describe the progressive accumulation of the abnormal conformer of the prion protein (PrPCWD) and spongiform degeneration in a single section of brain stem in Rocky Mountain elk (Cervus elaphus nelsoni) with chronic wasting disease (CWD). A section of obex from 85 CWD-positive elk was scored using the presence and abundance of PrPCWD immunoreactivity and spongiform degeneration in 10 nuclear regions and the presence and abundance of PrPCWD in 10 axonal tracts, the subependymal area of the fourth ventricle, and the thin subpial astrocytic layer (glial limitans). Data was placed in a formula to generate an overall obex score. Data suggests that PrPCWD immunoreactivity and spongiform degeneration has a unique and relatively consistent pattern of progression throughout a section of obex. This scoring technique utilizing a single section of obex may prove useful in future work for estimating the presence and abundance of PrPCWD in peripheral tissues and the nervous system in elk with CWD.
Introduction
Chronic wasting disease (CWD), a transmissible spongiform encephalopathy, has been reported in captive and free-ranging cervids.1,21,25,26 It is generally accepted that aberrantly folded prion protein (PrP) is associated with CWD and that this abnormal conformation can be detected with immunohistochemistry. 22 The degree of PrPCWD immunoreactivity (IR) and spongiform degeneration in the brain is thought to correlate with the duration of disease. It is speculated that the abundance of detectable PrPCWD IR and spongiform degeneration spreads and increases in the brain with time and disease progression. However, this relationship has not been clearly established in Rocky Mountain elk (Cervus elaphus nelsoni) as suggested in free-ranging 21 and experimentally infected mule deer (Odocoileus hemionus) 2 and captive white-tailed deer (Odocoileus virginianus). 11 We hypothesize that PrPCWD spreads throughout the regions in the obex of the brain stem at a rate that may correspond with the movement of PrPCWD through the nervous system and peripheral tissues. Herein we report the description of a detailed scoring technique, using a single section of the brain stem at the level of the obex, in elk with naturally occurring CWD. This scoring technique may be useful for gaining insight into the spread of PrPCWD and the pathogenesis of CWD in Rocky Mountain elk.
Materials and methods
Postmortem samples of brain stem, retropharyngeal lymph node, and palatine tonsil were obtained from ~300 free-ranging and 15,000 captive adult Rocky Mountain elk and were examined for the presence of PrPCWD IR using immunohistochemistry. 22 The free-ranging elk originated from northern Colorado and the captive elk originated from multiple herds throughout the Rocky Mountain region of the United States and Canada. Nearly all of these elk were euthanized by personnel affiliated with various state or federal wildlife agencies or by owners of the elk ranches, but a few were found dead. The elk that were found dead were all captive and had been observed for clinical signs before they died. Clinical signs included behavior changes, agitation, mild to moderate aggression especially when cornered or run through a chute, weight loss, excessive salivation, grinding of the teeth, and emaciation. Sex and age were recorded for many of the elk. Ages of elk were estimated by counting cemental rings of the first incisor teeth. 12 If fresh tissues were available, the PRNP genotype was determined. 16
Brain stems, retropharyngeal lymph nodes, and palatine tonsils were fixed in 10% neutral buffered formalin for 1 week. A section of obex (brain stem at the level where the fourth ventricle converges into the central canal of the spinal cord and contains the dorsal motor nucleus of the vagus nerve), medial retropharyngeal lymph node, and palatine tonsil from each elk were trimmed, placed in plastic cassettes, and allowed to fix in fresh formalin for another 2–3 days prior to processing. Cassettes were processed overnight using conventional methods in an automated tissue processor as previously reported. 22
Sections of brain stem were cut at 5 µm, and tissue ribbons were mounted on positively charged glass slides a and dried in a 65°C oven overnight. Slides were deparaffinized, placed in 99% formic acid b for 5 min at room temperature, and then rinsed in running tap water for 5 min. Slides were then autoclaved for 20 min at 121°C in citrated buffer, c cooled, and transferred into Tris buffer. d Staining was performed with an automated immunostainer at 37°C using an alkaline phosphatase red kit with amplification. d Tissues sections were immunolabeled with an anti-prion antibody kit (clone 99/97.6.1 d ), which uses a mouse monoclonal antibody derived from ascites for use on the aforementioned automated slide staining system. d Specific antibody activity is ~8 µg/mL in a Tris-based buffer. Monoclonal antibody F99/97.6.1 binds an epitope at residues 220–225, near the carboxyl terminus of the processed prion protein. 16 This monoclonal antibody was incubated on slides for 32 min at 37°C. The slides were rinsed and amplifier A (rabbit anti-mouse IgG heavy and light chains) was applied and incubated for 8 min at 37°C while amplifier B was run using an empty dispenser. The slides were rinsed, and the universal biotinylated secondary antibody was applied and incubated for 8 min at 37°C. Next, the slides were washed, and the streptavidin–alkaline phosphatase conjugate was applied and incubated for 12 min at 37°C with mixing. Fast Red A d and naphthol d were applied and incubated for 8 min at 37°C. Next, Fast Red B d was applied to the slide and incubated for 8 min at 37°C. Slides were counterstained with hematoxylin for 4 min at 37°C. Positive and negative control slides containing lymphoid tissues and brain were stained with each run. When used following the immunohistochemistry procedures as described by National Veterinary Services Laboratories (NVSL), the commercially available anti-prion antibody (clone 99/97.6.1 d ) kit will detect the prion protein in brain and lymphoid tissues in elk and deer with CWD. Routine hematoxylin and eosin–stained sections were examined for spongiform degeneration in each case. 19
A polyclonal antibody specific to glial fibrillary acidic protein (GFAP), an astrocytic-specific marker, was used on sections of obex from 2 elk from each score category. The GFAP stain was performed using a commercial kit d with no pretreatment. This polyclonal antibody was incubated on slides for 32 min at 37°C on an automated immunostainer with a basic alkaline phosphatase red kit. d
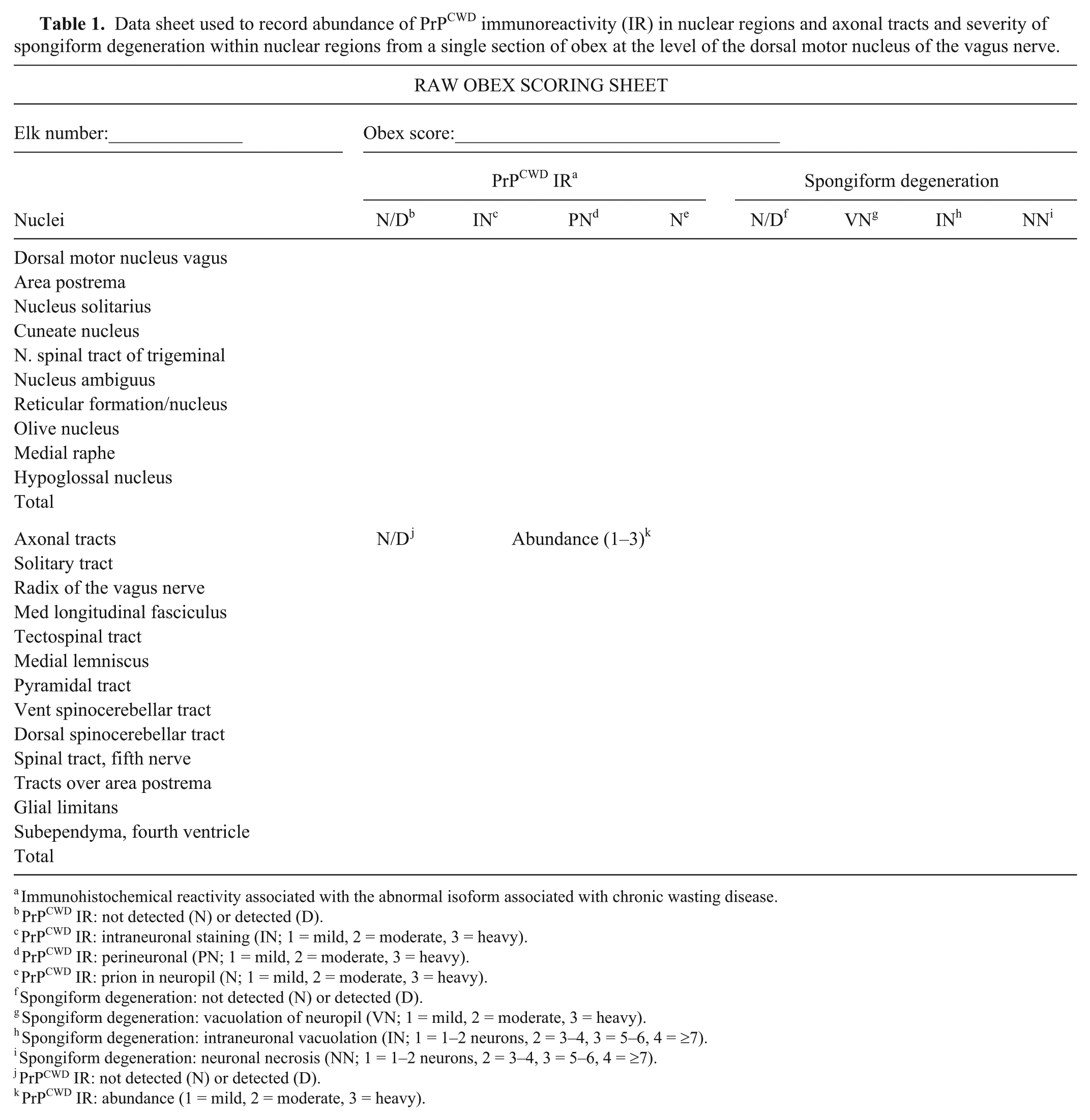
A single section of obex from each CWD-positive elk was scored (raw score) by evaluation of the presence and abundance of PrPCWD IR and spongiform degeneration in 10 nuclear regions and for the presence and abundance of PrPCWD IR in 10 axonal tracts, the subependymal area of the fourth ventricle, and the thin subpial astrocytic layer (glial limitans). PrPCWD IR was characterized by coarse granular red chromogen staining throughout these regions. This evaluation was done as follows: on low power (2×), and 10 nuclear areas of the obex including the dorsal motor nucleus of the vagus nerve (V), nucleus solitarius (S), area postrema (P), cuneate nucleus (C), nucleus of the spinal tract of the trigeminal nerve (T), nucleus ambiguus (A), reticular formation and nucleus (R), olive nucleus (O), medial raphe (MR), and the hypoglossal nucleus (H) were examined for presence of PrPCWD IR. 27 Next, the abundance of PrPCWD IR within each nucleus at 3 locations (intraneuronal, perineuronal, and within the neuropil) was evaluated subjectively and given a numeric score: 0 = not detected, 0.5 = trace, 1 = mild, 2 = moderate, and 3 = heavy (Figs. 1, 2). Lesions were bilaterally systemical, both sides were examined, and the average was recorded. Beaded linear and fine granular patterns, plaque formations of IR, and bright red granular material within Virchow–Robinson spaces were noted, but were not included in the scoring. Detectable PrPCWD IR also was evaluated in the axonal tracts: solitary tract (ST), radix of the vagus nerve (RV), axonal tracts associated with the area postrema (TP), medial longitudinal fasciculus (MF), tectospinal tracts (TS), medial lemniscus (ML), pyramidal tracts (PT), ventral spinocerebellar tracts (VS), dorsal spinocerebellar tracts (DS), and spinal tract of the trigeminal nerve (SF). Detectable PrPCWD IR in the glia limitans (GL) and in the loose tissue of the subependymal layer (SE) of the fourth ventricle was recorded with the axonal tracts. The abundance of PrPCWD IR within each of the axonal tracts and the GL and SE was scored subjectively and given a numeric score: 0 = not detected; 0.5 = trace; 1 = mild; 2 = moderate; and 3 = heavy (Figs. 3, 4).
Dorsal motor nucleus of the vagus nerve; Rocky Mountain elk (Cervus elaphus nelsoni; 132MM genotype), obex score = 1. The grading for the perineuronal PrPCWD immunoreactivity (IR) would be a 1 because only a few neurons are affected (thick arrow). The grade for the PrPCWD IR within the neuropil also would be a 1 (arrowhead). Note the beaded linear pattern of PrPCWD IR located within the neuropil that appears to be associated with an axon (thin arrow). Note the absence of intracytoplasmic PrPCWD IR within the neurons. Immunohistochemical staining: streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain.
Dorsal motor nucleus of the vagus nerve; Rocky Mountain elk (Cervus elaphus nelsoni; 132MM genotype), obex score = 9. Note the extensive amount of PrPCWD immunoreactivity (IR) located on the outer surface of neurons—numerical score of 3 (arrow). Note the extensive amount of PrPCWD IR within the neuropil—numerical score of 3 (arrowhead). Note the absence of intracytoplasmic PrPCWD IR within the neurons. Immunohistochemical staining: streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain.
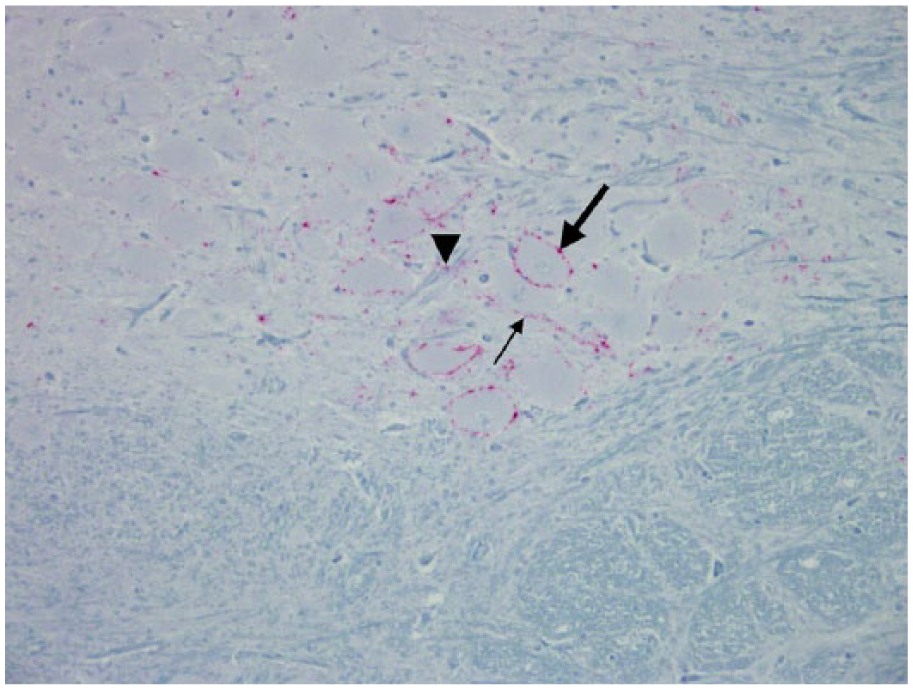
Axonal tract of the radix of the vagus nucleus; Rocky Mountain elk (Cervus elaphus nelsoni; 132MM genotype), obex score = 4. Note the minimal degree of PrPCWD immunoreactivity (IR) located on the outer surface of axons (arrows). This would be a PrPCWD IR abundance score of 1. Immunohistochemical staining: streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain.
Axonal tract of the radix of the vagus nucleus; Rocky Mountain elk (Cervus elaphus nelsoni; 132MM genotype), obex score = 8. Note the abundance of PrPCWD immunoreactivity (IR) located on the outer surface of axons (thin arrow). This would be an abundance score of 3. Also notice the extensive accumulation of PrPCWD IR around the axonal tract (thick arrow). Immunohistochemical staining: streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain.
Routine hematoxylin and eosin–stained sections were examined for spongiform degeneration in each case. The degree of spongiform degeneration including the degree of vacuolation within the neuropil, intraneuronal vacuolation, and neuronal necrosis was evaluated and recorded for each nucleus. Criteria used for scoring spongiform degeneration were the following: vacuolation of neuropil: 0 = not detected; 1 = <15 vacuoles; 2 = >15 but <35 vacuoles; and 3 = >35. Intraneuronal vacuolation was scored by counting the number of neurons containing vacuoles: 1 = 1–2 neurons; 2 = 3–4 neurons; 3 = 5–6 neurons; and 4 = ≥7 neurons. Neuronal necrosis was scored as follows: 1 = 1–2 necrotic neurons; 2 = 3–4; 3 = 5–6; and 4 = ≥7 (Figs. 5, 6). Criteria used for evaluating PrPCWD IR and spongiform degeneration in our study was similar to that found in the literature describing immunohistochemical labeling and spongiform degeneration for profiling lesions in domestic sheep with naturally occurring and experimental scrapie and bovine spongiform encephalopathy (BSE).3,4,7–9,13–15,24 Raw scores were recorded on a data sheet (Table 1). Following evaluation of these regions, the values were placed in a formula: A + B + C + D + E + F = raw obex score.
A = total number of nuclei with detectable PrPCWD IR.
B = average abundance of PrPCWD IR in nuclei. Add the 3 (intraneuronal, perineuronal, and neuropil) numerical scores for all 10 nuclear locations then divide by 10.
C = total number of axonal tracts with detectable PrPCWD IR including the GL and the SE.
D = average abundance of PrPCWD IR in the 10 axonal tracts including the GL and the SE. Add these abundance scores for each of the 12 locations and divide by 10.
E = total number of nuclei with spongiform degeneration.
F = average severity of spongiform degeneration. Add the numerical score for the 3 locations of spongiform degeneration for all 10 nuclear areas then divide by 10.
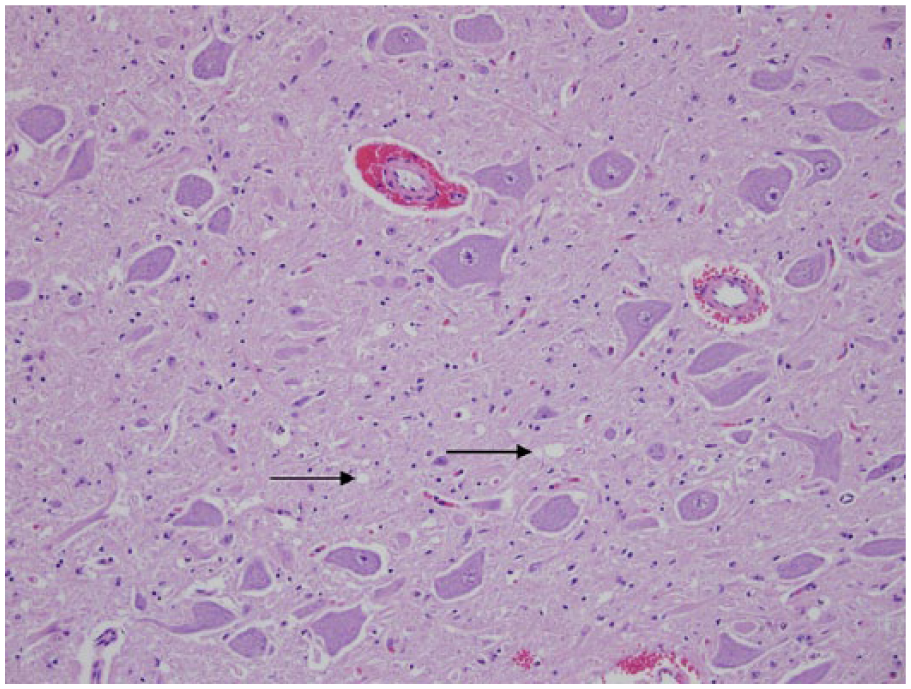
Dorsal motor nucleus of the vagus nerve; Rocky Mountain elk (Cervus elaphus nelsoni; 132MM genotype), obex score = 4. The degree of spongiform degeneration within the neuropil was mild (arrows). The neuropil vacuolation has a numeric score of 1; vacuolation of neurons was 0; and neuronal necrosis was 0. Hematoxylin and eosin.
Dorsal motor nucleus of vagus nerve; Rocky Mountain elk (Cervus elaphus nelsoni; 132MM genotype), obex score = 6. The degree of spongiform degeneration within the neuropil was considered to be moderate, with a numeric score of 2 (arrows); vacuolation of neurons was a numeric value of 1 (arrowhead); and neuronal necrosis was 0. Hematoxylin and eosin.
Data sheet used to record abundance of PrPCWD immunoreactivity (IR) in nuclear regions and axonal tracts and severity of spongiform degeneration within nuclear regions from a single section of obex at the level of the dorsal motor nucleus of the vagus nerve.
Immunohistochemical reactivity associated with the abnormal isoform associated with chronic wasting disease.
PrPCWD IR: not detected (N) or detected (D).
PrPCWD IR: intraneuronal staining (IN; 1 = mild, 2 = moderate, 3 = heavy).
PrPCWD IR: perineuronal (PN; 1 = mild, 2 = moderate, 3 = heavy).
PrPCWD IR: prion in neuropil (N; 1 = mild, 2 = moderate, 3 = heavy).
Spongiform degeneration: not detected (N) or detected (D).
Spongiform degeneration: vacuolation of neuropil (VN; 1 = mild, 2 = moderate, 3 = heavy).
Spongiform degeneration: intraneuronal vacuolation (IN; 1 = 1–2 neurons, 2 = 3–4, 3 = 5–6, 4 = ≥7).
Spongiform degeneration: neuronal necrosis (NN; 1 = 1–2 neurons, 2 = 3–4, 3 = 5–6, 4 = ≥7).
PrPCWD IR: not detected (N) or detected (D).
PrPCWD IR: abundance (1 = mild, 2 = moderate, 3 = heavy).
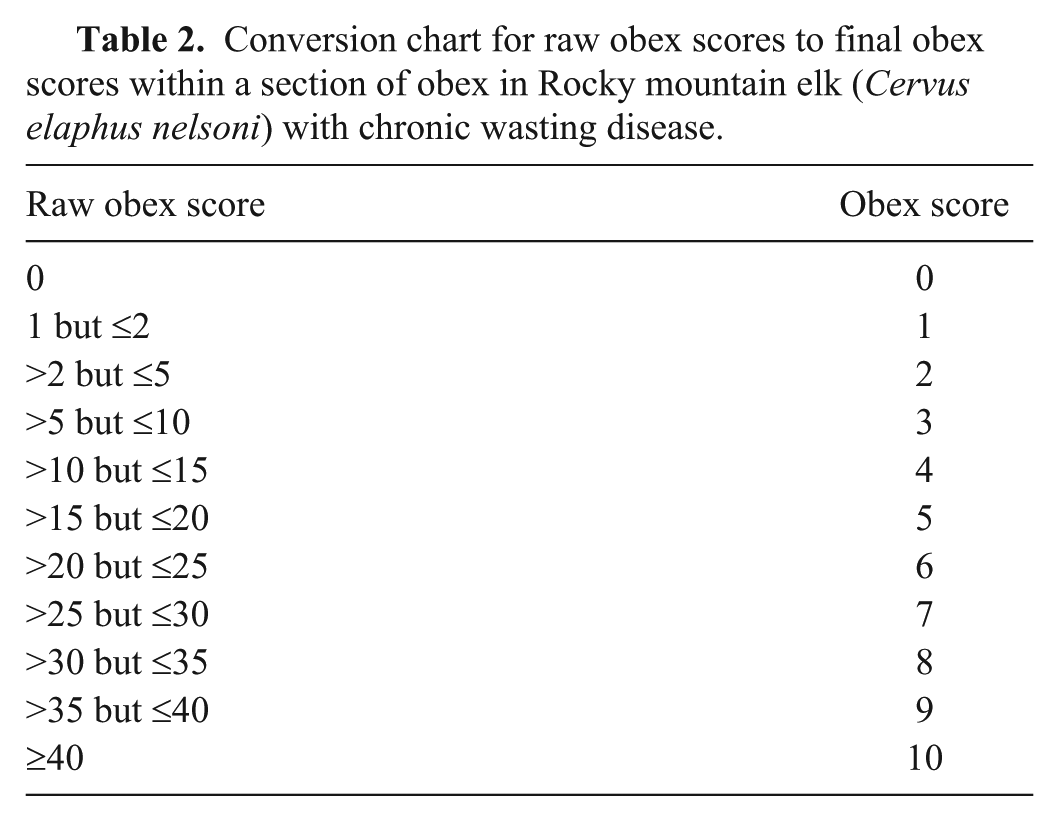
These 6 numbers are now added together (A + B + C + D+ E + F = raw obex score; e.g., 10 + {30/10} + 12 + {40/10} + 10 + {30/10} = 42, which indicates an elk in the terminal stages of CWD) to obtain the raw obex score. To simplify the scoring system, raw scores were grouped into 1 of 11 categories (Table 2).
Conversion chart for raw obex scores to final obex scores within a section of obex in Rocky mountain elk (Cervus elaphus nelsoni) with chronic wasting disease.
Results
Of the elk that were examined for the presence of PrPCWD IR, 323 were found to be positive (26 free-ranging and 297 captive). However, not all of the samples were suitable for use in our study. Suitable samples were those in which the section of obex contained all of the nuclear and axonal regions, and had minimal autolysis. Fifty-nine captive and 26 free-ranging elk were found to have samples meeting these requirements. Out of the 85 CWD-positive elk, 4 had PrPCWD IR in lymphoid tissues, but not in the obex. These elk were included in our study and were given an obex score of 0. Two captive elk had PrPCWD IR in the dorsal motor nucleus of the vagus nerve of the obex, but not in sections of retropharyngeal lymph nodes and palatine tonsil and were included in the study. The elk that did not have detectable PrPCWD IR in any of the examined tissues were considered to be free of CWD. Even though PrPCWD IR was not detected in any of these tissues from the “negative elk,” it does not totally rule out the presence of the CWD agent within these animals, it only indicates that the PrPCWD prion was undetectable with our immunohistochemical technique within these specific tissues.
If fresh tissues were available, the PRNP genotype was determined. 16 The 2 most common PRNP genotypes (132MM and 132ML) that have been reported in elk were found in our study.16,18 Genotype was determined in 49 of the elk (42 out of 49 (86%) 132MM; 6 out of 49 (12%) 132ML; and 1 (2%) 132LL; Table 3).
Summary of obex scores in the 3 different PRNP genotypes from 26 free-ranging and 59 ranch-raised Rocky Mountain elk (Cervus elaphus nelsoni) with chronic wasting disease.
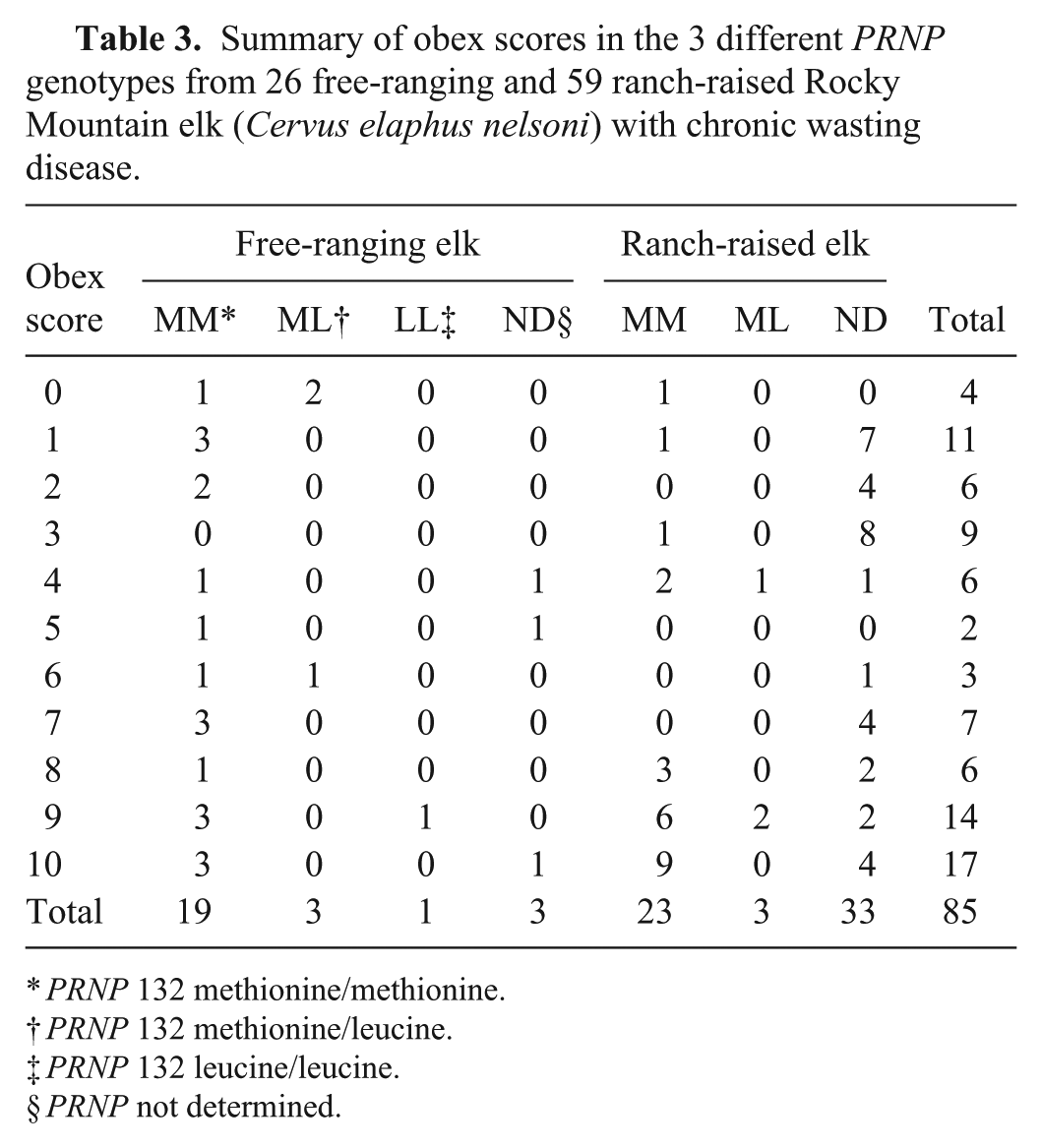
PRNP 132 methionine/methionine.
PRNP 132 methionine/leucine.
PRNP 132 leucine/leucine.
PRNP not determined.
The sex was recorded in 57 of the positive elk and included 39 (69%) females and 18 (32%) males. Sixty-four of the 85 (75%) positive elk were non-clinical and presented no observable signs; whereas 21 (25%) showed typical clinical signs of CWD at the time of euthanasia or were observed with clinical signs prior to death. The ages were estimated in 43 elk and ranged from 1.5 to 16 years. Thirty-one of these 43 (70%) elk were ≤5 years of age.
Clinical signs were not observed in any of the elk with an obex score of ≤8, and all of these elk were euthanized. Of the 14 elk with an obex score of 9, 7 showed clinical signs and, of these 14 elk, all were euthanized except for 2. Of the 17 elk with an obex score of 10, clinical signs were reported in 11. Of these elk, 10 were euthanized and 7 were found dead.
Description of obex scores
Eleven categories (0–10) or obex scores were formulated based on grouping of raw scores in elk that were positive for CWD. The following is a brief description of the highlights of the PrPCWD IR and spongiform degeneration in these categories. The pattern of detectable PrPCWD IR in the nuclei and axonal tracts and the severity of spongiform degeneration were progressive and similar in all 3 PRNP genotypes associated with the particular obex score (Table 4).
Summary of number of elk, presence or absences of clinical signs observed, locations of PrPCWD immunoreactivity and spongiform degeneration in specific nuclei, and presence of PrPCWD immunoreactivity in axonal tracts in the obex of 85 Rocky Mountain elk (Cervus elaphus nelsoni) with naturally occurring chronic wasting disease.*
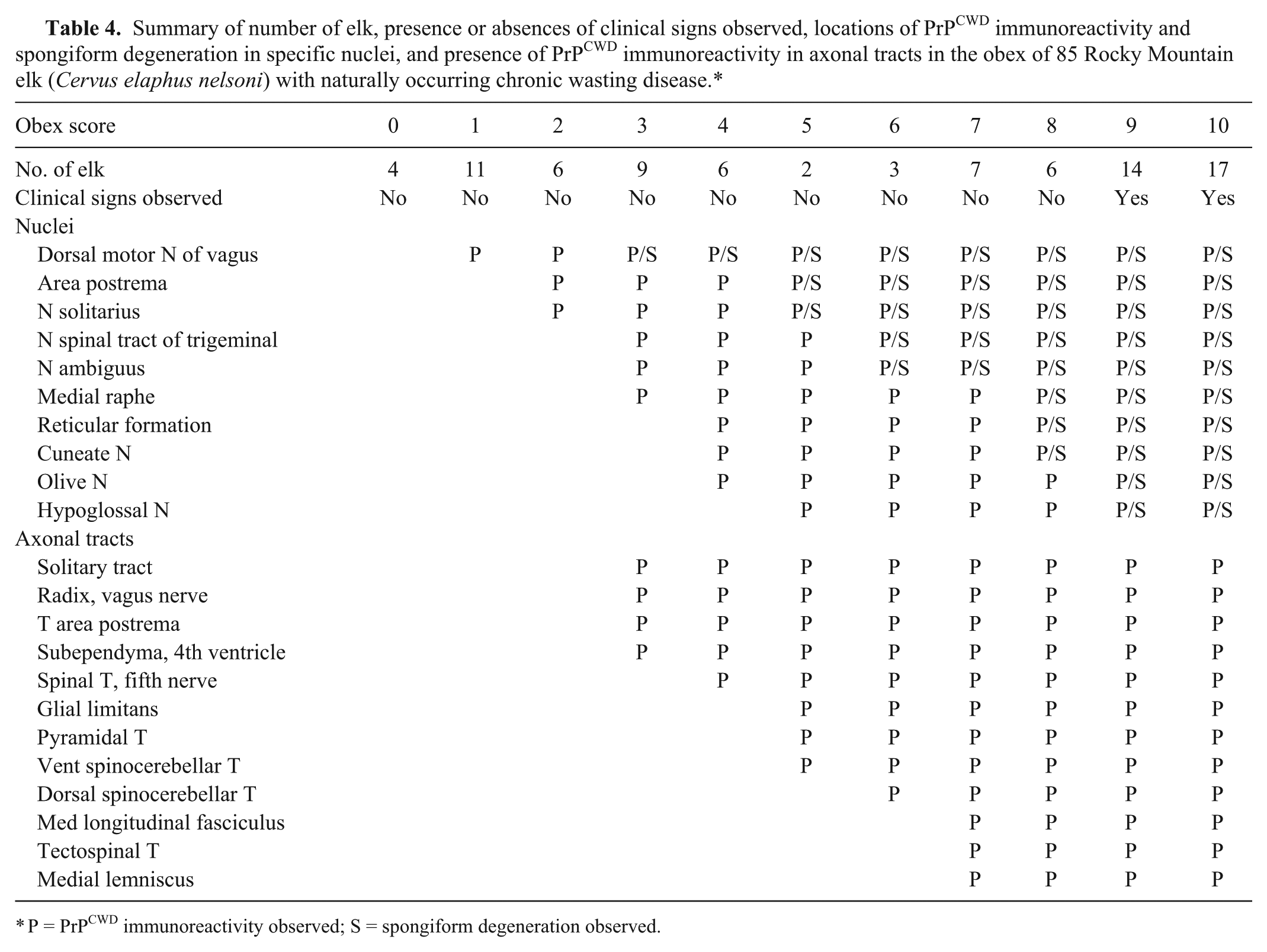
P = PrPCWD immunoreactivity observed; S = spongiform degeneration observed.
Obex score = 0 (raw score 0)
Four elk had PrPCWD IR in the medial retropharyngeal lymph node, but PrPCWD IR was not detected in the palatine tonsil or 5 different levels of the V.
Obex score = 1
Eleven elk had a raw obex score that varied from 1.05 to 1.2. Trace amounts of PrPCWD IR were detected in the lower aspects of the posterior half of the V. Minimal amount of PrPCWD IR was found around the periphery of several neurons and/or within the surrounding neuropil. In 1 elk, PrPCWD IR was exclusively found around several neurons; in 2 elk, PrPCWD IR was only found within the neuropil, and, in 8 elk, PrPCWD IR was found in both locations. PrPCWD IR within the neuropil and on the outer surface of the membranes of neurons was characterized by a coarse red granular material. PrPCWD IR was not found in any of the other nuclei or in axonal tracts. Spongiform degeneration was not present in any of the nuclei.
Obex score = 2
Six elk had a raw obex score that varied from 2.3 to 4.4. Minimal amounts of PrPCWD IR were detected in the V and P in all 6 elk. In 3 elk, PrPCWD IR also was present in the S.
Obex score = 3
Nine elk had a raw obex score that varied from 6.0 to 10.0. PrPCWD IR was detected in the V, P, and S in all 9; A in 4; T in 3; and MR in 2. Plaques of PrPCWD IR were found in the V and S. PrPCWD IR was present in the ST in all 9; RV in 5; and TP in 3. One of these elk also had a trace amount of PrPCWD IR in the SE. A minimal degree of spongiform degeneration was present in V in 3 and in the S in 1 elk.
Obex score = 4
Six elk had a raw obex score that varied from 10.3 to 14.2. PrPCWD IR was detected in the V, P, S, and T in 6; A and MR in 5; C in 3; and O and R in 1 elk. Plaques of PrPCWD IR were found in most of the nuclei. PrPCWD IR was present in the ST in 5; in the TP in 3; RV in 3; and in the SF and SE in 1 elk. A minimal degree of spongiform degeneration was present in the V in 4 of the elk.
Obex score = 5
Two elk had a raw obex score that was 19.15 and 19.7, respectively. PrPCWD IR was detected in the V, P, S, T, A, MR, and H in the elk with the raw score of 19.15. PrPCWD IR also was detected in the C and MR in the elk with the raw score of 19.7. Plaques of PrPCWD IR were found in most nuclei. PrPCWD IR staining was present in the ST, RV, PT, VS, SF, TP, GL, and SE in both elk. A minimal degree of spongiform degeneration was present in the neuropil of the V in the elk with a raw score of 19.15, and a moderate degree of spongiform degeneration was present in the V with minimal spongiform degeneration in the P and S in the elk with a raw score of 19.7. This latter elk did show clinical signs consistent with pneumonia and, at postmortem examination, a severe suppurative bronchopneumonia was found.
Obex score = 6
Three elk had a raw obex score that varied from 23.5 to 24.9. PrPCWD IR was detected in all of the nuclei except the O in 2 elk and the H in 1 elk. Plaques of PrPCWD IR were found in most of the nuclei, and PrPCWD IR was present in all axonal tracts except the MF, TS, and ML. A moderate degree of spongiform degeneration was present in the V with mild spongiform degeneration in the P, S, and T.
Obex score = 7
Seven elk had a raw obex score that varied from 25.6 to 28.8. PrPCWD IR was detected in all of the nuclear areas except for the O in 1 elk. One elk (PRNP genotype not determined) had a few neurons within the C in which intraneuronal PrPCWD IR was detected (Fig. 7). Up to this point, intraneuronal PrPCWD IR was not detected. PrPCWD IR was present in all of the axonal tracts. The primary difference between an obex score of 6 and 7 was the slight increase in the overall abundance of PrPCWD IR, and presence of intraneuronal PrPCWD IR. However, the degree of spongiform degeneration was similar.
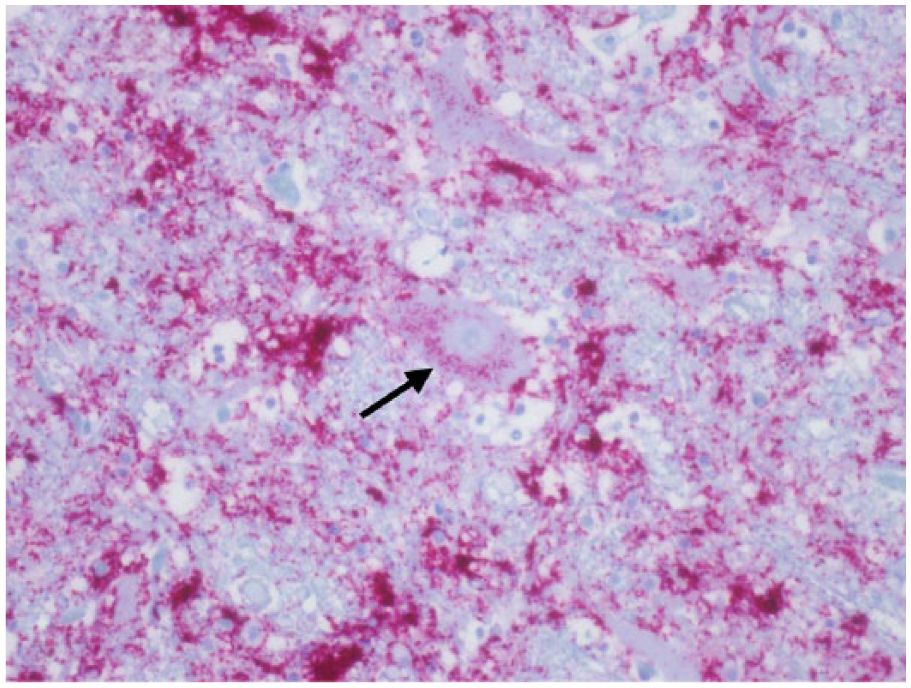
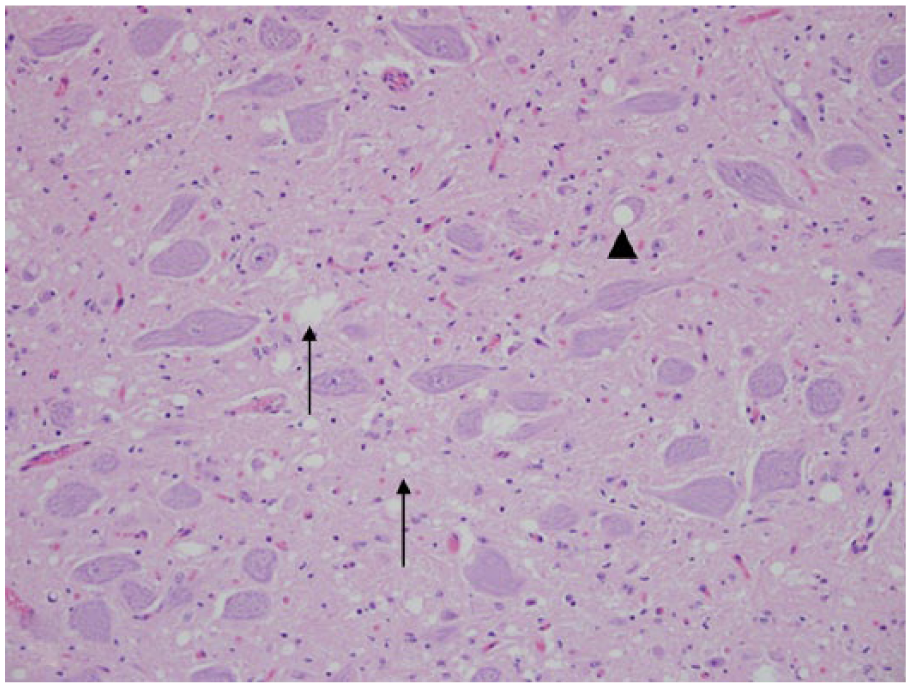
Cuneate nucleus; Rocky Mountain elk (Cervus elaphus nelsoni; 132MM genotype), obex score = 9. Note the intracytoplasmic PrPCWD immunoreactivity within a neuron (arrow), numeric score of 2. Immunohistochemical staining: streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain.
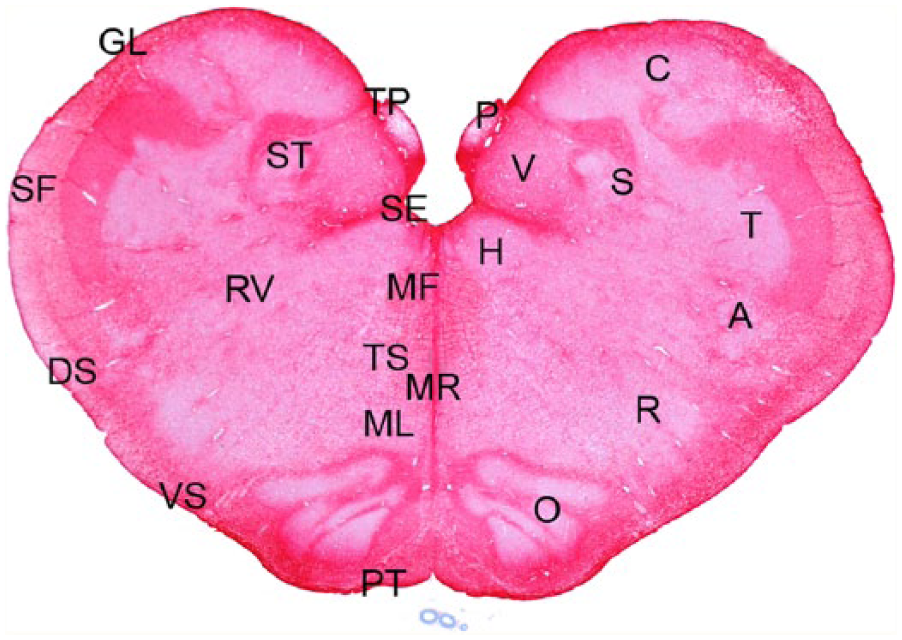
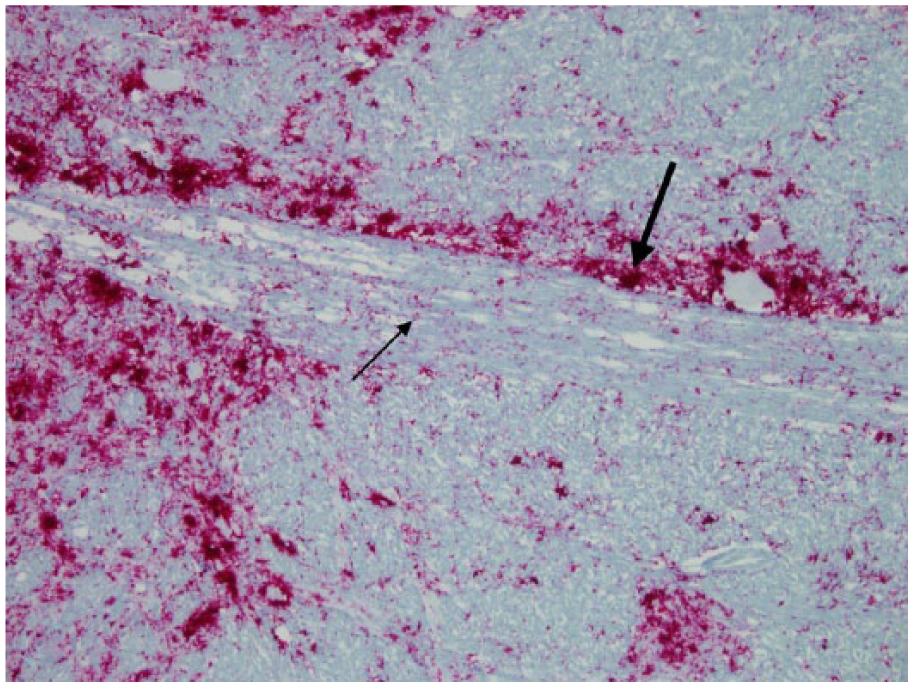
Obex; Rocky Mountain elk (Cervus elaphus nelsoni; 132MM genotype), obex score = 8. This stain demonstrated no difference in the abundance of astrocytes between control elk and elk with different obex scores. This stain also delineated the areas evaluated for scoring. Nuclei (right side): dorsal motor nucleus of the vagus nerve (V), nucleus solitarius (S), area postrema (P), cuneate nucleus (C), nucleus of the spinal tract of the trigeminal nerve (T), nucleus ambiguus (A), reticular formation/nucleus (R), olive nucleus (O, medial raphe (MR), and hypoglossal nucleus (H). Axonal tracts (left side): solitary tract (ST), radix of the vagus nerve (RV), tracts associated with the area postrema (TP), medial longitudinal fasciculus (MF), tectospinal tracts (TS), medial lemniscus (ML), pyramidal tracts (PT), ventral spinocerebellar tracts (VS), dorsal spinocerebellar tracts (DS), and spinal tract of the fifth nerve (SF). Astrocytic layer/glial limitans (GL) and subependyma (SE) of the fourth ventricle. Immunohistochemical staining, glial fibrillary acidic protein: streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain, commercial kit.d
Obex score = 8
Six elk had a raw obex score that varied from 30.7 to 34.5. PrPCWD IR was detected in all of the nuclear areas, and the pattern was similar as to that seen in elk with an obex score of 7. Plaques of PrPCWD IR were present in all nuclei and in all axonal tracts. A moderate to severe spongiform degeneration was found in the V with mild to moderate spongiform degeneration in 5–7 of the other nuclear regions. The primary difference between an obex score of 7 and 8 was an increase in abundance of PrPCWD IR and a slightly greater overall degree of spongiform degeneration.
Obex score = 9
Fourteen elk had a raw obex score that varied from 35.5 to 39.7. PrPCWD IR was detected in all of the nuclear areas with a pattern similar to elk with an obex score of 8, having plaques of PrPCWD IR in all of the nuclei. Ten of the 14 elk had neurons in which intraneuronal PrPCWD IR was detected. A moderate to severe spongiform degeneration was found in the V with mild to moderate spongiform degeneration in 6–9 of the adjacent nuclear regions. The primary difference between an obex score of 8 and 9 was heavier PrPCWD IR and a slightly greater overall degree of spongiform degeneration. Classical clinical signs were first reported in 7 of these elk with an obex score of 9.
Obex score = 10
Seventeen elk had a raw obex score that varied from 41.1 to 45.7. Moderate to heavy PrPCWD IR was detected in all of the nuclear areas and axonal tracts. The pattern was similar to elk with an obex score of 9 with numerous plaques in all of the nuclei. Intraneuronal PrPCWD IR was detected in all of the 17 elk. Intraneuronal PrPCWD IR was not detected in the V in any of the CWD-positive elk regardless of obex score. Severe spongiform degeneration was found in the DMNV with moderate to severe spongiform degeneration in all of the adjacent nuclear regions. The primary difference between an obex score of 9 and 10 was advanced spongiform degeneration in the nuclear regions. Clinical signs were reported in 12 of 17 of these elk.
The nuclear regions and axonal tracts were well delineated with the GFAP stain for astrocytic processes. The GFAP staining was equivalent in the control and CWD-positive elk, including elk with different obex scores.
Discussion
The pattern of spread of PrPCWD IR and spongiform encephalopathy in a section of obex in elk with naturally occurring CWD progresses in a relatively specific pattern and did not seem to vary with PRNP genotype. This spread and increase in abundance of PrPCWD IR as well as an increase in the severity of spongiform encephalopathy could be utilized to place elk in stages or categories by scoring these 22 sites within the obex (Figs. 9–12).
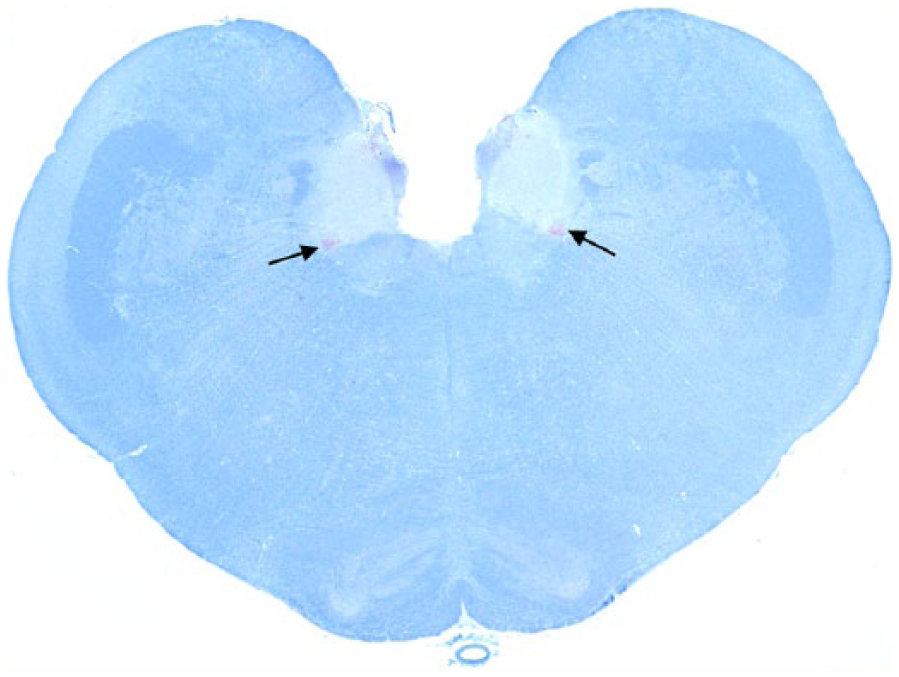
Obex; Rocky Mountain elk (Cervus elaphus nelsoni; 132 M/M genotype), obex score = 2. Note the minimal degree of PrPCWD immunoreactivity in the ventral lateral aspects of the dorsal motor nucleus of the vagus nerve (arrows). Immunohistochemical staining: streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain.
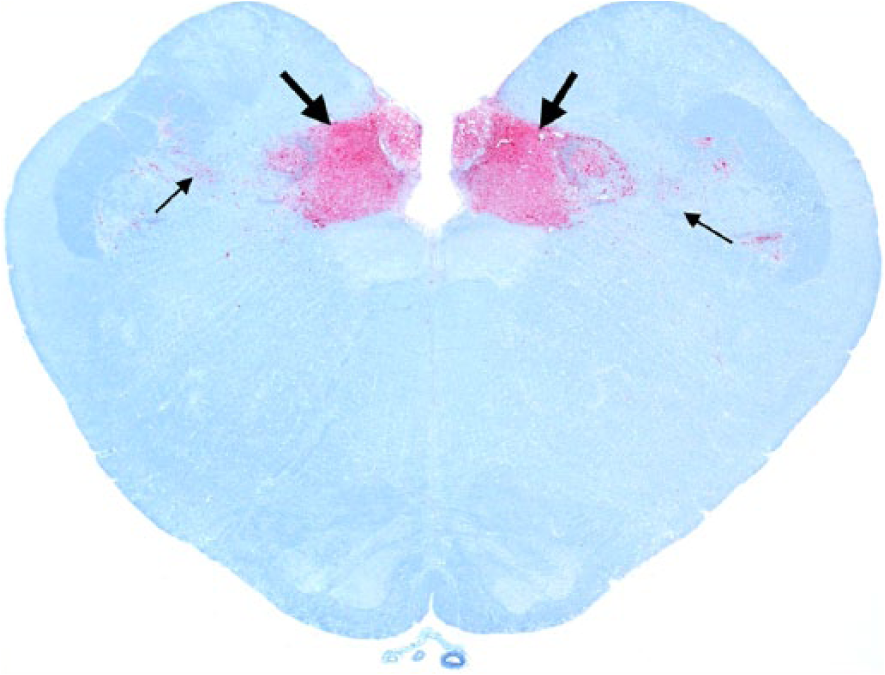
Obex; Rocky Mountain elk (Cervus elaphus nelsoni; 132MM genotype), obex score = 4. Note the intense PrPCWD immunoreactivity (IR) in the dorsal motor nucleus of the vagus nerve and solitarius nucleus (thick arrows) and minimal PrPCWD IR in adjacent nuclei (thin arrows). Immunohistochemical staining: streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain.
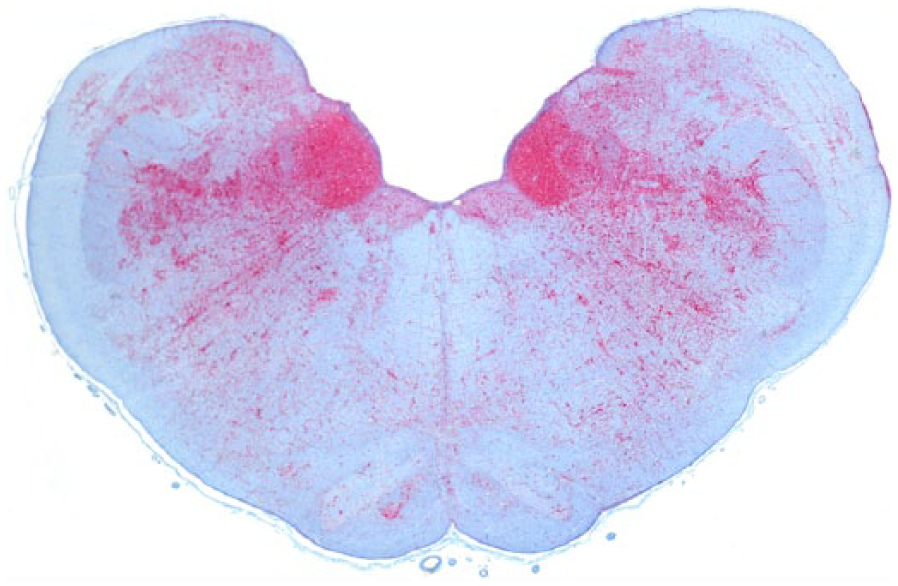
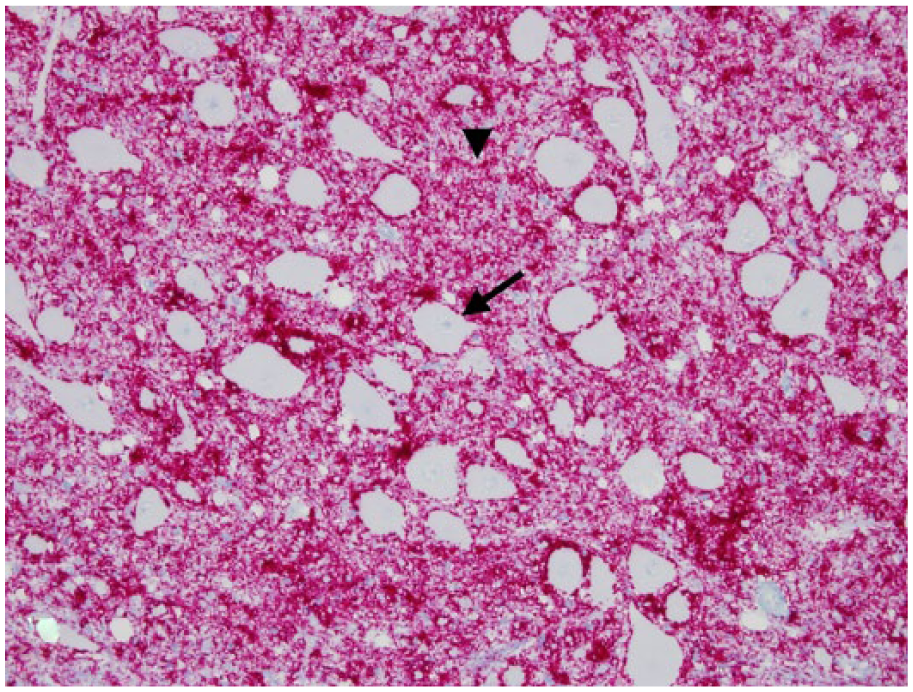
Obex, Rocky Mountain elk (Cervus elaphus nelsoni; 132MM genotype), obex score = 8. Note the intense PrPCWD immunoreactivity (IR) in the dorsal motor nucleus of the vagus nerve and solitarius nucleus and a moderate degree of PrPCWD IR in all of the adjacent nuclei. Immunohistochemical staining: streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain.
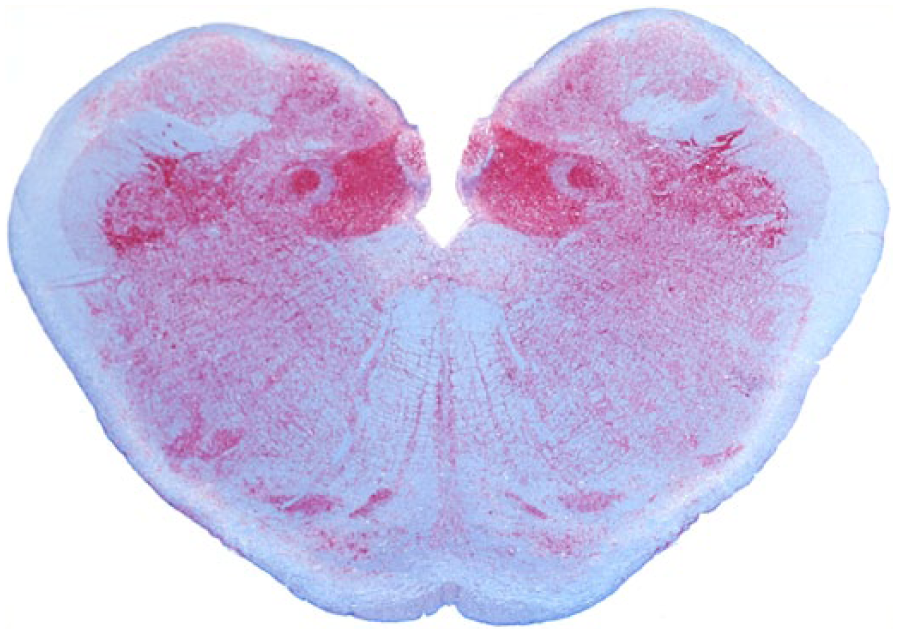
Obex; Rocky Mountain elk (Cervus elaphus nelsoni; 132MM genotype), obex score = 10. Note the intense PrPCWD immunoreactivity in all of the nuclei. Immunohistochemical staining: streptavidin–alkaline phosphatase method. Hematoxylin/bluing counterstain.
The correlation between intensity of the accumulation of prion and spongiform degeneration of the brain stem was remarkable. Clinical signs were not observed in elk with an obex score of 8 or less, and all of these elk were observed prior to euthanasia. Clinical signs were first observed in elk with an obex score of 9. Most of the elk with an obex score of 10 did show clinical signs. Perhaps if the caretakers of the elk had observed the elk more closely clinical signs would have been observed with earlier obex scores. However, many of these elk were free ranging and were euthanized at random for another project. The reason that clinical signs were not observed earlier no doubt could be associated with the experience of the caretakers; on the other hand, even with domestic sheep, clinical signs are not observed until late in the disease. It is the opinion of the authors that the reason for this finding is that neurons are not widely affected until late in the disease and clinical signs are probably more related to direct neuronal damage than just accumulations of the abnormal conformer of PrPCWD within the neuropil and astrocytes.
Four elk did not have detectable PrPCWD IR in 5 sequential sections of V and palatine tonsil, but did have detectable PrPCWD IR in the medial retropharyngeal lymph node. This pattern of PrPCWD IR has been reported in a previous study where 43 of 226 (19%) CWD-positive captive elk had staining exclusively in the lymphoid tissues.17,20 This pattern of PrPCWD IR is believed to represent early cases of CWD.
PrPCWD IR initially accumulated in the ventral aspects of the middle one-third to posterior half of the V in elk with an obex score of 1. This finding is similar to findings reported in elk, 20 free-ranging mule deer, 23 and ranch-raised white-tailed deer. 11 Early PrPCWD IR deposition was characterized by coarse granular deposits adjacent to the outer surface of the cell membranes of neurons and/or within the neuropil, and appeared to be associated primarily with synapses and astrocytes. It could not be determined which location was affected first. The significance of this finding is unclear, but may suggest that astrocytes and synaptic junctions are affected and perhaps accumulate PrPCWD before neurons.
The perineuronal PrPCWD IR was characterized by coarse red granular deposits that appeared to be adherent or attached to the outer membrane of perikarya and neurites. This pattern has been described in domestic sheep naturally and experimental infected with scrapie and BSE, and was thought to be associated with astrocytic processes as they attach to cell membranes of the perikarya and neurites.3,4,7 –9,13 –15,24
Astrocytes have numerous functions within the brain, and have connections to neurons, axons, and vessels. Astrocyte functions include monitoring and regulation of fluids, and electrolytes homeostasis of neurons and surrounding extracellular space, participation in the support of axonal tracts, monitoring and removal of excessive neurotransmitter release at synaptic junctions, protection and insulation of nodes of Ranvier, as well as other functions dealing with the blood brain barrier, healing, and repair. 28 Given the specific patterns observed in these early cases of CWD, and comparison with findings in domestic sheep with natural and experimental scrapie and BSE, it was speculated that astrocytes may be one of the first cell types affected by PrPCWD in elk. Surprisingly, the GFAP stain was not useful in detecting differences in the obex scores.
Another possible factor relating to this perineuronal PrPCWD IR could be associated with the multiple synaptic junctions located on membranes of the perikarya called boutons terminaux. 10 Abnormal prion protein has been reported to localize in synaptic junctions. 6 Therefore, this perineuronal pattern maybe associated with PrPCWD accumulation within synaptic junctions located on the membrane of neurons.
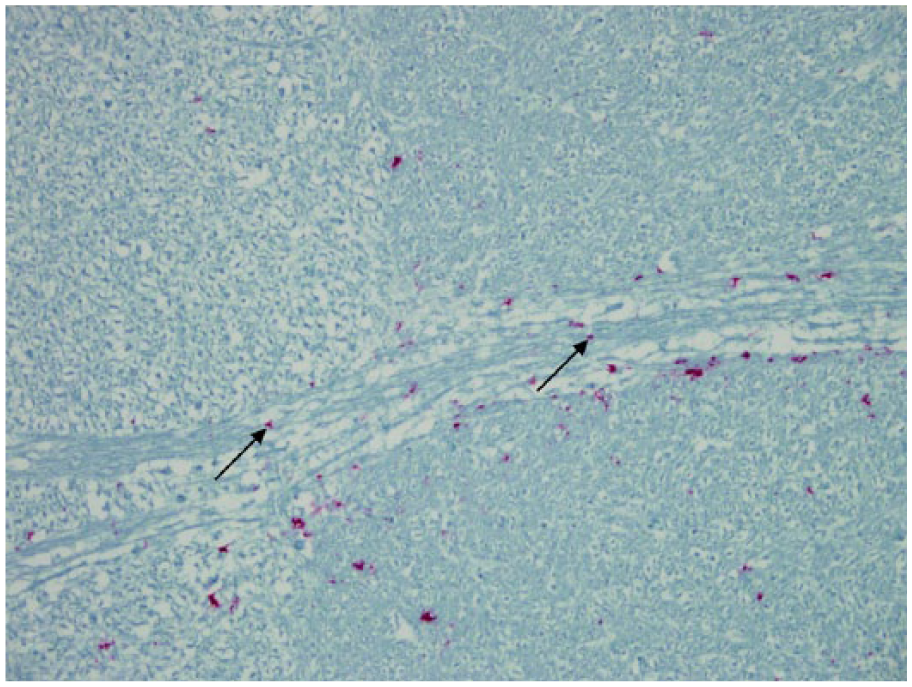
PrPCWD IR within axonal tracts was first observed in elk with an obex score of 3 and was consistent and heavily affected in the axon tracts of the S followed by axons from the V. Astrocytic processes can be associated with axons as supporting structures with oligodendritic cells covering axons and nodes of Ranvier. 28 The association of astrocytic PrPCWD IR within these axonal tracts may be the route of PrPCWD movement or travel within these tracts and perhaps the brain in general. PrPCWD IR was not observed within the axoplasm of these axons. It was assumed that the PrPCWD associated with these axons were exiting the brain, but the direction of prion movement could not be determined.
Another pattern of PrPCWD IR was formation of plaques. A few of these plaques had a nucleus in its center that was compatible in size and shape with astrocytic nuclei. These plaques of PrPCWD IR were first observed in the V in elk with an obex score of 2, therefore they formed rather early in the disease. It was speculated that these plaques could be associated, in part, with astrocytes that had their cytoplasm filled with PrPCWD. Plaques also have been associated with astrocytes in domestic sheep with natural and experimental scrapie as well as BSE.3,4,7,8,13–15,24 These findings further support early astrocytic involvement with PrPCWD metabolism and perhaps movement within the neuropil.
Intraneuronal PrPCWD IR was not observed until an obex score of 7 was reached. This may suggest that direct neuronal damage does not occur early in the disease. Following the intraneuronal accumulation of PrPCWD, clinical signs shortly followed. The lack of intraneuronal staining in the earlier stages of CWD is probably one reason for lack of clinical signs in these elk. It seemed odd that intraneuronal PrPCWD IR was not observed in the V in any of the elk regardless of obex score.
It has been documented that the amino acid sequence of the PRNP gene at codon 132 plays an important role in the duration of the incubation period in elk developing CWD.5,17 The 132MM genotype is the most common in free-ranging and captive elk and is associated with an estimated incubation period of ~2 years in experimentally infected elk. The 132ML genotype is not uncommon in captive and free-ranging elk, and the experimental incubation period in elk of this genotype is ~3.5 years.5,17 The PRNP genotype 132LL are rare (<4%) in free-ranging and captive elk and have an experimental incubation period of ~5–6 years or longer.5,17 The ratio of 132MM to 132ML to 132LL of elk in our study was roughly equivalent to the ratio found in free-ranging and captive populations of elk.16,18 Differences in patterns of PrPCWD IR deposition were not identified between the various PRNP genotypes with regard to progression, abundance, or severity of observed spongiform degeneration. However, the number of times and/or duration of exposure of these elk with the PrPCWD agent were not known. Even though the number of 132ML and 132LL elk with CWD was low, similar patterns of spread of PrPCWD IR and spongiform encephalopathy were observed in this section of obex. It was concluded that the PRNP genotypes of the elk did not significantly alter the pattern of progression and abundance of PrPCWD in these nuclei and axonal tracts and the severity of the spongiform degeneration. However, PRNP may have an influence on the speed of development of these lesions.
Data from our study suggests that the pattern of CWD progression and abundance of PrPCWD IR in these nuclei and axonal tracts, as well as the progression and severity of the spongiform degeneration have a unique and relatively consistent pattern throughout a section of brain stem at the level of the obex in Rocky Mountain elk with naturally occurring CWD (Table 4). This technique of scoring the obex may have potential usefulness as a basis for predicting the presence and abundance of PrPCWD in peripheral tissues and the central nervous system.
Footnotes
Acknowledgements
We thank the owners of captive elk (Dennis and Stephanie White and 3 anonymous owners) for allowing us to do rectal biopsies on their elk and, if positive cases were found, allowing us to euthanize and examine postmortem the suspected positive elk. We thank the numerous state and federal employees for euthanizing and collecting samples from elk that were killed in depopulation projects to try to eradicate CWD from captive herds in the United States and Canada. We thank Bob Zink and Todd Bass for cutting and staining the tissues. We thank Vicki Jameson, Crystal Meyerett, Tara Ruby, Amy Graham, Danielle Lagana, David Walter, Christy Wyckoff, and Jordan Spaak for assisting in the postmortem examinations of some of these elk. We thank Linda Hamburg and Desiree Lesiak for PRNP genotyping. We thank Jay Oaks, Charlie Kerlee, and Barbara Traut for assisting with photography. We thank Mary Kay Watry, Ben Bobowski, and John Mack for providing assistance with and access to free-ranging elk at Rocky Mountain National Park as part of the park’s elk management strategy.
Authors’ contributions
All authors contributed to conception and design of the study, critically revised the manuscript, and gave final approval. TR Spraker, T Gidlewski, MA Wild, K VerCauteren, and KI O’Rourke contributed to acquisition, analysis, and interpretation of data. JG Powers contributed to analysis and interpretation of data. B Cummings contributed to acquisition of data. TR Spraker drafted the manuscript and agrees to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
a.
Histobond adhesive slides, StatLab Medical Products, McKinney, TX.
b.
ACROS Organics, Morris Plains, NJ.
c.
pH 6.1, DAKO target retrieval solution; Dako Denmark A/S, Glostrup, Denmark.
d.
APK wash solution, NexES automated immunostainer, Basic alkaline phosphatase red kit with amplification, Anti-prion antibody kit; Ventana Medical Systems Inc., Tucson AZ.
Declaration of conflicting interests
The author(s) declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was partially funded by National Park Service project no. CSURM-98, Cooperative Agreement no. H1200040001 task order 123400070064; USDA/ARS Cooperative Agreement no. 58-5348-8-275/project no. 5348-32000-026-105; and Colorado State University Diagnostic Laboratory, College of Veterinary Medicine and Biomedical Sciences, Colorado State University, Fort Collins, Colorado.
