Abstract
A monoclonal antibody (mAb) was produced against immunoglobulin M (IgM) of South American camelids. A single radial immunodiffusion (SRID) assay and a competitive enzyme-linked immunosorbent assay (ELISA) were developed to measure IgM in serum samples. Isotype and specificity of the mAb were assessed. The performance of the SRID assay was preliminarily evaluated in terms of working range, plate stability over a 4-week period, and initial intra- and interassay variation. The concentration of IgM was determined in 55 samples by SRID assay and ELISA, and results were not significantly different by t-test (0.64 ± 0.19 mg/ml for the SRID assay, and 0.58 ± 0.24 mg/ml for ELISA; P = 0.1489). The mAb was shown to be stable over the 4-week evaluation period, and the SRID assay was reproducible when tested in triplicate for intra-assay variability and in quadruplicate for interassay variability, with a percentage coefficient of variation of less than or equal to 5%. Also, the SRID assay proved to be sensitive enough to measure IgM levels in undiluted serum samples, and had a good correlation with ELISA. The current study is intended to submit a preliminary report of a mAb against IgM of South American camelids, and suggest the future potential of the mAb developed for diagnostic application, including use in the SRID assay.
The breeding of South American camelids (SCs) has become a worldwide practice. Although little is known about their immunological status, these species have been reported to suffer from many infectious diseases1,11,15 and immunodeficiencies.3,10 In this context, an appropriate sanitary control is required in order to prevent zoonotic diseases among consumers, breeders, and farmers in general. Furthermore, infectious diseases and immunodeficiencies in these animals could result in considerable economic loss. Therefore, early diagnosis appears to be a convenient strategy to adopt.
Both immunoglobulin G (IgG) and IgM are secreted in great amounts in the colostrum, and absorption difficulties have been reported to be an important cause of failure in the passive transfer of immunoglobulin in SC newborns. 6 In fact, newborns with low serum immunoglobulin levels have a higher risk of early death. 8 Previous reports have analyzed failure in the passive transfer of immunoglobulin by measuring serum immunoglobulins through single radial immunodiffusion (SRID) in SC newborns.16–18 As previously described, all camelids (including SCs) have a particular type of immunoglobulin, which is devoid of light chains, and is known as a heavy-chain antibody (HCAb). 14 The serum immunoglobulin pool consists of 13% IgM, 65% conventional IgG (IgG1, 150 kDa), and 22% HCAbs (IgG2 and IgG3, 90 and 80 kDa, respectively). This complex structure may become an obstacle in producing standards for quantitative techniques—for example, for calibration curves—and particularly for SRID assays, because the existence of IgG with various molecular weights might lead to multiple precipitation rings when an anti-IgG is used and hence to misinterpretation of results. Given this complexity, to date, there is no commercial kit available to correctly quantify immunoglobulin in SC serum. For these reasons, it was decided to produce and characterize a monoclonal antibody (mAb) against the IgM of SCs, and to use it for the development of a SRID assay and an enzyme-linked immunosorbent assay (ELISA), both assays designed to quantify IgM from SCs. Immunoglobulin M was chosen as the potential analyte, because SCs only have IgM as a conventional antibody, and its heavy chain is substantially different from the other immunoglobulins, thus avoiding any cross-reactivity of mAb. The performance of both immunoassays was preliminarily checked.
The use of mAb in precipitation assays has already been described, and is said to yield specific and reproducible results. 7 However, previous articles have indicated the need to use more than 1 mAb.2,13 In the current report, the production of a single mAb capable of interacting with its specific antigen and yielding visible and measurable precipitation rings is described. The precision, reproducibility, and sensitivity of the SRID assay were assessed on a preliminary basis, and its suitability and performance were compared against those obtained with a competitive ELISA, also developed in-house. Unlike ELISA, the SRID assay can also be used to make a field diagnosis, allowing the operator to conduct the test without any complex laboratory equipment.
Llama (Llama glama) IgM was purified from normal llama serum (NLS, defined as serum from young healthy animals with no history of disease for at least 90 days) by ammonium sulfate precipitation at 30% concentration. Pellets were then resuspended in 7.5 ml of phosphate buffered saline (PBS), and processed by a size-exclusion chromatography (SEC) a using PBS as the mobile phase. Fractions of 5 ml were collected, and absorption was measured in a spectrophotometer b at 280 nm. The chromatogram showed 2 different peaks (Fig. 1), namely IgM (first peak) and other serum proteins (second peak). Fractions of interest were run in a 10% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) under reducing conditions and stained with 0.1% Coomassie brilliant blue red-tinted solution c (Fig. 1, panel). Total protein concentration was measured using a commercial kit based on the bicinchoninic acid (BCA) method, c and relative purity was analyzed by densitometry using a specific software. d Fractions were concentrated to a final protein concentration of 2.15 mg/ml by ultrafiltration e and stored at 4°C in PBS with 0.05 M trehalose and 0.5 M NaCl. Purified IgM was used as the antigen to coat ELISA plates, to produce SRID and ELISA standards, and to prepare µ-heavy chain as the immunogen of choice for mAb production.
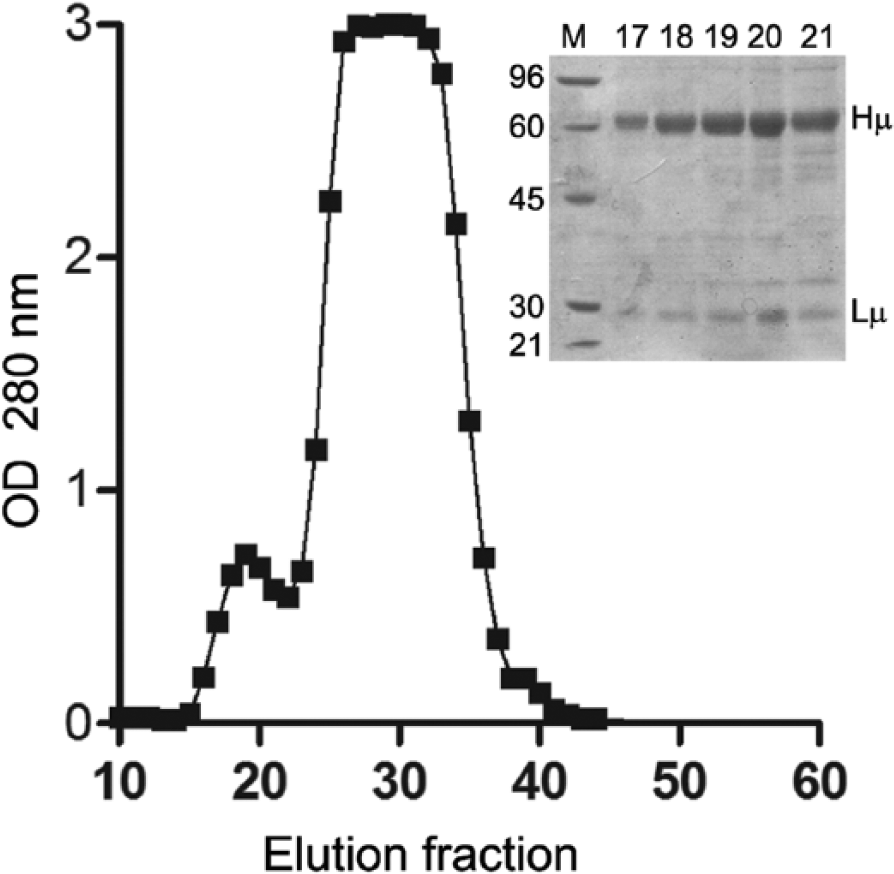
Chromatogram of the size-exclusion chromatography. The upper-right panel shows the sodium dodecyl sulfate–polyacrylamide gel electrophoresis analysis of fractions 17–21 under reducing conditions, which contained mainly immunoglobulin (Ig)M. Lane M corresponds to molecular weight markers, and bands corresponding to IgM heavy-chain (Hµ) and light-chain (Lµ) are indicated on the right. OD = optical density.
The IgM heavy chain was obtained by reduction alkylation of IgM, as described previously. 12 Briefly, the purified IgM was first dialyzed against 0.5 M Tris–HCl (pH 8) and then reduced with 0.2 M 2-mercaptoethanol for 2 hr at room temperature. Iodoacetamide f was added in a concentration of 130 mg/ml, and the solution was incubated at 4°C for 30 min. Then, the µ-heavy chain was separated by SEC. g The purity of these fractions was analyzed by 10% SDS-PAGE, while their protein concentration was assessed by the BCA method. c This protocol yielded protein with acceptable purity for the immunization of animals (data not shown).
Monoclonal antibody was produced in BALB/c mice. All animal handling operations were performed following the standard guidelines set down by the School of Pharmacy and Biochemistry, University of Buenos Aires, Argentina. Four female mice were intraperitoneally inoculated once with 250 µg of purified µ-heavy chain in complete Freund adjuvant (CFA), h and then boosted weekly 3 additional times using 10 µg of total protein in CFA. Animals were euthanized in a CO2 chamber, and their spleen was removed and disaggregated to obtain a splenocyte suspension. Fusion was performed using 50% polyethylene glycol with an average molecular weight of 3,500 (PEG 3500) and the NS0 mouse myeloma cell line, in accordance with previously reported protocols 5 in a 5:1 ratio (splenocytes:NS0 cell line). After 2 weeks, hybridomas were screened for antibody production using an indirect ELISA, and then cloned by the limit dilution technique. The absence of reactivity against IgG1, IgG2, and IgG3 was checked by the same ELISA. Briefly, screening was performed, and cross-reactivity was studied by coating microplates with 1 µg of the corresponding antigens (IgM, IgG1, IgG2, or IgG3, purified following the procedure described elsewhere 4 ) for 1 hr at 37°C, blocking overnight with 3% bovine serum albumin (BSA) in PBS and then incubating 50 µl of culture supernatants (1 hr at 37°C). Bound antibodies were detected using a commercial antibody (goat anti-mouse horseradish peroxidase [HRP]-labeled, i diluted 1:10,000), developed with 3,3′,5,5′-tetramethylbenzidine (TMB) substrate j and stopped with the addition of 50 µl of 4N H2SO4. Optical density was measured at 450 nm in a microplate reader. k The mAb production protocol yielded 6 specific clones with high specificity toward IgM, as shown in Table 1.
Specificity of monoclonal antibodies anti–immunoglobulin (Ig)M.
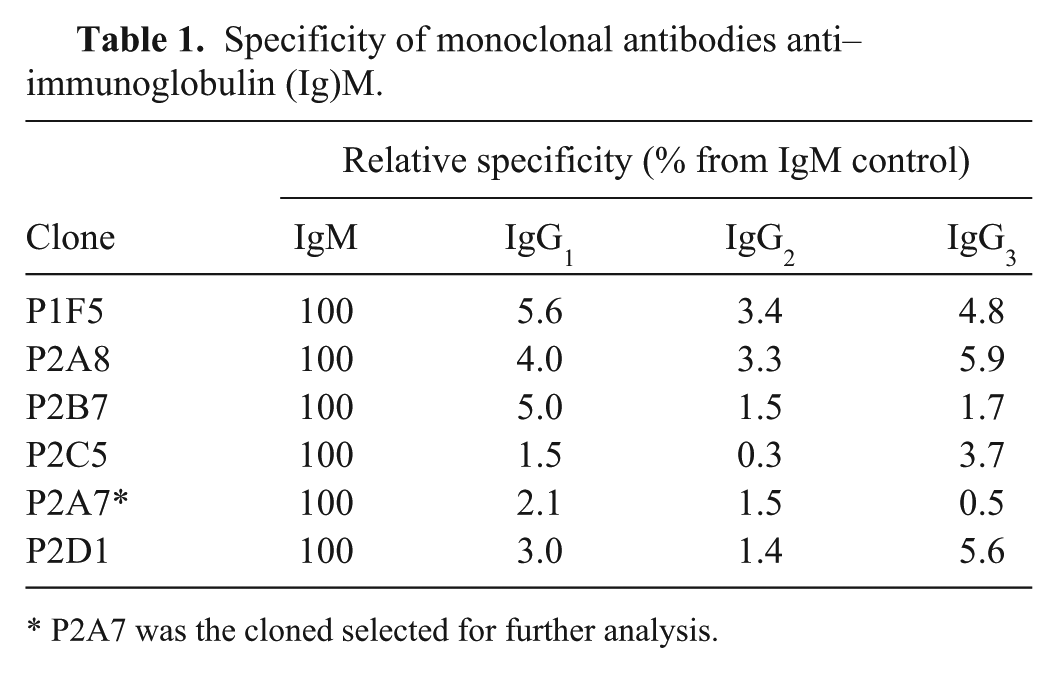
P2A7 was the cloned selected for further analysis.
A single clone (P2A7) was selected for further production of anti-IgM because of its stability, high reactivity, and remarkable specificity. Clone P2A7 was characterized as mouse IgM–anti-llama IgM in an ELISA conducted as described for supernatant screening, but using specific anti-mouse isotype antibodies (anti-mouse IgG1, anti-mouse IgG2a, anti-mouse IgG2b, anti-mouse IgG3, and anti-mouse IgM, all HRP-labeled i ). Because of the low production of mAb in culture supernatants, ascitic fluid was produced by priming 4 BALB/c mice with incomplete Freund adjuvant 15 days before injecting 1 × 106 P2A7 cells in sterile PBS. After 7–10 days, ascitic fluid was collected, and the mice were euthanized.
Monoclonal antibodies were purified from ascitic fluid as described in the literature. 9 Briefly, ascitic fluid was centrifuged at 3,500 × g, followed by ammonium sulfate precipitation at 50% saturation. Pellets were resuspended in PBS, dialyzed against 20 mM Tris–HCl (pH 8.5), and then loaded into a precast ion-exchange matrix l using a fast protein liquid chromatography system. m The column was equilibrated with 20 mM Tris–HCl (pH 8.5), and bound proteins were eluted with a linear gradient from 0% to 100% of 20 mM Tris–HCl and 1 M NaCl (pH 8.5). The protein concentration of each fraction was determined by BCA, c and relative purity was assessed by 10% SDS-PAGE followed by Coomassie blue staining. The purification of mAb from mouse ascitic fluid yielded fractions containing approximately 1.26 mg/ml of highly pure immunoglobulin (>95%).
The SRID media was composed of 1.2% agarose, f 3% PEG 6000, n 10% sucrose, n 0.05%, Tween-20, 0.05% sodium azide, and 40 µg/ml of mAb. The mixture was poured in plates up to 1.2 mm of thickness, and allowed to gel at room temperature. Three-millimeter diameter wells were cut, and 10 µl of IgM standards—corresponding to IgM at 1.40, 1.20, 0.86, 0.70, 0.54, 0.38, and 0.27 mg/ml—were loaded, as well as 10 µl of NLS samples. Plates were then incubated for 72 hr at room temperature, and the diameter of precipitation rings was measured using a calibrated magnifying glasso used for scale comparison purposes.
Good linearity (r2 = 0.9935) was observed in the SRID assay over the entire range of the standard curve (Fig. 2A), suggesting its suitability to test undiluted samples, as serum IgM concentration is expected to measure approximately 1 mg/ml in NLS. For assay validation purposes, the intra- and interassay variability of the SRID assay was tested, both of which proved to have a percentage coefficient of variation (%CV) of less than 5% (Table 2). Furthermore, the SRID assay was observed to remain stable for at least 4 weeks, with a %CV of only 1.5% (data not shown). Intra-assay variation was preliminarily evaluated by measuring in triplicate 4 aliquots of purified IgM within the standard curve range (at 0.50, 0.75, 1.00, and 1.40 mg/ml) in the same plate. Interassay variability was tested by measuring 10 NLS samples ranging from 0.72 to 0.46 mg/ml of IgM, in 3 different plates incubated at the same time. The overall %CV was calculated by averaging individual %CV values for each sample. Stability was checked by testing the same serum sample in different plates once a week for 4 weeks. All SRID plates were prepared as described, stored at 4°C, and analyzed as described.
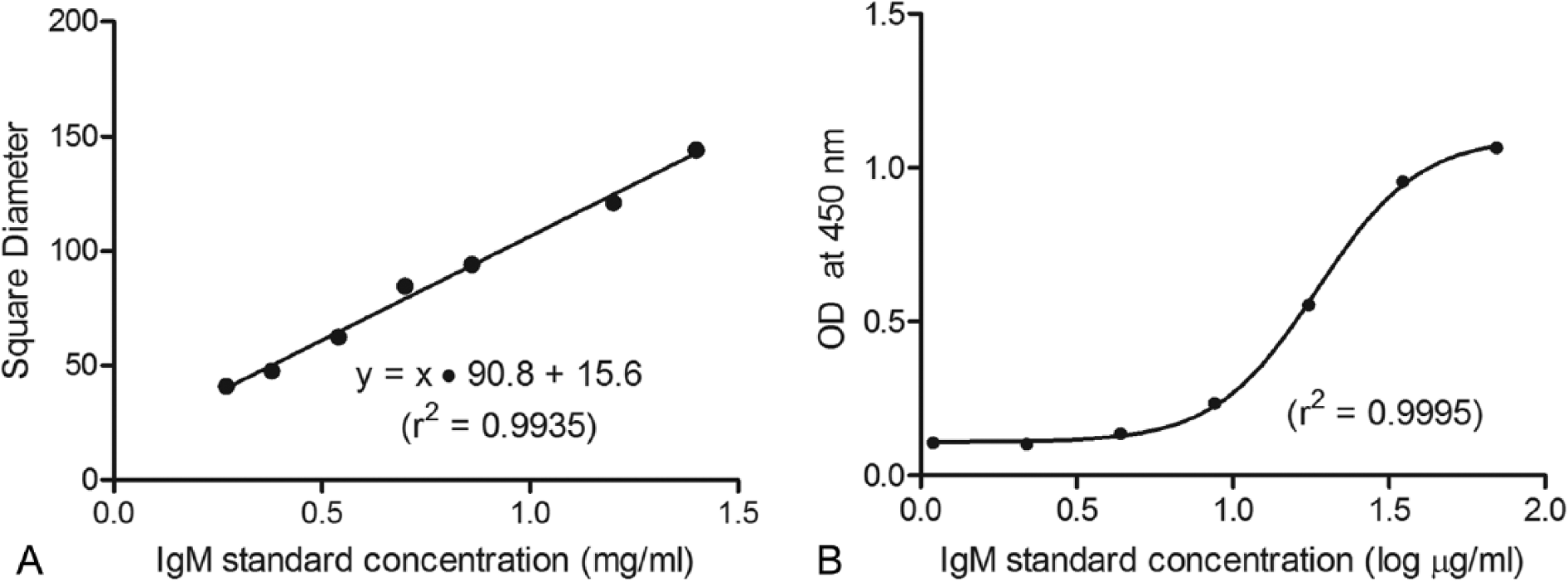
Calibration curve for single radial immunodiffusion (
Intra-assay variation of serum immunoglobulin M measured by single radial immunodiffusion.*
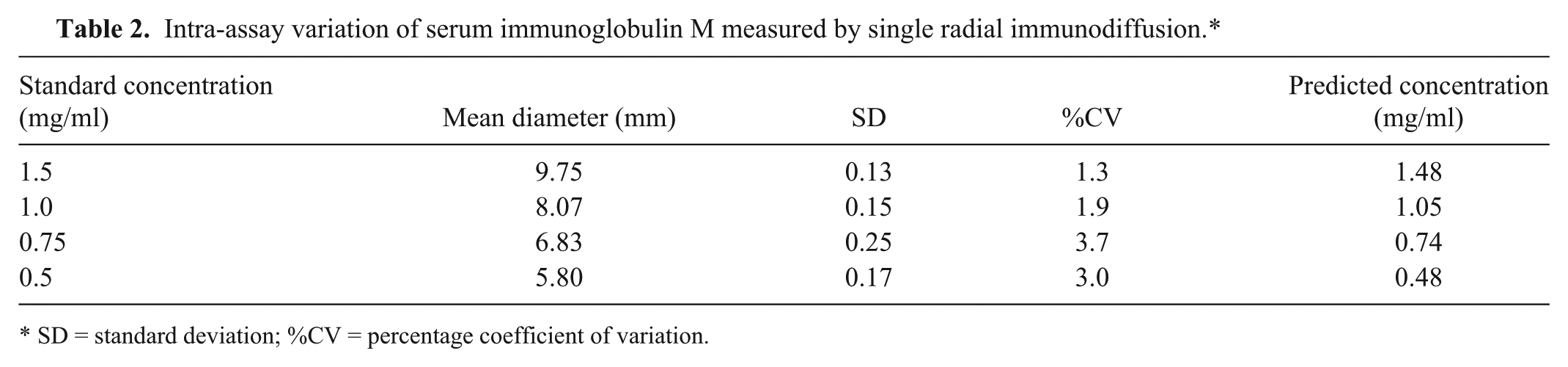
SD = standard deviation; %CV = percentage coefficient of variation.
A competitive ELISA was conducted to quantify IgM by coating wells with 1 µg of llama IgM purified as described for 1 hr at 37°C, blocking overnight with 3% BSA in PBS at 4°C. In parallel, NLS samples or llama IgM standards—corresponding to IgM at 70.00, 35.00, 17.50, 8.75, 4.37, 2.19, and 1.09 µg/ml—were preincubated overnight at 4°C with a dilution of 1:5,000 of self-produced mAb anti-llama IgM. After preincubation, samples and standards were added to the plate and incubated for 1 hr at 37°C. Bound immunoglobulins were detected using goat anti-mouse antibody, HRP conjugated, i at 1:10,000 for 1 hr at 37°C. Reactivity was developed by using 100 µl of TMB j and stopped with the addition of 50 µl of 4N H2SO4. Optical density was measured at 450 nm in a microplate reader. k The nonlinear regression of the ELISA standard curve (Fig. 2B) had excellent fit (r2 = 0.9995).
In order to test these assays and compare their performance, 55 NLS samples were tested both by SRID and competitive ELISA. The average value of IgM was 0.64 ± 0.19 mg/ml for the SRID assay, and 0.58 ± 0.24 mg/ml for ELISA. A linear regression was performed using a Deming approach, and a slope of 0.9466 ± 0.1579 was obtained (Fig. 3). Residues were analyzed by the Bland–Altman method (not shown).
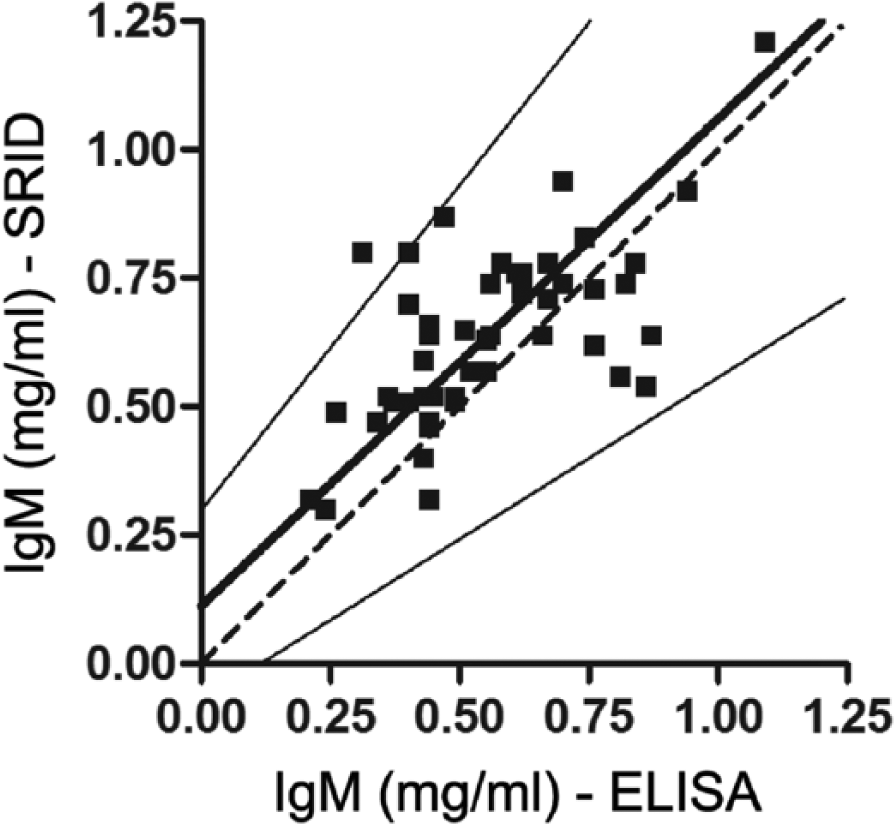
Deming regression of immunoglobulin (Ig)M concentrations obtained by single radial immunodiffusion (SRID) and enzyme-linked immunosorbent assay (ELISA). The thick line stands for the Deming regression of the experimental data, while the dashed line shows the ideal fit. Thin lines show the 95% confidence interval of the regression.
The current study describes the production of a mAb specific for the IgM of SCs, which might be suitable for SRID and ELISA for IgM quantitation purposes. In a preliminary evaluation, the mAb was shown to be stable over the 4-week evaluation period, and reproducible when tested in triplicate for intra-assay variability and in quadruplicate for interassay variability. Also, the SRID assay was adequate to measure IgM levels in undiluted serum samples. Furthermore, its comparison with the competitive ELISA assay showed a good correlation, and also that the SRID assay may be a simpler, cheaper, and more suitable method for field diagnosis. To conclude, this is a preliminary report on a mAb anti-IgM that can be regarded as a simple, effective method to quantify antibodies in SCs. Because there is a correlation between low levels of serum antibodies and early death among SC newborns, this mAb and these techniques might stand as suitable methods to evaluate the effectiveness of passive transfer of immunity in SC newborns through field diagnosis, and therefore help prevent unnecessary deaths. Further work will be necessary to fully characterize the performance of this assay and gain deeper insight into IgM levels and their significance in a clinical context.
Footnotes
a.
Ultrogel Aca 34 matrix, Pall Corp., in a glass column of 3 cm × 45 cm, GE Healthcare Bio-Sciences AB, Uppsala, Sweden.
b.
Ultrospec 1100 pro, GE Healthcare Bio-Sciences AB, Uppsala, Sweden.
c.
Coomassie brilliant blue R-250 solution, BCA protein assay kit; Pierce Biotechnology Inc., Rockford, IL.
d.
Scion image software, Scion Corp., Frederick, MD.
e.
Amicon Millipore filter, Merck & Co. Inc., Whitehouse Station, NJ.
f.
Sigma-Aldrich, St. Louis, MO.
g.
Sephadex G-100 matrix in a glass column of 1 cm × 100 cm, GE Healthcare Bio-Sciences AB, Uppsala, Sweden.
h.
Freund adjuvant, Pierce Biotechnology Inc., Rockford, IL.
i.
Bethyl Laboratories Inc., Montgomery, TX.
j.
TMB, Invitrogen Corp., Carlsbad, CA.
k.
Multiskan EX microplate reader, Thermo Fischer Scientific Inc., Waltham, MA.
l.
Q-Sepharose, GE Healthcare Bio-Sciences AB, Uppsala, Sweden.
m.
ÄKTA Purifier, GE Healthcare Bio-Sciences AB, Uppsala, Sweden.
n.
Anedra S.A., San Fernando, Argentina.
o.
Scale magnifying glass, Spi, Osaka, Japan.
Declaration of conflicting interests
The author(s) declared no potential conflict of interests with respect to the research, authorship, and/or publication of this article.
Funding
The authors received financial support from Agencia de Promoción Científica y Técnica (ANPCyT; grant PICT 2008-1565).
