Abstract
Chinese Shar-Pei dogs have a high prevalence of hypocobalaminemia and are commonly presented with clinical signs suggestive of severe and long-standing gastrointestinal disease such as diarrhea, vomiting, and/or weight loss. The aim of the current study was to evaluate serum concentrations of inflammatory markers, markers for intestinal disease, and immunological markers in Shar-Peis with hypocobalaminemia or normocobalaminemia (serum cobalamin concentrations within the reference interval). Serum samples from Shar-Peis were collected from various parts of the United States. Serum concentrations of inflammatory markers (i.e., C-reactive protein [CRP], calprotectin [CP], and S100A12), hyaluronic acid (HA, a marker for cutaneous mucinosis), and analytes commonly altered in chronic intestinal diseases (i.e., albumin, zinc, alpha1-proteinease inhibitor [α1PI], immunoglobulin [Ig]A, and IgM) were compared between Shar-Peis with hypocobalaminemia and Shar-Peis with normocobalaminemia. Serum concentrations of CRP, CP, S100A12, HA, zinc, and cα1-PI concentrations did not differ between hypocobalaminemic and normocobalaminemic Shar-Peis (P > 0.05). Serum concentrations of albumin were significantly lower in hypocobalaminemic Shar-Peis (median: 2.5 g/dl) than in normocobalaminemic Shar-Peis (median: 2.9 g/dl; P < 0.0001). Higher serum IgA concentrations and lower serum IgM concentrations were observed in hypocobalaminemic Shar-Peis (median: 1.7 g/l and 0.8 g/l, respectively) than in normocobalaminemic Shar-Peis (median: 0.7 g/l and 1.9 g/l, respectively; both P < 0.0001). In conclusion, no difference was found in serum concentrations of CRP, CP, S100A12, and HA between hypocobalaminemic and normocobalaminemic Shar-Peis whereas some differences were observed in analytes (e.g., albumin, IgA, and IgM) that may be altered in patients with chronic enteropathies.
Introduction
Cobalamin (i.e., vitamin B12) is a complex water-soluble vitamin, which is involved in many metabolic processes such as RNA/DNA synthesis, hematopoiesis, and fatty acid metabolism. A malabsorption of cobalamin can be caused by a variety of gastrointestinal disorders. Based on serum sample submissions to Gastrointestinal Laboratories in both North America and the United Kingdom, the Chinese Shar-Pei dog has been described as having a high prevalence of marked or severe hypocobalaminemia.4,8
Shar-Peis with marked hypocobalaminemia commonly present with clinical signs suggestive of severe and longstanding gastrointestinal disease such as diarrhea, vomiting, and/or weight loss. 4 It has been shown that hypocobalaminemia in Shar-Peis is associated with hyperhomocysteinemia, which suggests that the function of the intracellular cobalamin-dependent enzyme (i.e., methionine synthase) is impaired. 12 In this context, hyperhomocysteinemia has been described in human beings with chronic inflammatory diseases such as rheumatic disease, 30 cardiovascular and/or end-stage renal disease, 32 and inflammatory bowel disease (IBD). 26
Serum concentrations of C-reactive protein (CRP), a recognized acute phase reactant 2 ; calprotectin (CP), a marker of granulocytic inflammation15,22; and S100A12, a sensitive marker of neutrophilic inflammation, 20 have been described in dogs with chronic gastrointestinal diseases and were observed to be increased in dogs with IBD15,19 (Heilmann RM, Allenspach K, Procoli F, et al.: 2011, Calgranulin concentrations in dogs with inflammatory bowel disease. J Vet Intern Med 25:1486. Abstract). However, to the authors’ knowledge, such inflammatory markers (i.e., CRP, CP, and S100A12) have not yet been reported in Shar-Peis with hypocobalaminemia.
Hyaluronic acid (HA) has been suggested as another potential marker for inflammation. 14 Interestingly, increased serum HA concentrations have been described in Shar-Peis with cutaneous mucinosis when compared to healthy controls, and it has been proposed that this condition is a consequence of a genetic defect involving HA.24,34 Therefore, it appears reasonable to measure serum HA concentrations in Shar-Peis with hypocobalaminemia.
A higher production of cell surface HA has been documented on mucosal endothelial cells in human patients with IBD when compared with healthy controls. 21 Along those lines, low serum cobalamin concentrations have also been documented in both human and canine patients with chronic enteropathies such as IBD.1,33 We hypothesize that certain serum analytes (e.g., albumin, 1 zinc [Gingerich KK, Parnell NK, Moor GE: 2008, Serum magnesium and zinc concentrations in dogs with inflammatory bowel disease. J Vet Intern Med 22:747–748. Abstract], and canine alpha1-proteinease inhibitor11,17), which have been reported to be affected in dogs with severe and longstanding intestinal disease might be altered in Shar-Peis with hypocobalaminemia.
In human beings, immunoglobulin (Ig)A has been suggested to play a role in the pathogenesis of chronic intestinal diseases leading to cobalamin deficiency. 2 In dogs, decreased concentrations of IgA and IgM in serum have been observed in Shar-Peis with a suspected primary immune-deficiency syndrome.23,25 However, these 2 immunoglobulins have not been documented in Shar-Peis with hypocobalaminemia. The aims of the current study were to compare serum concentrations of inflammatory markers, markers for chronic intestinal disease, and immunological markers in Shar-Peis with hypocobalaminemia and normocobalaminemic (serum cobalamin concentrations within the reference interval), and to evaluate if an inflammatory phenotype exists in Shar-Peis with hypocobalaminemia.
Materials and methods
Sample collection
For the purpose of the current study, serum samples from Shar-Peis were collected between March 1, 2006 and December 1, 2009. The protocol for collection of serum samples from healthy dogs was reviewed and approved by the Clinical Research Review Committee at Texas A&M University (CRRC#2003-51, CRRC#2007-30; College Station, Texas). These serum samples had been collected from Shar-Peis from various parts of the United States, and the owner of each enrolled dog completed a questionnaire, which included questions concerning the signalment and the current health status of the dog. Samples were shipped on ice overnight to the Gastrointestinal Laboratory at Texas A&M University. All serum samples were then stored at −80°C and, depending on the amount of serum available, were used to assay all biomarkers within 3 months from the time of sample collection. Some of the samples were collected from dogs that also had been used for a genome-wide association study of cobalamin deficiency in the Shar-Pei as reported elsewhere. 10 Not all analytes were evaluated in all samples (Table 1).
Number (n) and proportion of hypocobalaminemic (Hypo-COB) and normocobalaminemic (Normo-COB) Chinese Shar-Peis that were included in the current study are listed for each test performed.*
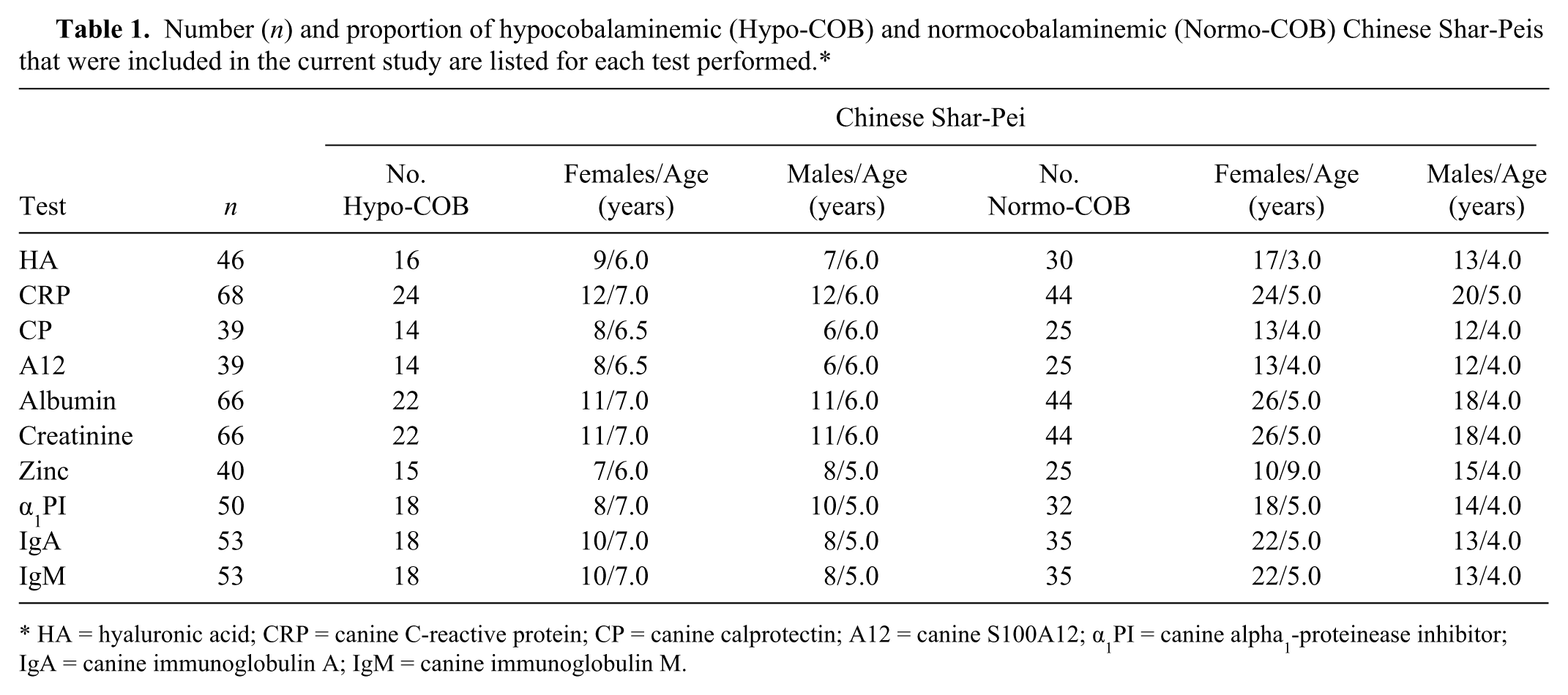
HA = hyaluronic acid; CRP = canine C-reactive protein; CP = canine calprotectin; A12 = canine S100A12; α1PI = canine alpha1-proteinease inhibitor; IgA = canine immunoglobulin A; IgM = canine immunoglobulin M.
Concentrations of serum cobalamin
Serum cobalamin concentrations in Shar-Peis were measured using an automated chemiluminescence assay a with a reference interval of 251–908 ng/l. b Only dogs that were normocobalaminemic or that had an undetectable serum cobalamin concentration (<150 ng/l; these dogs were considered to be markedly hypocobalaminemic; Table 1) were included.
Concentrations of serum inflammatory markers
Serum CRP concentrations were quantified in Shar-Peis with and without hypocobalaminemia (Table 1) using a commercial enzyme-linked immunosorbent assay (ELISA) kit c with a reference (ref.) interval of 0–7.6 mg/l. d Serum calgranulin concentrations were measured using an in-house ELISA for canine CP 13 (ref. interval: 0.9–11.9 mg/l; Heilmann RM, et al.: 2011, Calgranulin concentrations in dogs with inflammatory bowel disease) and an in-house radioimmunoassay for canine S100A12 (ref. interval: 33.0–233.0 μg/l). 16
Concentrations of serum hyaluronic acid
Serum HA concentrations were measured in samples from Shar-Peis (Table 1) with and without hypocobalaminemia by use of a commercially available ELISA kit. e Because of the lack of an in-house control interval for canine serum HA concentrations, serum from 7 healthy German Shepherd Dogs was used to provide an approximate control range. Because of the small sample size, serum HA concentrations of healthy German Shepherd Dogs (controls) were compared to measurements of healthy control dogs of other studies 35 (Seki M, Asano K, Sakai M, et al.: 2008, Clinical significance of serum hyaluronic acid in canine hepatic diseases. J Vet Intern Med 22:805. Abstract). The protocol for collection of serum samples from healthy German Shepherd Dogs using a questionnaire was reviewed and approved by the Clinical Research Review Committee at Texas A&M University (CRRC#2005-35).
Concentrations of serum chronic intestinal disease markers
Serum albumin (ref. interval: 2.4–4.5 g/dl) concentrations were measured for Shar-Peis with and without hypocobalaminemia using an automated clinical chemistry analyzer. f An external laboratory (Texas A&M Veterinary Medical Diagnostic Laboratory) was used to measure serum zinc concentrations (ref. interval: 0.7–2.0 ppm) g in Shar-Peis with and without hypocobalaminemia. Finally, serum α1PI concentrations were measured using an in-house radioimmunoassay (ref. interval: 732–1,802 mg/l). 17
Concentrations of selected serum immunoglobulins and serum creatinine
Serum IgA and IgM concentrations were quantified by ELISAs using canine-specific commercial kits. h Serum creatinine (ref. interval: 0.5–1.4 mg/dl) concentrations were measured in Shar-Peis with and without hypocobalaminemia using an automated clinical chemistry analyzer. f
Data analysis
To conduct statistical analyses, a commercial software package i was used. A Mann–Whitney U test for nonparametric data was used to compare serum CRP, CP, S100A12, HA, albumin, zinc, α1PI, IgA, IgM, and creatinine concentrations between hypocobalaminemic and normocobalaminemic Shar-Peis. In addition, a Kruskal–Wallis test with a Dunn posttest for multiple comparisons among nonparametric data sets was used to compare serum HA concentrations between hypocobalaminemic Shar-Peis, normocobalaminemic Shar-Peis, and healthy German Shepherd Dogs (controls). A Fisher exact test was used to evaluate if marked hypocobalaminemia in Shar-Peis is associated with decreased serum albumin concentration as well as an increased IgA-to-IgM ratio; the odds ratio (OR) and the 95% confidence interval (CI) were also calculated. Receiver operating characteristic curves were used to determine the optimum serum albumin concentration and IgA-to-IgM ratio that distinguished hypocobalaminemic Shar-Peis from normocobalaminemic Shar-Peis. Significance for all tests was set at P < 0.05.
Results
For all different biomarkers (i.e., HA, CRP, CP, S100A12, zinc, and α1PI), hypocobalaminemic Shar-Peis were not significantly older than normocobalaminemic Shar-Peis (P > 0.05), except for the markers albumin and creatinine (both: P = 0.0375), and IgA and IgM (both: P = 0.0496; Table 1). Gender-specific differences were not identified between the 2 groups of Shar-Peis for all different biomarkers (P > 0.05).
Serum inflammatory markers
Concentrations of serum CRP, CP, and S100A12 were not significantly different between hypocobalaminemic and normocobalaminemic Shar-Peis (Table 2; Fig. 1). Fifty percent of hypocobalaminemic Shar-Peis had serum CP concentrations above the upper limit of the reference interval, 43% had serum S100A12 concentrations above the suggested upper limit of the reference interval, and 29% of hypocobalaminemic Shar-Peis had a serum CRP concentration above the upper reference limit.
Medians, ranges, and P values for comparison of the different analytes between hypocobalaminemia (Hypo-COB) and normocobalaminemic (Normo-COB) Chinese Shar-Peis.*
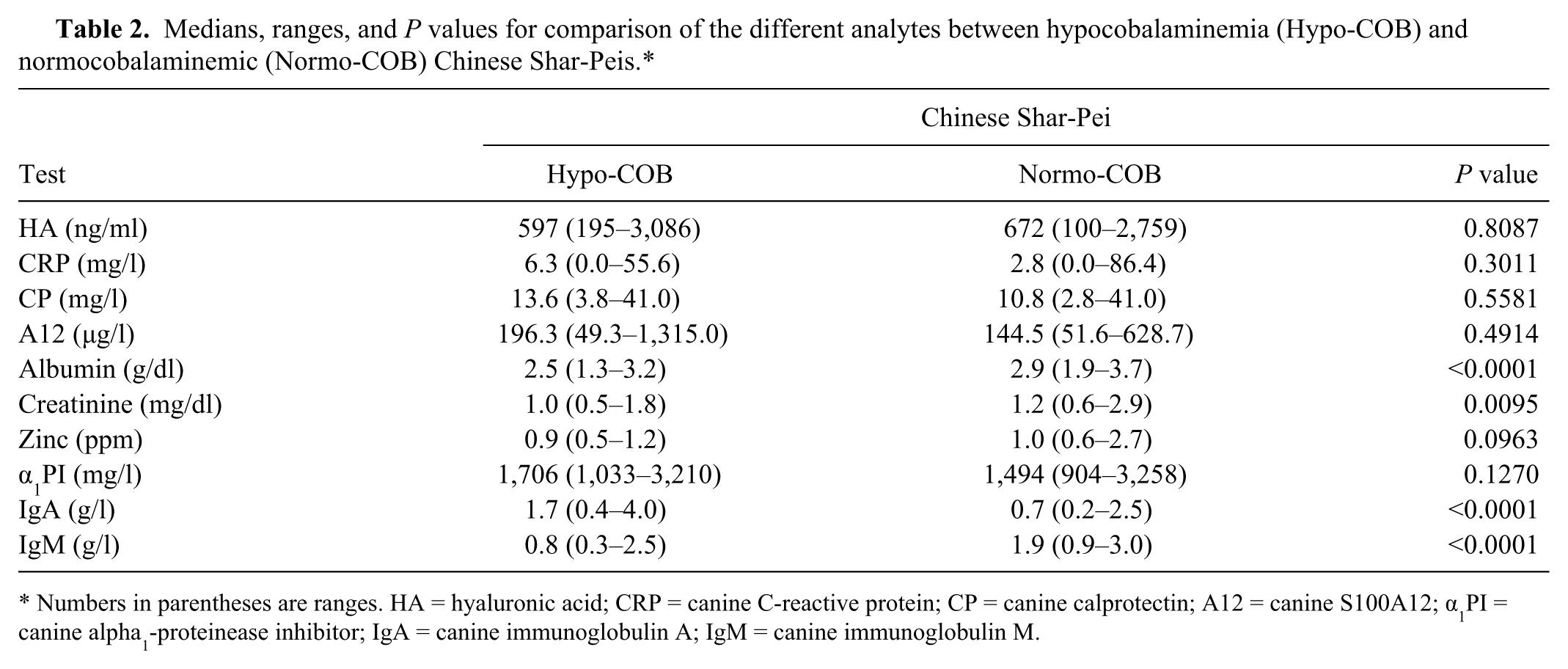
Numbers in parentheses are ranges. HA = hyaluronic acid; CRP = canine C-reactive protein; CP = canine calprotectin; A12 = canine S100A12; α1PI = canine alpha1-proteinease inhibitor; IgA = canine immunoglobulin A; IgM = canine immunoglobulin M.
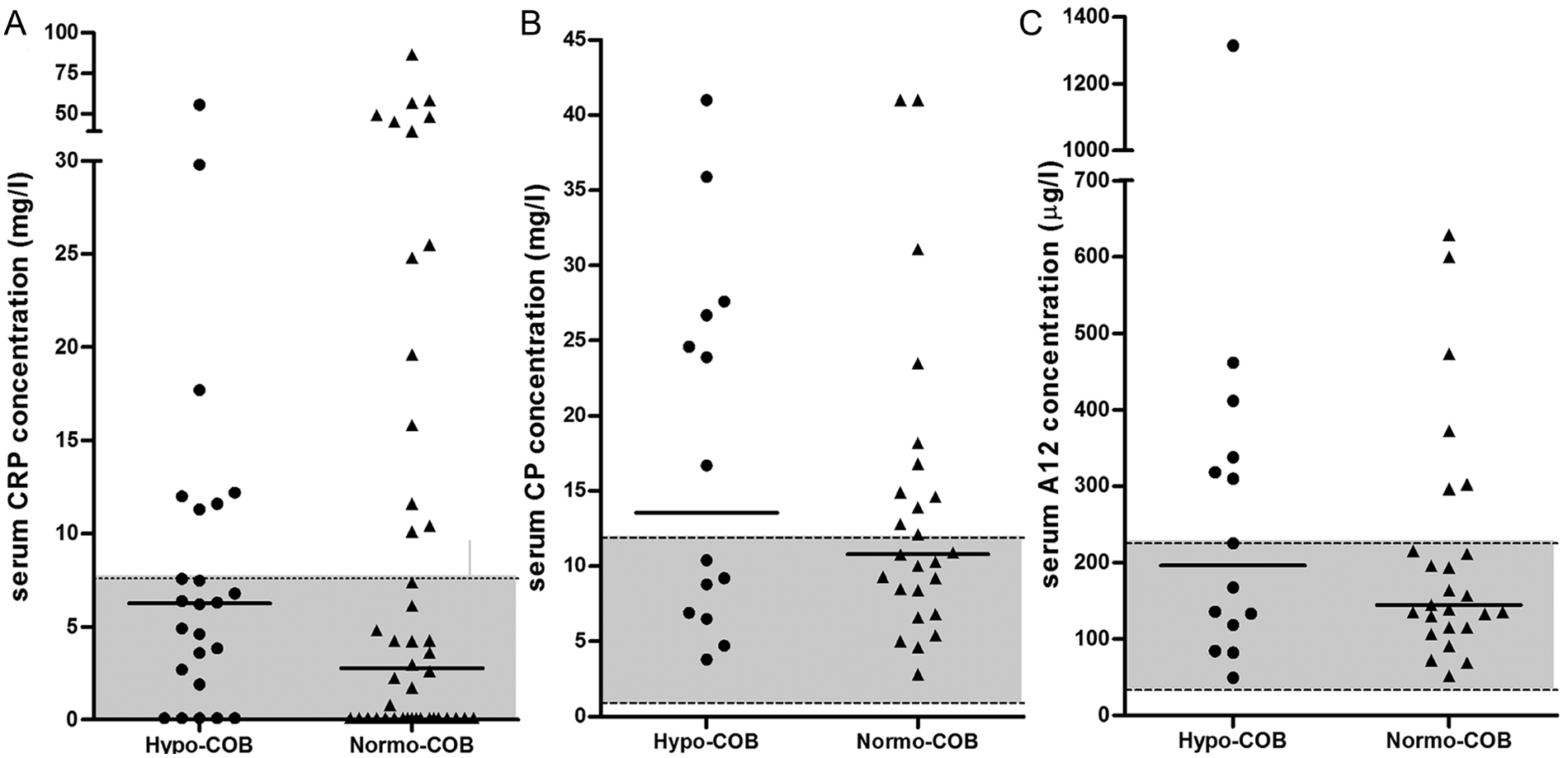
Comparison of (
Serum HA concentrations
No significant difference of serum HA concentrations was identified between hypocobalaminemic Shar-Peis and normocobalaminemic Shar-Peis (Table 2). However, serum HA concentrations differed among groups of Shar-Peis with and without hypocobalaminemia and healthy German Shepherd Dogs (P = 0.0156). The posttest showed that concentrations of serum HA were significantly higher in both hypocobalaminemic Shar-Peis (median: 597 ng/ml) and normocobalaminemic Shar-Peis (median: 672 ng/ml) when compared to healthy German Shepherd Dogs (median: 227 ng/ml; for both P < 0.05; Fig. 2).
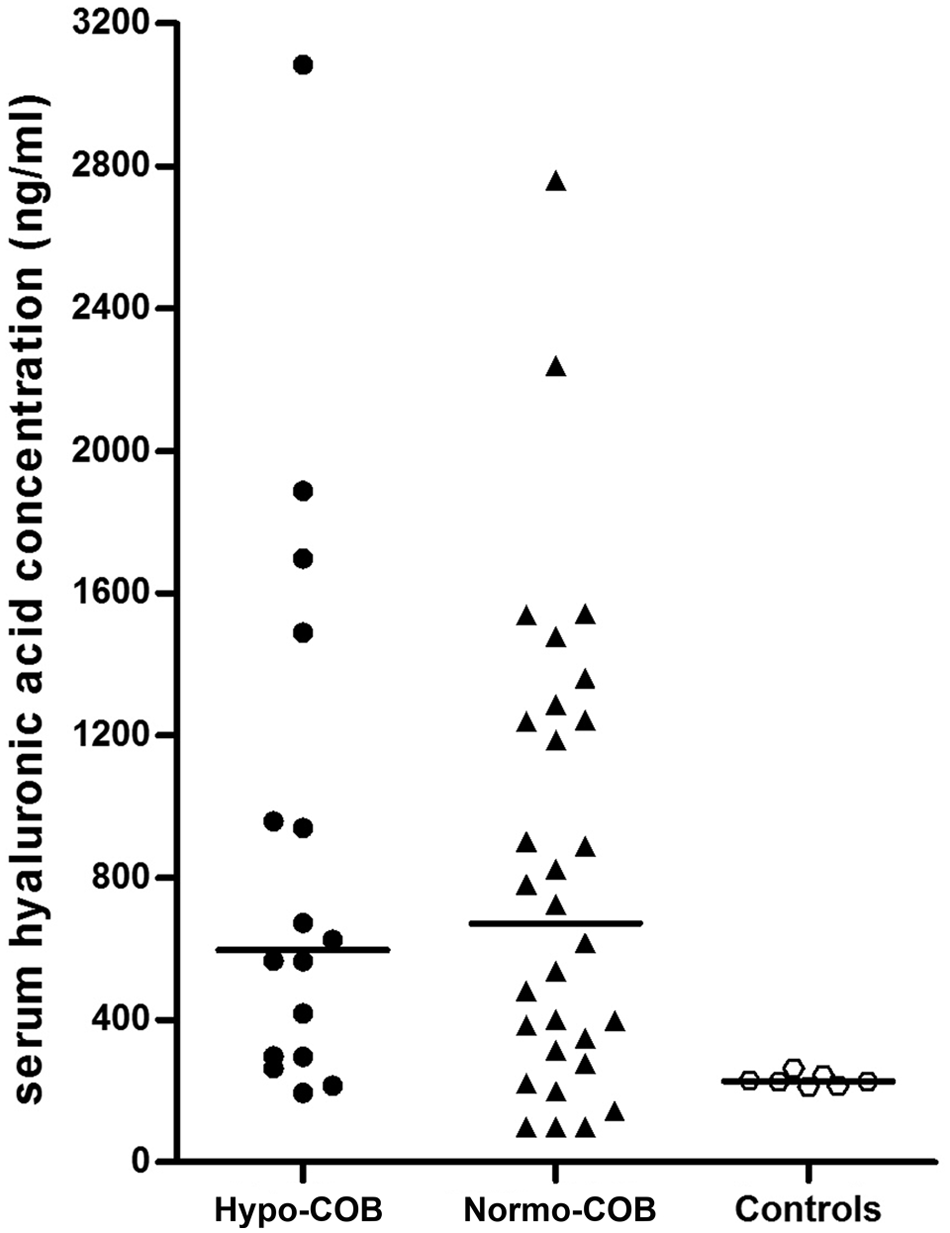
Comparison of serum hyaluronic acid concentrations between hypocobalaminemic (Hypo-COB) Chinese Shar-Peis, normocobalaminemic (Normo-COB) Chinese Shar-Peis, and healthy German Shepherd Dogs (Controls).
Serum markers of chronic intestinal disease
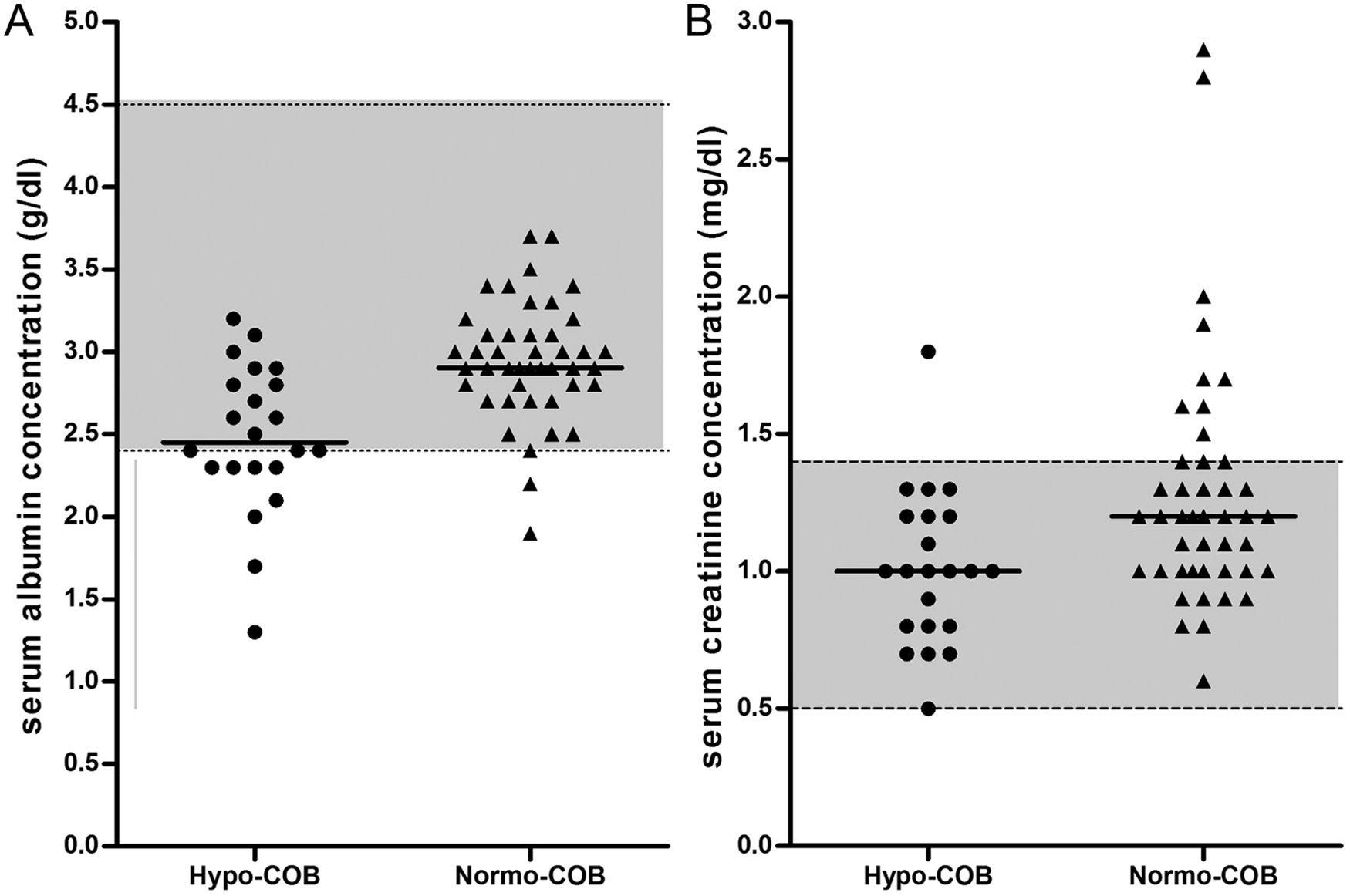
Serum concentrations of albumin were significantly lower in hypocobalaminemic Shar-Peis when compared with normocobalaminemic Shar-Peis (Table 2; Fig. 3). Approximately 57% (n = 8) of hypocobalaminemic Shar-Peis and 4.5% (n = 2) of normocobalaminemic Shar-Peis had a serum albumin concentration below the lower limit of the reference interval (<2.4 g/dl), and hypocobalaminemia was significantly associated with hypoalbuminemia (OR: 12.0, 95% CI: 2.8–63.4; P = 0.0015). A low (<2.4 g/dl) serum albumin concentration distinguished hypocobalaminemic Shar-Peis from normocobalaminemic Shar-Peis with a sensitivity of 36% and specificity of 96%. Hypocobalaminemic Shar-Peis also had lower serum zinc concentrations than normocobalaminemic Shar-Peis, but this difference was not significant (Table 2; Fig. 4). Serum concentrations of α1PI were not different between hypocobalaminemic and normocobalaminemic Shar-Peis (Table 2).
Comparison of (
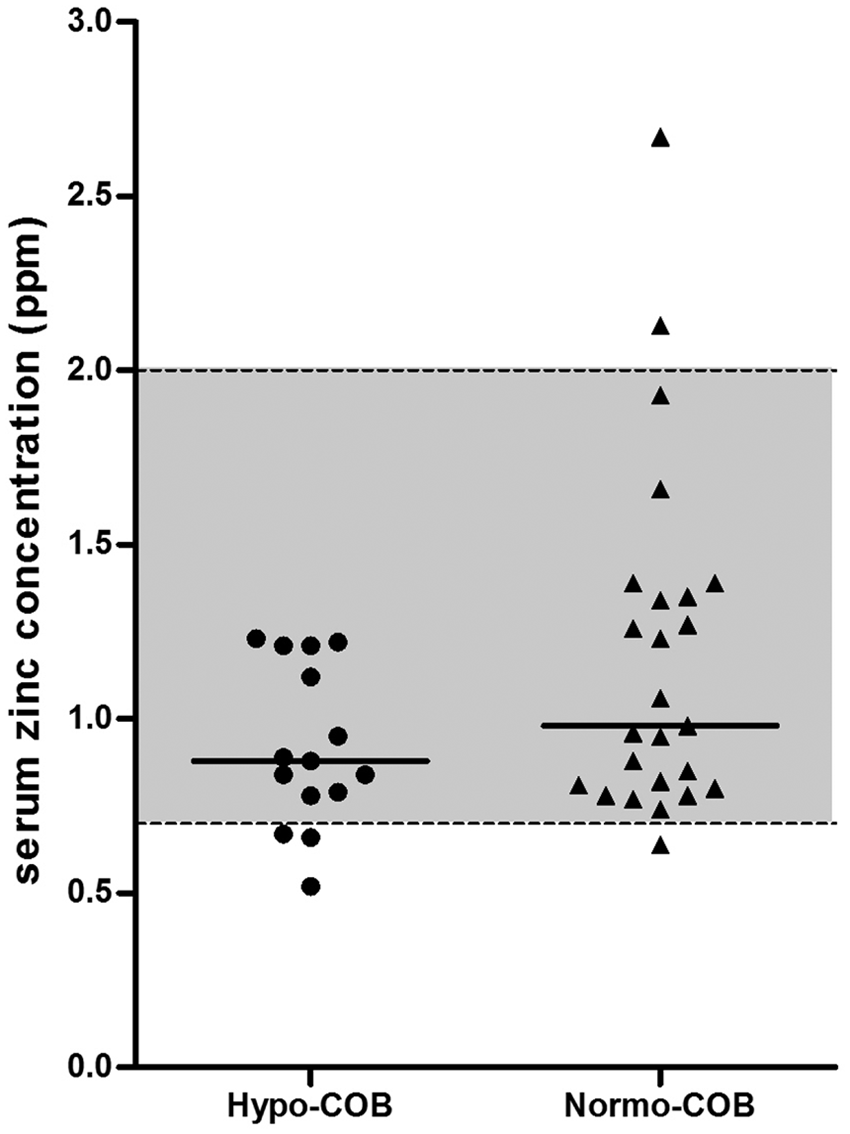
Comparison of serum zinc concentrations between hypocobalaminemic (Hypo-COB) and normocobalaminemic (Normo-COB) Chinese Shar-Peis. The dashed horizontal lines indicate the limits of reference interval (0.7–2.0 ppm) for serum zinc concentrations in dogs.
Serum immunoglobulin and creatinine concentrations
Serum IgA concentrations were significantly higher in hypocobalaminemic Shar-Peis than in normocobalaminemic Shar-Peis (Table 2; Fig. 5). In contrast, serum IgM concentrations were significantly lower in hypocobalaminemic Shar-Peis than in normocobalaminemic Shar-Peis (Table 2; Fig. 5). The IgA-to-IgM ratio was significantly higher in hypocobalaminemic Shar-Peis (median: 2.3, range: 0.6–7.8) than in normocobalaminemic Shar-Peis (median: 0.40, range: 0.1–1.9; P < 0.0001), and hypocobalaminemia was associated with an IgA-to-IgM ratio of >1.5 (OR: 88.4, 95% CI: 9.4–830.9; P < 0.0001). A high (>1.5) IgA-to-IgM ratio distinguished hypocobalaminemic Shar-Peis from normocobalaminemic Shar-Peis with a sensitivity of 72% and a specificity of 97%.
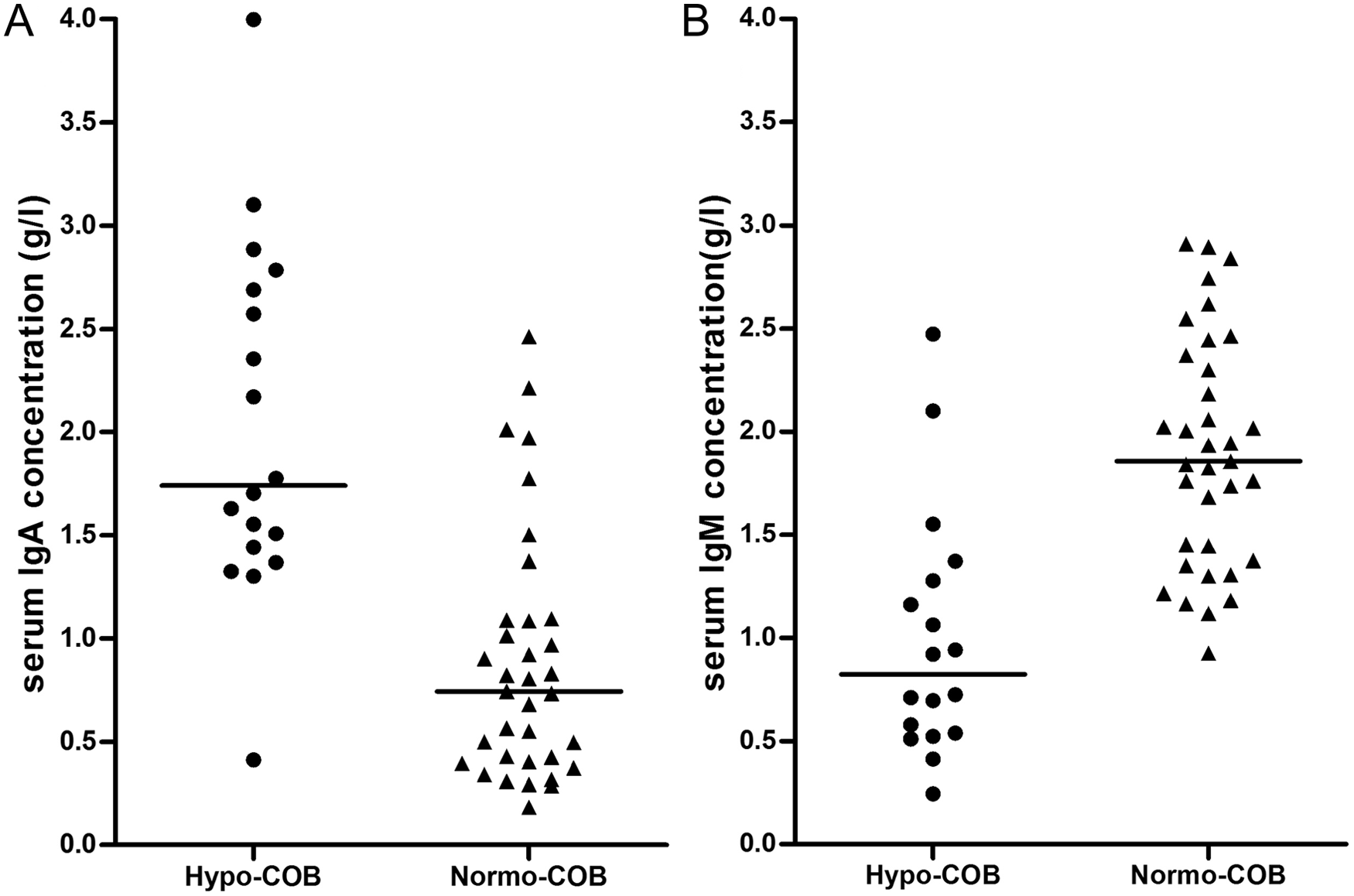
Comparison of (
Concentrations of serum creatinine were significantly lower in hypocobalaminemic Shar-Peis when compared to normocobalaminemic Shar-Peis (Table 2; Fig. 3). However, this difference did not appear clinically relevant because normocobalaminemia was not associated with hypercreatininemia (OR: 0.19, 95% CI: 0.02–1.57; P = 0.1459).
Discussion
The Shar-Pei has been described as having a high prevalence of marked hypocobalaminemia, and clinical signs of hypocobalaminemic Shar-Peis are suggestive of severe and longstanding gastrointestinal disease such as diarrhea, vomiting, and/or weight loss. 4 The current study assessed serum concentrations of inflammatory markers, markers for chronic intestinal disease, and immunological markers in Shar-Peis with and without hypocobalaminemia, and whether an inflammatory phenotype exists in Shar-Peis with hypocobalaminemia.
Serum concentrations of the inflammatory markers CRP, the calgranulins (i.e., CP and S100A12), and HA did not differ significantly between hypocobalaminemic and normocobalaminemic Shar-Peis. This finding might suggest that hypocobalaminemia in Shar-Peis is not associated with other potential diseases in this breed, such as cutaneous mucinosis and Shar-Pei fever. However, further studies are needed to determine serum cobalamin concentrations in Shar-Peis with confirmed cutaneous mucinosis and/or Shar-Pei fever.
It is interesting to note that all 3 inflammatory markers (CRP, and the calgranulins [i.e., CP and S100A12]) were increased in both hypocobalaminemic and normocobalaminemic Shar-Peis. Serum CRP concentrations were increased above the upper limit of the reference interval in 29% and 31%, respectively, whereas 50% and 44% of the hypocobalaminemic and normocobalaminemic Shar-Peis, respectively, had serum CP concentrations above the suggested upper reference limit. In contrast, we found serum S100A12 concentrations to be more frequently increased above the suggested reference limit in hypocobalaminemic Shar-Peis (43%) compared with normocobalaminemic Shar-Peis (24%), although this difference did not reach significance (P > 0.05; data not shown). The increase in inflammatory markers in the current study in both hypocobalaminemic and normocobalaminemic Shar-Peis would suggest that an inflammatory phenotype exists in both groups of Shar-Peis and is not associated with hypocobalaminemia in this breed. The high proportion of dogs with a serum CP concentration above the reference interval in the groups of Shar-Peis with and without hypocobalaminemia is interesting because increased serum CP concentrations have also been observed in dogs with chronic enteropathies. 15 However, further studies to investigate serum CRP and calgranulins in dogs (e.g., Shar-Peis) with chronic enteropathies and diseases other than chronic enteropathies (e.g., sepsis or arthritis) are currently underway and needed.
Hyperhyaluronic acidemia was not associated with hypocobalaminemia in Shar-Peis in the present study. It is possible that the increased HA concentrations in both Shar-Peis with and without hypocobalaminemia reflect a high production of cell surface HA on mucosal endothelial cells as has been documented in human patients with IBD. 21 Although only a few control dogs (healthy German Shepherd Dogs) were used in the current study, serum HA concentrations in these dogs were comparable to historical controls (median: 244.12 μg/l) and dogs with hepatic diseases (median: 59.17 ng/ml 35 ; Seki M, et al.: 2008, Clinical significance of serum hyaluronic acid in canine hepatic diseases), while Shar-Peis with and without hypocobalaminemia had much higher serum HA concentrations similar to Shar-Peis reported by others. 35 Elevated serum HA concentrations together with increased concentrations of the calgranulins, and especially CP, in serum from both Shar-Peis with and without hypocobalaminemia could be speculated to reflect compromised gastrointestinal health. For instance, HA deposition within the gastrointestinal tract can lead to low-grade inflammation and malabsorption of vitamins such as cobalamin. However, further studies with larger groups of dogs are needed to confirm this hypothesis.
Hypocobalaminemic Shar-Peis had lower serum albumin concentrations, and hypoalbuminemia was more frequently detected in hypocobalaminemic compared to normocobalaminemic Shar-Peis. This result appears of significance given that hypocobalaminemia in dogs with chronic enteropathies has been shown to be associated with hypoalbuminemia.1,15 Hypoalbuminemia could also occur due to renal protein loss, 7 and chronic kidney disease is frequently associated with an increased serum creatinine concentration. Chronic kidney disease as a cause of hypoalbuminemia cannot be definitively ruled out but seems rather unlikely given that, in 21 (95%) hypocobalaminemic Shar-Peis, the serum creatinine concentration was within the reference interval. In human beings, hypoalbuminemia has also been associated with the inflammatory process (negative acute phase protein) or amyloidosis 29 and an increased concentration of protease inhibitors (e.g., α1PI) has been hypothesized to contribute to the pathogenesis of amyloidosis. 31 Along these lines, we have shown in a previous study that hypoalbuminemia is associated with a decreased serum α1PI concentration in hypocobalaminemic Yorkshire Terriers and hypothesized that serum α1PI concentrations might have prognostic implications in dogs with chronic small intestinal disease. 11 However, in the present study, serum concentrations of α1PI were not significantly different between hypocobalaminemic and normocobalaminemic Shar-Peis suggesting that hypocobalaminemia in Shar-Peis is not associated with altered serum α1PI concentrations. Because albumin and α1PI are assumed to be lost through the gastrointestinal tract at the same rate and because α1PI was not shown to be an acute phase protein in dogs,6,9 the altered availability of albumin and α1PI in serum raises the question of whether another source of α1PI (such as inflammatory cells) might have an impact on the serum α1PI concentration. Therefore, further studies are needed to investigate the relationship of serum albumin and serum as well as fecal α1PI in Shar-Peis and in dogs with chronic gastrointestinal disease and gastrointestinal protein loss.
Serum concentrations of creatinine were significantly lower, albeit numerically only slightly different, in hypocobalaminemic Shar-Peis compared with normocobalaminemic Shar-Peis. Increased serum creatinine concentrations have been observed in 1 (5%) hypocobalaminemic Shar-Pei and in 9 (20%) normocobalaminemic Shar-Peis. However, hypocobalaminemia was not significantly associated with normocreatininemia, and the difference between the median serum creatinine concentrations in both groups was less than the critical difference in creatinine that is required to be considered clinically relevant. 18 A study that investigated renal amyloidosis in dogs showed that serum creatinine concentrations were 3-fold higher in Shar-Peis with renal amyloidosis compared to non–Shar-Peis with renal amyloidosis. 28 Furthermore, the study revealed that hypoalbuminemia occurs more often in non–Shar-Peis with renal amyloidosis than in Shar-Peis with renal amyloidosis. 28 Based on the results of the present study, it may be speculated that hypocobalaminemia in Shar-Peis is likely not associated with renal amyloidosis. However, given the lack of urine samples and/or renal biopsies for analysis and further phenotypic characterization of the Shar-Peis enrolled in this study, the results of the present study do not allow any definitive conclusions.
Amyloid deposition has been reported in Shar-Peis and non–Shar-Peis with renal amyloidosis. 28 Because extrarenal amyloid depositions have been shown to more commonly occur in Shar-Peis compared to non–Shar-Peis with renal amyloidosis, it is possible that amyloid deposition in the gastrointestinal tract, pancreas, and central nervous system 28 affects cobalamin metabolism and/or malabsorption of nutrients such as cobalamin and zinc. Concurrently, serum zinc concentrations were also numerically lower in hypocobalaminemic Shar-Peis compared with normocobalaminemic Shar-Peis, although this difference was not significant. Thus, investigation of the effect of intramural amyloid and HA deposition within the gastrointestinal tract on malabsorption of certain nutrients in Shar-Peis with chronic enteropathies warrants further research.
Hypocobalaminemic Shar-Peis had higher serum IgA concentrations and, in contrast, lower serum IgM concentrations than in normocobalaminemic Shar-Peis. The results of the current study are in contrast to lower serum IgA concentrations in Shar-Peis as reported previously.23,25 This finding supports the indication that IgA deficiency 23 and the primary immunodeficiency syndrome in Shar-Peis 25 are not associated with hypocobalaminemia in Shar-Peis. Interestingly, in human patients with plasma cell dyscrasias, a link between serum IgA and the pathogenesis of cobalamin deficiency has been suggested in that IgA may have an anti–intrinsic factor-like activity or be involved in other mechanisms that have an impact on normal cobalamin absorption. 3 In dogs, high serum IgA concentrations have been observed in patients with inflammatory diseases such as meningitis–arteritis. 27 Although speculative, an increased serum IgA concentration could be caused by a breakdown of mucosal immunity and microbe tolerance, and may argue for an inflammatory phenotype in hypocobalaminemic Shar-Peis. Along these lines, in serum, increased IgA and decreased IgM concentrations have been described in human beings with type 2 diabetes mellitus when compared with healthy controls, and this finding was considered to be due to low-grade systemic inflammation. 5 Similar trends for these 2 immunoglobulins, best reflected by the IgA-to-IgM ratio, were seen in hypocobalaminemic Shar-Peis in the present study. However, these results warrant further investigation of the gastrointestinal immunoglobulin secretion in Shar-Peis with hypocobalaminemic.
It is important to acknowledge the limitation of this study. During the process of collecting the samples for both groups, it was not possible to conduct biochemistry, hematology, and urine analysis. That data would have been useful to connect our findings to other potential diseases in this breed. Nevertheless, the present results might lead to cobalamin analysis being considered in research investigations with Shar-Peis that have been diagnosed with cutaneous mucinosis and/or Shar-Pei fever.
In summary, Shar-Peis have a high prevalence of marked hypocobalaminemia, and clinical signs are suggestive of severe and longstanding gastrointestinal disease such as diarrhea, vomiting, and/or weight loss. No difference was found in serum concentrations of CRP, CP, S100A12, and HA between Shar-Peis with and without hypocobalaminemia. However, the results of the current study suggest that an inflammatory phenotype may exist in both hypocobalaminemic and normocobalaminemic Shar-Peis. Hypocobalaminemic Shar-Peis also had higher serum IgA concentrations, lower serum IgM concentrations, and serum albumin concentrations compared with normocobalaminemic Shar-Peis. These findings may suggest that hypocobalaminemia in Shar-Peis is not associated with other highly prevalent diseases in Shar-Peis (such as cutaneous mucinosis and Shar-Pei fever). However, further studies are needed to determine serum cobalamin concentrations in Shar-Peis with confirmed cutaneous mucinosis and/or Shar-Pei fever. Future research also needs to clarify if hypocobalaminemia is a cause or consequence of chronic gastrointestinal disease in Shar-Peis.
Footnotes
Acknowledgements
Presented in part at the Annual Forum of the American College of Veterinary Internal Medicine in Montreal, Quebec, Canada (June 2009), and in Denver, Colorado (June 2011).
a.
Immulite 2000, Siemens Healthcare Diagnostics Inc., Deerfield, IL.
c.
C-reactive protein, ELISA kit; Tridelta Development Ltd., Maynooth, Ireland.
e.
Hyaluronic acid, ELISA kit, Echelon Biosciences, Salt Lake City, UT.
f.
Albumin and creatinine, automated clinical chemistry analyzer; Stanbio Laboratory, Boerne, TX.
g.
Texas Veterinary Medical Diagnostic Laboratory, College Station, TX.
h.
IgA and IgM ELISA kits, Bethyl Laboratories Inc., Montgomery, TX.
i.
GraphPad Prism5, GraphPad Software Inc., La Jolla, CA.
Declaration of conflicting interests
Romy Heilmann, Jan Suchodolski, and Jörg Steiner have submitted a patent application that includes the S100A12 immunoassay used in this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
