Abstract
Various staining methods are available for reptilian species blood cell quantification. However, these methods have shown inaccurate differentiation limitations. The current study evaluates staining effects and blood cell counting results using an alternative method, counting blood cells diluted with 0.45% sodium chloride solution and stained with crystal violet. Blood samples from 8 red-eared slider turtles (Trachemys scripta) were collected. Red and white blood cell counts were performed using different methods: the unstained method, the Unopette method, Liu stain, and crystal violet method using blood cells diluted in various sodium chloride solution osmolarities. The staining properties and blood cell count results were compared. The crystal violet method using blood cells diluted in 0.45% sodium chloride solution delivered the best staining and counting results among all of the tested methods, with the lowest average coefficient of variance. The proposed method can easily be performed, serving as a feasible method for blood cell counting in chelonians.
Introduction
Hematology is a part of the laboratory diagnostic evaluation in patients of all species, including fish, 5 reptiles,1,3,7 and birds.2,4 Various staining solutions and methods have been adapted from mammalian manual counting methods to perform blood cell quantification in reptilian species, including the May-Grünwald/Giemsa stain, 6 Natt–Herrick method, 4 and the Unopette test. 2 These methods have shown limitations, including difficulty in differentiating thrombocytes and small lymphocytes in the Natt–Herrick method6,7 or difficulty in distinguishing white blood cells (WBCs) from debris, immature red blood cells (RBCs), and blood parasites. 2 Inconsistency in blood cell counts obtained between the 2 chambers of the Neubauer hemocytometer have been observed using the Unopette test, assumedly caused by diluting pipette errors. 2 The Natt–Herrick method reveals higher variability in blood cell counts when compared to the Unopette test.2,7
Obtaining accurate differentiation between lymphocytes and thrombocytes and precise cell counts are essential to whether a method is practicable. It is therefore necessary to develop a method that is easy to perform and capable of meeting the requirements.
The objective of the current study is to develop a simple staining method and compare its staining effects and blood count results against other methods. The proposed method, crystal violet for cells diluted in 0.45% sodium chloride solution, may provide an easy and suitable application for clinical use in chelonian blood cell counting.
Materials and methods
Animals and samples
Eight healthy adult red-eared slider turtles (Trachemys scripta) certified by laboratory and physical examination were used in the present study. The turtles were kept at the Taipei Zoo (Taipei, Taiwan) and were placed in an outdoor enclosure with a pool to swim. Shade was provided to avoid direct sunlight. The turtles were fed every day at 2
Blood cell counting
Red and white blood cell counts were performed within 1 hr after collection using a hemocytometer a with different staining methods, including the unstained method, the Unopette b method, Liu stain c method, and the crystal violet d method. Normal saline (0.9% sodium chloride) was used to dilute the blood cells in the unstained method. Blood cells stained with Liu stain or crystal violet were diluted using various osmolarity solutions, including pure distilled water, or 0.15%, 0.45%, 0.75%, and 0.9% sodium chloride solutions. The cells in distilled water or 0.15% sodium chloride solution were swollen in appearance, and the damaged membrane made cells indistinct. Cells in 0.75% or 0.9% sodium chloride solution brought faint staining results, owing to inadequate penetration of the stains. Cells in 0.45% sodium chloride solution still maintained membrane integrity while counting and were found to have the best staining property with the proper amount of stains entering the cells. Liu stain and crystal violet for blood cells diluted in 0.45% sodium chloride solution were therefore employed in all of the subsequent experiments. After the cells were diluted and mixed well in each stain solution, the hemocytometer chambers were charged and stood 10 min before counting.
All cell preparations were performed by a single technician. Each sample from each method was counted 4 times and performed by 4 different people who were blind to the staining methods.
Four large corner squares (1 mm2) were counted in the WBC count. The 4 small corner squares plus the central square (1/25 mm2) were counted in the RBC count. Blood cells were diluted to 1:100 ratio in the Unopette method based on the instruction manual. In the other methods, blood was diluted to 1:75 ratio using sodium chloride solution, which provided adequate RBC and WBC dispersion and allowed the technicians to perform RBC and WBC counts at the same time. Because the dilution ratio, the square measure (1 mm2 for the large square and 1/25 mm2 for the small square), and the depth of the chamber (0.1 mm) of the hemocytometer were known, the total WBC and RBC count for each mm3 (equivalent to 1 μl) of blood for each method could be calculated using the following formulas.
Counting formulas
1. Unstained method
Ten microliters of blood was gently mixed with 740 μl of normal saline (0.9% sodium chloride solution).
(1a) Total WBC = total hemocytometer count ÷ 4 (4 large squares) ÷ 0.1 (depth of the chamber) × 75 (dilution rate).
(1b) Total RBC = total hemocytometer count × 5 (reaching to 1-mm2 area) ÷ 0.1 (depth of the chamber) × 75 (dilution rate).
2. Unopette method
(2a) Total WBC = total hemocytometer count ÷ 4 (4 large squares) ÷ 0.1 (depth of the chamber) × 100 (dilution rate).
(2b) Total RBC = total hemocytometer count × 5 (reaching to 1-mm2 area) ÷ 0.1 (depth of the chamber) × 100 (dilution rate).
3. Liu stain method
Prepare 1:75 diluted stained blood cell in 0.45% sodium chloride solution: 0.4 ml of Liu stain A is mixed with 9.8 ml of normal saline and 9.8 ml of distilled water to produce solution A; 0.4 ml of Liu stain B is mixed with 9.8 ml of normal saline and 9.8 ml of distilled water to produce solution B. Ten microliters of blood is gently mixed with 370 μl of solution A for 5 min, then mixed with 370 μl of solution B for another 5 min.
(3a) Total WBC = total hemocytometer count ÷ 4 (4 large squares) ÷ 0.1 (depth of the chamber) × 75 (dilution rate).
(3b) Total RBC = total hemocytometer count × 5 (reaching to 1-mm2 area) ÷ 0.1 (depth of the chamber) × 75 (dilution rate).
4. Crystal violet method
Prepare 1:75 diluted stained blood cells in a 0.45% sodium chloride solution: 0.1 ml of R1 oxalated crystal violet sodium is mixed with 9.95 ml of normal saline and 9.95 ml of distilled water to produce solution C. Ten microliters of blood is then gently mixed with 740 μl of solution C for 5 min.
(4a) Total WBC = total hemocytometer count ÷ 4 (4 large squares) ÷ 0.1 (depth of the chamber) × 75 (dilution rate).
(4b) Total RBC = total hemocytometer count × 5 (reaching to 1-mm2 area) ÷ 0.1 (depth of the chamber) × 75 (dilution rate).
Data analysis
Each sample from each method was counted 4 times by 4 different people. Mean WBC and RBC count values were calculated and shown as mean ± standard deviation (SD). The variation rate for the 4 counts of each sample from each method is shown as the coefficient of variation (CV). Eight CV values were obtained from 8 samples in a given method. These 8 CV values were further calculated for the average (average CV, shown as mean ± SD) in regard to a single method (Tables 1, 2).
White blood cell (WBC) count results obtained using the following methods: unstained, Unopette, Liu, and crystal violet in red-eared slider turtles (Trachemys scripta).*
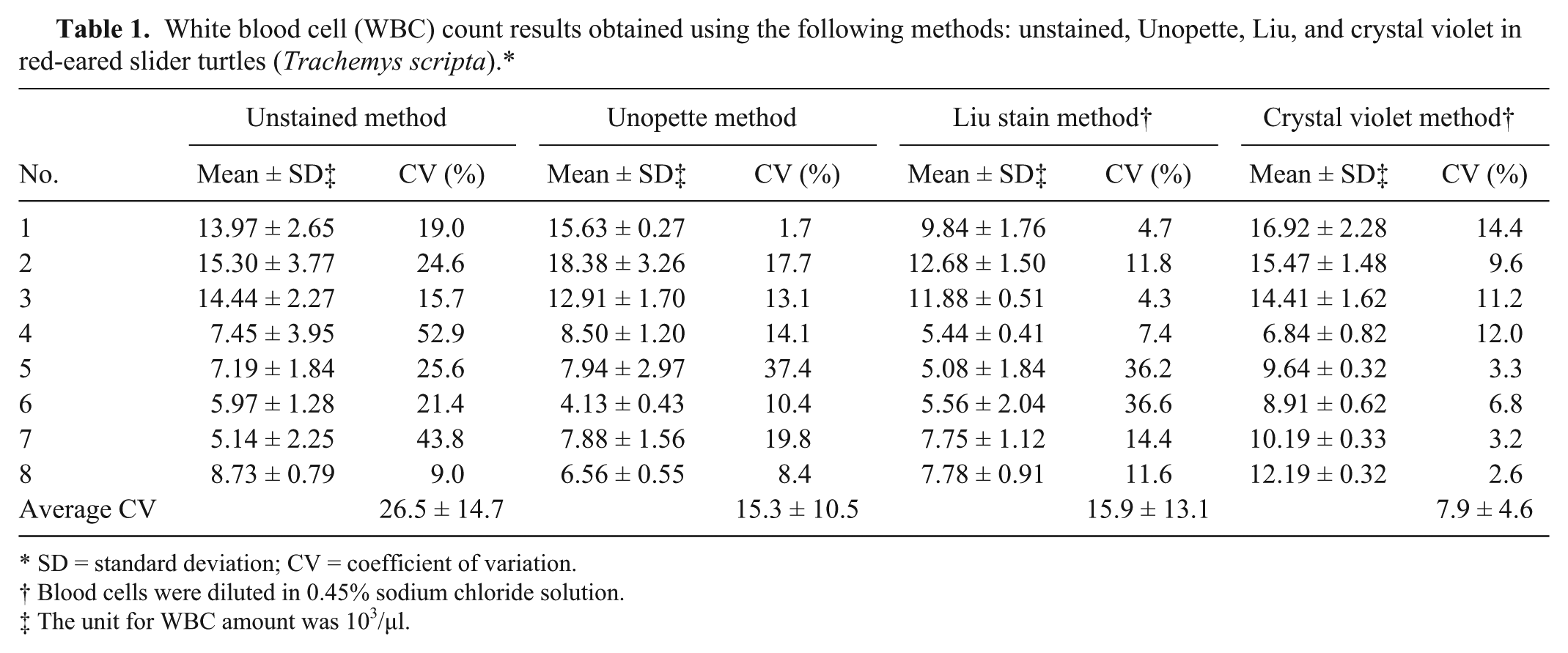
SD = standard deviation; CV = coefficient of variation.
Blood cells were diluted in 0.45% sodium chloride solution.
The unit for WBC amount was 103/μl.
Red blood cell (RBC) count results obtained using the following methods: unstained, Unopette, Liu, and crystal violet in red-eared slider turtles (Trachemys scripta).*
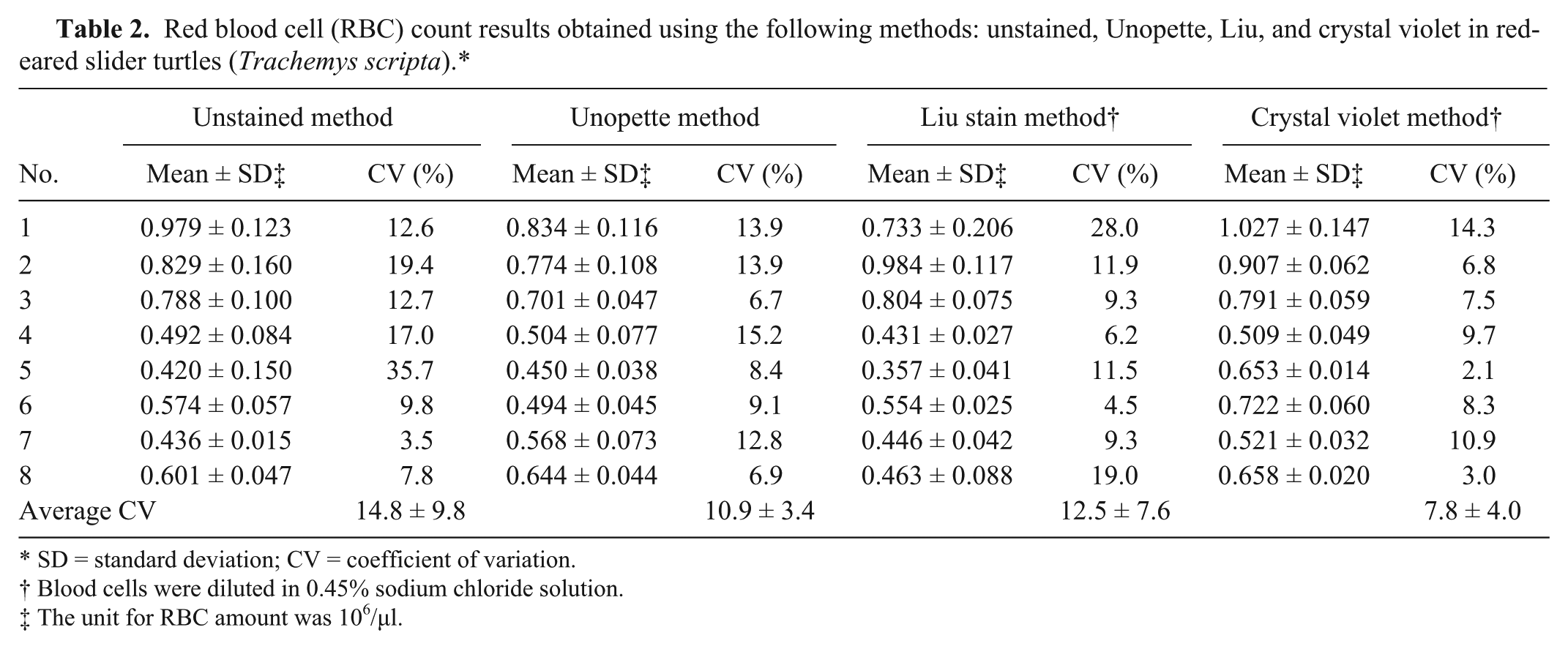
SD = standard deviation; CV = coefficient of variation.
Blood cells were diluted in 0.45% sodium chloride solution.
The unit for RBC amount was 106/μl.
Results
The unstained method exhibited colorless cells. The RBCs had very pale nuclei that could not be clearly identified. Granulocytes could be identified, but the granules were not stained well and presented a vague appearance. The lymphocyte nuclei were also unidentifiable. The thrombocytes were pale and hard to identify from the background (Fig. 1). The unstained blood cell appearance led to high average CV for both WBC and RBC counts.
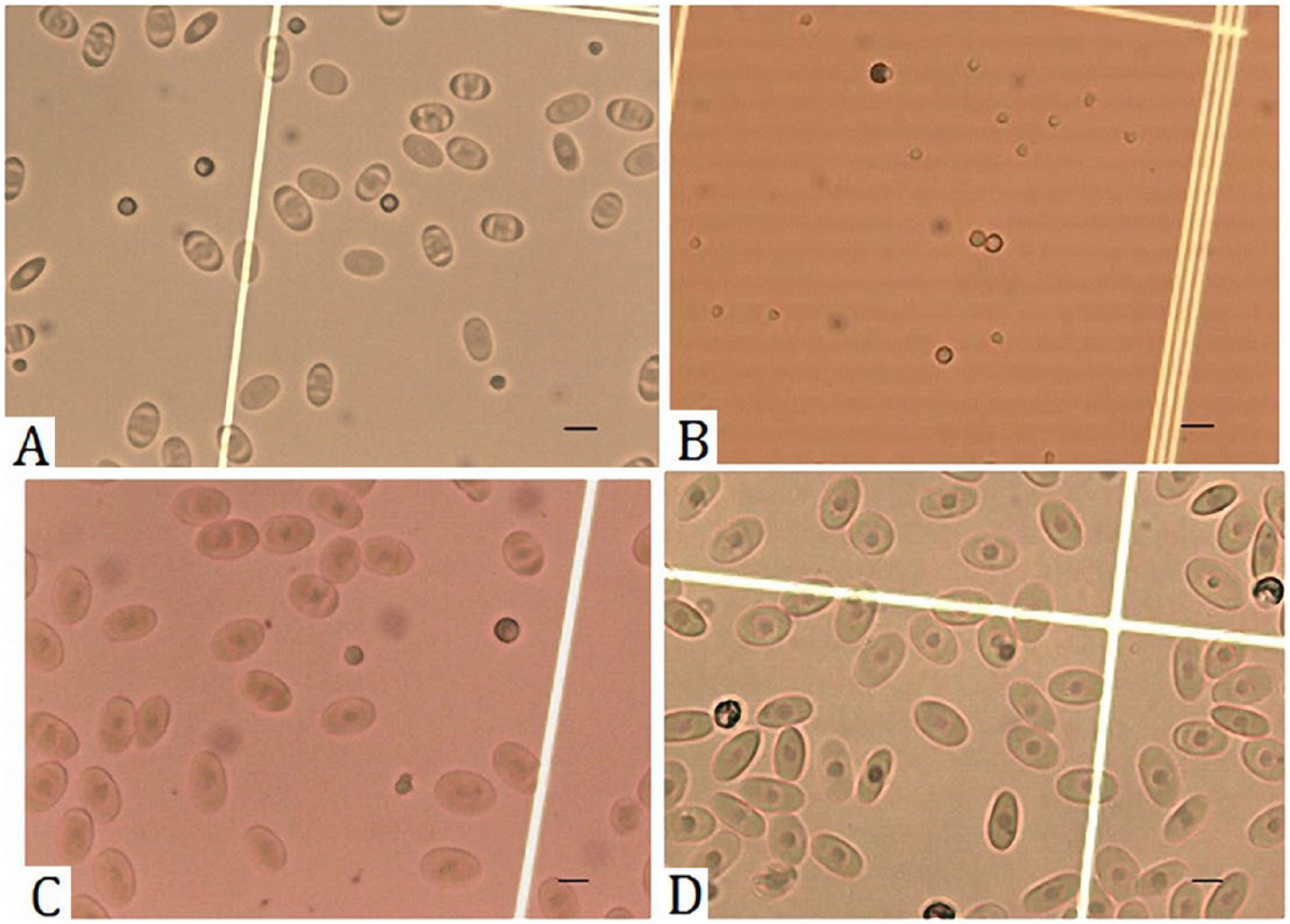
Blood cell staining results.
The Unopette method lysed the erythrocyte membrane, which led the RBCs to have fairly clear nuclei but very pale cell membranes. The RBC nuclei were easily confused with the lymphocytes and thrombocytes. The granulocytes were clearly stained (Fig. 1). The Unopette method staining properties may cause blood cell misclassification, leading to incorrect blood counts.
The Liu stain method (cells diluted in 0.45% sodium chloride solution) displayed fairly clear staining results in which both RBC membranes and nuclei could be identified. The granulocytes were clearly stained, as well. However, the lymphocytes and thrombocytes appeared similar and were difficult to differentiate (Fig. 1).
The crystal violet method (cells diluted in 0.45% sodium chloride solution) had the best staining results. The RBCs had dark violet nuclei, clearly identified cell membranes, with the cytoplasm stained light violet. The granulocytes were clearly stained (Figs. 1, 2). Lymphocyte nuclei were stained dark violet, and the thrombocytes had a similar appearance. However, the thrombocyte nuclei and cytoplasm had greater transparency than the lymphocytes (Fig. 3). These results verified that the crystal violet method had the best staining results among the 4 methods.
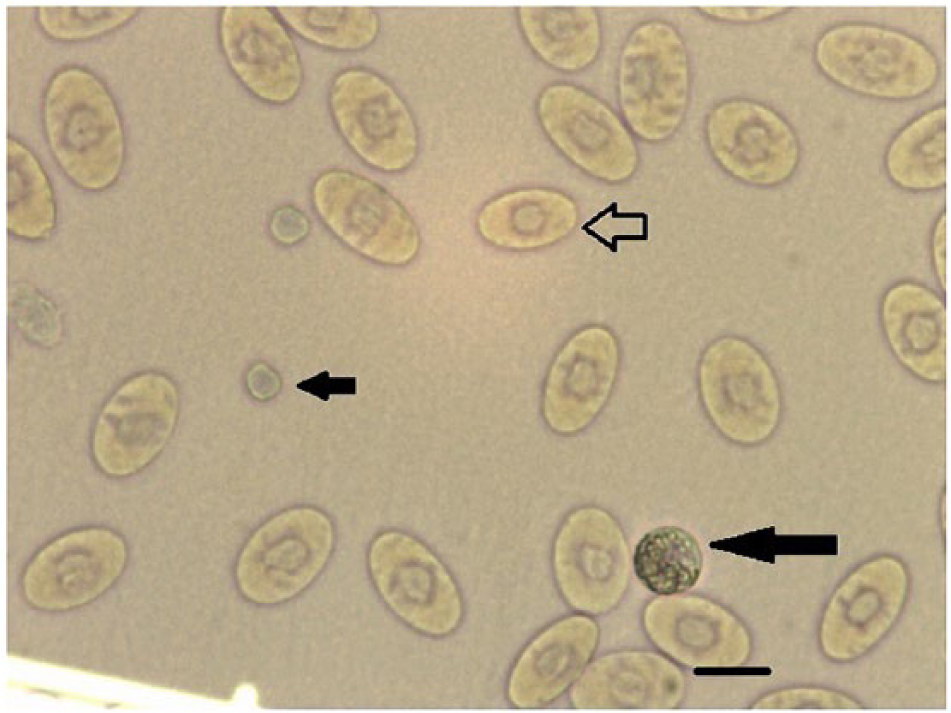
Crystal violet staining for blood cells diluted in 0.45% sodium chloride solution. Erythrocytes (open arrow), granulocytes (long solid arrow), and thrombocytes (short solid arrow) are indicated. Scale bars = 10 μm.
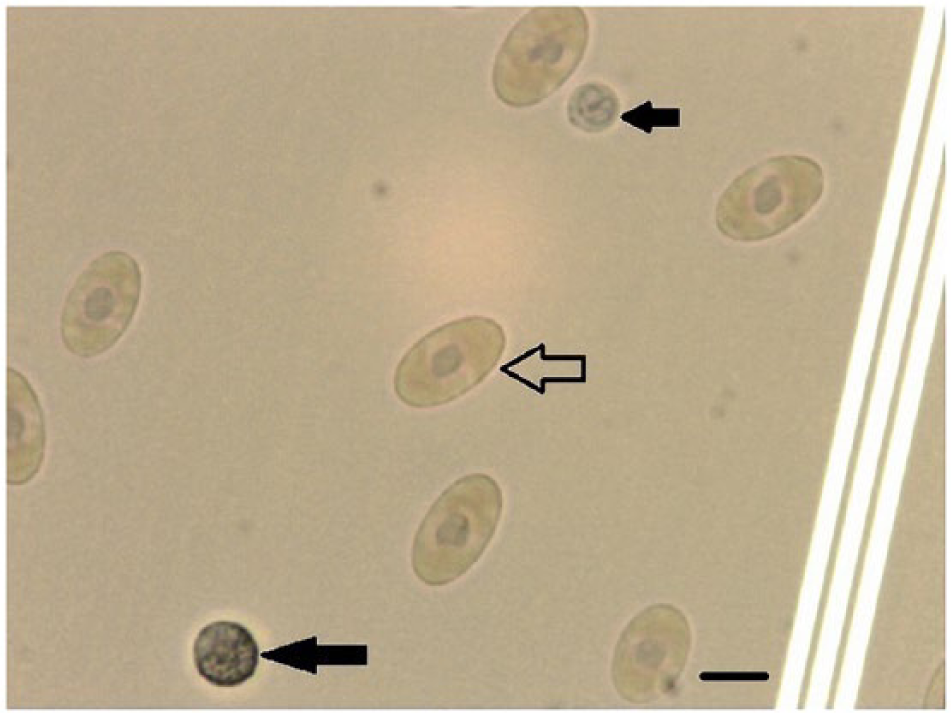
Crystal violet staining for blood cells diluted in 0.45% sodium chloride solution. Erythrocytes (open arrow), lymphocytes (long solid arrow), and thrombocytes (short solid arrow) are indicated. Scale bars = 10 μm.
The average CV in WBC count for the unstained, Unopette, Liu stain, and crystal violet methods was 26.5 ± 14.7, 15.3 ± 10.5, 15.9 ± 13.1, and 7.9 ± 4.6, respectively; showing that the crystal violet method had the lowest average CV (both mean value and SD; Table 1). The average CV in RBC count for the unstained, Unopette, Liu stain, and crystal violet methods was 14.8 ± 9.8, 10.9 ± 3.4, 12.5 ± 7.6, and 7.8 ± 4.0, respectively (Table 2). The crystal violet method also showed low average CV (the lowest mean value and the second low SD). In both WBC and RBC counts, the unstained method exhibited the highest average CV (both mean value and SD), indicating that colorless blood cells easily produced high counting deviations.
Discussion
Hematology in reptiles and birds is challenging and time consuming because the nuclei of erythrocytes and thrombocytes make cell counting impossible using automated instruments. A number of manual counting methods have been developed to achieve accurate results. The current study found that the crystal violet for the blood cells diluted in 0.45% sodium chloride solution had considerably low CVs regarding both RBC and WBC counts. Erythrocytes could be clearly identified, and lymphocytes, thrombocytes, and granulocytes could easily be distinguished. The staining procedures were simple and quick to perform. Because crystal violet is an inexpensive and ubiquitous reagent that can be obtained easily in most laboratories, it may serve as an easy and convenient staining method for blood cell counting in chelonians.
This developed staining method allowed for readily counting the total amounts of leukocytes, erythrocytes, and thrombocytes simultaneously in a hemocytometer chamber. However, discrimination among heterophils, basophils, eosinophils, monocytes, and lymphocytes should be further achieved through blood smear film.
Four blood cell counts were performed for each turtle using 4 different technicians who were blind to the staining methods. This approach reduced the influence of personal prejudice, making the results more reliable.
The blood cell staining results obtained using various sodium chloride–diluted osmolarities were examined in the present study. The stain under normal saline, 0.9% sodium chloride solution could not adequately penetrate the cell and clearly stain the nuclei. The solution with extremely low osmolarity or the pure distilled water resulted in cell swelling or cell membrane vanishing, making differentiation difficult. Liu stain or crystal violet for blood cells diluted in 0.45% sodium chloride solution left the cell membranes integral and obtained the best staining results. Therefore, this sodium chloride solution osmolarity was employed in the following experiments while using Liu stain or crystal violet stain.
The unstained method did not stain the cells, resulting in confusion when counting erythrocytes, lymphocytes, and thrombocytes, thus having the highest average CV in both counts.
The Unopette method used in the study was the Unopette test 5855, which was recommended for chelonians by the Becton-Dickinson agent in Taiwan. The Unopette test 5877, a stain that was more commonly used for birds and reptiles in other studies,2,3 is not available in Taiwan due to discontinued production. The Unopette Test 5855, a diagnostic reagent for human leukocytes and plates, might be acceptable as a replacement for the Unopette test 5877, a reagent for heterophils and eosinophils but not for all leukocytes, 2 in chelonian blood cell counting. The Unopette test 5855 stained granulocytes clearly, but the lymphocytes were easily confused with thrombocytes and erythrocyte nuclei. Therefore, a separate blood smear and differential count for the above cells were needed. Trying to achieve a direct WBC count using Unopette may cause higher variability.
The Liu stain method had similar staining results compared with the crystal violet method when the blood cells were diluted in 0.45% sodium chloride solution, but the cells were stained slightly paler. This may be improved by elevating the stain concentration and extending the staining time. Further experiments are needed for comparison.
Differentiating lymphocytes and thrombocytes in chelonian and avian species is challenging.6,7 By using the crystal violet method (cells diluted in 0.45% sodium chloride solution), there was better discrimination between these 2 types of cells. Further studies employing different osmolarities and staining time may be necessary to see if a more exacting staining quality could be obtained.
Natt–Herrick solution is commonly used to obtain reptile and avian blood cell counts. According to a previous study, WBC counts performed in 2,000 erythrocytes of blood smears with Natt–Herrick solution were not consistent with the results obtained from 1,000 erythrocytes of blood smears. 7 Natt–Herrick solution was not adequate for differentiation between lymphocytes and thrombocytes in the tartaruga (or Amazon turtle, Podocnemis expansa). 7 Moreover, the CV value for total leukocyte counts in birds with the Natt–Herrick method was also higher than those with the Unopette method. 2 The Natt–Herrick method was not included in the current study. Further studies should include the Natt–Herrick method in comparison with the crystal violet method. It would be more appropriate if a variety of species other than the red-eared slider turtles could be tested in future studies.
Footnotes
Acknowledgements
The authors thank the staff in the Medical Center of the Taipei Zoo.
a.
Neubauer hemocytometer, Canemco Inc., Gore, Quebec, Canada.
b.
Unopette test 5855, BD, Franklin Lakes, NJ.
c.
ASK Tonyar Biotech, Tao Yuan, Taiwan.
d.
bioMérieux, Marcy l’Etoile, France.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
