Abstract
Rats (Rattus spp.) are among the most damaging invasive species worldwide. The accidental introduction of rats has caused significant detriment to native flora and fauna, crops, structures, and human livelihoods. Rats are vectors of disease and carriers of various zoonotic parasites. Capillaria hepatica (syn. Callodium hepaticum) is a parasitic nematode found primarily in rodents but is known to infect over 140 mammal species, including human beings and several species of domestic animals. In this case study, the presence of C. hepatica infection in black rats on Diego Garcia, British Indian Ocean Territory, is reported. Liver samples from 20 black rats (Rattus rattus) were collected during a concurrent population density estimation study. Histology revealed 15 (75%) of the rats sampled had a current or previous infection with C. hepatica. In addition, a larval cestode compatible in size and shape with Cysticercus fasciolaris, the larval stage of Taenia taeniaeformis of cats, was found in 3 (15%) of the rats sampled. The high prevalence of C. hepatica infection in rats on Diego Garcia has implications for human health given the high population density of rats found on the island.
Rats (Rattus spp.) are among the most damaging invasive species worldwide and pose a particularly insidious threat to island ecosystems. Accidental introduction of rats to islands has had significant detrimental consequences to native flora and fauna.12,15 In addition, rats are responsible for crop and stored food damage and damage to dwellings and structures. 5 As vectors of disease, they are most well-known for spreading bubonic plague during the Middle Ages. 16 However, they are also carriers of various zoonotic parasites.2,7,18
Capillaria hepatica (syn. Callodium hepaticum) is a parasitic nematode common to rodents and frequently reported in Norway rats (Rattus norvegicus), although infection has been documented in more than 140 mammal species (including human beings, dogs, cats, and horses) worldwide (Schmidt S. Untersuchungen zum Vorkommen von Capillaria hepatica und Metazestoden der Cyclophyllida bei Wildmäusen in Deutschland [Studies on the occurrence of Capillaria hepatica and metacestodes of Cyclophyllida in wild mice in Germany] [thesis]. Leipzig, Germany: Faculty of Veterinary Medicine, Leipzig University; 2001. 145 p. German).4,6 The life cycle of C. hepatica is direct, requiring a single host. Embryonated eggs are ingested, larvae hatch in the intestines, and the larvae migrate to the liver where they mature, breed, and lay unembryonated eggs. The encapsulated and unembryonated eggs are not shed by the host and become exposed to the environment through host carcass decomposition, or from scavengers or predators consuming the host and then shedding the eggs in their feces. Adult nematodes are short lived in the liver and usually die within 26–72 days.4,5,14 Optimal environmental conditions, such as adequate temperature and moisture, are required for eggs to embryonate. 6
Capillaria hepatica infection in human beings can result in capillariasis, which can only be diagnosed by liver biopsy. In a review, 163 human cases of infection were documented worldwide in which 72 cases resulted in hepatic capillariasis; 60% of which were in children under 8 years of age. 6 Symptoms range from mild to severe, but misdiagnosis can be fatal. 6 The current case study describes the finding of C. hepatica infections in black rats (Rattus rattus) on Diego Garcia, British Indian Ocean Territory.
Diego Garcia is a 44-km2 atoll on the southern edge of the Chagos Archipelago, British Indian Ocean Territory, and is located approximately 7° south of the equator. The climate is tropical with an average temperature of 27°C. Mean annual rainfall is 362 cm. The eastern portion of the island is a restricted nature preserve, and the western side consists of a United States Naval Support Facility. Vegetation consists primarily of coconut palm (Cocos nucifera) stands with a scattered understory of Neisosperma oppositifolia, Morinda citrifolia, and Hernandia sonora. Black rats were likely inadvertently introduced several centuries ago when European ships first landed on the atoll. 17
A 1-hectare grid (11 × 11 traps, 10 m between traps) in a mixed forest canopy approximately 1 km from the cantonment on the northwest side of the island was established. Black rats were trapped with commercial snap traps baited with peanut butter and fresh coconut during a concurrent rat population density study. Postmortem examinations were performed in the field within 1 hr of collection.
One hundred twenty-eight black rats (66 males and 62 females) were collected. On gross examination, 37 rats (29%) had no visual lesions or abnormalities in the liver. Livers from 91 rats (71%) had lesions characterized by 1–10-mm irregular yellow to gray regions within the liver parenchyma (Fig. 1). Another gross lesion was characterized by mottling, with increased lobular architecture or pallor suggestive of lipidosis (Fig. 2). Liver samples from 20 rats (9 males, 11 females) were collected, preserved in 90% ethanol, and refrigerated until shipped to Colorado State University (Fort Collins, Colorado) for histological examinations. Selected fixed tissues were trimmed, placed in cassettes, processed overnight, embedded in paraffin, sectioned at 5 µm, and stained with hematoxylin and eosin. a Sections were examined under light microscopy. Of these 20 livers, 17 had gross lesions as described above; whereas 3 livers had no gross lesions. Histological examination showed 15 (75%) livers to be infected with C. hepatica: 9 with active infections (Figs. 3, 4) with small to extensive sheets of eggs (Fig. 5). Following deposition of eggs, a fibrous capsule was formed around the unembryonated eggs (Fig. 6). Six of the 20 rats did not have an active infection but did have dead adult parasites with no evidence of egg production within the liver parenchyma (Fig. 7). Of the 9 rats with active C. hepatica infections, 3 had additional cysts compatible in size, shape, and location with Cysticercus fasciolaris, the larval form of Taenia taeniaeformis (Fig. 8), a tapeworm commonly found in domestic cats.1,8,9 Of the 3 livers with no visible gross lesions on histological examination, 1 had a small focus of fibrosis surrounding a dead nematode and the other 2 had no lesions. The liver that had increased lobular architecture on gross examination had no significant lesions.
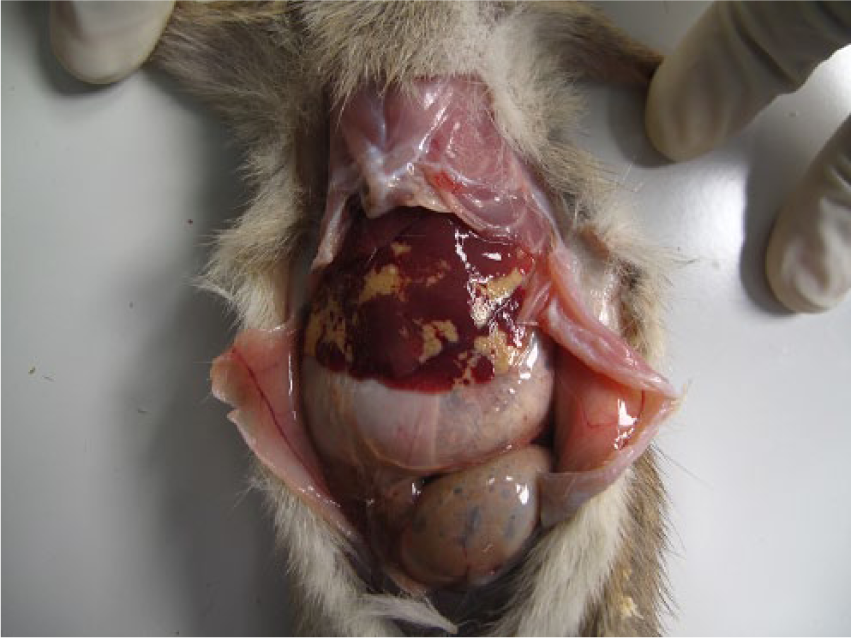
Gross photograph of the liver from a black rat from Diego Garcia, British Indian Ocean Territory. The multifocal pale yellow lesions are heavily infected regions of Capillaria hepatica.
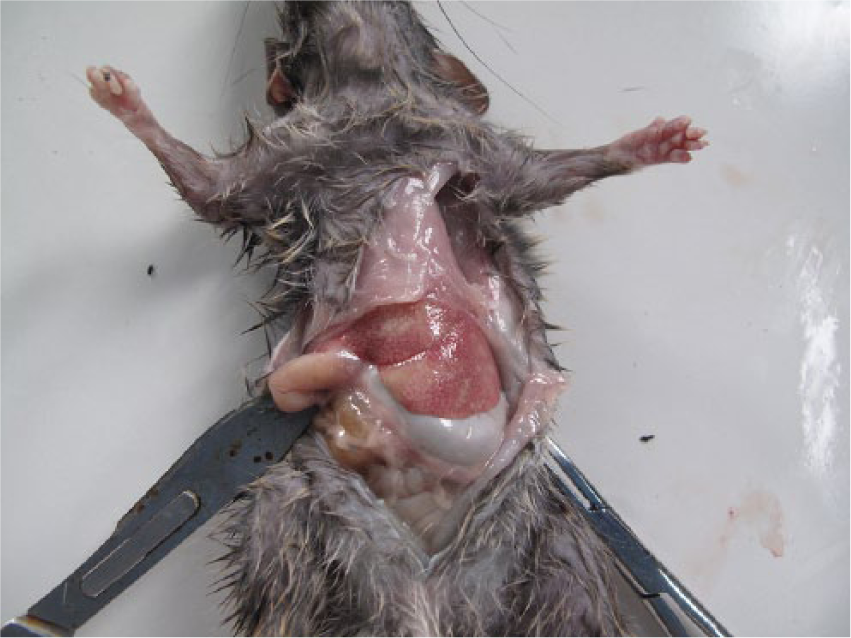
Gross photograph of the liver, with extensive, slightly irregular regions of pallor suggestive of lipidosis in a black rat from Diego Garcia, British Indian Ocean Territory.
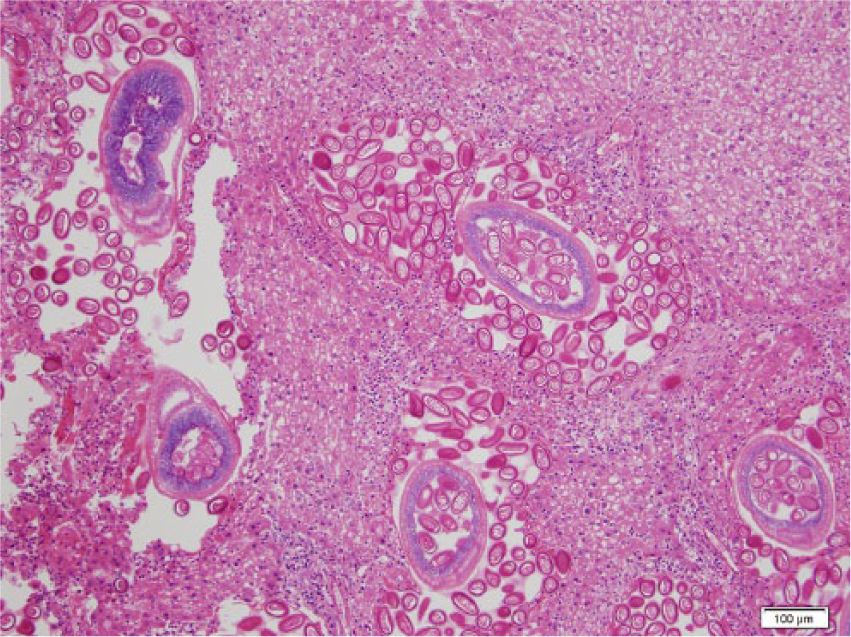
Photomicrograph of the liver of a black rat showing adult female Capillaria hepatica migrating through the parenchyma and depositing eggs. Hematoxylin and eosin stain. Bar = 100 µm.
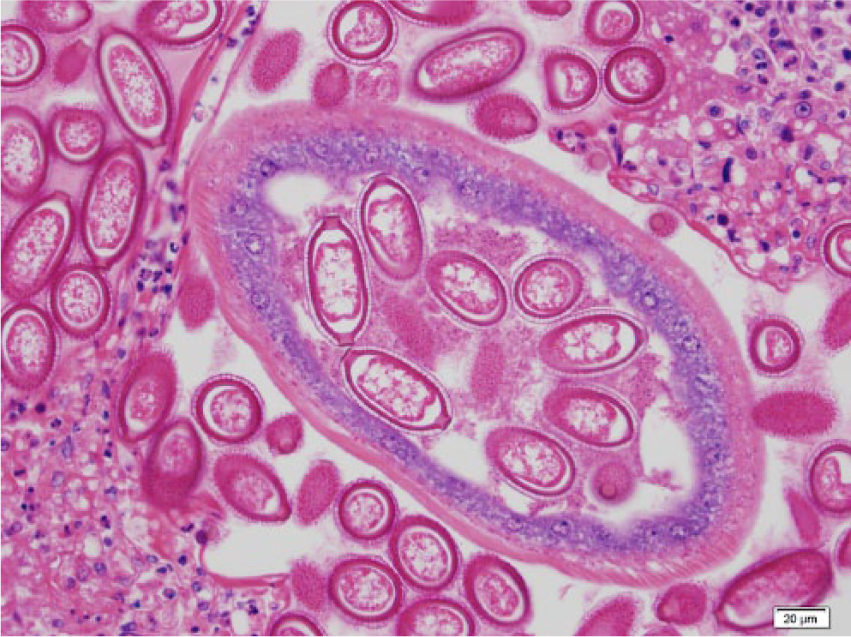
Photomicrograph of the liver of a black rat showing an adult Capillaria hepatica with eggs in her uterus with the classical double polar caps. Hematoxylin and eosin stain. Bar = 20 µm.
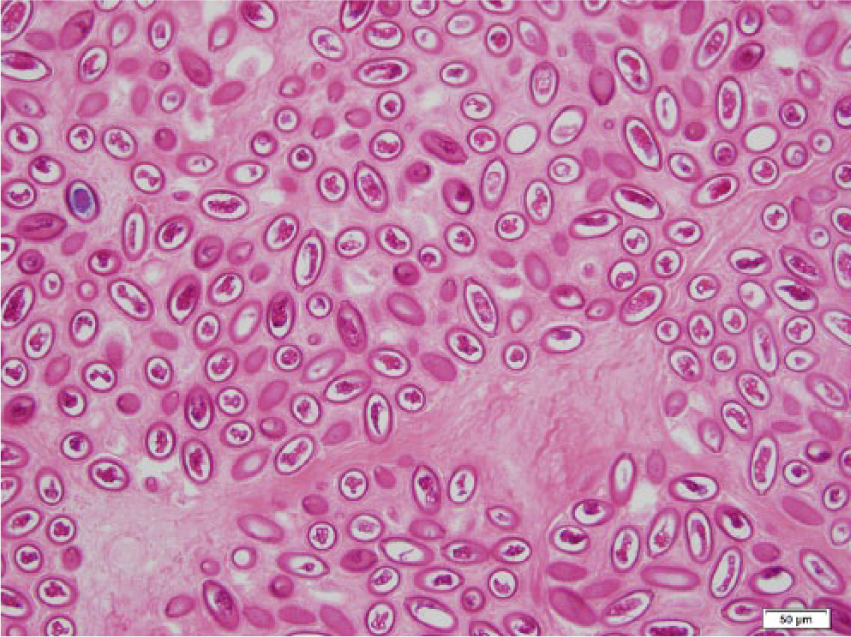
Photomicrograph of the liver of a black rat showing sheets of double polar eggs of Capillaria hepatica. Hematoxylin and eosin stain. Bar = 50 µm.
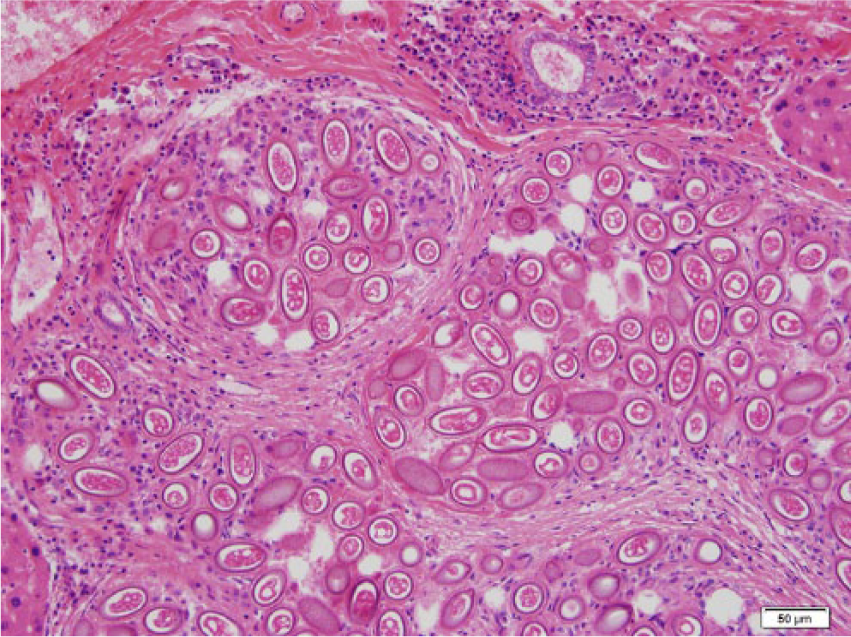
Photomicrograph of the liver of a black rat showing deposits of double polar eggs of Capillaria hepatica surrounded by fibrosis. Hematoxylin and eosin stain. Bar = 50 µm.
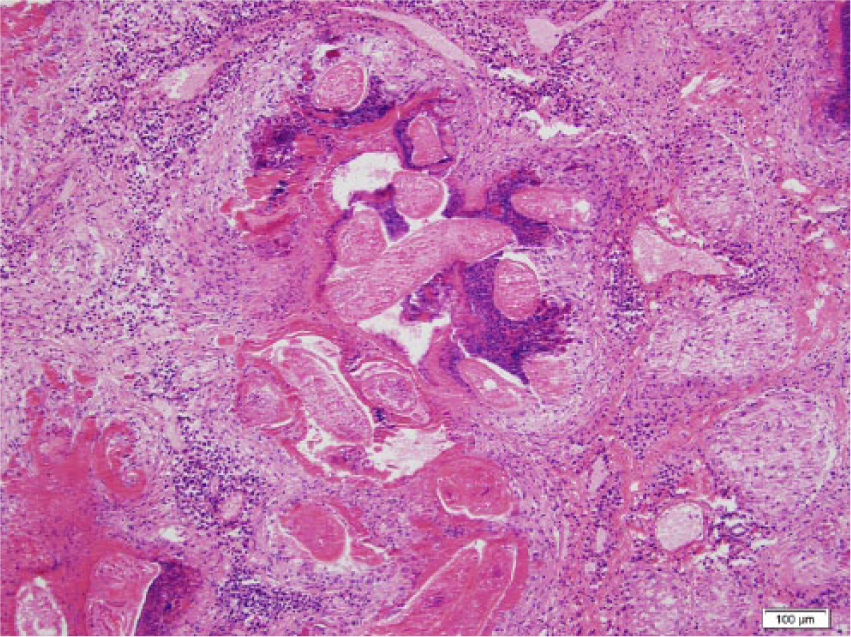
Photomicrograph of the liver of a black rat showing a dead adult Capillaria hepatica. Hematoxylin and eosin stain. Bar = 100 µm.
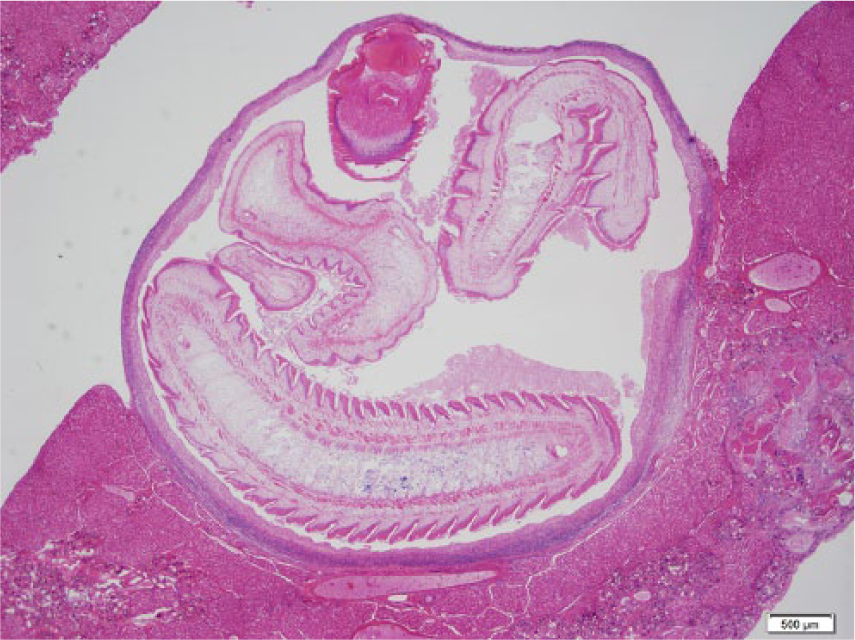
Photomicrograph of the liver of a black rat with a larval stage for Taenia taeniaeformis (Cysticercus fasciolaris) infection. Hematoxylin and eosin stain. Bar = 500 µm.
Overall, parasitic infection of black rats on Diego Garcia, particularly with C. hepatica, was high despite a relatively small sample size. The reported prevalence of C. hepatica in black rats is 5.4–46%.11,13 The 6 rats with livers containing dead nematodes indicative of prior infection may have been resistant to infection, developed immunity to the parasites, or reflect adult parasite mortality as a result of the relatively short life span of the parasite. Ingestion of embryonated eggs is the only route of infection with C. hepatica, and eggs only embryonate on exposure to the environment following decay of a host or excretion by predators and/or scavengers. As black rats are the only mammalian wildlife species on Diego Garcia, it is likely that infection is chronic and circulating within the rat population, probably maintained through cannibalism and cannibalistic scavenging. It was interesting to note several cases of C. fasciolaris (a cestode found in domestic cats) infection within the liver of 3 rats. It is possible that domestic cats may be playing a role in these infections; however, most of the feral cats have been removed from the island.
Parasitic infections in human beings due to C. hepatica are rare, but are typically associated with unsanitary conditions (i.e., contaminated drinking water) or a high prevalence of rodents. 6 Drinking water on Diego Garcia is via rain catchment and is sanitized, thus an unlikely source of infection. However, the rat density on Diego Garcia is extremely high (up to 187/ha in some habitats [Proceedings of the 26th Vertebrate Pest Conference, 3–6 March, 2014, Kona, Hawaii]). Extensive rodent control is implemented in the cantonment but not near contractor housing. Improper refuse containment, as well as household gardens and forested areas near contractor housing, provides habitat and a consistent food source for rats (E. Necessario, pest control supervisor, Diego Garcia, personal communication, 2014). Exposure to high rat densities could potentially put the personnel residing on Diego Garcia at risk of infection. Because ingestion of infected soil is a route of infection, particularly among children, gardening can result in contaminated dirt on vegetables being inadvertently consumed.3,10,13 A more comprehensive study of the extent of C. hepatica infection in rats throughout Diego Garcia may provide more information on potential regional differences in infection relative to differing rat densities relative to habitat types.
Footnotes
Acknowledgements
This article is dedicated to the memory of Daniel S. Vice: fisherman, biologist, husband, father, and friend. The authors wish to thank E. Necessario, M. Ferrer, J. Samson, R. Embate, G. Estrada, and F. Ebona for assistance in the field. Special thanks to T. Kuzmina for assistance in parasite identification and to 2 anonymous reviewers for their helpful comments on an earlier draft of this article.
a.
Tissu-Tek VIP 6, Sakura Finetek USA Inc., Torrance, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research was funded through U.S. Department of Defense Agreement number 13-73-15-6576-IA.
