Abstract
Within hours of chick delivery, acute mortality and mucosal lesions were reported on 2 northeast Georgia broiler farms that had applied a ferric sulfate litter amendment product. Histological evaluation of the larynx, tongue, and surrounding stroma revealed multifocal areas of necrosis or degeneration of the oral mucosa, acute focal necrotizing cellulitis, and the presence of a brown-black pigmented material adhered to affected epithelial and mucosal surfaces. Multifocal to diffuse ventricular koilin degeneration and acute hemorrhage was also demonstrated in association with pigmented adherent material on affected surfaces. Perls iron stain revealed that adherent material on affected tissues was strongly positive for iron. An experiment was designed to reproduce clinical signs, lesions, and mortality using the same litter amendment product. The ferric sulfate litter amendment was confirmed as the causative agent.
Keywords
Two cases from 2 northeast Georgia broiler integrators were submitted for diagnostic workup within 15 days of each other. Both companies reported an unspecified high level of mortality within hours of chick placement. In the first submission of 45 live chicks, 19 were dead on arrival, and all were severely dehydrated. Pooled tissues from the upper digestive and respiratory tracts were submitted for histological examination. The second submission involved fixed tissue from chicks with gross lesions involving the oral cavity, upper digestive and respiratory tract, and ventriculus.
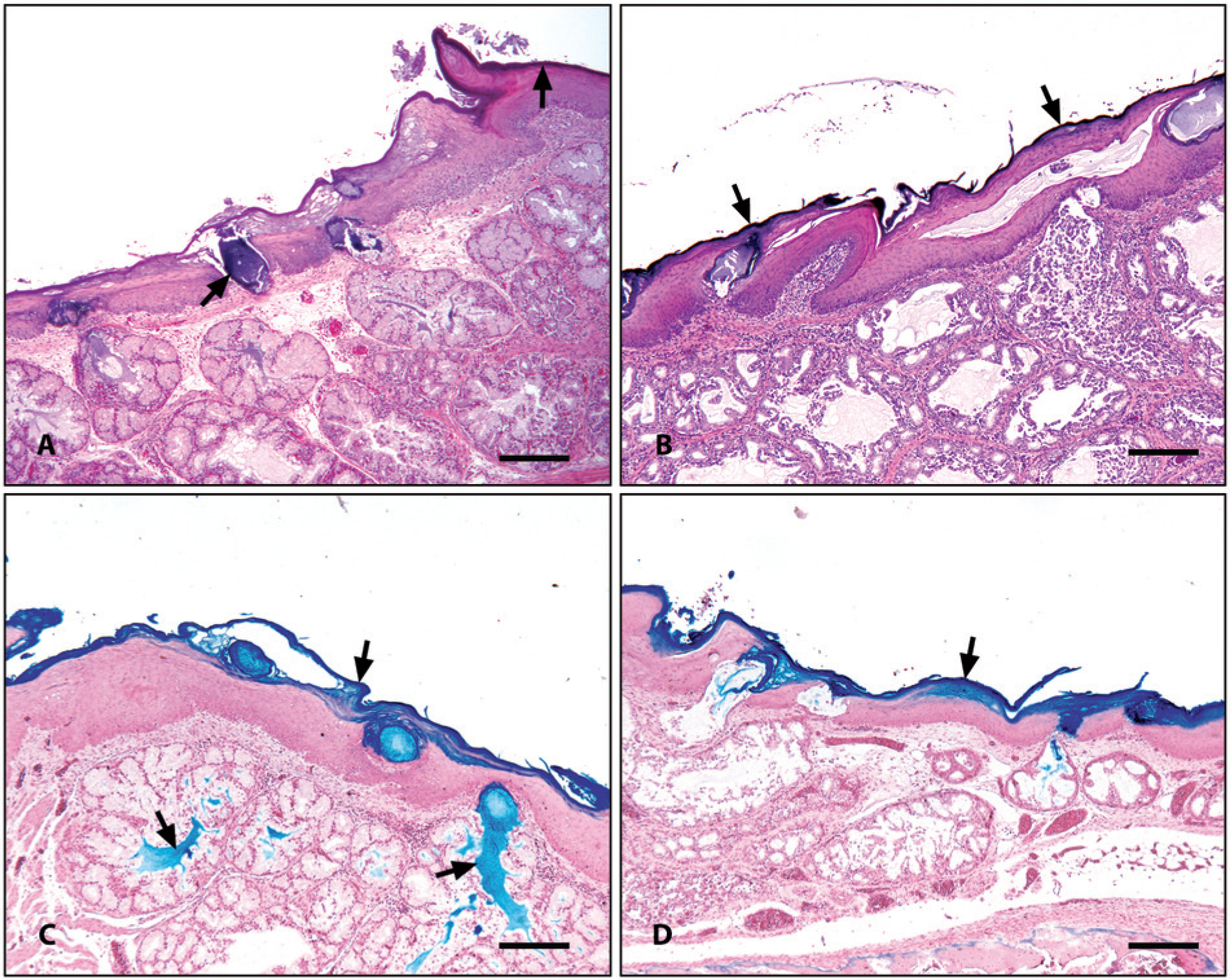
Microscopic examination of the oral cavity, tongue, and larynx demonstrated multiple areas of necrosis and epithelial degeneration with adherent brown-black pigmented material (Fig. 1A) in affected areas in both cases. The ventriculus submitted in the second case demonstrated multifocal to diffuse koilin degeneration and brown-black pigmented material on the surface of the koilin, suggestive of a chemical insult. Additionally, 1 section of larynx had severe necrosis with heterophils, macrophages, and edema in the stroma between the larynx and subcutaneous tissues. Common to both cases was the reported use of a ferric sulfate–based litter amendment product (FS) prior to chick placement. Litter amendment products are commonly used in situations where litter is reused for multiple flocks, with the intention of mitigating ammonia within poultry houses, and phosphorus runoff following land application of used litter. 1 Staining of affected tissues with Perls iron revealed the presence of iron in the adherent material detected in both cases. An experiment was designed to reproduce the clinical signs, mortality, and gross and histologic lesions associated with FS.
One-day-old broiler chicks.
Five 50-ft2 (4.65-m2) pens containing used softwood shaving–based poultry litter at Southern Poultry Research Inc. (Athens, Georgia) were assigned a litter treatment to represent various recommendations from the manufacturer and distributor of FS for application of the product. a Pen 1 remained untreated. Pen 2 was amended 6 days before chick placement using 100 lb per 1,000 ft2 (45 kg/92.9 m2) of FS, spread evenly and incorporated into the litter using gentle raking, per distributor recommendations b for rate and timing of application in broiler houses. Pen 3 received FS at 150 lb per 1,000 ft2 (68 kg/92.9 m2), spread unevenly throughout the pen 24 hr prior to chick placement, representing the timing and high-level application rate recommended on the product bag. Uneven application was conducted in an attempt to replicate the frequent inaccuracies in application occurring in the field after mass application of litter amendment products. Pen 4 was treated 24 hr prior to chick placement with FS at 75 lb per 1,000 ft2 (34 kg/92.9 m2) and a conventional sodium bisulfate litter amendment product (SB) c at 25 lb per 1,000 ft2 (11 kg/92.9 m2), spread unevenly throughout the pen. This pen represented the mixture of products and application used on the farm from the second clinical case. Pen 5 was treated 24 hr prior to chick placement with FS at 150 lb per 1,000 ft2 (68 kg/92.9 m2) and 25 lb per 1,000 ft2 (11 kg/92.9 m2) of SB, applied unevenly. This pen served to determine if the SB combined with a high level of FS may exacerbate lesions or mortality. The combined application of these products is not endorsed by either litter amendment product company. Pen treatment groups are summarized in Table 1.
Treatment pens based on litter amendment type, level applied, incorporation of product into the litter, evenness of application, and days prior to chick placement.*

No additive = used litter, no litter amendments added; FS 100 = ferric sulfate litter amendment added at 100 lb/1,000 ft2 (45 kg/92.9 m2); FS 150 = ferric sulfate litter amendment added at 150 lb/1,000 ft2 (68 kg/92.9 m2); FS 75 + SB = ferric sulfate litter amendment added at 75 lb/1,000 ft2 (34 kg/92.9 m2), mixed with 25 lb/1,000 ft2 (11 kg/92.9 m2) of sodium bisulfate litter amendment; FS 150 + SB = ferric sulfate litter amendment added at 150 lb/1,000 ft2 (68 kg/92.9 m2), mixed with 25 lb/1,000 ft2 (11 kg/92.9 m2) of sodium bisulfate litter amendment; dash (—) = not applicable.
Four hundred broiler chicks obtained from a local integrator d at hatch, were evenly distributed into the 5 pens in accordance with industry density standards, and given access to food and water ad libitum. Twenty chicks per pen were harvested every 12 hr over a 48-hr time period, euthanized with CO2, and examined postmortem by the same investigator. Any mortality present at any sampling point was automatically included in the group of 20 birds to be necropsied. At necropsy, oral lesions were measured in millimeters using a digital caliper e and attributed a nominal value based on a previously described scoring system. 4
Lesion scores were compared between treatment groups at each time point using the Kruskal–Wallis test. Follow-up pairwise comparisons of lesion scores were performed using the Dunn procedure. The percentages of affected birds were compared between groups at each time point using an exact test for independence. The significance of pairwise comparisons between individual groups was adjusted using the Bonferroni procedure to limit the overall type I error probability to 5%. All tests assumed a 2-sided alternative hypothesis, and
Tissues from the oral mucosa, tongue, esophagus, larynx, and ventriculus were collected from 3 chicks per group per sampling point for microscopic examination. After routine tissue processing, the tissues were examined microscopically. In addition, sections of oral cavity, esophagus, and ventriculus were stained with Perls iron.
Within minutes of placement, chicks exposed to FS consumed the product and demonstrated gasping, violent head shaking, and in some cases, mortality. No clinical signs or gross lesions were observed in the no-additive control group of chicks. Grossly, the oral cavity of FS-exposed chicks displayed rust-colored mucus adherent to underlying tissue that appeared to be eroded, suggestive of a caustic burn. Lesions were extensive and variable, involving any combination of the lingual surface, hard and soft palates, laryngeal mound, mechanical papillae of the glottis, and esophagus. A statistical analysis of oral lesion scores revealed a significant difference between groups at 12 hr (
Mean oral lesion scores for chickens exposed to different litter additives. Twenty birds from each group were evaluated at each time point with a total of 80 birds in each group.*
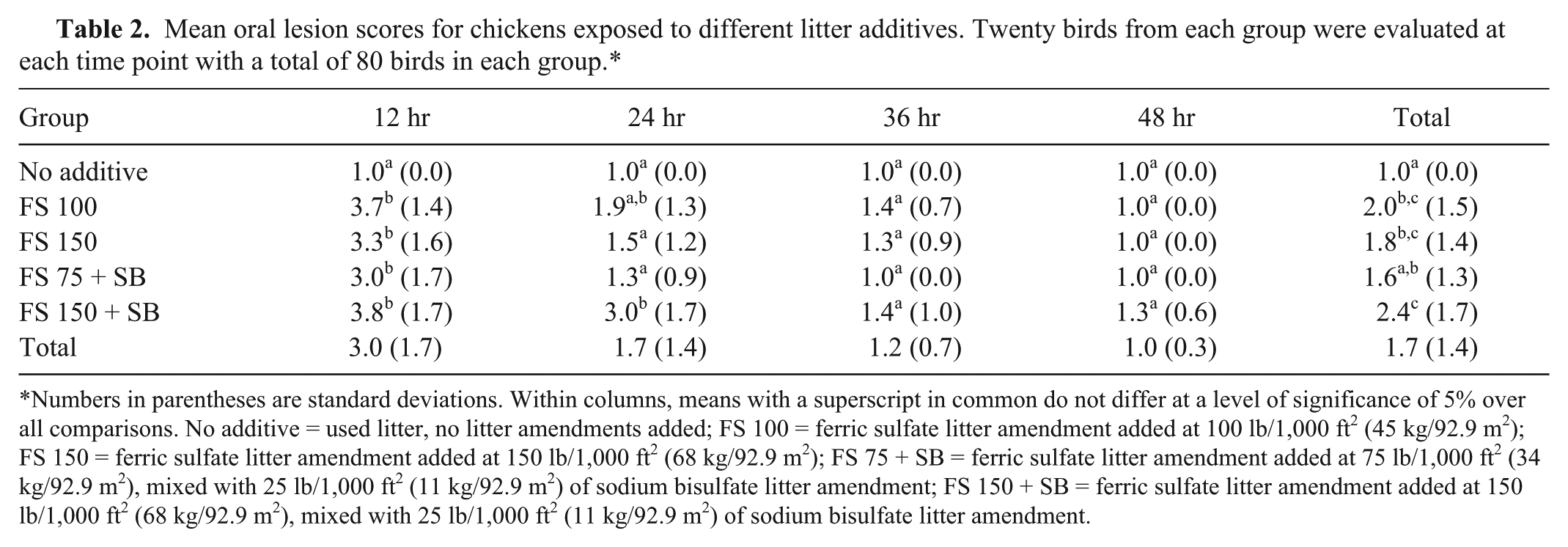
Numbers in parentheses are standard deviations. Within columns, means with a superscript in common do not differ at a level of significance of 5% over all comparisons. No additive = used litter, no litter amendments added; FS 100 = ferric sulfate litter amendment added at 100 lb/1,000 ft2 (45 kg/92.9 m2); FS 150 = ferric sulfate litter amendment added at 150 lb/1,000 ft2 (68 kg/92.9 m2); FS 75 + SB = ferric sulfate litter amendment added at 75 lb/1,000 ft2 (34 kg/92.9 m2), mixed with 25 lb/1,000 ft2 (11 kg/92.9 m2) of sodium bisulfate litter amendment; FS 150 + SB = ferric sulfate litter amendment added at 150 lb/1,000 ft2 (68 kg/92.9 m2), mixed with 25 lb/1,000 ft2 (11 kg/92.9 m2) of sodium bisulfate litter amendment.
Microscopic evaluation of the oral mucosa of the control chickens revealed no lesions. The oral mucosa in chicks from all FS-exposed groups had multifocal to diffuse brown-black pigmentation with associated epithelial degeneration in the beak and tongue, and clefting of the soft palate epithelium (Fig. 1B). There were multiple areas of mild to severe esophageal epithelial degeneration, with multiple dilated glandular openings forming cysts with pigment in their lumen and into the local mucin. The epithelium of the laryngeal opening also exhibited brown-black pigmentation with degeneration and edema at the hilus. Eight sections of trachea demonstrated extensive pigmentation of the respiratory epithelium along with multiple areas of ulceration while 3 sections had no lesions. The nasal sinus attached to only 1 section of submitted oral mucosa had extensive focal areas of epithelial necrosis and ulceration, and also exhibited pigmentation as seen in other tissues. Koilin degeneration and clefting were seen in the ventriculi of birds from the untreated control group. Ventriculi from all groups exposed to FS had brown-black pigment on the koilin surface, with a number of areas exhibiting clefting and vacuolation. Some areas demonstrated a sharp line of demarcation between eosinophilic and amphophilic areas in the koilin layer, suggesting a chemical insult. Tissue sections with brown-black pigment from FS-exposed groups stained positive with Perls iron staining (Fig. 1C, 1D).
A previous account of exposure to a different iron-based poultry litter amendment, namely ferrous sulfate heptahydrate, reported elevated mortality in commercial broiler chicks. 7 The previous case involved different findings on gross pathology, including pale musculature, friable livers, and swollen and discolored kidneys. None of the clinical signs herein reported were described in the ferrous sulfate case. Although it is possible for ferric sulfate to be reduced to ferrous sulfate in the presence of moisture, the striking differences between case reports suggest an alternate, yet undetermined cause of acute chick mortality. Furthermore, although the aforementioned study explored the effects of a similar compound in 1-day-old broiler chicks, the product was administered by gavage, which does not demonstrate voluntary consumption of the product under conditions mimicking commercial broiler production. The design of the current study aimed to demonstrate that the day-old chicks would in fact be attracted to, and consume, the product when applied as per the manufacturer and distributor recommendations tested in this experiment, resulting in lesions and mortality as reported in the 2 field cases in northeast Georgia. The consumption of the product by the day-old chicks, as opposed to the delivery of a similar product by means of gavage as in the previous report, 7 could help to explain the differences in observed gross and microscopic pathology and the absence of clinical signs in the aforementioned study. The amount of product consumed per chick could not be measured using the current study’s design, but the presence of lesions in all exposed groups when the product was applied using any of the manufacturer or distributor recommendations tested in this study confirms its role in inducing lesions under simulated commercial conditions. A future study modeled after previous designs5,7 could serve to determine if different dosages of product could induce different degrees of lesions and clinical signs.
Gasping and head shaking, responses generally associated with respiratory distress, 3 were observed in groups exposed to FS and signaled the possibility for hypoxia resulting in chick mortality. As tracheas were not harvested from all FS-exposed groups in the initial sampling, a separate group of day-old chicks was exposed to the product for the sole purpose of ruling out tracheal involvement. Ten chicks were placed in a 25 cm × 30 cm box with 10 g of the ferric sulfate product spread evenly in the bottom of the box. Three of the 10 birds perished within 5 min of voluntary consumption of the product, and tracheas collected from all chicks 1 hr postexposure were submitted for microscopic evaluation. Microscopic tracheal lesions were not detected in any of the chicks sampled within 1 hr postexposure, including chicks that died within minutes of consuming the product (data not shown), suggesting lack of direct tracheal involvement as a cause of mortality. While 8 of the 11 tracheas from the pen trial had lesions and pigment present, this may not be representative of treatment groups at the given time points given that only 3 birds per group were selected for microscopic examination. Another possible explanation for the exhibited behaviors could be that these are nociceptive responses exhibited after FS-induced irritation. Head shaking is a documented response to oral stimulation, albeit not exclusively related to nociception. 2 A more in-depth investigation that includes quantification of iron levels in internal organs and physiological parameters would be necessary for insight on a possible cause of death, for example, systemic iron toxicity causing oxidative stress from reactive oxygen species. 6
Pigmented material staining positive with Perls iron stain was present in all groups exposed to FS, as was initially observed in the 2 clinical cases. Degenerative glossitis, stomatitis, and multifocal to diffuse koilin degeneration of the ventriculus were observed in the clinical and experimental chickens. The presence of mild koilin degeneration in the untreated control group is likely attributed to unrelated factors. The absence of pigmented material adherent to these lesions differentiated this group from groups exposed to FS.
Although statistical differences in oral lesion scores did exist between groups at the 12- and 24-hr time points, no conclusions can be made regarding best application practices of the FS product based on this experiment. The FS product was however consumed by day-old chicks and did result in oral lesions in all groups exposed at 12 and 24 hr postexposure, with lesions being mild to completely absent in 36- and 48-hr samplings, suggesting recovery from the initial insult. Adherent ferric ion–containing pigment on affected tissues, its absence in the untreated control group, and the lack of clinical signs and mortality in the untreated group confirm the FS amendment as the primary cause of lesions and mortality in both the experimental and the 2 clinical cases. In the field, clinical signs associated with toxicity of similar products are often displayed only by a very small percentage of birds,4,7 and for this reason it was deemed necessary to expose a significant number of chicks to the product in question with the goal of obtaining a modest percentage of affected birds. The mortality obtained in the current study in exposed chicks was well above the U.S. industry goals of <1% mortality in the first 7 days of life, with ferric sulfate–exposed birds exceeding this figure in the first 12 hr. Any case of elevated chick mortality where an FS litter amendment was used should be evaluated for possible FS-induced oral lesions. Oral lesions in surviving birds may impair their ability to consume feed and water thus affecting economic performance, confirmation of which requires a study that is longer in duration and approach.
Footnotes
Acknowledgements
The authors thank Fieldale Farms Corp. for generously donating chicks and litter amendment products for the in vivo experiment.
a.
Klasp (Fe2(SO4)3·9(H2O)), Kemira Water Solutions Inc., Bartow, FL.
b.
Manure Safety Solutions LLC, Manning, IA.
c.
PLT (NaHSO4), Jones-Hamilton Co., Walbridge, OH.
d.
Fieldale Farms Corp., Baldwin, GA.
e.
Lowes Home Improvement, Mooresville, NC.
f.
Stata version 12.1, StataCorp LP, College Station, TX.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was sponsored by the Department of Population Health, University of Georgia and Fieldale Farms Corp., Baldwin, Georgia.
