Abstract
Bluetongue is a potentially fatal arboviral disease of domestic and wild ruminants that is characterized by widespread edema and tissue necrosis.
Since the initial recognition of BTV in North America around 1950, ongoing surveillance has identified only BTV serotypes 10, 11, 13, and 17 to be present throughout central and western regions of the United States.5,11,13 In contrast, numerous other BTV serotypes (including serotypes 1–6, 9–14, 17, 19, 22, and 24) have been identified in the southeastern United States and Caribbean islands.5,9,18 Importantly, BTV serotype 2 (BTV-2) was recently identified in a dairy heifer from the northern Sacramento Valley of California,12,13 the first detection of this virus serotype in the western part of the United States since the initial isolation from cattle in Florida in 1982.3,6,7 The genome of this BTV-2 isolate from California (BTV-2 USA2010/CA) was subsequently sequenced, and initial BLAST analysis (http://www.ncbi.nlm.nih.gov/blast/Blast.cgi) suggested that the virus may be a reassortant that is most related to the strains BTV-2 USA1999/FL and BTV-6 USA2006 isolated from Florida and the southeastern United States, respectively. 12 To further characterize the BTV-2 USA2010/CA isolate, the current study compiled a database of complete BTV genome sequences available from GenBank as well as additional previously unpublished sequences of 3 BTV isolates from the United States and Panama (Table 1). Alignments and phylogenetic analysis were then performed to better understand the relationships of these BTV strains and the putative origin of this particular BTV-2 strain isolated from California.
Newly sequenced
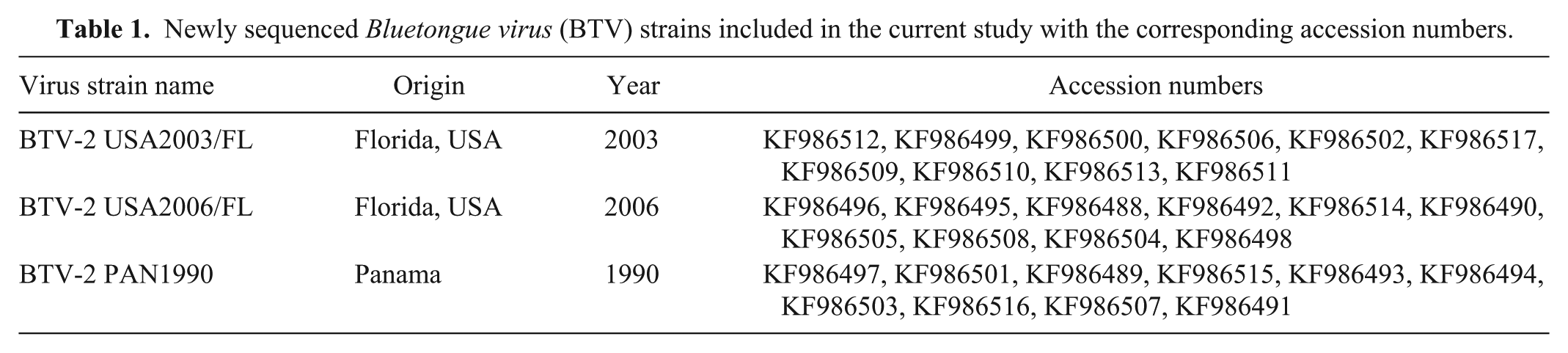
Virus strains were propagated in baby hamster kidney (CCL 10)
a
or bovine pulmonary artery endothelial (CCL 209)
a
cells, and viral double-stranded RNA (dsRNA) purified from extracted total RNA by a previously described method.20 A whole genome sequence-independent amplification procedure was performed as previously described.
15
Briefly, restriction sites were removed from the anchor primer PC3-T7restrict (5′p-GTTCAGCCTGACCACGTTAATACGACTCACTATATTTTTATAGTGAGTCGTATTA-OH3′), to facilitate sequencing and then purified by high-performance liquid chromatography. The PC3-T7restrict primer (600 ng) was ligated to dsRNA (500 ng) in the ligation reaction mix as described previously
15
for a final volume of 90 μl. Ligation was performed at 37°C for 16 hr, and then purified using a commercial kit
b
following the protocol recommended by the manufacturer. The cleaned and ligated product (8 μl) was supplemented with 25 pmol PC2restrict primer (5′p-ACGTGGTCAGGCTGAAC-3′) and 1 μl of 300 mM methyl mercury hydroxide and allowed to denature for 10 min. The mixture was placed on ice, and 1.5 μl of 1 M β-mercaptoethanol was added and allowed to neutralize for 1 min. The copy DNA (cDNA) was then generated using cloned
Library preparation and sequencing was performed using a commercial platform e with the corresponding commercially available kits, f following the protocols recommended by the manufacturer. Briefly, approximately 1 mg of viral cDNA was fragmented, barcoded, and quantitated. f Template generation, enrichment, and sequencing of the template were performed on the appropriate instruments. e
Standard flowgram format files were imported to commercial software g for contig creation. Partial contigs were assembled and blasted to determine reference sequences that were then used for reference assemblies. Multiple sequence alignments of the nucleotide sequences were performed using the multiple alignment fast Fourier transform method followed by in silico translation of the open reading frames and alignment of the predicted proteins for each genomic segment. Distance relationships were calculated from the nucleotide and protein alignments. Although a comprehensive analysis was performed with all full-length gene sequences available from GenBank, for simplicity, only the most relevant, closely related strains and U.S. prototype BTV strains are shown (Table 2).
Percent pairwise identity relative to the
CA = California; FL = Florida; USA = United States of America; RSA = Republic of South Africa; PAN = Panama; MTQ = Martinique; NA = data not available. Boldfaced values indicate highest genetic identity with BTV-2 USA2010/CA. GenBank accession numbers for the represented BTV strains are available in supplementary Table 1.
Overall, the California isolate shared the highest identity with BTV-2 USA1982/OnaB (Table 2). Seven of the 10 BTV genome segments (M4-S10) share 98% or greater sequence identity with the OnaB strain, and 5 of these 7 share 100% identity with the predicted proteins (Table 2). Segments L2 and L3 were more closely related to BTV-2 USA2006/FL and BTV-2 USA2003/FL, respectively (Table 2). The L1 gene and encoded VP1 protein was most conserved with BTV-6 USA2006 isolated from the southeastern United States (Table 2). Thus, results from the analysis indicate that the BTV-2 USA2010/CA isolate is most closely related to BTV-2 strains from the southeastern United States and Florida region. The BTV-2 PAN1990 isolate shared close identity to the BTV-2 strains analyzed, yet branched distinctly from both the Republic of South Africa (RSA) prototype and U.S. strains (trees not shown).
The first identification of BTV-2 in the United States was in 1982.3,6,7 At that time, two BTV-2 isolates with distinct RNA electrophoretic mobilities were identified from the town of Ona, Florida and designated as OnaA and OnaB. 14 The OnaA strain was the first isolated, and was identical to the South African BTV-2 prototype. 14 However, after the initial isolation in September and October of 1982, there were no further isolations of the OnaA strain. The Ona B strain was isolated at the same site in October and November of 1982. 14 Electrophoretic mobility changes were identified in multiple genome segments, and subsequent sequencing confirmed mutations in genes encoding VP7, NS1, and NS2. 14 Additional isolations of OnaB were made in Florida and later in Alabama through 1985.3,4 However, BTV-2 was not identified again in the United States until 1999 when it was isolated from sheep in Florida. 14 When compared to the original 1982 isolates, the 1999 BTV-2 isolate most resembled OnaB, suggesting that OnaB had persisted in the region. 14 The OnaB strain was found to be closely related genetically to 2 other BTV strains of other serotypes circulating in Florida at the same time, specifically BTV-13 (AY855268) and BTV-17 (AY855269), indicating that this virus had reassorted with other BTV serotypes in the area. 14 Collectively, the findings from these previous studies suggest that BTV-2 was perhaps first introduced to the United States in 1982 and circulated for a short period of time as the original OnaA strain before quickly diverging genetically to the OnaB strain, and eventually reassorting with other BTV strains circulating in the region.
The California BTV-2 USA2010/CA isolate was identified in the blood of a heifer during an epidemiological surveillance study of 4 dairy farms in northern Sacramento Valley that took place in 2010.12,13 This finding was novel because it represents the documented instance of BTV-2 being detected in a region of the United States beyond the southeast. 12 There is, to date, no approved vaccine for BTV-2 in the United States. Although some of the animals from the surveillance study suffered from ulcerative stomatitis and coronitis, other BTV serotypes were also present on the farm concurrently, making any association of disease with this particular strain of BTV-2 speculative and its clinical significance uncertain.12,13 Unfortunately, although the heifer was apparently born on the farm in California, there was no detailed history available regarding movement of other livestock onto the premises. Without comprehensive ongoing surveillance, and only limited data available, it remains unclear how this novel strain of BTV-2 was introduced to California. The current phylogenetic analysis showed that the BTV-2 USA2010/CA isolate, as well as BTV strains from the southeastern United States, grouped distinctly from the BTV-2 RSA prototype and the original U.S. OnaA strain. This suggests that the BTV-2 USA2010/CA isolate is closely related to more recent circulating BTV-2 strains from the southeastern United States.
Footnotes
Acknowledgements
The authors thank Drs. Lee Cohnstaedt, D. Scott McVey, and Raymond Rowland for review of this article.
a.
American Type Culture Collection, Manassas, VA.
b.
QIAquick gel extraction, Qiagen Inc., Valencia, CA.
c.
AMV Reverse Transcriptase, Life Technologies, Grand Island, NY.
d.
TakaRa Ex
e.
Ion Torrent OneTouch, OneTouch ES, Personal Genome Machine; Life Technologies, Grand Island, NY.
f.
Ion Xpress Plus fragmentation library kit, Xpress barcode adapters, Ion library quantitation kit, OneTouch 200 template kit v2, Ion PGM sequencing 400 kit, Ion 314 chip; Life Technologies, Grand Island, NY.
g.
Geneious 6.0, Biomatters Ltd., Auckland, New Zealand.
Authors’ note
Mention of trade names or commercial products in this report is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the U.S. Department of Agriculture (USDA), Agricultural Research Service (project 5430-32000-006-00D).
