Abstract
The current report describes the diagnostic features in 10 cases of acute fatal canine leptospirosis with minimal renal and hepatic changes that may present a diagnostic challenge for the pathologist. Most affected dogs were less than 6 months of age and had a biochemical profile consistent with hepatorenal dysfunction. Clinical signs consisted of vomiting, depression, icterus, dehydration, diarrhea, and anorexia. All dogs died or were humanely euthanized within 3–7 days after the onset of clinical disease. Necropsy findings included pulmonary edema with hemorrhages, icterus, renal and hepatic pallor and swelling, and gastric edema with hemorrhage. Despite severe azotemia, histological changes in the kidneys were subtle in all dogs, and included mild renal tubular simplification, with single-cell necrosis and attenuation, along with minimal interstitial lymphoplasmacytic inflammation, edema, and hemorrhage. Hepatic lesions included scattered hepatocellular single-cell necrosis and hepatocellular dissociation. Prominent extrarenal lesions typically associated with uremia including vascular fibrinoid necrosis in multiple organs, pulmonary mineralization with occasional fibrinosuppurative exudation, and gastric mineralization were also present. Postmortem diagnostic confirmation was based on the detection of leptospiral antigen on fresh renal samples by fluorescent antibody test and on the demonstration of intact spirochetes in sections of kidneys using immunohistochemical staining. Acute fatal canine leptospirosis occurred as a fulminant hepatorenal disease affecting mainly young dogs, and the diagnosis was dependent on the recognition of the subtle renal changes with confirmation via fluorescent antibody testing or immunohistochemical staining.
Leptospirosis is a zoonotic disease caused by distinct serovars of Leptospira interrogans.7,11–13 Since the advent of the bivalent antileptospiral vaccine that protects against serovars Canicola and Icterohaemorrhagiae, there has been an epidemiologic shift in canine leptospirosis in the United States such that infection is now more often associated with serovars Grippotyphosa and Pomona, with variations according to the geographical area.2,3,7,15,16,22 Pathological features of subacute and chronic canine leptospirosis are well described in the veterinary literature, with renal changes consisting of severe lymphoplasmacytic and neutrophilic tubulointerstitial nephritis in subacute cases and interstitial fibrosis with less severe inflammation in chronic cases.2,3,7,9,12,14,15,20–22 The antemortem diagnosis of leptospirosis typically relies on the detection of high serum antibody titers using the microscopic agglutination test (MAT) in dogs with acute onset of renal disease and azotemia. 5 However, in the past few years, cases of acute canine leptospirosis affecting young dogs have been identified that present with fulminant hepatorenal disease. These dogs have low or negative leptospiral antibody titers, presumably due to the short duration of infection, and subtle renal and hepatic lesions, making the diagnosis of leptospirosis more challenging. The current study briefly describes the clinicopathological and diagnostic features in 10 cases of acute fatal canine leptospirosis.
Cases were retrieved from the Athens Veterinary Diagnostic Laboratory (University of Georgia, Athens, Georgia) archives from January 1990 to December 2012. Clinical and necropsy reports, as well as hematoxylin and eosin–stained slides from all cases were reviewed, and available data were noted for each dog. All dogs had been positive for leptospiral antigen using fluorescent antibody test (FAT) on fresh samples of kidney obtained at necropsy. Immunohistochemical staining (IHC) for Leptospira spp. was performed from available paraffin-embedded sections of kidney using a primary rabbit polyclonal antibody at a dilution of 1:100 that was pooled from 6 different leptospiral serotypes: Bratislava, Canicola, Icterohaemorrhagiae, Grippotyphosa, Hardjo, and Pomona. a
The signalment and the clinical and biochemical findings from the retrieved cases are summarized in Table 1. Seven cases (70%) were diagnosed during the summer months, with 2 cases (20%) occurring during the winter and only 1 case (10%) during the spring. Clinical signs were rapidly progressive and lasted from 3 to 7 days. Blood work results were available from only 5 cases, and all dogs had neutrophilic leukocytosis (values were not available). Vaccination history, serology, and urinalysis information were not available. Gross and histological findings and the diagnostic confirmation for each case are summarized in Table 2 and Figures 1–6. Histological changes in the kidneys were similar and subtle in all 10 cases. There was proximal tubular injury characterized by attenuation of the brush border (tubular simplification) with resultant relative expansion of the tubular lumen (Fig. 1). Necrotic epithelial cells were often observed within the tubular lumen and were more easily appreciated than individual in situ necrotic cells (Fig. 2). As a result from necrosis, the tubular epithelium was attenuated as it spread to cover the denuded basement membrane (Fig 2). In addition, there was minimal to mild, scattered accumulations of interstitial lymphocytes and plasma cells (Fig. 2), with variable degrees of interstitial edema and hemorrhage, and occasional vascular wall mineralization of small arterioles and venules. Hepatic lesions included scattered single-cell hepatocellular necrosis in all 10 cases and hepatocellular dissociation in 8 cases (cases 2–9; Fig. 3). Canalicular bile plugs were not observed in any of the examined cases, and there was no evidence of erythrophagocytosis.
Signalment, clinical and biochemical findings, and serology in 10 cases of acute fatal canine leptospirosis.*
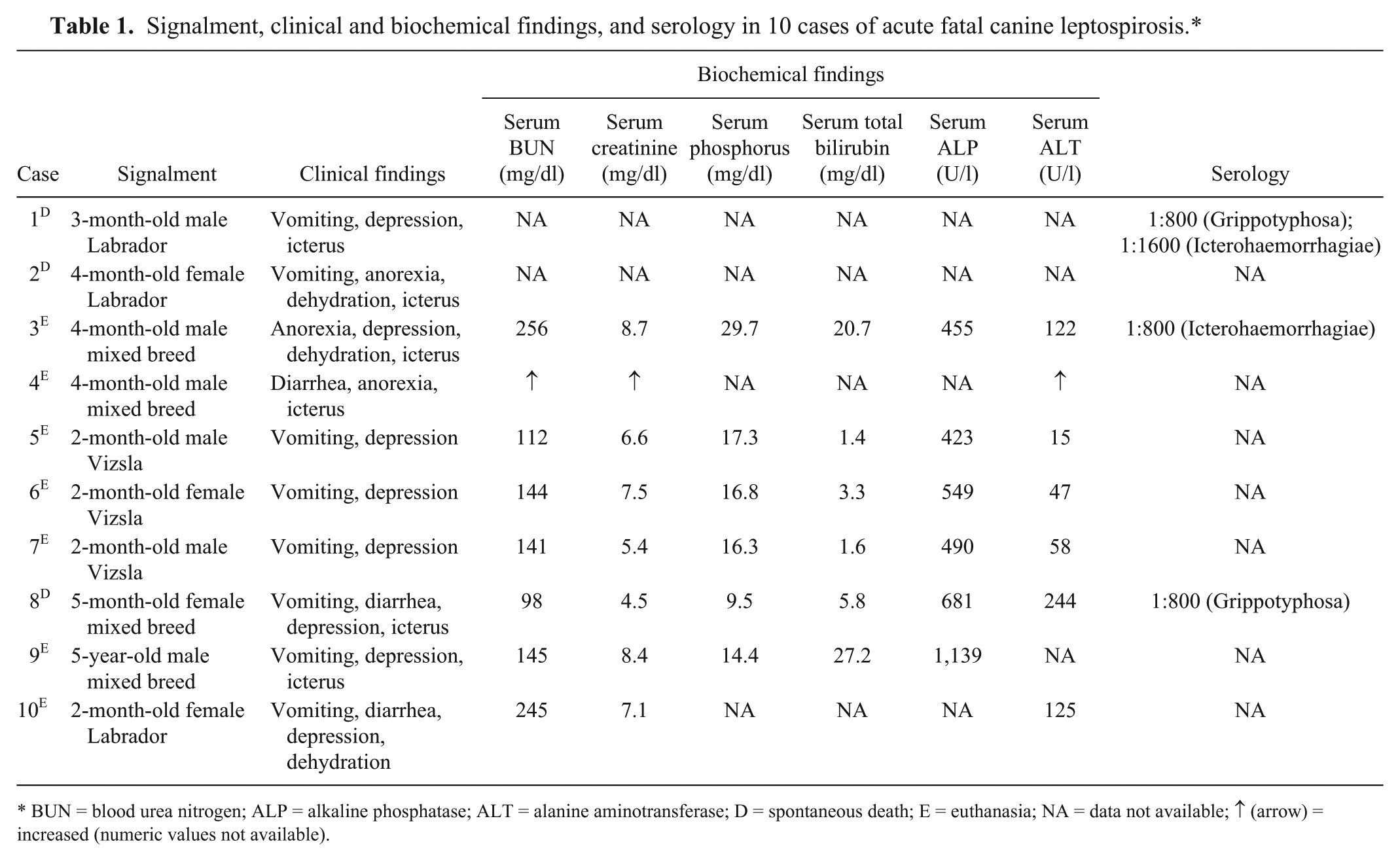
BUN = blood urea nitrogen; ALP = alkaline phosphatase; ALT = alanine aminotransferase; D = spontaneous death; E = euthanasia; NA = data not available; ↑ (arrow) = increased (numeric values not available).
Gross pathological findings, extrarenal and extrahepatic histological changes, and diagnostic confirmation in 10 cases of acute fatal canine leptospirosis.*
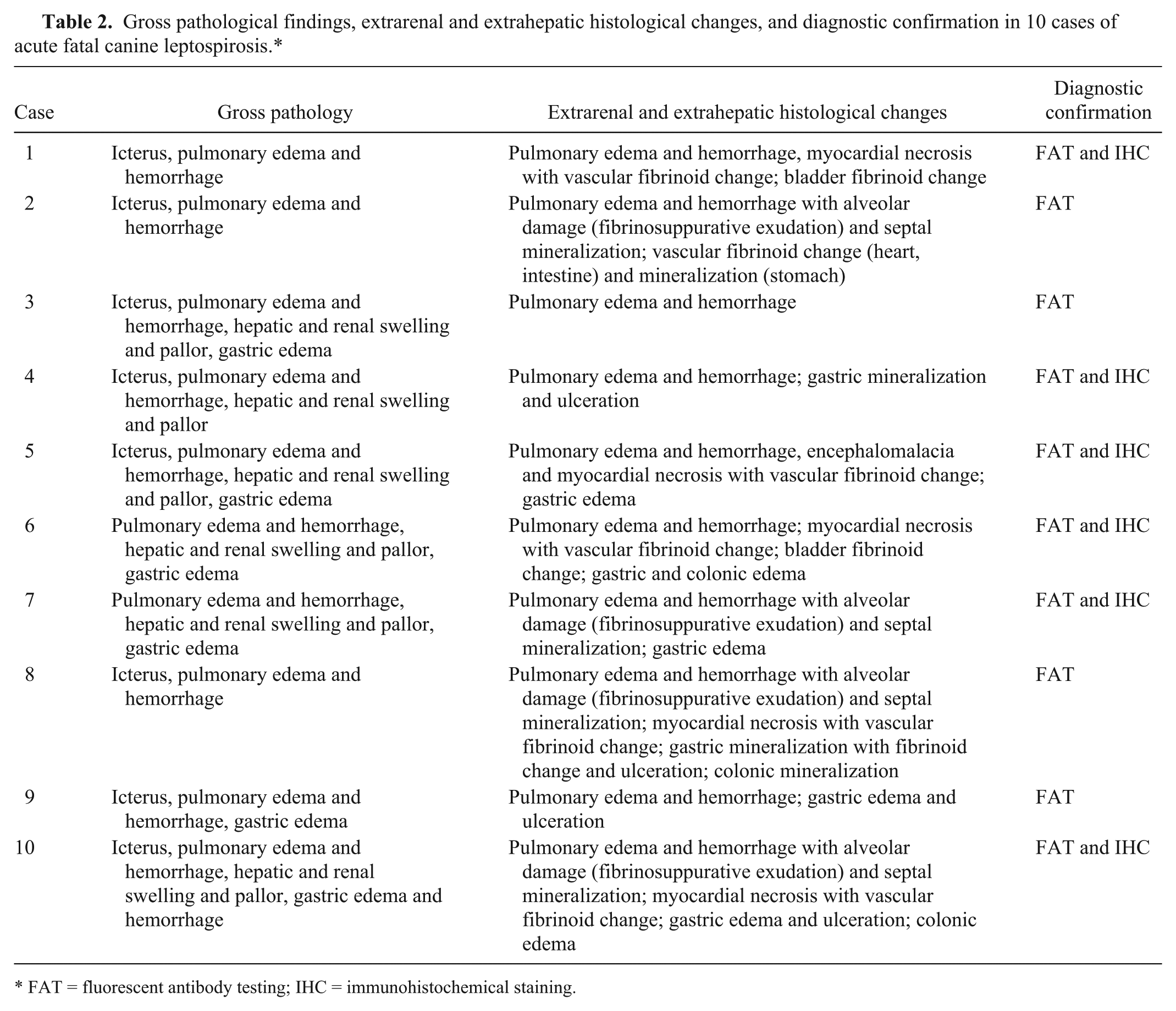
FAT = fluorescent antibody testing; IHC = immunohistochemical staining.
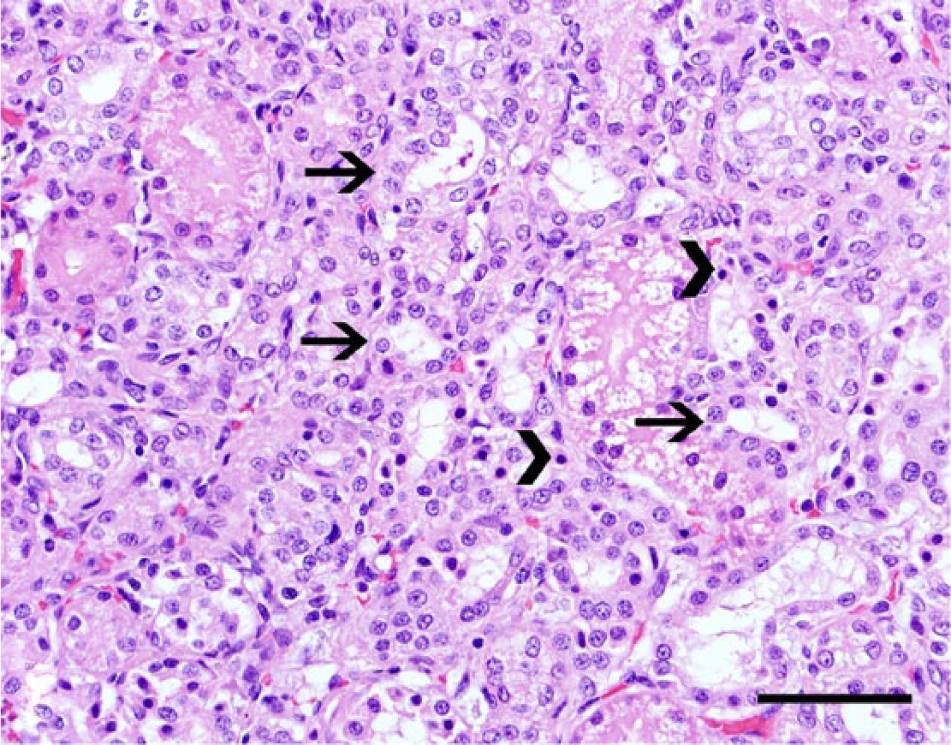
Kidney, dog no. 5. Tubular dilation and simplification of the proximal tubules due to brush border loss (arrows) and scattered necrotic epithelial cells with hypereosinophilic cytoplasm and pyknotic nuclei (arrowheads). Hematoxylin and eosin. Bar = 50 µm.
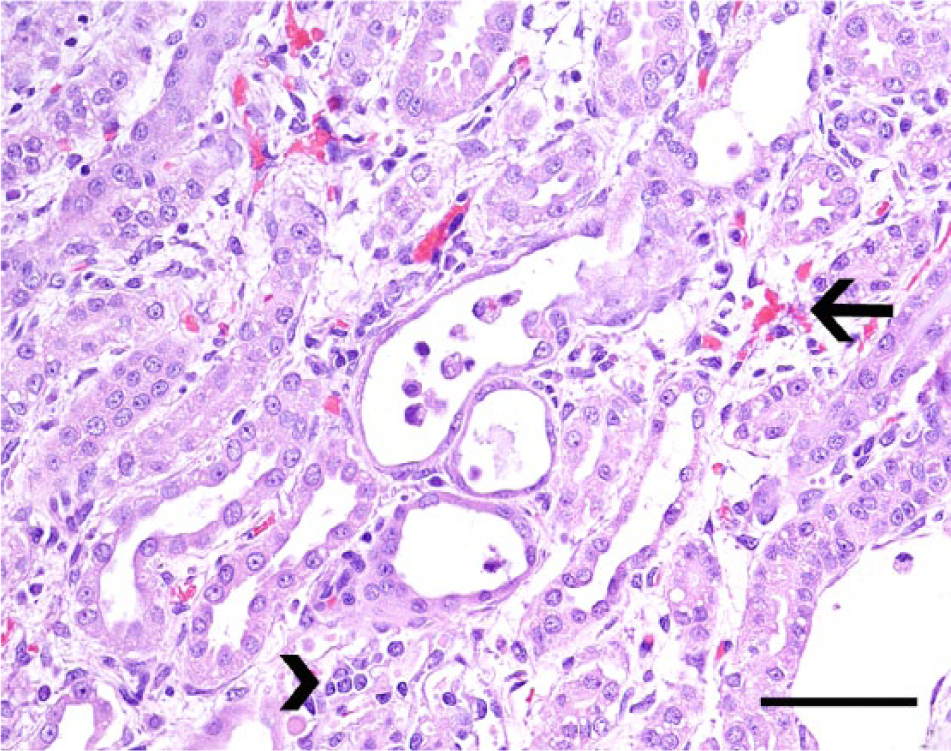
Kidney, dog no. 5. Renal tubules are partially occluded by necrotic intraluminal debris (center) and are lined by flattened epithelial cells. The renal interstitium is widened by edema fluid, small areas of hemorrhage (arrow), and minimal numbers of scattered lymphocytes and plasma cells (arrowhead). Hematoxylin and eosin. Bar = 50 µm.
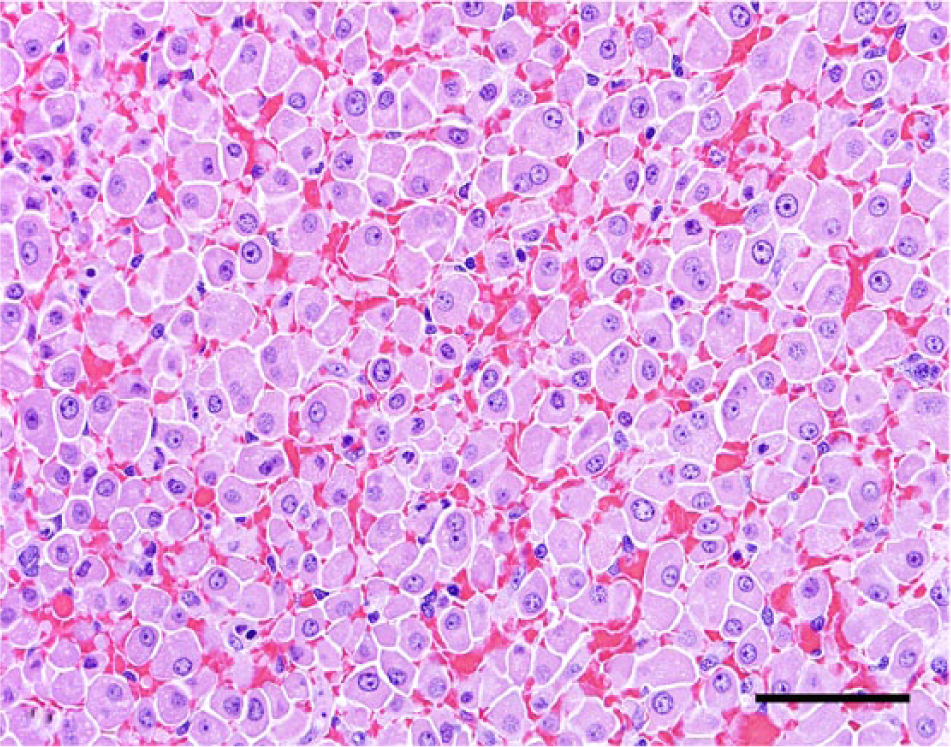
Liver, dog no. 2. The normal hepatocyte cord architecture has been disrupted due to diffuse hepatocellular dissociation. Hematoxylin and eosin. Bar = 50 µm.
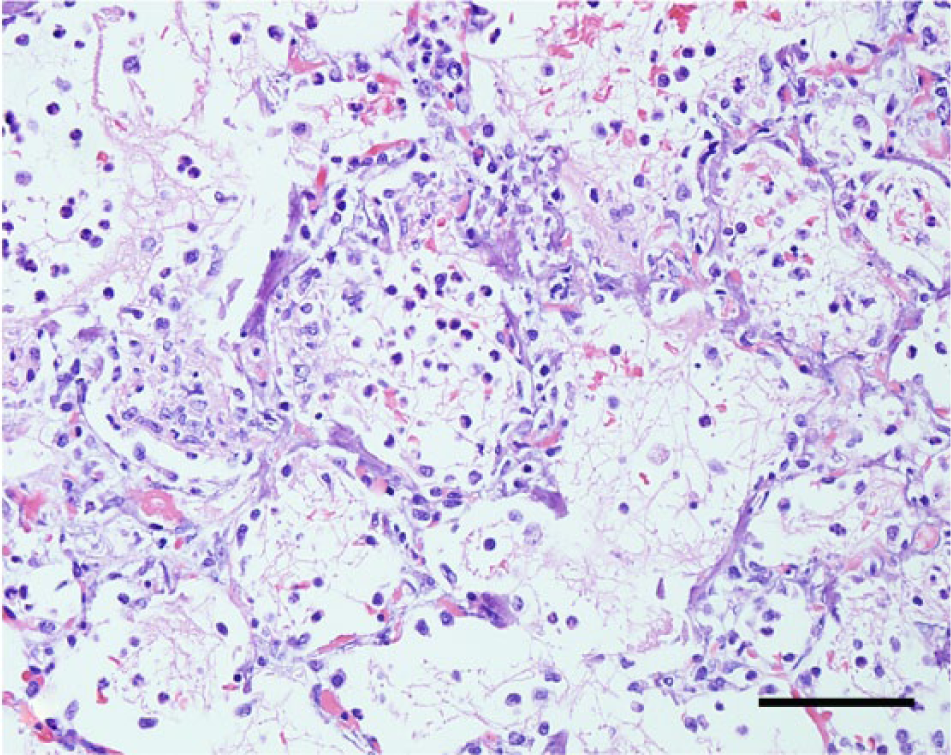
Lung, dog no. 7. The pulmonary parenchyma contains extensive areas of septal mineralization and alveolar expansion by neutrophils and macrophages admixed with fibrin, edema fluid, and erythrocytes. Hematoxylin and eosin. Bar = 100 µm.
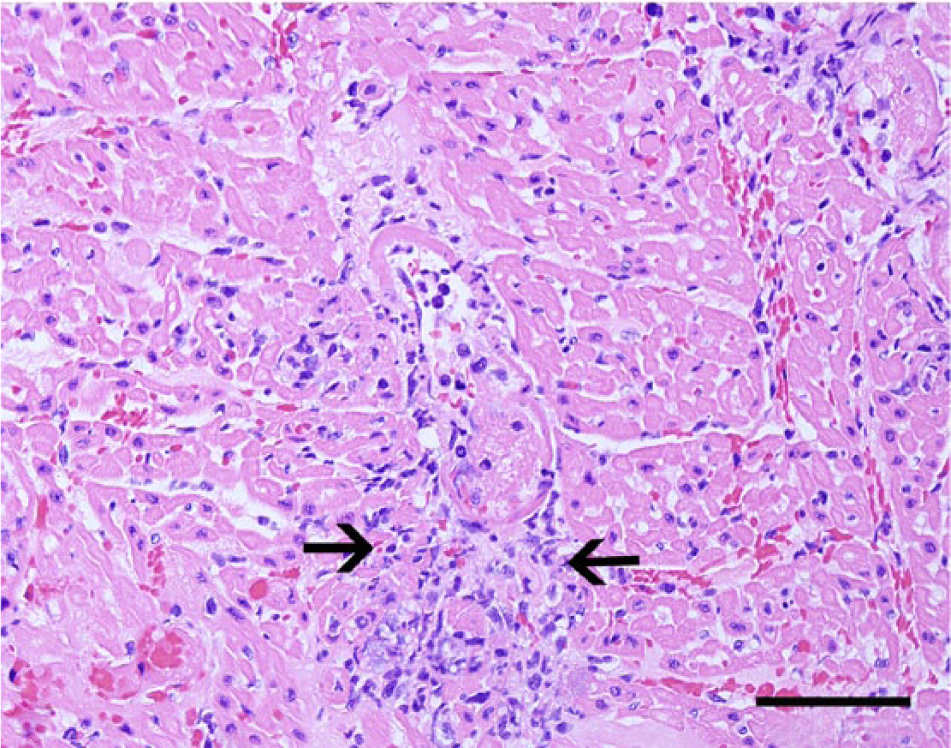
Heart, dog no. 6. A small cardiac arteriole has undergone fibrinoid vascular necrosis (center). The vascular wall is expanded by fibrin and neutrophils, and there is a small area of myofiber necrosis (arrows). Hematoxylin and eosin. Bar = 100 µm.
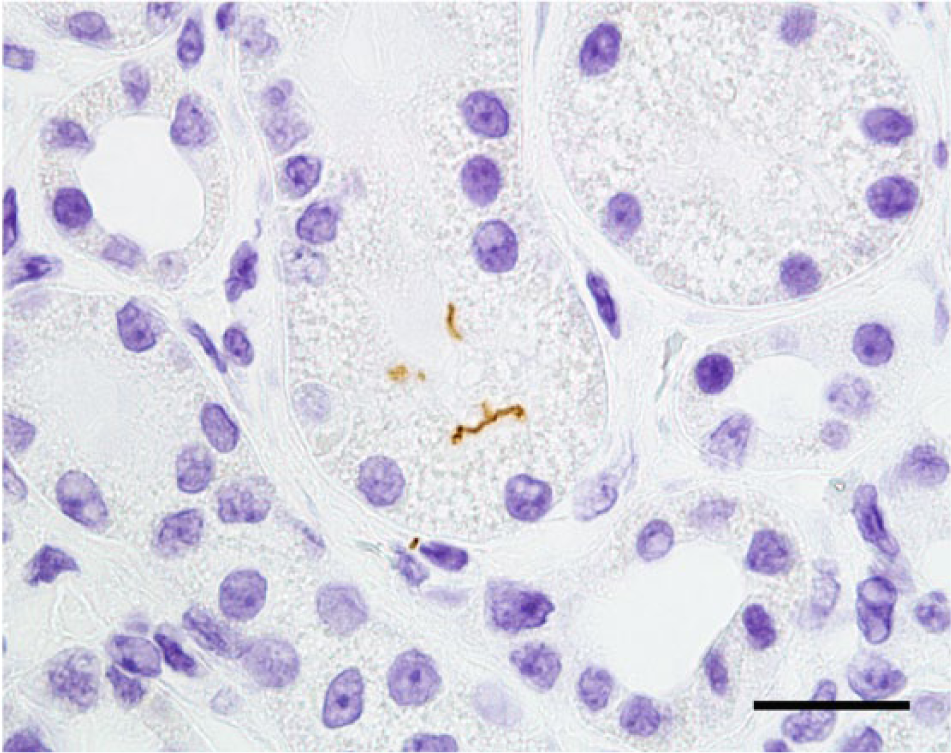
Kidney, dog no. 4. Positive immunostaining showing 2 intact spirochetes in the renal tubular epithelium. Immunohistochemical staining for Leptospira spp. antigen; streptavidin–biotin–peroxidase method, Gills II hematoxylin counterstain. Bar = 25 µm.
The majority (90%) of cases of acute fatal leptospirosis in this study were observed in young dogs less than 6 months of age. Clinical disease was acute and severe, but histological changes in the kidneys and liver were subtle despite clinical evidence of severe hepatorenal disease. Consistent with the acute nature of the clinical disease, leptospiral titers when available were considered low, 3 making the clinical diagnosis difficult. Similarly, postmortem diagnosis by FAT and IHC was difficult in most cases due to the low number of leptospiral organisms in the kidney. Although the cases in this study may share some similarities with other cases of acute or subacute canine leptospirosis described elsewhere, the young age of affected dogs, the uniformly fatal outcome, and the subtle renal and hepatic changes are unique.
Age, sex, and breed have been regarded as potential risk factors for canine leptospirosis according to large population studies in the United States and Canada, which demonstrate an increased risk of infection in middle-aged, sexually intact male hounds, herding, working, or mixed breed dogs.2,9,15,21,22 These individuals are more likely to be active and spend time outdoors, increasing a potential environmental exposure to the infectious agent. 21 The reason behind the age difference observed in this study is unknown, but it has been recognized that clinical disease in cases of leptospirosis may vary according to the infecting serovar and the individual host susceptibility, making such atypical infections a reflection of the exposure of immunologically naïve dogs to a newly emerging agent.2,7,9,15,18 Therefore, it is possible that infection with an emergent leptospiral serovar could have led to the disease in the young dogs of this report.3,10 No sex predisposition was observed in this study, and conclusions regarding breed predisposition could not be made based on the small number of affected dogs. Similar to what has been reported in other studies, the majority of the cases in the current study were diagnosed during the summer months.20,22
Canine leptospirosis may occur as a hyperacute, acute, subacute, or chronic disease.2,3,7–9,12,14,15,17,20,21 Subacute and chronic cases have been well described in numerous publications, but hyperacute and acute disease has not been similarly characterized.2,3,7–9,12,14,15,20,21 Hyperacute leptospirosis has been briefly described as a fulminant, septicemic disease occurring mainly in puppies infected by serovar Icterohaemorrhagiae that develop hemorrhagic disease characterized by mucosal hemorrhages, hematemesis, epistaxis, and melena. 14 Because renal changes have not been addressed in cases of hyperacute leptospirosis, it is difficult to draw any detailed comparison between the cases in the present study and the hyperacute cases. 14 Affected dogs in the current study did present with rapidly progressive and acute clinical signs, but no clinicopathological evidence of a primarily hemorrhagic disease was observed, and although no blood work information on plasma coagulation parameters was established, the pulmonary hemorrhages in the affected individuals was attributed to the disseminated vascular damage (fibrinoid necrosis) rather than to a primary coagulative disorder. 13 Furthermore, hemorrhagic foci in the lungs were less severe than the disseminated pulmonary hemorrhage reported in cases of canine and human pulmonary hemorrhagic syndrome associated with leptospirosis. 10 Acute canine leptospirosis has been reproduced experimentally in puppies inoculated with Leptospira kirschneri serovar Grippotyphosa and was characterized by icterus and renal changes consisting of interstitial inflammation and tubular degeneration and necrosis.8,14 The natural disease observed in the dogs of the present study is similar to these experimental acute cases. 8
Similar to the dogs in the current study, disseminated vascular fibrinoid necrosis may occur in dogs with uremia and most frequently affects small arterioles or muscular arteries in the gastric submucosa, tongue, colon, gall bladder, urinary bladder, kidneys, small intestine, and myocardium.12,13 In addition to the pulmonary edema, hemorrhage, and mineralization, 4 dogs also developed changes morphologically consistent with acute pulmonary alveolar injury that manifests in human beings and other species as a consequence of septicemia or endotoxemia. 16 A third possibility for the vascular lesions observed in the dogs described herein would include acute severe systemic hypertension (malignant hypertension) secondary to renal failure.12,13 The overall pathological findings in the dogs closely resemble the changes described in cases of acute septicemic leptospirosis (Weil’s disease) in human beings.1,19 Individuals with Weil’s disease develop systemic and fulminant illness characterized by severe icterus, fever, headache, hemorrhages, and acute renal failure.1,19 Detailed pathological descriptions on this disease are scarce, 1 but patients dying during the early stages of disease develop gross changes characterized by icterus and multiple hemorrhagic foci. Surprisingly and similarly to what was observed in the 10 dogs in the current report, human patients develop subtle renal lesions consisting of scattered tubular necrosis, vasculitis, and occasional mild interstitial inflammation despite clinical evidence of renal failure. 1 Thus, it is possible that the 10 cases of acute fatal canine leptospirosis in the present study may actually represent a septicemic or endotoxemic disease similar to Weil’s disease, and these features may suggest that leptospiral septicemia or endotoxemia could also have played a role in the endothelial damage in these dogs. 7 It is unknown whether one or a combination of these mechanisms could have contributed to the vascular fibrinoid necrosis, but regardless of the pathogenesis, it is likely that the vascular lesions and consequent increased vascular permeability culminated in tissue hemorrhage and necrosis in all affected dogs. 7
Major differences were observed between the renal changes in the cases described herein when compared with cases of subacute and chronic canine leptospirosis described by other authors.2,8,9,15 The classic renal changes of subacute canine leptospirosis consist of moderate to severe lymphoplasmacytic and neutrophilic tubulointerstitial nephritis, with tubular degeneration, necrosis, and mineralization.2,8,9,15 In chronic cases, inflammation is less severe and is accompanied by interstitial fibrosis and tubular atrophy (C Brown, personal observation). In the present study, only widely scattered small foci of inflammation with minimal to mild tubular necrosis were observed, similar to what has been described in cases of Weil’s disease. 1 Despite clinicopathological evidence of severe renal dysfunction, renal lesions in these 10 cases were often so mild that they could be easily overlooked. It is well recognized in human nephropathology that histologically subtle renal changes (such as tubular simplification) are often associated with acute renal failure and profound renal functional abnormalities. 4 Therefore, the subtle changes observed in the present study could account for the observed azotemia.
Icterus was observed in 80% of the affected dogs in the current study, with no evidence of hemolysis or post-hepatic biliary obstruction. It has been well established that icterus in cases of canine leptospirosis occurs secondarily to hepatocellular damage caused by leptospiral toxins. 7 While hepatocellular dissociation may occur as a postmortem change, it has also been associated with loss of E-cadherin membrane expression in human beings affected by leptospirosis, indicating that the dissociation may be a genuine lesion and not only a postmortem finding associated with autolysis as previously suggested. 6 Thus, icterus may be attributed to a possible defective bilirubin uptake and conjugation by damaged hepatocytes in the affected dogs.7,14
Serum antibody titers in dogs experimentally infected with Leptospira spp. are first detected 5–13 days postinoculation. 3 Leptospiral titers determined in 3 cases in the present study were low and not clearly distinguishable from post-vaccinal titers, consistent with the acute nature of this disease. 3 Therefore, serologic tests may not be useful for antemortem or postmortem diagnostic purposes in these cases. 11 Although FAT was positive in 100% of the cases and it seems to be a reliable test to confirm infection during necropsy, the amount of detected antigen in fresh samples of kidney was reported as very low. Similarly, IHC results were occasionally reported as negative on the necropsy report. Careful reexamination of renal tissues from all cases in this report revealed rare intact spirochetes in 4 cases originally interpreted to be negative. Rare globular staining was observed in interstitial macrophages in some cases that also had intact spirochetes, but this scant globular staining was not considered specific for a diagnosis of leptospirosis. This pattern of leptospiral immunostaining is clearly different than that observed in cases of subacute leptospirosis, where globular positive staining, presumed to represent degraded bacteria, is abundant within tubular lumina, tubular epithelial cells, and interstitial macrophages, and clumps of intact spirochetes are often observed within tubular lumina. 7
In summary, naturally occurring acute fatal canine leptospirosis in the current study occurred primarily in dogs less than 6 months of age and was characterized by a fatal hepatorenal disease with subtle hepatic and renal changes and severe extrarenal lesions typically associated with uremia. All cases shared pivotal pathological features with the acute septicemic human leptospirosis (Weil’s disease), which suggests that acute fatal canine leptospirosis may represent a septicemic or endotoxemic disease. The accurate diagnosis depends on the recognition of the subtle renal lesions and successful diagnostic confirmation by FAT or IHC.
Footnotes
Acknowledgements
The authors wish to thank A. Wayne Roberts and Rachel Steffens at the College of Veterinary Medicine (CVM), The University of Georgia (UGA) for performing the fluorescent antibody test, and Abbie Butler (CVM-UGA) for her outstanding support with the immunohistochemistry.
a.
ARA-050, CAA-010, ICA-020, GRA-020, SJA-060, POA-010, National Veterinary Services Laboratories, Ames, IA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) declared that they received no financial support for their research and/or authorship of this article.
