Abstract
Vascular hamartomas are non-neoplastic developmental anomalies of vessels. Cases of cerebral vascular hamartomas have been previously reported in dogs and cats. A 4-week-old Freiberger foal had shown persistent problems with breathing and swallowing since birth, and bilateral laryngeal paralysis was diagnosed. The foal subsequently developed left sided facial nerve paralysis and a secondary corneal ulcer in the left eye. Necropsy revealed a pinkish mass in the obex region of the brain. The mass was further investigated by histology and immunohistochemistry. Histologically, the mass consisted of many thin-walled, blood-filled vascular structures of variable diameter involving the white matter of the obex. The lining cells were immunohistochemically positive for factor VIII (von Willebrand factor) interpreted as endothelial cells. The endothelial lining showed also variable immunoreactivity for smooth muscle actin and vimentin. Normal neural parenchyma labeled with antibodies directed against glial fibrillary acidic protein and neuron-specific enolase was present between the vascular proliferations. A diagnosis of focal vascular hamartoma in the obex was made. The development of clinical signs is attributed to the compression of the surrounding neural parenchyma.
Vascular hamartomas are nonneoplastic developmental anomalies of vessels. The term hamartoma implies the presence of the lesion at birth, with subsequent parallel growth with that of the animal. 10 In horses, vascular hamartomas are uncommon, and have been reported in the skin and subcutis,4,8 in the tendon sheath, 12 and in the ovary. 13 Vascular hamartomas in the central nervous system have been reported in the brain of cats and dogs9,15 and in the thoracic spinal cord of a Welsh Cob filly. 7 The current study describes a vascular hamartoma in the obex of a Freiberger foal with unusual clinical presentation.
A 1-day-old Freiberger foal was presented to the Equine Department of the Vetsuisse Faculty, University of Zurich, Switzerland for evaluation of abnormal nursing behavior and weakness. Initial clinical examination was unremarkable apart from a weak suckle reflex and increased recumbency. Hematology was within normal limits, and a biochemistry panel revealed mild hypoproteinemia (40 g/l, reference [ref.] range: 45–47) and hyperglobulinemia (13 g/l, ref. range: 9–12), mildly elevated concentration of blood urea nitrogen (4.5 mmol/l, ref. range: 3.5–4.0), and moderate creatinine kinase (230 U/l, normal: <50) and gamma-glutamyl transferase (49 U/l, ref. range: 9–17). An immunoglobulin (Ig)G test a revealed an IgG concentration of 400 mg/dl. A diagnosis of failure of transfer of passive immunity and inappropriate nursing behavior was made. The foal received a transfusion of equine fresh frozen plasma (750 ml), broad spectrum antimicrobial coverage (penicillin G sodium, 30,000 IU/kg, every 6 hr, intravenously [IV]; amikacin, 25 mg/kg, every 24 hr, IV), and a single dose of vitamin E and selenium (25 mg/ml and 1 mg/ml, respectively; 2.5 ml, intramuscularly) and tetanus antitoxin (3,000 IU, subcutaneously). A nasogastric feeding tube was placed, and the foal was fed mare’s milk on a regular basis. After 24 hr, the IgG test was repeated and was found to be between 400 and 800 mg/dl. Thus, a second transfusion of fresh frozen plasma (750 ml) was performed.
On the second day of hospitalization, the foal showed signs of respiratory distress, labored breathing, an elevated respiratory rate of 36 breaths per minute, and slightly cyanotic mucus membranes. Mild bilateral mucous nasal discharge was present, and the foal coughed spontaneously. Auscultation of the lungs revealed crackles and wheezing bilaterally. The foal developed hyperlactatemia (6.2 mmol/l, normal: <4), and venous blood gas analysis showed an increased oxygen extraction and hypercapnia (pO2, 24 mmHg, ref. range: 37–56; pCO2, 66 mmHg, ref. range: 38–48; sO2, <60%, ref. range: 93–100). Repeated hematology revealed marked leukopenia of 1,900/μl (normal: 5,000–12,000/μl) with hyperfibrinogenemia (4 g/l; normal: <3 g/l). Intranasal O2 supplementation was commenced. Radiographic examination of the thorax showed signs consistent with aspiration pneumonia, and ultrasonographic evaluation of the lung showed bilaterally comet tails consistent with pleural roughening but no evidence of lung consolidation. Endoscopy of the respiratory tract revealed a narrow pharynx, bilaterally thickened arytenoid cartilages, and minimal arytenoid abduction resulting in a severely narrowed rima glottidis. Additionally, the foal exhibited an intermittent dorsal displacement of the soft palate (DDSP). A diagnosis of bilateral laryngeal paralysis with secondary aspiration pneumonia was made. In addition, left sided facial nerve paralysis and a left corneal ulcer was present. Due to progression of the respiratory distress, a tracheotomy was performed. Meloxicam (0.6 mg/kg, every 24 hr, IV), omeprazole (4 mg/kg, every 24 hr, orally [PO]), and an antibiotic eye ointment (neomycin 3.5 mg/g and bacitracin 250 IU/g; left eye, every 24 hr) were given. Over the course of the night, the foal became dysphagic. A course of tapered dexamethasone (0.1 mg/kg initially then 0.05 mg/kg, every 24 hr, IV) over 6 days provided no improvement. Several attempts to remove the tracheal tubus failed, and signs of dysphagia persisted. Repeated endoscopy showed persistent bilateral laryngeal paralysis. Culture of a tracheobronchial aspirate revealed mild growth of Pasteurella caballi, Enterobacter sp., and Escherichia coli. After 8 days, the tracheotomy tube could be removed without the development of respiratory distress. Due to progression of the corneal ulcer, the left eye was enucleated. The foal was switched to oral medications (doxycycline, 10 mg/kg, every 12 hr, PO; meloxicam, 0.6 mg/kg, every 24 hr, PO; omeprazole continued). The foal was fed from a bucket, and normal suckling behavior was closely monitored. Twenty-five days after admission, the foal was discharged. It was advised to feed the foal with milk replacer from a bucket, foal pelleted ration, and hay under close monitoring for evidence of dysphagia. Additionally, the foal received trimethoprim–sulfadimidine (30 mg/kg, twice a day, PO), meloxicam, and omeprazole.
Following discharge, the foal exhibited repeated episodes of dyspnea and collapse 1–3 times per day and, 4 days later, was re-presented to the Equine Department, Vetsuisse Faculty of the University of Zurich. In between these episodes, the foal was bright and alert. Signs of dysphagia, coughing, and nasal discharge were not reported by the owner. On presentation, the foal was quiet but alert. Physical examination was unremarkable apart from auscultation of the lungs that revealed severe bilateral crackles and wheezes, worst in the cranioventral lung fields. The left sided facial nerve paralysis was unchanged. Repeated upper airway endoscopy revealed similar findings to those seen initially although the arytenoid cartilages appeared to be a little less swollen. There was still no abduction of the right and left arytenoid cartilages. Intermittent DDSP was evident, and collapse of the left arytenoid cartilage medially obstructing the rima glottidis could be produced by forcing breathing. Overnight, the foal exhibited an episode of severe respiratory distress that was unresponsive to intranasal O2 insufflation, and a second tracheotomy was performed. Over the next 4 days the condition of the foal showed no signs of improvement. Due to poor prognosis, the foal was euthanized.
Necropsy revealed a 1 cm × 1.5 cm, dark red, well-demarcated mass predominantly in the white matter of the obex. The location of the mass was intra-axial without meningeal involvement. On cut section, it was mainly confined to the right ventrolateral area of the white matter. The brain and cranial spinal cord were fixed in 4% formalin and embedded in paraffin wax according to standard procedures. Formalin-fixed, paraffin-embedded materials from these regions were further investigated by histology (hematoxylin and eosin) and special stains (van Gieson Elastica) on 3-μm sections. The laryngeal and pharyngeal areas were completely normal by macroscopic inspection.
Immunohistochemistry was performed to detect factor VIII (A0082, b rabbit polyclonal), smooth muscle actin (SMA; M0851, b mouse monoclonal), desmin (N1526, b mouse monoclonal), vimentin (M7020, b mouse monoclonal), glial fibrillary acidic protein (GFAP; Z0334, b rabbit polyclonal), and neuron-specific enolase (NSE; M087301, b mouse monoclonal). Immunohistochemistry was performed on an automated immunostainer. b Antigen retrieval was performed for all Abs except SMA (no pretreatment) and consisted of incubation in the microwave (98°C, 20 min) with citrate buffer. b After antigen retrieval and washing in phosphate buffered saline (PBS; pH 8.0), the sections were immersed in a peroxidase blocking solution b (10 min), washed in PBS, and incubated with the primary Abs at room temperature (RT). The Ab dilutions and incubation periods at RT were as follows: factor VIII (1:300, 40 min), SMA (1:400, 60 min), vimentin (1:100, 30 min), desmin (prediluted, 40 min), GFAP (1:500, 10 min), and NSE (1:150, 20 min). Detection of all Ab reactions was continued using a streptavidin peroxidase detection kit according to the manufacturer’s protocol. b Finally, the slides were developed using 9-ethylcarbazol-3-amine substrate solution (RT, 10 min), and counterstained with hematoxylin.
Microscopically, the mass was located predominantly in the ventrolateral white matter of the obex and consisted of many thin-walled, blood-filled vascular proliferations (Fig. 1). The adjacent gray matter was compressed by the mass. The vascular spaces were of variable diameter and lined by cuboidal to flattened spindle-shaped endothelial cells (Fig. 2). Numerous vessels were partially or totally occluded by fibrin thrombi. These fibrin thrombi were exclusively in the lumina of the vascular channels but not within the normal neural parenchyma. In between the vascular proliferations, a variable amount of hemosiderin-laden macrophages was present. There was no acute hemorrhage present within and/or adjacent to the hamartoma. The white matter in between the vascular channels and adjacent to the hamartoma was affected by rarefaction characterized by axon swelling and degeneration, presence of gitter cells, and focal dystrophic calcification. These lesions in the white matter were interpreted as malacia consistent with an effacing and compressive lesion caused by the vascular hamartoma. Hamartomatous vessels were negative by van Gieson Elastica whereas preexisting meningeal arteries showed a distinct black staining of the lamina elastica interna (not shown). The other investigated areas of the brain and cranial spinal cord were histologically unremarkable.
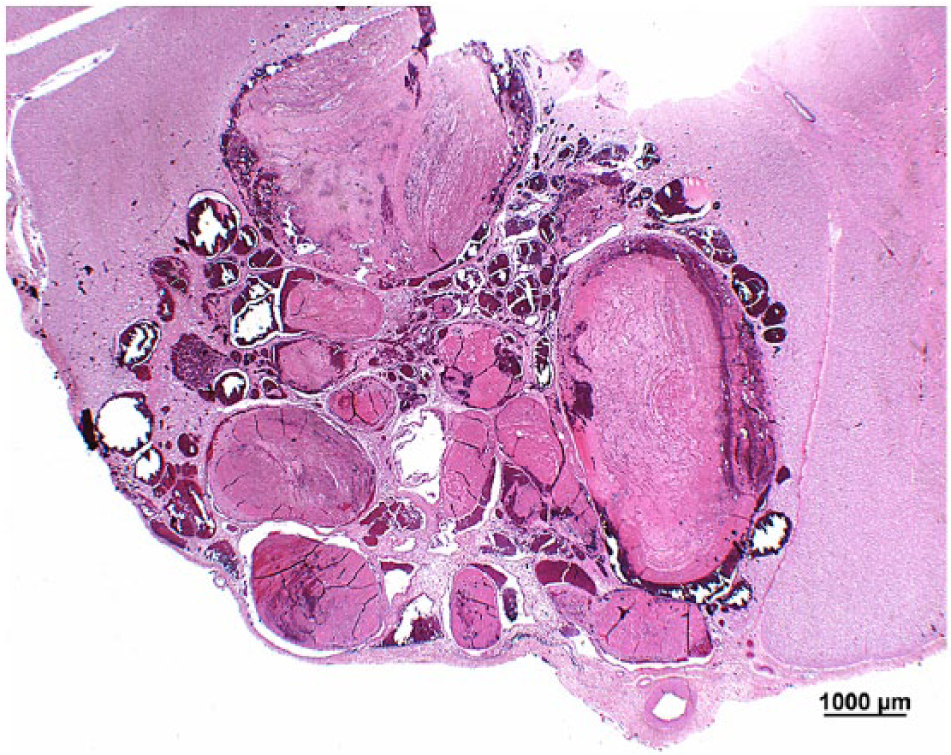
Foal, obex, vascular hamartoma. Multiple thin-walled vascular channels of variable diameter are filled with erythrocytes or fibrin thrombi. The hamartoma is surrounded by the meninges and the adjacent neuropil. Hematoxylin and eosin. Bar = 1,000 μm.
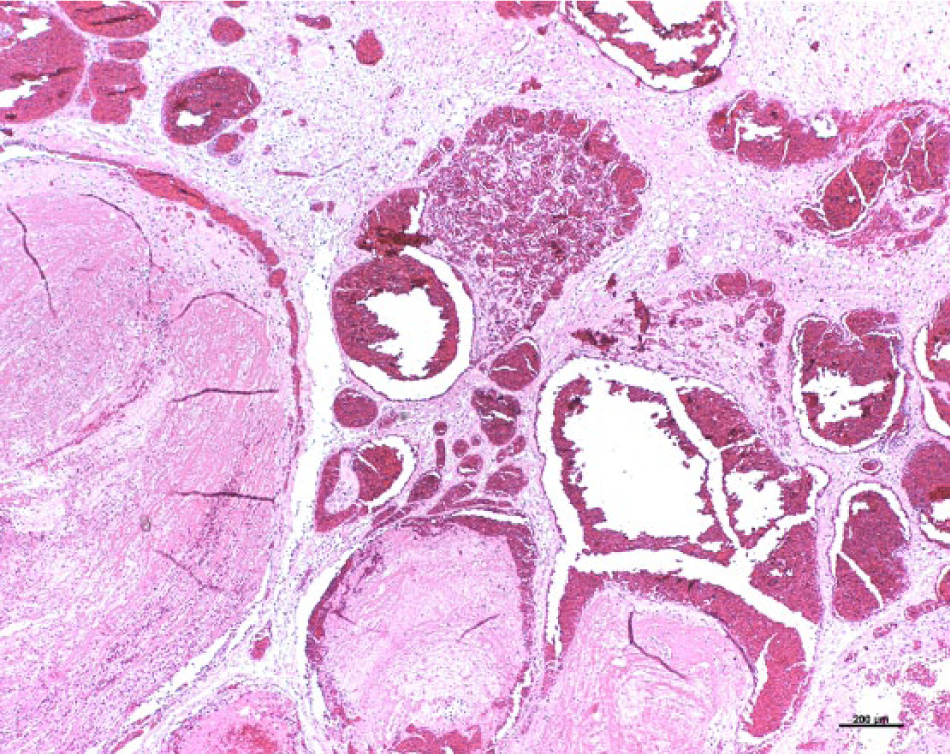
Foal, obex, vascular hamartoma. Multiple thin-walled vascular channels of variable diameter containing erythrocytes are lined by endothelial cells. Larger vessels contain fibrin thrombi. The surrounding white matter shows rarefaction and axonal swelling. Hematoxylin and eosin. Bar = 200 μm.
By immunohistochemistry, the endothelial lining of the vascular hamartoma was strongly positive for factor VIII (Fig. 3A). Thrombosed vessels showed irregular labeling with factor VIII in their lumina. The endothelial lining was also often positive for SMA (Fig. 3B), and rare thick-walled vessels showed additional distinctive labeling of their media. The endothelium and the stroma surrounding the hamartomatous vessels but not their medial layer were positive for vimentin (Fig. 3C). Immunoreactivity of the endothelial lining for vimentin and SMA suggests pericyte origin. 1 Vessel walls were occasionally positive for desmin, indicating myopericyte components. 1 Glial fibrillary acidic protein–positive astrocytic processes (Fig. 3D) and NSE-positive neurons (not shown) were observed between the hamartomatous vessels labeling the brain stem parenchyma.
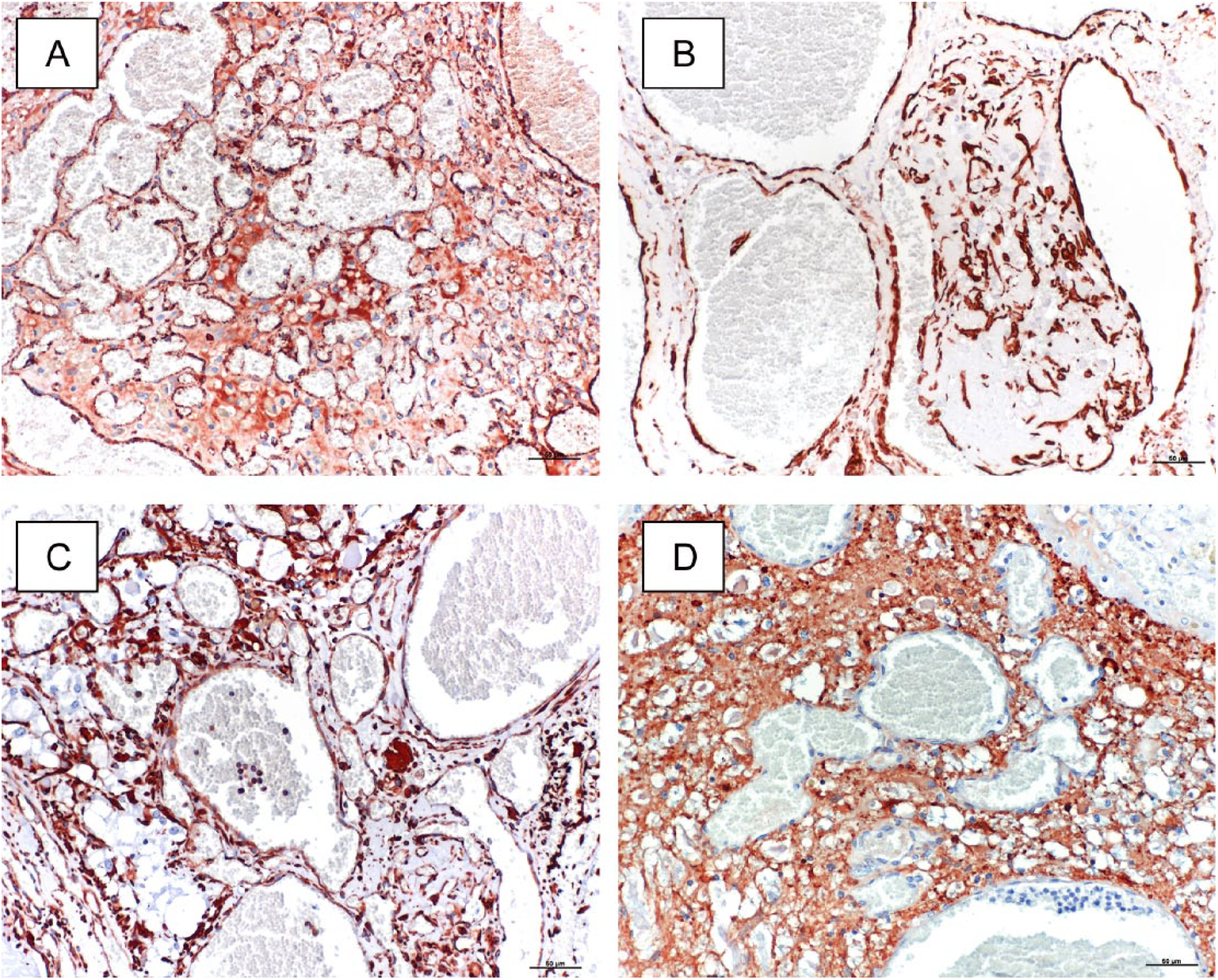
Foal, obex, vascular hamartoma.
Cerebral and cerebellar vascular hamartomas have been reported in cats,9,16 dogs,14,15 and a calf 2 ; however, the calf was diagnosed as a disseminated cavernous hemangioma. A cerebellar vascular hamartoma affected a 16-month-old cat with clinical signs suggestive for a cerebellar lesion such as nystagmus, ataxia, and high-stepping gait. Magnetic resonance imaging (MRI) and fine-needle aspiration of the abnormal tissue was performed, but the final diagnosis of a vascular hamartoma was made by histology after euthanasia. 16 Computed tomography (CT) was performed in a geriatric 11-year-old cat with neurological signs and revealed a mass in the thalamocortex. 9 The diagnosis of a vascular hamartoma was made histologically postmortem. Examination by CT revealed a well-demarcated mass in the frontal lobe, striatum, and thalamus of a 7-year-old dog resulting in the diagnosis of a cerebral vascular hamartoma postmortem. 14 Both imaging techniques (MRI and CT) were able to identify a mass but differential diagnosis included neoplasia and less likely granuloma. Postmortem histology confirmed the diagnosis of a vascular hamartoma. Vascular hamartomas are congenital malformations but often cause no clinical signs until later in life as illustrated in the geriatric cat suffering from a thalamocortic vascular hamartoma but with an onset of neurological symptoms at the age of 11 years. 9 In contrast, the foal in the current report presented much earlier, with clinical symptoms apparent shortly after birth; however, the localization and nature of the primary lesion remained unclear because diagnostic imaging techniques other than laryngeal endoscopy were not performed. The localization of the vascular hamartoma in the obex of the foal can be linked to the clinical signs observed because nuclei of the cranial nerves V, VII, IX, X, and XII originate from this brain region. These innervate muscles have afferent nerves that are responsible for coordinating the process of swallowing and the innervation of the facial muscles. 6 It is likely that pressure caused by the hamartoma compromised nerve function leading to dysphagia, impaired airway function with subsequent dyspnea, and facial nerve paralysis followed by corneal ulcer development due to keratoconjunctivitis sicca. Another explanation for the dyspnea could be damage to the formatio reticularis, in particular nuclei responsible for in- and expiration. 6 In contrast, previous reports in cats and dogs indicated a predilection for vascular hamartomas in the telencephalon and thalamocortex.9,14,15
Immunohistochemical patterns of the vascular hamartoma in the geriatric cat 9 and in the foal were similar with endothelial lining cells strongly positive for factor VIII (endothelial cells) and a second population positive for SMA indicating pericyte origin. The presence of normal neuronal tissue between the hamartomatous vessels is considered the main feature for differentiating this non-neoplastic lesion from a hemangioma. 15 A hemangioma is considered as a true benign neoplasm of endothelial cells. Some authors use the term angioma to refer to a vascular hamartoma. 10 Immunohistochemistry is useful for the correct classification of vascular hamartomas as demonstrated in a study on the brains of 5 dogs. 15
Vascular hamartomas in the spinal cord have been reported in a 10-month-old Nubian goat with progressive hind limb paraparesis, 11 a dog, 5 a Hereford calf, 3 and in a Welsh Cob filly with ataxia. 7 The hamartoma in the thoracic spinal cord of the goat led to widespread rarefaction and Wallerian degeneration due to compression as seen in the present case affecting the obex of the foal. In contrast to the present histological features with the predominance of thin-walled vascular proliferation resembling venous structures, the vascular hamartoma components in the goat consisted of a single endothelial layer lined by thick fibroelastic walls. These walls consisted of a broad tunica elastica staining black with van Gieson as well as smooth muscle fibers and connective tissue strongly labeled with desmin and actin but lacking vimentin immunoreactivity. In contrast, the vascular structures in the foal were structurally more similar to veins and might be classified as venous malformation as previously reported in cerebral vascular hamartomas in dogs and cats.9,14–16 A venous malformation in the thoracic segment of the spinal cord was diagnosed in an 8-month-old Welsh Cob filly with progressive ataxia in the hindquarters. 7 Secondary lesions such as adjacent malacia and compression most likely caused the clinical symptoms as reported in the present case. In addition, multiple hemorrhage and thrombosis as often present in and/or adjacent to vascular hamartomas might contribute to clinical signs. 14 In the present case, malacia, thrombi, and previous hemorrhage (hemosiderin-laden macrophages) was obvious. However, there was no acute hemorrhage adjacent to the vascular hamartoma. In summary, congenital malformations such as vascular hamartomas in the brain and/or spinal cord might be considered as a rare differential diagnosis in foals with central nervous signs, nerve deficits, and ataxia.
Footnotes
Acknowledgements
The authors are grateful to the laboratory technical staff of the Institute of Veterinary Pathology (in particular Sabina Wunderlin), Vetsuisse Faculty, University of Zurich for technical help.
a.
SNAP foal IgG test, IDEXX Switzerland AG, Liebefeld-Bern, Switzerland.
b.
Dako Schweiz AG, Baar, Switzerland.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
