Abstract
Five horses originating from 4 different California race tracks were submitted to the California Animal Health and Food Safety Laboratory for necropsy and diagnostic workup. The 5 horses had a history of sudden collapse and death during exercise. In all of them, massive hemoperitoneum and hemorrhages in other cavities or organs were observed. The liver from these 5 animals and from 27 horses that had been euthanized due to catastrophic leg injuries (controls) were subjected to a rodenticide anticoagulant screen. Traces of brodifacoum, diphacinone, or bromadiolone were detected in the 5 horses with massive bleeding (5/5), and no traces of rodenticides were detected in control horses (0/27). Other frequent causes of massive hemorrhages in horses were ruled out in 4 of the cases; one of the horses had a pelvic fracture. Although only traces of anticoagulants were found in the livers of these horses and the role of these substances in the massive bleeding remains uncertain, it is speculated that exercise-related increases in blood pressure may have reduced the threshold for toxicity of these anticoagulants.
In 2014, anticoagulant rodenticides (ARs) that are currently available include first-generation ARs (FGARs) such as warfarin, diphacinone, and chlorophacinone, and second-generation ARs (SGARs) including brodifacoum, bromadiolone, difethialone, and difenacoum. Second-generation ARs were developed to counter resistance of rodent target species to warfarin; such rodenticides are more toxic and have longer half-lives than FGARs. 10 The popularity of ARs for rodent pest control, particularly SGARs, has led to an increased number of cases of accidental poisoning in wild animals, farm animals, and pets. 16 Animals intoxicated with ARs frequently develop hemorrhagic diathesis, and are occasionally found dead without premonitory clinical signs being observed. 5 The main effect of these substances is due to inhibition of the prothrombin complex (coagulation factors II, VII, IX, and X), as a result of interference with the action of vitamin K.
Several of the FGARs have medical utility in human beings and other animal species. In horses, some of these compounds have shown limited effectiveness in the treatment of thrombotic diseases or diseases suspected of being of thrombotic origin, such as navicular disease, thrombophlebitis, chronic laminitis, disseminated intravascular coagulation, and verminous aneurysm.2,3,5 However, the use of these substances is not without risks. Complications include epistaxis, gastrointestinal bleeding disorders, and susceptibility to hematomas. 17 Few cases of AR intoxication have been reported in horses, and all of these were individual cases that had clinical coagulopathy and were associated with the consumption of large amounts of bait, but none of those presented with massive hemorrhages.1,9
A race horse with a history of sudden death was submitted for postmortem examination and diagnostic work up. Necropsy findings included massive hemoperitoneum and bleeding in the thorax and pericardium. An AR screen revealed the presence of diphacinone in the liver. This prompted routine testing for ARs in cases of exercise-related, extensive fatal hemorrhage, and 4 more horses tested positive for the AR screen in a relatively short period of time. We present here the results of that investigation, which includes a cluster of 5 cases of acute death occurring during or immediately after strenuous exercise, associated with massive bleeding in body cavities, and presence of traces of ARs in the liver.
Five dead geldings were received in a 1-year period from 4 different race tracks in California. Information on signalment and clinical history of individual horses is presented in Table 1. Briefly, 4 Thoroughbreds and 1 Quarter horse of ages between 3 and 16 years were studied. Three of the horses died during a race, a fourth one died during training, and the 16-year-old died while working as a pony horse accompanying a race horse during its warmup prior to the race (ponying).
Clinical history and main necropsy findings in 5 horses with traces of anticoagulant rodenticides.
A full necropsy was performed on all 5 horses. Samples of liver, spleen, kidneys, lungs, heart, gastrointestinal tract, skeletal muscle, and adrenal gland, plus the whole brain were collected for histology. The samples were fixed by immersion in 10% buffered formalin (pH 7) for a minimum of 24 hr. The whole brains were fixed for approximately 1 week after which they were coronally sliced at approximately 0.5-cm intervals and immersed in fresh formalin for an additional week. Subsamples from parietal cortex, corpus striatum, thalamus, midbrain at the level of anterior colliculi, pons, cerebellar peduncles, cerebellum, and medulla at the level of the obex were obtained. All samples were processed routinely for histology to obtain hematoxylin and eosin–stained, 4-μm-thick sections.
A heavy metal screen for 9 metals (lead, manganese, iron, mercury, arsenic, molybdenum, zinc, copper, and cadmium) was performed on livers of 3 of the horses by inductively coupled plasma–atomic emission spectrometry (ICP-AES). a Briefly, a 1-g liver sample was digested overnight in a mixture of nitric and hydrochloric acids at 70°C. After cooling to room temperature, the digested samples were treated with a mixture of hydrochloric acid and water, and introduced into the nebulizer of the ICP. Each metal was individually detected and quantitated by comparison with an external calibration solution. The quality of the analytical run was controlled through the use of Standard Reference Materials (SRMs) containing certified concentrations of each analyte. The reporting limits in tissues for these metals are 1 ppm for lead, mercury, and arsenic; 0.4 ppm for molybdenum; 0.3 ppm for cadmium; 0.2 ppm for iron; 0.1 ppm for zinc and copper; and 0.04 ppm for manganese.
Liver samples from the 5 horses and liver samples from 27 racehorses euthanized due to catastrophic musculoskeletal injuries, were also analyzed for warfarin, coumachlor, bromadiolone, brodifacoum, diphacinone, chlorophacinone, and difethialone using a previously published method modified for tissue analysis. 11 Briefly, samples were initially screened for the presence of ARs by liquid chromatography–tandem mass spectrometry (LC-MS/MS). b According to this method, any positive anticoagulant sample was then quantitated by high-performance liquid chromatography (HPLC) c using either ultraviolet diode array detection d (diphacinone, chlorophacinone, and difethialone) or fluorescence detection e (warfarin, coumachlor, bromadiolone, and brodifacoum). Limits of quantitation for these anticoagulants vary according to their sensitivity to ultraviolet or fluorescence detection. In tissue, such limits are 0.01 ppm for brodifacoum; 0.05 ppm for bromadiolone, warfarin, and coumachlor; and 0.25 ppm for chlorophacinone, diphacinone, and difethialone. Anticoagulants that were determined to be positive by LC-MS/MS, but were below the reporting limit by HPLC, were defined as trace. For dicoumarol determination, 5 g of liver from all horses were diluted in acidic water and extracted with ethyl acetate. The extract was passed through a solid phase extraction column f and analyzed using HPLC with diode array detection. Quantification of dicoumarol is done by comparing any sample peak with a standard curve utilizing dicoumarol standard material. The method reporting limit is 0.05 ppm.
In addition, several other tests were performed following California Animal Health and Food Safety Laboratory System standard operating procedures, including brain cholinesterase activity (horses 1 and 3), Salmonella sp. polymerase chain reaction on liver and small intestine (horses 1–3), aerobic culture on liver and/or lung (horses 1–3), and Sarcocystis neurona immunohistochemical staining (horse 1).
The proportion of horses with hemoperitoneum that had detectable AR in the liver was compared to the proportion of 27 control horses that had detectable AR in the liver using Fisher exact test. g A summary of gross, histological, and toxicological results is presented in Table 1. Briefly, all 5 horses had hemoperitoneum (Fig. 1) and at least 2 of the following changes: hemothorax, hemopericardium, and mesenteric, muscular, diaphragmatic, and/or pulmonary hemorrhages. The hemoperitoneum, hemothorax, and hemopericardium were characterized by the presence of large quantities (approximately 8–20 liters in the abdomen, 4–10 liters in the thorax, and 0.5 liters in the pericardial sac) of mostly unclotted blood free in the corresponding cavity. Detailed dissection and gross examination of the main thoracic, abdominal, and pelvic blood vessels did not reveal the presence of tears or other vascular lesions. Horse 1 had acute pelvic fracture and extensive hemorrhages.
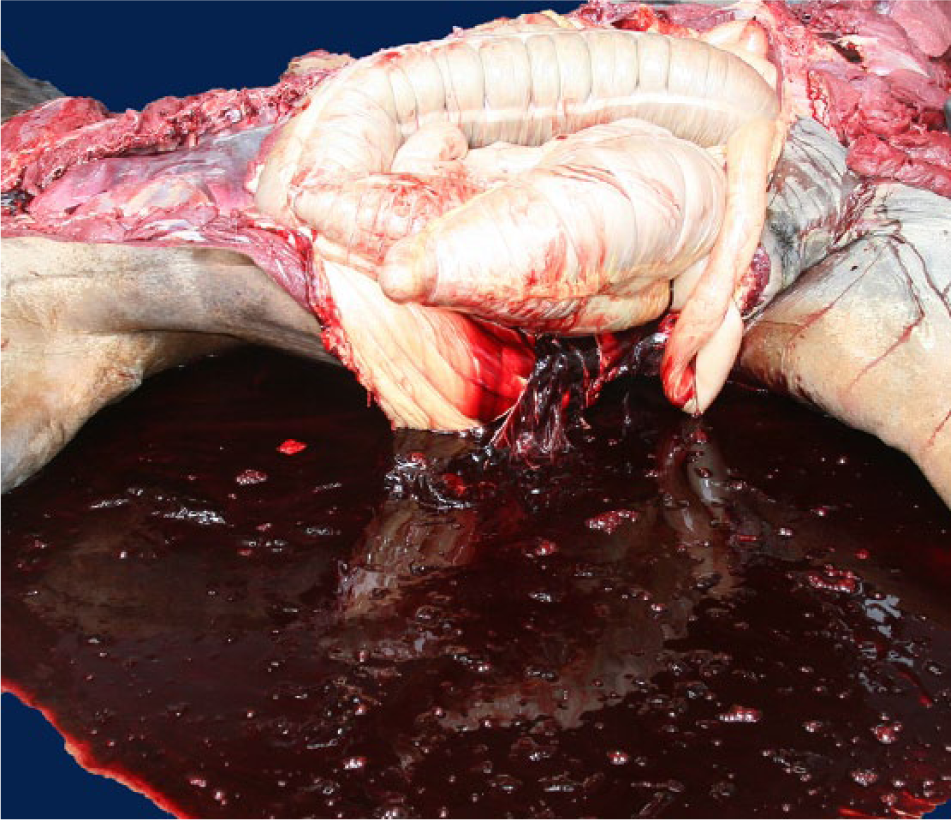
Horse 3. Large amounts of mostly unclotted blood are present in the abdominal cavity.
Histological examination was unremarkable in horse 3, and 1 or more of the following changes were evidenced in the other 4 horses: acute pulmonary congestion and edema (horses 2 and 4), acute pulmonary hemorrhages (horse 5), splenic hemosiderosis (horses 2 and 3), mild multifocal cardiac myofiber hyper contraction bands (horse 3), mild focal arterial mineralization in anterior mesenteric artery (horse 3), and mild and focal cerebral perivascular cuffing characterized by lymphocytes surrounding a few veins of the basal ganglia (horse 3). No other histological abnormalities were observed in other tissues of the 5 horses examined. These changes were considered incidental and most likely not associated with the cause of death.
Traces of brodifacoum were found in the liver of horses 1–3, whereas traces of diphacinone were found in horse 4, and traces of bromadiolone were detected in horse 5. No traces of ARs were detected in any of the control horses. The detected concentrations of all of these ARs were below the laboratory reporting limits and were therefore not quantified and reported as trace amounts. The probability of detectable AR was significantly (P < 0.0001) higher in horses with hemoperitoneum than in control horses.
Hydroxyethyl promazine sulfoxide, the major metabolite of the tranquilizer acepromazine, was found in aqueous humor of horse 1. Liver heavy metals and brain cholinesterase activity were within normal limits in the horses tested. All other tests performed were unremarkable or negative.
The most significant finding in all 5 horses in the current study was severe hemoperitoneum, accompanied by a variable combination of hemothorax, hemopericardium, and mesenteric, muscular diaphragmatic, and/or pulmonary hemorrhages, associated with the presence of traces of AR in the liver. Causes of hemoperitoneum previously reported in animals include traumatic rupture of the liver, spleen, and kidneys; tumors of liver, spleen, and ovaries; manual ablation of the corpus luteum; rupture of major arteries; mesenteric injury; intestinal migration of Strongylus vulgaris larvae; thrombocytopenia; and intoxication with anticoagulant rodenticides.4,8,15 In horses, the most frequent causes of hemoperitoneum are trauma, neoplasia, uterine artery rupture, and mesenteric injury. Hemoperitoneum of undetermined cause (idiopathic) has also been frequently described in horses, and it is either the most important or the second most important form of hemoperitoneum described in this species.6,7 Because only traces of AR were found in the horses of the present study, we cannot completely rule out that the cases presented herein were idiopathic, and that the presence of AR in these horses represented only incidental findings.
With the exception of AR intoxication and thrombocytopenia, all of the causes mentioned above were ruled out based on gross and/or microscopic examination of the tissues. Although thrombocytopenia cannot completely be ruled out, it is unlikely that this was the cause of hemorrhage in these horses because this finding is more commonly observed associated with disseminated intravascular coagulation and some disseminated neoplasias, 2 neither of which were seen in these horses. Horse 1 had a pelvic fracture and although no torn blood vessels were found, we cannot completely rule out that a broken bone fragment produced a vascular tear that was not detected during postmortem examination.
The trace amounts of AR detected in the liver of the 5 horses with massive internal hemorrhages, the absence of AR in 27 control horses, and the elimination of other causes of internal hemorrhages suggest an association between AR exposure and acute fatal internal bleeding into the body cavities, particularly the abdominal cavity. Because all 5 horses presented with sudden death, no antemortem coagulopathy testing could be performed. However, because it was thought that it was possible that more horses were subclinically affected with AR poisoning, partial thromboplastin time (PTT) and prothrombin time (PT) was performed on 30 clinically healthy horses from the same stable of horse 4, and the results were unremarkable (data not shown). This suggests that the traces of AR found in the cases presented herein were individual medical conditions in a few horses and not a population problem. The origin of the AR could not be determined. Investigation of the AR used for pest control in 2 of the stables did not reveal that the drugs detected in the animals had been used.
Despite the statistically significant association between severe hemorrhage and detection of trace amounts of anticoagulants in these horses, it is unlikely that these ARs were solely responsible for the severe hemorrhages observed, although the threshold for diagnostic liver concentrations of ARs has not been determined for horses or other species. It is possible that other unknown factors acted as predisposing factors or worked synergistically with the AR to produce those hemorrhages. The 5 horses died while undergoing exercise, suggesting there is a common factor or factors triggered by exercise. The nature of the potential associated factor(s) triggered during exercise is unknown, but it is perhaps related to increase in blood pressure and/or release of vasoactive amines or other chemical mediators that may result in a lowered intoxication threshold for the ARs detected in these animals.
A number of drugs have been suggested to increase the toxic effects of warfarin and other ARs in horses, including anti-inflammatory drugs, such as phenylbutazone and oxyphenbutazone, and other drugs such as heparin, phenytoin, salicylates, quinidine, potentiated sulfas, and steroid hormones.9,14 Two of the horses in the current study had been reported to have received phenylbutazone within 48 hr before their deaths and, although there is no history of phenylbutazone administration in other horses, it is common practice to administer this drug to horses prior to racing. It may be possible that these drugs also contribute to lowering the intoxication threshold for the ARs identified in these animals.
Intoxication with warfarin-based AR has been occasionally described in horses, with disseminated hemorrhagic lesions in the subcutis, muscles, pericardial sac, meninges, liver, kidneys, gastrointestinal tract, and urinary bladder.1,9 Hemoperitoneum was not mentioned in any of the animals included in the 2 studies referred to. However, in all of those animals that survived, it is possible that the horses had hemoperitoneum but that it went undetected. Abdominal distention is not a frequent clinical finding in animals with that condition, even with massive amounts of blood in the abdomen. 12
Anticoagulant rodenticides are the most widely used pesticides to control rodents, although there is an ongoing effort to reduce their availability and use due to non–target animal exposures (U.S. Environmental Protection Agency: 2008, Risk mitigation decision for ten rodenticides. Available at: www.epa.gov/oalj/filings/Reckitt_HrgReq_Ex03.pdf). Domestic animals are usually accidentally intoxicated by ingestion of baits that are made highly palatable by the addition of saccharose.3,16 Brodifacoum and bromadiolone are SGARs, whereas diphacinone is a less toxic and less persistent FGAR. Regardless of the group they belong to, these compounds inhibit vitamin K1 epoxide reductase activity in the liver, thereby reducing circulating vitamin K–dependent clotting factors II, VII, IX, and X. The high potency of brodifacoum in the horse has been reported, in which the ingestion of 0.125 mg/kg body weight (approximately 1 kg of commercial bait per horse) resulted in depression, anorexia, weight loss, plus severe hypocoagulability and hemogram alterations, including low hematocrit, hemoglobin, erythrocyte, and platelet counts. 3
In wildlife, the presence of ARs at various concentrations is frequently identified, and most of these animals do not die of AR toxicity. Little is known about the sub-lethal, chronic effects of these compounds, and what kind of interactions may occur between these compounds and other factors. 13
In conclusion, the results of the current study suggest that there may be a clinically significant association between strenuous exercise-related, acute, fatal hemorrhages in body cavities and the detection of trace amounts of AR in horses. It is likely that other yet unknown risk factors may play a synergistic role and contribute to internal bleeding.
Footnotes
Acknowledgements
The authors acknowledge Dr. Scott Stanley for providing the results obtained at the Equine Analytical Chemistry Laboratory of the California Animal Health and Food Safety Laboratory System.
a.
Fisons Accuris, Applied Research Laboratories, Palm Beach, FL.
b.
Thermo LXQ, Thermo Scientific, San Jose, CA.
c.
Agilent 1100 series, Agilent Technologies Inc., Santa Clara, CA.
d.
Diode array detector (Agilent G1315B), Agilent Technologies Inc., Santa Clara, CA.
e.
Fluorescence detector (Waters 474), Waters Corp., Milford, CA.
f.
Florisil, JT Baker, Center Valley, PA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
