Abstract
An antigen-capture enzyme-linked immunosorbent assay (ELISA) was developed for use as a presumptive screening test for detection of Salmonella enterica serovar Enteritidis and other group D Salmonella in poultry hatchery environments. A mixture of 2 monoclonal antibodies that recognize different forms of the lipopolysaccharide O-antigen was used for specific detection of group D Salmonella. The performance of the ELISA was evaluated in comparison to standard Salmonella culture procedures. Culture for each sample included nonselective enrichment with buffered peptone water and primary selective enrichment and delayed secondary enrichment with both tetrathionate and Rappaport–Vassiliadis broths. One thousand fifty-seven samples were collected from poultry hatcheries over a 5-year period (received in 85 submissions), and S. Enteritidis was recovered from 106 (10%) of them. The diagnostic sensitivity and specificity of the ELISA relative to culture were 97.2% and 99.6%, respectively, on a sample basis and were both 100% on a submission basis. Delayed secondary enrichment increased the number of S. Enteritidis culture and ELISA-positive samples as compared to nonselective enrichment and primary selective enrichment by 25%. A significantly higher (P < 0.05) number of S. Enteritidis culture- and ELISA-positive results were obtained from Rappaport–Vassiliadis broth than from tetrathionate broth or buffered peptone water cultures. The results indicate that this ELISA procedure may be useful for screening poultry hatchery environmental samples for the presence of S. Enteritidis.
Introduction
Salmonella enterica serovar Enteritidis is a leading cause of human salmonellosis in many countries worldwide.6,7 Epidemiological investigations have implicated raw or undercooked eggs as the vehicle of transmission of S. Enteritidis to consumers in a high percentage of outbreaks. 2 Intact eggs may become directly contaminated with S. Enteritidis during egg formation as a result of infections of the reproductive tissues of laying hens or indirectly by penetration of the organism through the eggshell following exposure to an environment contaminated with S. Enteritidis.6,15 Identification of contaminated hatcheries is a principal objective in efforts to reduce transmission of S. Enteritidis infection to susceptible individuals.
In the current monitoring system in Canada, fluff samples are taken routinely from poultry premises for Salmonella culture under the authority of a provincial program. If S. Enteritidis is found, it is reported to the Canadian Food Inspection Agency by the province, the identified premise is cleaned and disinfected following depopulation, and environmental samples are taken to prove S. Enteritidis freedom prior to repopulation.
Bacteriological examination of samples collected from poultry hatcheries is the standard method for monitoring these establishments for the presence of S. Enteritidis and other Salmonella. 26 However, cultural procedures for isolation of Salmonella are labor intensive, expensive to perform, and time-consuming.1,13 Thus, the development of alternative methods that would detect S. Enteritidis more efficiently1,13 would be useful in surveillance programs that aim to identify hatcheries that are contaminated with S. Enteritidis and in monitoring the cleanup and disinfection of S. Enteritidis–contaminated premises.
Salmonella Enteritidis and the avian pathogen S. enterica serovar Gallinarum biotype Pullorum (S. Pullorum) are members of group D in the internationally recognized Kauffmann–White Salmonella classification scheme. 19 In this classification scheme, members of the same group have similar lipopolysaccharide (LPS) O-antigen structure. The structure of the S. Pullorum LPS O-antigen has been determined to be indistinguishable from that of S. Enteritidis,4,9,20 and anti-LPS monoclonal antibodies (mAbs) that were specific for S. Pullorum, S. Enteritidis, and other Salmonella group D serovars have been produced. 4
In the present study, an antigen-capture enzyme-linked immunosorbent assay (ELISA) that was based on a combination of 2 mAbs that react with different S. Pullorum LPS O-antigen epitopes was developed as a presumptive screening test for detection of S. Enteritidis in poultry hatchery environmental samples. Although the LPS of many strains of group D Salmonella contain significant amounts of these 2 epitopes, the LPS of some strains contain significant amounts of one or the other but not both of the epitopes, 4 and thus a mixture of the 2 mAbs was used. The performance of the ELISA was evaluated in comparison to standard cultural isolation procedures using naturally contaminated field samples.
Materials and methods
Samples
The samples analyzed in the present study were obtained from poultry hatcheries in various Canadian provinces, and samples were taken after cleaning and disinfection. The samples were collected using sterile sponges a (4 cm × 7 cm × 0.6 cm) premoistened in 1% buffered peptone water (BPW) b from the following sites: hatchers, furnaces, vents, drains, counters, floors, air ducts, ceilings, fans, lights, trolleys, tables, and shipping docks. Once a surface had been sampled, the sponge was placed in a sterile bag, c kept cool in containers by ice-packs, and shipped to the Canadian Food Inspection Agency, Ottawa Laboratory (Fallowfield), Animal Health Microbiology diagnostic laboratory for testing for the presence of Salmonella. All samples collected from an individual hatchery on the same day and submitted together for testing were referred to as one submission.
Culture methods
Fifty milliliters of BPW was added to each bag containing a sponge, the contents of the bag were agitated in a stomacher d for 2 min, and the bag and contents were incubated at 35°C for 18–24 hr for nonselective enrichment (NSE). Using sterile 10-µl disposable loops, e a loopful of the NSE culture was streaked onto brilliant green sulfadiazine (BGS) agar b and xylose–lysine–tergitol-4 (XLT4) agar b to obtain isolated colonies. The plates were incubated at 35°C for 20–24 hr and examined for colonies with morphology typical of Salmonella spp. 22 The plates were then incubated at 35°C for an additional 20–24 hr and reexamined. Up to 3 typical colonies were selected from each culture plate for presumptive and confirmed Salmonella spp. identification.
Two milliliters and 0.2 ml of the NSE culture was inoculated into 18 ml of Hajna tetrathionate (TT) broth b and 20 ml of Rappaport–Vassiliadis (RV) broth, f respectively, and the broths incubated at 42°C for 20–24 hr21,22 for primary selective enrichment (PSE). A loopful of each of the TT and RV PSE cultures was streaked onto BGS and XLT4 agars as described previously. The plates were incubated and examined for colonies typical of Salmonella spp. as described previously.
The remainder of the TT and RV PSE culture broths was incubated at room temperature (22 ± 2°C) for 5–7 days. One milliliter of the TT culture was inoculated into 9 ml of TT broth, and 0.1 ml of the RV culture was inoculated into 10 ml of RV broth.22,25 The freshly inoculated TT and RV broths were incubated at 42°C for 18–24 hr for delayed secondary enrichment (DSE), and a loopful of the TT and RV DSE cultures was streaked onto BGS and XLT4 agars as described previously. The plates were incubated and examined for colonies typical of Salmonella spp. as described previously.
Colonies resembling Salmonella spp. on BGS and XLT4 agar were subcultured to trypticase soy agar, b and then presumptively identified, biochemically, by inoculation to triple sugar iron agar, b lysine iron agar, b and urea agar b slants along with a positive agglutination with rabbit Salmonella O antiserum poly A-I b and Salmonella group D antiserum. a Individual isolates were further identified using either a commercial identification kit g or a commercial identification system, h both used according to the manufacturer’s instructions. Salmonella isolates were submitted to the Salmonella OIE Reference Centre, Public Health Agency of Canada, Guelph, Ontario, for serotyping and phage typing. The NSE, PSE, and DSE culture broths were stored at 4°C for up to 10 months prior to ELISA testing.
ELISA reagents, control antigens, and test samples
Hyperimmune polyclonal sera against S. Pullorum was produced in rabbits by the inoculation of a mixture of formalin-killed cells of S. Pullorum strain 11, a standard antigenic type strain, and S. Pullorum strain 77, a variant antigenic type strain. The inoculation mixture contained approximately 1 × 109 colony forming units (CFU)/ml of each strain. Rabbits were immunized according to a protocol published previously. 14 Blood was collected 11 days after the last vaccination, and the serum was separated and stored at −20°C. All experiments involving animals were approved by the local Animal Care Committee under the guidelines of the Canadian Council on Animal Care.
A combination of 2 murine mAbs, M1801 and M1797, produced in-house, 4 were used in the ELISA procedure. Both mAbs react specifically with Salmonella group D LPS O-antigen but recognize different group D LPS O-antigen epitopes. 4 The 2 mAbs were tissue culture derived in medium containing 10% fetal calf serum. Information on the isotype of the mAbs was published previously. 4
Two different cell concentrations of S. Pullorum strain 11 (7.5 × 106 CFU/ml and 2.5 × 106 CFU/ml) and of S. Pullorum 77 (1 × 107 CFU/ml and 2.5 × 106 CFU/ml) were used to provide a strong positive and a weak positive control with mAbs M1801 (mean optical density [OD] 1.20 and 0.43, respectively; Table 1) and M1797 (mean OD 1.26 and 0.41, respectively; Table 1). Salmonella enterica serovar Typhimurium American Type Culture Collection 14028, a group B Salmonella strain, was used as a negative control. Concentrations of control antigens and antibody reagents were optimized in preliminary experiments using checkerboard titration (data not shown). Salmonella Enteritidis Ottawa Laboratory Fallowfield strain 1863 was included in the determination of the analytical sensitivity of the ELISA.
Enzyme-linked immunosorbent assay (ELISA) reactivity of positive and negative control antigens with a mixture of monoclonal antibodies M1801 and M1797.
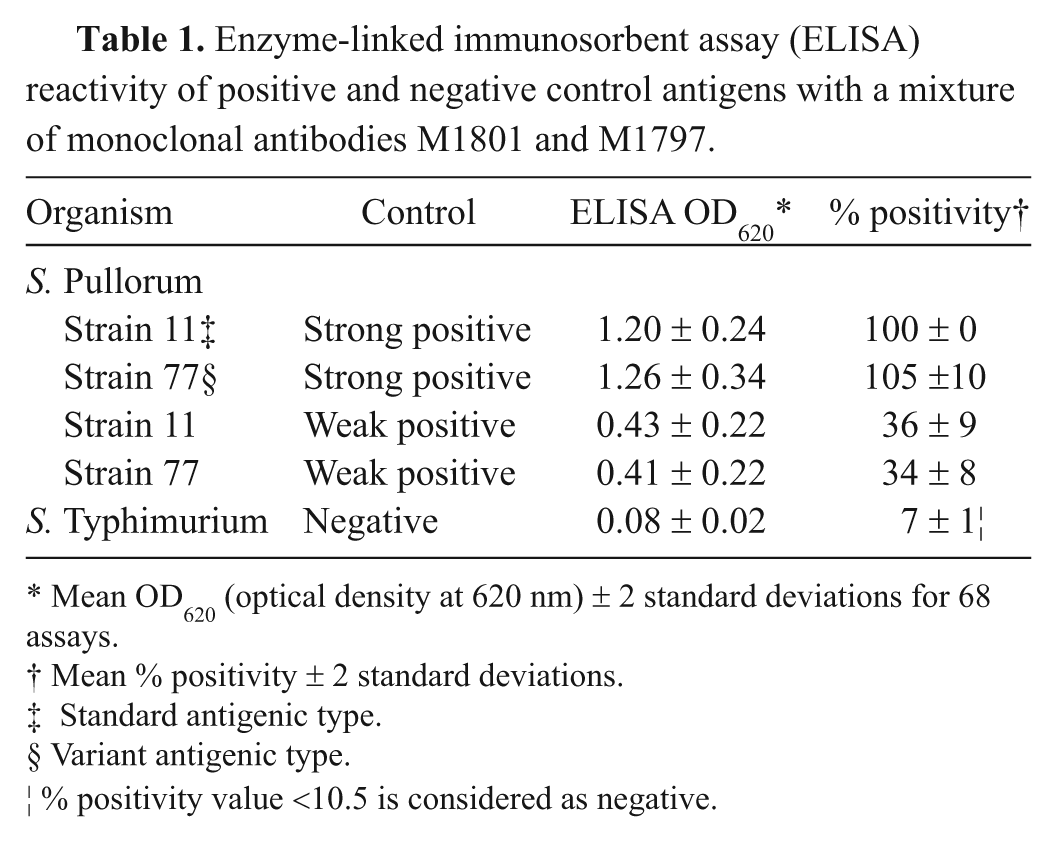
Mean OD620 (optical density at 620 nm) ± 2 standard deviations for 68 assays.
Mean % positivity ± 2 standard deviations.
Standard antigenic type.
Variant antigenic type.
% positivity value <10.5 is considered as negative.
Volumes of approximately 1.0 ml of the NSE (BPW), PSE (TT and RV), and DSE (TT and RV) broths (total of 5 broths for each sample) and the control cultures were heated for 15 min at 100°C approximately 18 hr before testing, stored at 5°C overnight, and brought to room temperature before ELISA testing.
ELISA procedure
A double antibody sandwich ELISA format was used. One hundred microliters of a 1-in-20,000 dilution of the rabbit polyclonal anti-Salmonella serum in 0.06 M carbonate buffer (pH 9.6), containing sodium bicarbonate i and sodium carbonate, j was added to each well of a 96-well polystyrene plate k and incubated for 18 hr at room temperature. Plates were washed l four times with 0.01 M phosphate buffer containing monobasic sodium phosphate, i dibasic sodium phosphate, i 0.15 M NaCl, m and 0.05% Tween 20 n (PBST). One hundred microliters of each control antigen preparation and each culture enrichment broth sample was added to 2 wells, and the plates were incubated for 1 hr at room temperature. The plates were washed as described previously, 100 µl of a mixture of mAb M1801 diluted 1:50 in PBST and mAb M1797 diluted 1:20 (or 1:30 depending on the mAb tissue culture lot) in PBST, and the plates were incubated for 1 hr at room temperature. The plates were washed as previously described, 100 µl of horseradish-peroxidase–conjugated goat anti-mouse immunoglobulin G (H+L) o diluted 1:20,000 in PBST was added to each well, and the plates were incubated for 1 hr at room temperature. The plates were washed, 100 µl of 3,3N,5,5N-tetramethylbenzidine/hydrogen peroxide substrate p was added to each well, and the plates were shaken for 10 min. The OD at 620 nm was determined using a microplate reader. q
The absorbance values were normalized to provide a more accurate comparison of results from day-to-day by expressing the OD values as a percentage of the S. Pullorum strain 11 strong positive control that was included in each assay. 10 A relative percent positivity (%P) was calculated using the following formula:

An ELISA result was interpreted as positive if the %P value for the test sample was ≥ 10.5. A %P value of ≥10.5 was selected as it provided the highest agreement (lowest number of false-positive and lowest number of false-negative results, calculated using the kappa statistic) between the ELISA and the S. Enteritidis culture results.
Statistical analysis
Chi-square tests of homogeneity were used to compare the number of samples positive for S. Enteritidis by culture and the number of samples positive for group D Salmonella by ELISA at different enrichment stages and to compare the type and number of enrichment broths positive for S. Enteritidis by culture and positive for group D Salmonella by ELISA broths. Data were considered to be statistically significant when P < 0.05. The kappa statistic was calculated and used to determine the agreement between the culture and ELISA tests. A kappa value of >0.7 was considered excellent agreement. Statistical calculations were performed using a commercially available software package. r
Results
ELISA reactivity for control antigens
The mean ELISA OD results for the 5 control antigens with the mixture of mAbs M1801 and M1797 are shown in Table 1. Ninety-six percent (328/340) of the OD values for the 5 control antigens in 68 assays were within 2 standard deviations of the mean. The analytical sensitivity of the ELISA was determined from an antigen titration of the 3 control strains and an S. Enteritidis strain. As shown in Figure 1, the detection limit of the ELISA was in the range of 5 × 105–5 × 106 CFU/ml for the 2 S. Pullorum strains and the S. Enteritidis strain.
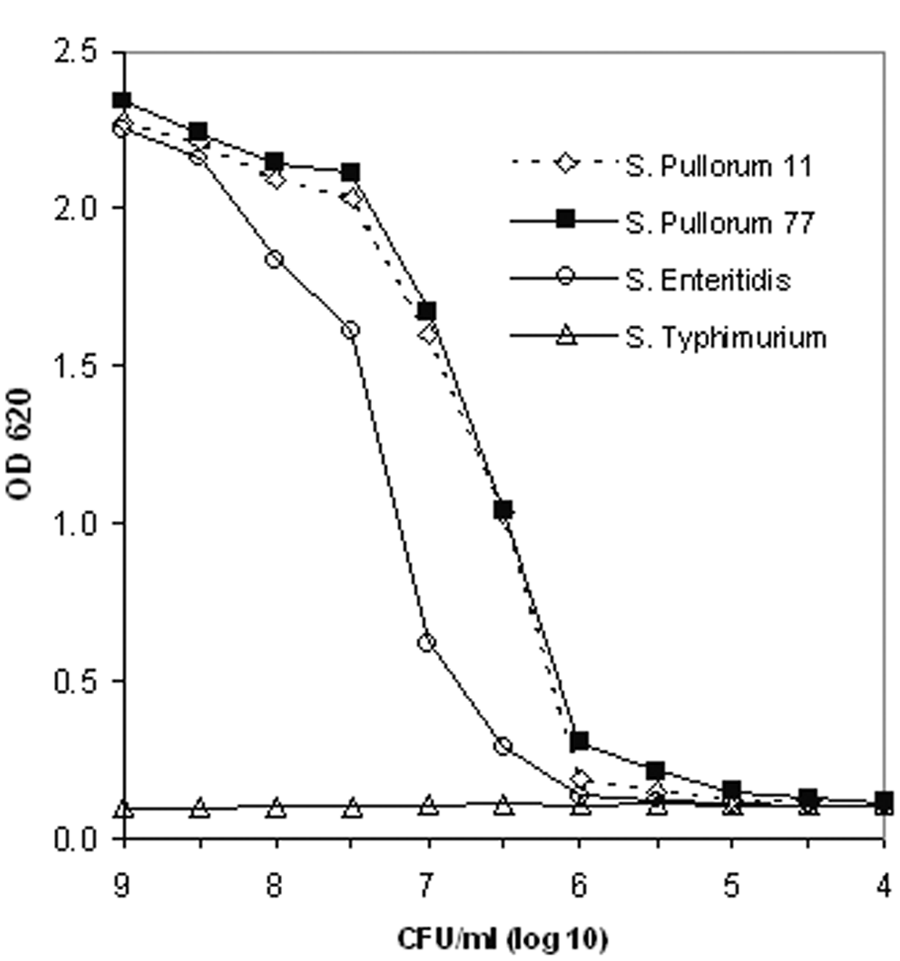
Detection limit of the Salmonella group D enzyme-linked immunosorbent assay for Salmonella enterica serovar Gallinarum biotype Pullorum strains 11 and 77, Salmonella enterica serovar Enteritidis Ottawa Laboratory Fallowfield strain 1863, and Salmonella enterica serovar Typhimurium American Type Culture Collection strain 14028. OD = optical density; CFU = colony forming units.
Isolation of S. Enteritidis and non–group D Salmonella by culture
A total of 1,057 poultry hatchery environmental samples that were received in 85 submissions were included in the current study. Salmonella Enteritidis was recovered by culture from 106 (10.0%) of the samples and was the only group D Salmonella isolated. However, 5 of the 106 samples were initially negative for S. Enteritidis by culture and positive by ELISA. Upon further testing, which involved identification of additional colonies on the original BGS and XLT4 agar plates from NSE, PSE, and DSE cultures, S. Enteritidis was isolated from these 5 samples. Salmonella Enteritidis was recovered from at least 1 sample from 46 (54%) of the 85 submissions. In a submission where there was only 1 S. Enteritidis–positive sample (the sample was initially ELISA positive but S. Enteritidis culture negative), the presence of S. Enteritidis was confirmed after further testing.
The number of samples that were culture positive for S. Enteritidis at the various stages of enrichment is shown in Table 2. Salmonella Enteritidis was isolated from 3 (2.8%), 8 (7.5%), and 26 (24.5%) of the samples only at the NSE, PSE, and DSE stages, respectively. Significantly more (P < 0.01) S. Enteritidis culture–positive samples were detected by the DSE stage as compared to the PSE stage.
Number of samples positive for Salmonella enterica serovar Enteritidis by culture and number of samples positive for group D Salmonella by enzyme-linked immunosorbent assay (ELISA) at various culture enrichment stages.*
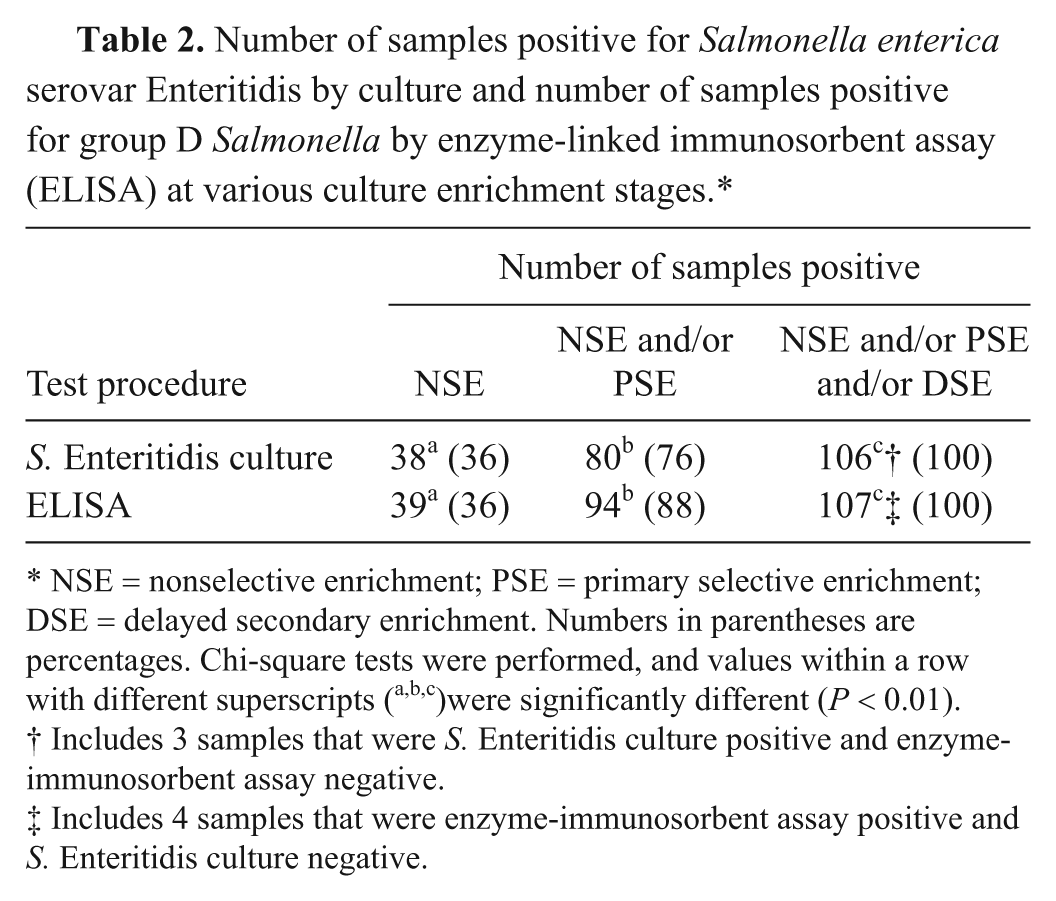
NSE = nonselective enrichment; PSE = primary selective enrichment; DSE = delayed secondary enrichment. Numbers in parentheses are percentages. Chi-square tests were performed, and values within a row with different superscripts (a,b,c)were significantly different (P < 0.01).
Includes 3 samples that were S. Enteritidis culture positive and enzyme-immunosorbent assay negative.
Includes 4 samples that were enzyme-immunosorbent assay positive and S. Enteritidis culture negative.
The type and number of enrichment broths from which S. Enteritidis was isolated at the 3 stages of enrichment are shown in Table 3. The recovery of S. Enteritidis was significantly higher (P < 0.01) from the RV broth at the DSE stage than from the earlier stages from either RV or TT broth. Salmonella Enteritidis was isolated only from RV broth with 35 samples and only from TT broth with 6 samples.
Type and number of enrichment broths positive for Salmonella enterica serovar Enteritidis by culture and positive for group D Salmonella by enzyme-linked immunosorbent assay (ELISA) at 3 culture enrichment stages.*
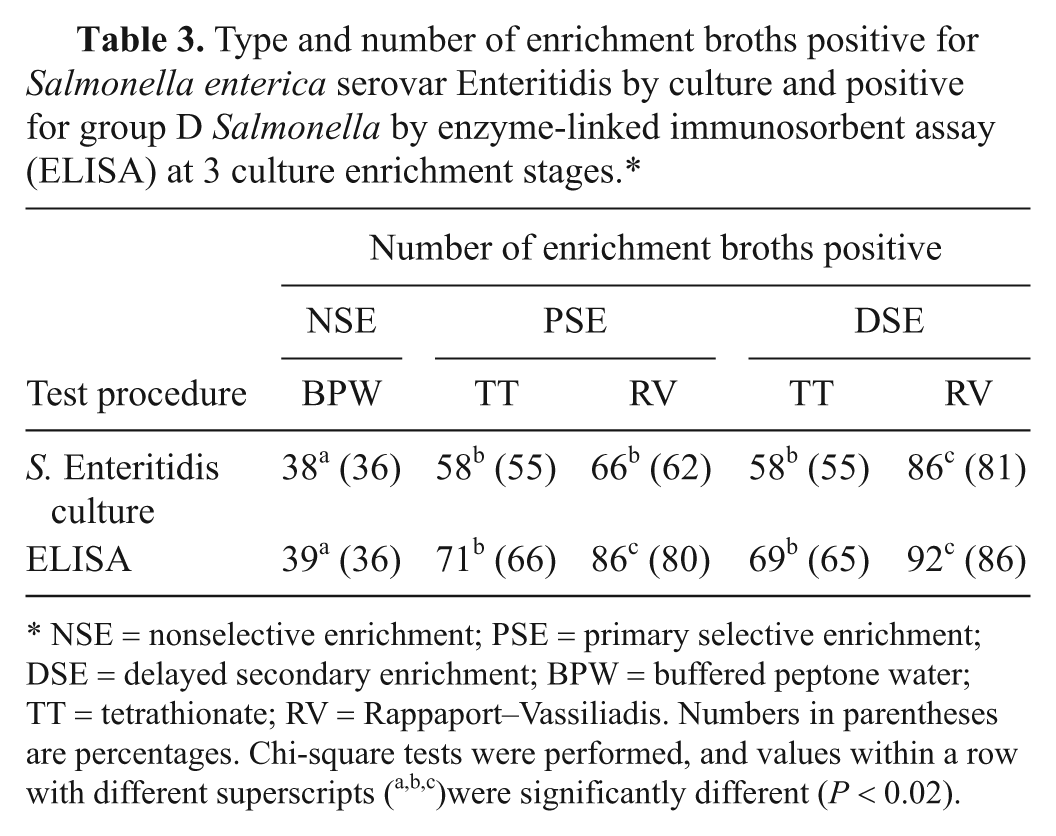
NSE = nonselective enrichment; PSE = primary selective enrichment; DSE = delayed secondary enrichment; BPW = buffered peptone water; TT = tetrathionate; RV = Rappaport–Vassiliadis. Numbers in parentheses are percentages. Chi-square tests were performed, and values within a row with different superscripts (a,b,c)were significantly different (P < 0.02).
The phage types (PT) of the S. Enteritidis isolates recovered from the 106 samples were PT8, PT9b, PT13, PT13a, PT23, PT51, and an atypical PT. One S. Enteritidis isolate was untypeable by the phage typing scheme.
Non–group D Salmonella were isolated from 132 (12.4%) of the 1,057 samples. Salmonella Enteritidis and non–group D Salmonella were both isolated from 27 (20.5%) of the 132 samples. A total of 14 non–group D Salmonella serovars, as confirmed by the OIE Salmonella Reference Centre, were identified: S. Agona, S. Heidelberg, S. Kiambu, S. Typhimurium, S. Typhimurium var. 5-, S. Tumodi (group B); S. Infantis, S. Mbandaka, S. Montevideo, S. Tennessee, S. Thompson (group C1); S. Hadar, S. Kentucky (group C2-C3); and S. Senftenberg (group E4).
ELISA reactivity for field samples
All 1,057 samples were tested by both culture and ELISA. Of these, 103 samples were ELISA positive (at least 1 culture enrichment broth positive) and S. Enteritidis culture positive. Three samples were ELISA negative and S. Enteritidis culture positive, and 4 samples were ELISA positive and S. Enteritidis culture negative. Nine hundred forty-seven samples were ELISA negative and S. Enteritidis culture negative. There was at least 1 ELISA-positive sample for each of the 46 submissions with at least 1 S. Enteritidis culture–positive sample, and there were no ELISA-positive samples for the 39 submissions with no S. Enteritidis culture–positive samples.
The diagnostic sensitivity and diagnostic specificity of the ELISA relative to culture were 97.2% (95% confidence interval [CI]: 91.3%, 99.3%) and 99.6% (95% CI: 98.8%, 99.9%), respectively, on a sample basis. The kappa statistic was 0.98 (95% CI: 0.94, 0.99). On a submission basis, the diagnostic sensitivity and specificity of the ELISA were both 100% in comparison to culture.
The number of samples positive by ELISA at the various stages of enrichment is shown in Table 2. When results were compared where S. Enteritidis was detected only in a single enrichment broth, the ELISA was positive for 1 (0.9%), 4 (3.7%), and 13 (12.3%) of the samples only at the NSE, PSE, and DSE stages, respectively. Of the ELISA-positive samples, significantly (P < 0.01) more were detected by the DSE stage of enrichment than by the PSE stage (94 vs. 107, respectively). Similarly, of the culture-positive samples, significantly (P < 0.01) more were detected by the DSE stage of enrichment than by the PSE stage (80 vs. 106, respectively).
The type and number of enrichment broths that were positive by ELISA are shown in Table 3. A significantly higher (P ≤ 0.01) number of RV broths were positive by ELISA at both the PSE and DSE stages as compared to TT broths. The ELISA was positive with at least 1 RV broth and at least 1 TT broth for 99 (92.5%) and 73 (68.2%) of the samples, respectively.
Discussion
In the present study, the use of DSE significantly increased the isolation of S. Enteritidis from poultry hatchery environmental samples. This result is consistent with the results from previous studies, which showed that DSE improves the recovery of Salmonella from poultry and poultry environments,22,23,25 swine and swine environments,5,16,18 and cattle. 17 The basis for the enhanced isolation of Salmonella with DSE is not clear. A previous study 25 suggested that the extended incubation time with DSE may allow injured or low numbers of Salmonella to multiply to detectable levels and reduce the level of competing organisms in the culture and thus provide more favorable growth conditions for Salmonella. However, in another study, 22 using a selective medium containing novobiocin to inhibit competing flora, DSE was more effective for the recovery of Salmonella than PSE with samples that were not exposed to disinfectants or other adverse conditions prior to culture. Many of the samples included in the present study may have contained injured S. Enteritidis cells as they were collected after cleaning of the hatchery facilities and application of a disinfectant. Results of another study 11 suggested that resistant Salmonella variants are selected during prolonged incubation in selective enrichment media and that these variants are able to initiate rapid growth when subcultured into fresh media.
Salmonella Enteritidis was isolated more frequently using RV broth than TT broth in the present study. The RV broth has a lower pH (5.2 ± 0.2 vs. 7.6 ± 0.2 for TT), contains a different inhibitory dye (malachite green vs. brilliant green), has a higher salt concentration, and has relatively fewer nutrients as compared to TT broth.8,24 In addition, the inoculum size is 10 times lower (0.1 ml vs. 1.0 ml) for RV than for TT. Any of these factors, alone or in combination, may have contributed to the higher recovery of S. Enteritidis from RV broth. While a similar observation has been previously reported, 12 the scientific basis for this enhanced recovery is not known and would require further investigation. Nevertheless, in the present study, S. Enteritidis was isolated from TT broth only with 6 samples. Thus, to enhance the recovery of Salmonella, the inclusion of more than one selective enrichment broth should be considered regardless of the culture method used. This is particularly relevant in situations where greater isolation sensitivity is required to minimize the risk of reintroducing S. Enteritidis into S. Enteritidis–free poultry such as in restocking premises that have been cleaned, disinfected, tested, and reported free of this zoonotic pathogen.
Previously, several ELISAs were developed for the detection of group D Salmonella, including S. Enteritidis.1,3,13 With one exception in which a specific component was not identified, the target antigen with these assays was LPS, and mAbs or polyclonal antibodies were used for detection. These assays were evaluated with pure cultures of group D Salmonella and other bacteria, various foods, and other samples artificially inoculated with group D Salmonella and other bacteria, or with field samples.
The assay developed in the present study was based on detection of group D Salmonella LPS O-antigen epitopes. Two mAbs, produced against S. Pullorum LPS, were used in combination to detect the 2 different LPS O-antigen forms recognized in both S. Pullorum 4 and S. Enteritidis. 20 The assay was compared to culture, with more than 1,000 field samples collected from the environment of poultry hatcheries. Salmonella Enteritidis was the only group D Salmonella recovered, and excellent agreement (kappa = 0.98) was observed between the ELISA and culture results. Five samples were S. Enteritidis culture positive only after retesting following a positive ELISA result. Initially, non–group D Salmonella were isolated from all 5 of these samples and may have been present in larger numbers than S. Enteritidis. Four samples were ELISA positive and culture negative, and these may have contained nonviable S. Enteritidis or other group D Salmonella, an organism other than group D Salmonella with an antigen that cross-reacted with one or both of the mAbs, or S. Enteritidis or other group D Salmonella not detected by culture. Three samples were ELISA negative and S. Enteritidis culture positive. Salmonella Enteritidis was isolated from only 1 of the 5 enrichment broths from each of these 3 samples and may have been present at a low concentration not detectable by ELISA.
The use of the ELISA as a presumptive screening test would allow a faster turnaround time for reporting results than that for culture. For culture-negative samples and with the culture procedure used in the present study, the ELISA results on the DSE broths could be available at least 2 days before the culture results, if the ELISA testing was done on the same day as plating to selective agar. If suspicious colonies were found, a minimum of at least 2 additional days would be required to obtain a negative culture result. For culture-positive samples, a presumptive positive result would be available at least 2–9 days earlier by ELISA than by culture, considering that significantly more of the positive samples were detected at the PSE stage by ELISA than by the culture. Culture testing would be required to confirm all ELISA-positive results.
In summary, a mAb-based ELISA was developed for the detection of group D Salmonella and validated in comparison to standard culture procedures using field samples. The validation results indicate that the antigen-capture ELISA could be used as a reliable screening test for the detection of S. Enteritidis in poultry hatchery environmental samples. In addition, the use of DSE and RV media both significantly enhanced recovery and detection rates of S. Enteritidis from these samples.
Footnotes
Acknowledgements
The authors gratefully acknowledge the expert contributions of S. Duff, E. Falardeau, K. Habib, C. Elmgren, K. Arnold, and J. Widdison and thank the Canadian Food Inspection Agency operations staff for collection of the field samples.
a.
SOLAR-CULT® sampling sponges, Qualicum Scientific, Ottawa, Ontario, Canada.
b.
BD Difco™, BD, Franklin Lakes, NJ.
c.
Whirl-Pak® bag, Nasco International Inc., Fort Atkinson, WI.
d.
Stomacher® 40, Seward Laboratory Systems Inc., Port Saint Lucie, FL.
e.
Inoculating loops, VWR International, Mississauga, Ontario, Canada.
f.
Oxoid Co., Nepean, Ontario, Canada.
g.
API® 20E, BioMérieux, St. Laurent, Quebec, Canada.
h.
Vitek® GNI+ cards and Vitek® Jr., BioMérieux, St. Laurent, Quebec, Canada.
i.
Fisher Scientific Ltd, Nepean, Ontario, Canada.
j.
J. T. Baker, Phillipsburg, NJ.
k.
Nunc Maxisorp™, Fisher Scientific Ltd, Nepean, Ontario, Canada.
l.
Labsystems Wellwash 384, Fisher Scientific Ltd, Nepean, Ontario, Canada.
m.
EMD Chemicals, Gibbstown, NJ.
n.
Sigma-Aldrich, St. Louis, MO.
o.
Jackson ImmunoResearch Laboratories, West Grove, PA.
p.
Kirkegaard & Perry Laboratories, Gaithersburg, MD.
q.
Multiskan Ex, Fisher Scientific Ltd, Nepean, Ontario, Canada.
r.
Minitab Statistical Software, Minitab Inc., State College, PA.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
This research was supported by the Canadian Food Inspection Agency.
