Abstract
Pine needle abortion is a naturally occurring condition in free-range cattle caused by the consumption of pine needles from select species of cypress, juniper, pine, and spruce trees. Confirmatory diagnosis of pine needle abortion has previously relied on a combined case history of pine needle consumption and detection of isocupressic acid in a sample from the dam. Stable metabolites of isocupressic acid include agathic acid, dihydroagathic acid, and tetrahydroagathic acid, which have been shown to be present in the serum of mature animals for a few days following consumption of pine needles. As maternal serum is infrequently submitted for diagnosis of cattle abortions, a diagnostic assay capable of confirming isocupressic acid exposure in other matrices would be desirable. To the authors’ knowledge, no previous investigations have indicated whether these stable metabolites of isocupressic acid cross the placenta or are detectable in fetal tissues. Therefore, the presence of agathic acid, dihydroagathic acid, and tetrahydroagathic acid was evaluated using gas chromatography–mass spectroscopy on fetal thoracic fluid and stomach contents collected from 2 aborted bovine fetuses with a recent herd history of pine needle consumption by the dams and a subsequent abortion outbreak in the herd. Only tetrahydroagathic acid was detected in the fetal thoracic fluid and fetal stomach contents. The current study encourages diagnosticians to collect fetal thoracic fluids to permit the detection of tetrahydroagathic acid in cases of suspected pine needle abortion.
The first circumstantial reports of pine needle abortion (PNA) occurred in Canadian cattle in the 1920s (Bruce EA: 1927, Astragalus serotinus and other stock poisoning plants of British Columbia. Dominion of Canada, Department of Agriculture Bulletin 88). Additional suspected cases of PNA in Canadian cattle were reported in the 1940s and 1950s. 15 Later experimentation reproduced signs of PNA by feeding pine needles from ponderosa pine (Pinus ponderosa) and lodgepole pine (Pinus contorta) trees 15 although the authors of the experiment later determined that some pine needles may be safe for consumption. Pine needle abortion was well-characterized in the 1970s as an inducible clinical syndrome following the consumption of pine needles from ponderosa pine trees. 21 Consumption of conifer foliage has been associated with clinical signs of third trimester abortion, 16 stillbirths, weak calves, and retained placental membranes.15,19 Later experimentation in the 1990s has characterized the physiological effects of PNA.2,3,5 The current economic impact of cattle abortions and death loss due to PNA is estimated to cost the U.S. cattle industry between $4.5 million 13 and $20 million annually (Miner JL, Staigmiller RB, Peterson MK, et al.: 1987, Montana pine needles cause abortion in beef cattle. Montana Agricultural Experiment Station, Montana State University, Bozeman, MT, vol. 4, pp. 6–9).
Several species of conifer trees in North America have been implicated as a cause of PNA, either circumstantially or by virtue of isocupressic acid (ICA) content in foliage or bark. Such species include Monterey cypress (Cupressus macrocarpa), 19 common juniper (Juniperus communis), 9 Formosan juniper (Juniperus formosana),6,9,10 Utah juniper (Juniperus osteosperma), 12 Rocky Mountain juniper (Juniperus scopulorum),6,9,10 Engelmann spruce (Picea engelmannii), 8 lodgepole pine (Pinus contorta),9,11 pinyon pine (Pinus edulis and Pinus monophylla), 8 Jeffrey pine (Pinus jeffreyi),10,11 ponderosa pine (Pinus ponderosa), 20 and Monterey pine (Pinus radiata). 8 Historically, cattle have been known to consume conifer foliage when limited forage is available. Cattle in mountainous regions of the western United States, in particular, may become stranded for prolonged periods without access to adequate forage during inclement weather conditions. Forage specialists in Montana have concluded that diminished alternative forage availability, snow cover, inclement weather, and reduced grazing time increases consumption of pine needles. 17
Diterpene labdane acids such as ICA have been identified in the needles of select species of cypress, pine, juniper, and spruce trees at concentrations known to produce an abortifacient effect.6,7,23 After consumption of pine needles, ICA and oxidative metabolites of ICA are readily absorbed into the bloodstream where they are further oxidized by hepatic dehydrogenase enzymes resulting in detectable serum concentrations of agathic acid (AA), dihydroagathic acid (DHAA), and tetrahydroagathic acid (THAA) for a few days. 8 Agathic acid,6,8,14,20 DHAA,6,8,14,20 and THAA 8 have been consistently demonstrated in the serum of cows following either intravenous injection with ICA or consumption of ICA-containing pine needles.6,14,20 Experimental investigations suggest ICA exposure may alter cellular steroidogenesis thus decreasing progesterone production in vivo 3 and in vitro. 22 Despite these reports, ICA and ICA metabolites are considered to affect caruncular vasculature via antagonism of estrogen-sensitive calcium channels resulting in calcium influx into arteriolar smooth muscle, vasoconstriction, fetal anoxia, death, and subsequent abortion.2–5
Two frozen fetuses were presented to the Iowa State University Veterinary Diagnostic Laboratory (Ames, Iowa) for postmortem evaluation in November 2011. The 2 mid-gestation fetuses (crown–rump length measurement approximately 32 cm and 36 cm, respectively) were presented from a herd of 250 Red Angus purebred cows grazing native pasture on semiopen range in western Nebraska. These cows had been exposed to bulls for natural service approximately 100–180 days prior to the abortion outbreak. The owner found the fetuses while riding pastures to check cattle near a midday rest area along a conifer tree line. The frozen fetuses were submitted after more abortions were confirmed by the attending veterinarian during their annual pregnancy evaluation following the breeding season. The veterinarian indicated that the body condition of most cows was considered inadequate (4.0–4.5/9).
During pregnancy evaluation in late October, the attending veterinarian had identified multiple cows with indications of recent abortion or failing pregnancy just 10 days after removal from pasture. Fetal membranes exhibiting varying degrees of autolysis or deterioration were seen hanging from the vulva of at least 10 animals. On transrectal palpation of the uterus, several other nonpregnant cows had signs of a recent abortion and metritis (i.e., crepitus, cervical enlargement, uterine enlargement, and mucopurulent discharge from the vulva without visible fetal membranes). Other cows were marked for reexamination because the current pregnancy exhibited abnormalities suggestive of potential abortion. For the abnormal pregnancies, ballottement revealed a palpable fetus appropriately sized for 3.5 to 4 months gestation but the uterine artery of the gravid horn exhibited limited fremitus, a thin uterine wall, minimal palpable amniotic fluid, and lack of fetal movement. Eighteen out of 250 (7.2%) were confirmed nonpregnant during the October round-up. A reevaluation was requested on other animals. A second comingled herd from the same semiopen range pasture was reported to have experienced roughly the same incidence of nonpregnant cows.
When the owner returned to reevaluate the pasture, the owner noticed that the lower limbs of Ponderosa pine trees were missing needles. The owner recalled that cattle had stripped the lower limbs of these ponderosa pine trees 3 years prior at which time a higher incidence of nonpregnant cows had been discovered on pregnancy evaluation. At that time, no investigation into the cause of the abortions had been pursued.
The owner’s comments suggested that PNA should be considered in the differential diagnosis for the cause of abortion. Necropsy sample collections were adapted to include samples reflective of the dam’s serum (which was not available). It was hypothesized that ICA metabolites might be detectible if pine needles had been consumed by the dam. Therefore, fetal thoracic fluid and stomach contents were collected in addition to routine fetal tissue collection from the brain, heart, kidney, liver, lung, skeletal muscle, spleen, and thymus.
Initial diagnostic evaluation of the fetuses included gross and histological examination, immunohistochemical staining for Bovine viral diarrhea virus and Bovine herpesvirus 1, polymerase chain reaction assays for Leptospira species, culture for Campylobacter species in fetal stomach contents, and routine bacterial culture on lung and liver tissue. Microscopic examination did not reveal any significant lesions in any formalin-fixed tissues (brain, lung, heart, skeletal muscle, liver, kidney, spleen, thymus) from either fetus. Results for all of the above diagnostic assays were considered negative.
Because no evidence for the cause of abortion was derived from routine studies and the case history suggested exposure of the cows to pine needles, fetal thoracic fluid and fetal stomach contents that had been stored in an ultralow (–80°C) freezer were shipped to the Poisonous Plant Research Laboratory, U.S. Department of Agriculture, Agriculture Research Service (Logan, Utah) for detection of ICA metabolites.
The samples were extracted using procedures similar to those for bovine sera. The extract was derivatized and analyzed by gas chromatography–mass spectrometrya,b (GC-MS) as previously described.6,24 From the fetal fluids, only the ICA metabolite identified as THAA was detected at a retention time of 15.87–15.88 min and confirmed from the mass spectrum as detailed in Figure 1. The resulting spectrum is considered positive for THAA (as the definitive pattern for the di-trimethylsilane derivative parent and fragment ions are identical to the spectrum of the THAA standard eluted at the same retention time). The mass spectrum is provided in the lower portion of Figure 1. Later calculations provide an estimated concentration of the compound based on the area under the curve versus a known concentration of standard. In Figure 2B, a reagent blank run confirms an acceptable sample-to-noise ratio and identifies any potential confounding peaks in the reagents versus the ICA metabolite standards (Fig. 2A). In Figure 2C, a negative control with the same matrix does not provide detection of the compounds of interest. The fetal thoracic fluid sample in Figure 2D has a solitary peak corresponding to the THAA retention time. GC-MS chromatograms demonstrating fragment ion constructs provide confirmation of ICA metabolites. For THAA, the fragments ions m/z = 364 and 467 are used in the reconstructed ion chromatogram (Fig. 2B–2D). Results indicated that THAA was present in the fetal thoracic fluid and fetal stomach contents of 2 aborted fetuses at concentrations of 64 ppb and 139 ppb in fetal stomach contents and 562 ppb in fetal thoracic fluid. No ICA, AA, nor DHAA were detected in fetal stomach contents or fetal thoracic fluids. The lack of detection of these and other ICA metabolites may be explained in part by the ready metabolism of ICA and its metabolites by hepatic dehydrogenase enzymes and by esterases, reductases, and oxidases in the rumen of the dam 6 as detailed in Figure 3. Rumen esterases hydrolyze acetyl ICA and succinyl ICA to ICA. Rumen reductases convert ICA to imbricatoloic acid, which is in turn oxidized back into ICA or into subsequent ICA metabolites such as AA and DHAA. Isocupressic acid, AA, and DHAA readily solubilize and equilibrate across the rumen wall with the serum stores until metabolized by hepatic dehydrogenase activity with the end product, thus far reported, being THAA.
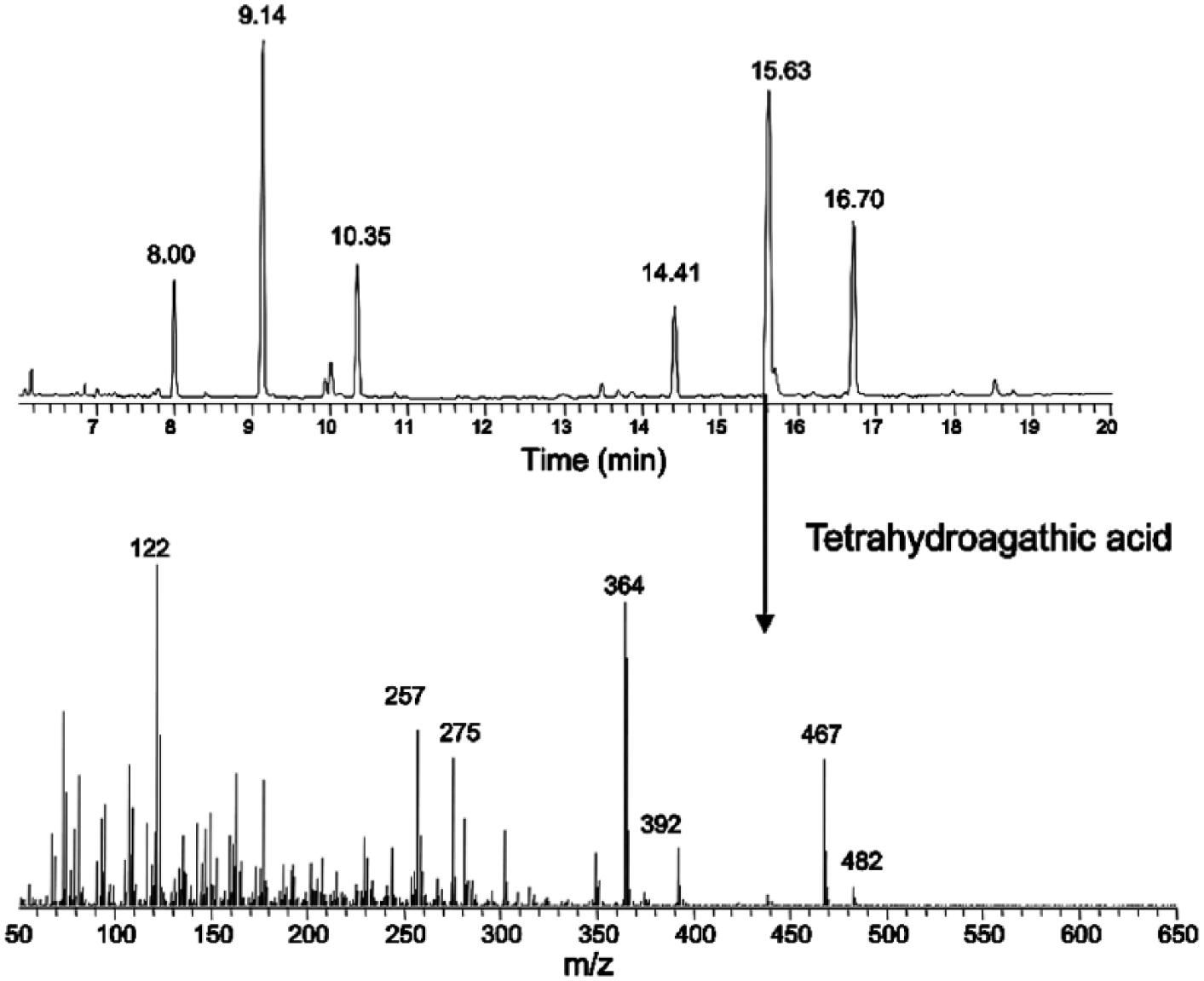
Gas chromatography–mass spectroscopy results for compound at peak retention time of 15.87–15.88 min confirm the presence of a di-trimethylsilane derivatized tetrahydroagathic acid.
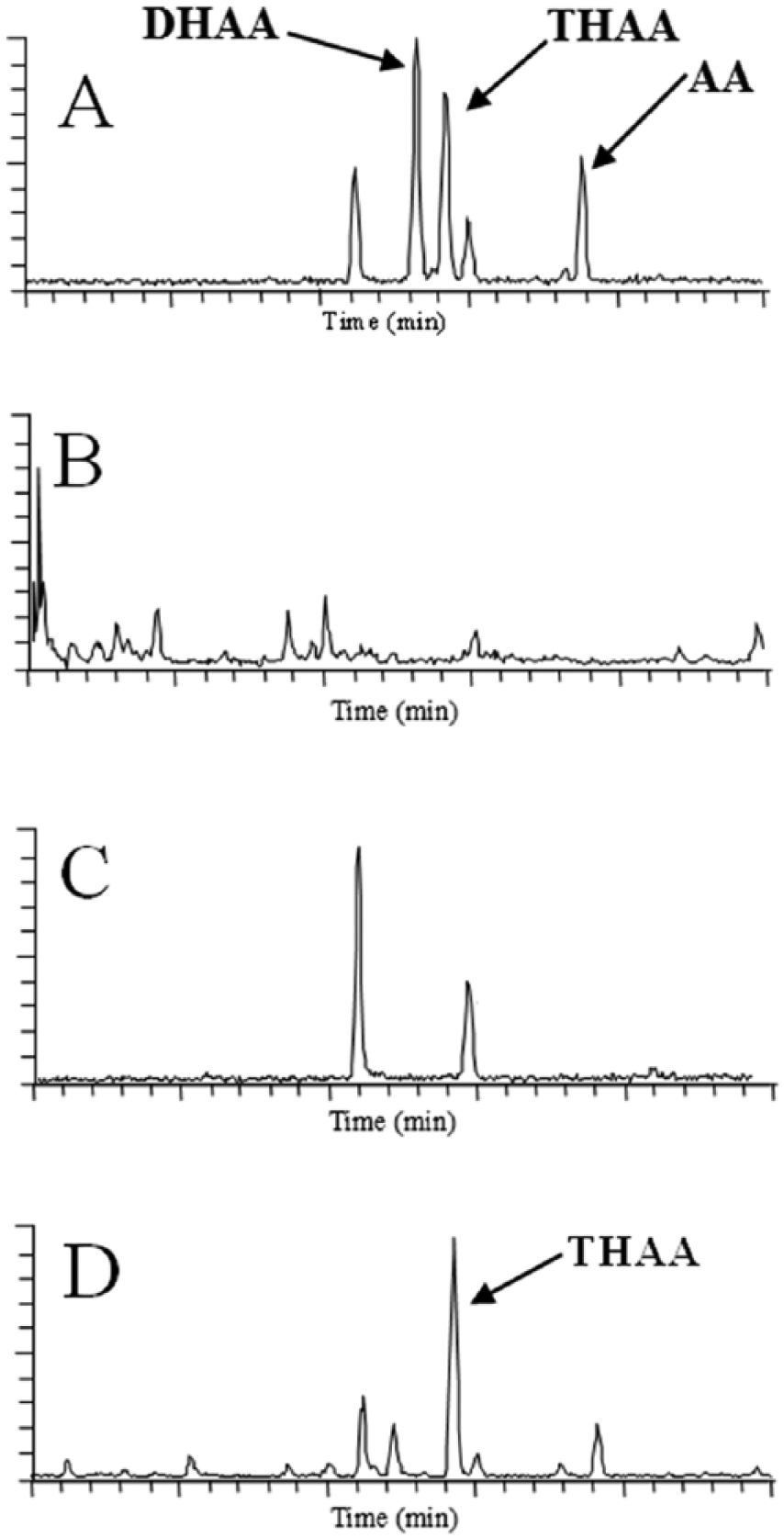
The ion chromatograms from the gas chromatography–mass spectroscopy in this figure are reconstructed ion chromatograms for agathic acid (AA), dihydroagathic acid (DHAA), and tetrahydroagathic acid (THAA) standards (
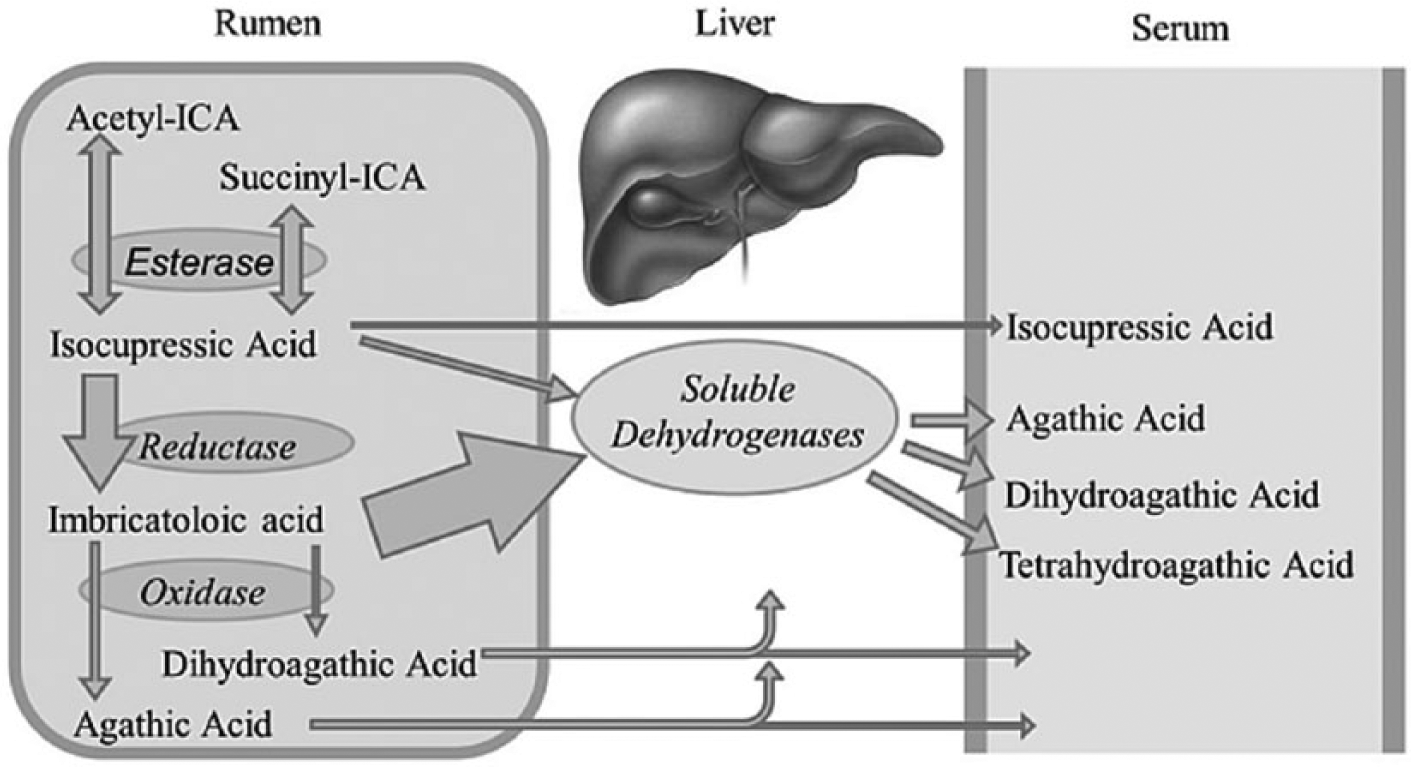
Absorption and metabolism of isocupressic acid to form tetrahydroagathic acid (adapted from Gardner et al. 1999 8 ).
The current study describes PNA resulting in the loss of approximately 125–140-day gestation fetuses. In previous experiments, 18 abortions occurred in 40% of cows gestating 157 days when fed ponderosa pine needles in experimental diets; however, no abortions occurred in the cows gestating 116 days. At the time of the current diagnostic investigation, there were no previous publications indicating that stable ICA metabolites cross the placenta. Similarly, there were no earlier reports indicating that THAA could be detected in fetal samples. Subsequent investigations of cattle abortion storms in California (Gardner DR, Panter KE, Aduriz G, et al.: 2013, Pine needle abortion in cattle update: metabolite detection in sera and fetal fluids from abortion case samples. In: Proceedings of the 9th international symposium on poisonous plants, HohHot, China) and Spain (Gardner et al.) 1 have been pursued and documented by the current group of authors to demonstrate the utility of THAA detection in fetal fluids to confirm PNA. Previous progress in this area of study had been primarily halted in part due to the limited number of laboratories with the analytical methods for detection of ICA or ICA metabolites. Other limiting factors include few diagnostic submissions requesting PNA rule-out and mixed perceptions regarding the utility of ICA or ICA metabolite detection. Previous experiments have relied on detection of ICA or ICA metabolites in the rumen contents and dam serum, respectively. Unfortunately, dam serum was not available for diagnosis in the present case. Similarly, dam serum is not frequently presented along with the fetus to diagnostic laboratories. Isocupressic acid detection in rumen contents may be considered questionable given the relatively short half-life of ICA in rumen contents compared to the extended duration of time that may elapse between intoxication and subsequent abortion. This would result in rumen metabolism of ICA prior to detection of an aborted fetus. Because ICA is metabolized to AA, DHAA, and THAA over the prolonged time period, detection of ICA metabolites are recommended as a preferred biomarker of pine needle consumption and subsequent PNA. The results of the current study support this diagnostic strategy.
A presumptive diagnosis of PNA was applied to the present case as a result of the supporting evidence provided by case history, the presence of THAA in fetal tissues, and the negative results from macroscopic evaluation, microscopic evaluation, and other diagnostic assays. The field investigation confirms that 1) PNA can be confirmed despite the absence of maternal serum; 2) THAA is detectible in fetal thoracic fluid following pine needle consumption and subsequent PNA; 3) THAA can be detected in fetal stomach contents, albeit at lesser concentrations, to confirm PNA; and 4) natural pine needle consumption can induce second trimester abortions.
Additional work is warranted to evaluate the sensitivity and limit of detection in fetal-derived matrices for THAA via GC-MS. The current investigation provides strong evidence that either ICA or ICA metabolites cross the placenta and enter into fetal tissues. Further investigation is needed to determine if THAA or other ICA metabolites are present in the fetal sera, liver, or other fetal tissues. Likewise, additional work is needed to determine whether the fetal liver further oxidizes circulating ICA, AA, and DHAA to THAA. Nonetheless, detection of THAA in the most commonly presented abortion diagnostic sample, the fetus, is a progressive step toward reevaluating the prevalence of PNA. Therefore, an updated review of PNA economic impacts may be warranted if prevalence estimates change as a result of the recent advances in diagnostic methods described herein.
Footnotes
Acknowledgements
The primary author would like to thank Dr. Aileen Keating and Dr. Rachel Derscheid for their guidance in preparing this publication.
a.
Gas chromatography–mass spectroscopy: Polaris Q MS and a Trace GC from Thermo Electron, Thermo Fisher Scientific Inc., Waltham, MA.
b.
Column: DB-5MS, Agilent Technologies Inc., Santa Clara, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
