Abstract
Three spherical opaque-white tumor nodules were found in close proximity to each other in the liver of a breeding sow, postslaughter, at a veterinary food inspection. The tumor nodules were circumscribed and histologically consisted of discrete hepatocellular and cholangiocellular nests, in association with polygonal-to-oval–shaped cells with slight cellular atypia. Immunohistochemically, all cellular components were negative for carcinoembryonic antigen, but positive for p53. Both cholangiocytes and oval-shaped cells were immunoreactive to anti-cytokeratin antibodies AE1/AE3 and MNF116. In addition, cholangiocytes were exclusively immunoreactive to anti-cytokeratin antibody CAM5.2, and hepatocytes were positive for MNF116 and hepatocyte paraffin 1. All neoplastic cells were positive for the hepatic progenitor cell markers, α-1-fetoprotein, sal-like protein 4, and epithelial cell adhesion molecule. From these results, the present case was diagnosed as hepatocholangiocellular adenoma, arising from epithelial cells of the canals of Hering.
Primary hepatic tumors originate from all cellular components of liver tissue. Epithelial tumors of the liver and biliary tracts, including hepatocellular and cholangiocellular carcinomas, have been reported for dogs, cats, cattle, horses, sheep, and pigs. Hepatocellular and cholangiocellular carcinomas are predominantly detected in dogs and cats, respectively. 10 Tumors containing neoplastic hepatocellular and cholangiocellular elements are rare, 2 with 3 benign cases reported in pigs.11,17 To date, the cellular origin of this type of tumor has yet to be investigated. In the current report, a porcine case of multiple occurrences of hepatocholangiocellular adenoma and the subsequent investigation of cellular origin are presented.
Veterinary food inspection of a healthy breeding sow, postslaughter, at a slaughterhouse in Kanagawa Prefecture, Japan in January 2011, revealed 3 nodules. The nodules were spherical in shape, 1–2 cm in diameter, and in close proximity to each other, at the cut surface of the liver (Fig. 1). No apparent macroscopic abnormalities were observed for any other organ. The liver tissues containing the tumors were fixed in 10% neutral buffered formalin (pH 7.4), and a pathological examination was conducted. Postfixation, the nodules were described as well circumscribed, spherical, and opaque-white at the cut surface. Notably, 1 nodule had formed cysts. Following paraffin embedding, 3-μm thick sections were prepared and subjected to staining using hematoxylin and eosin, periodic acid–Schiff (PAS), and Alcian blue, or immunohistochemical analysis.
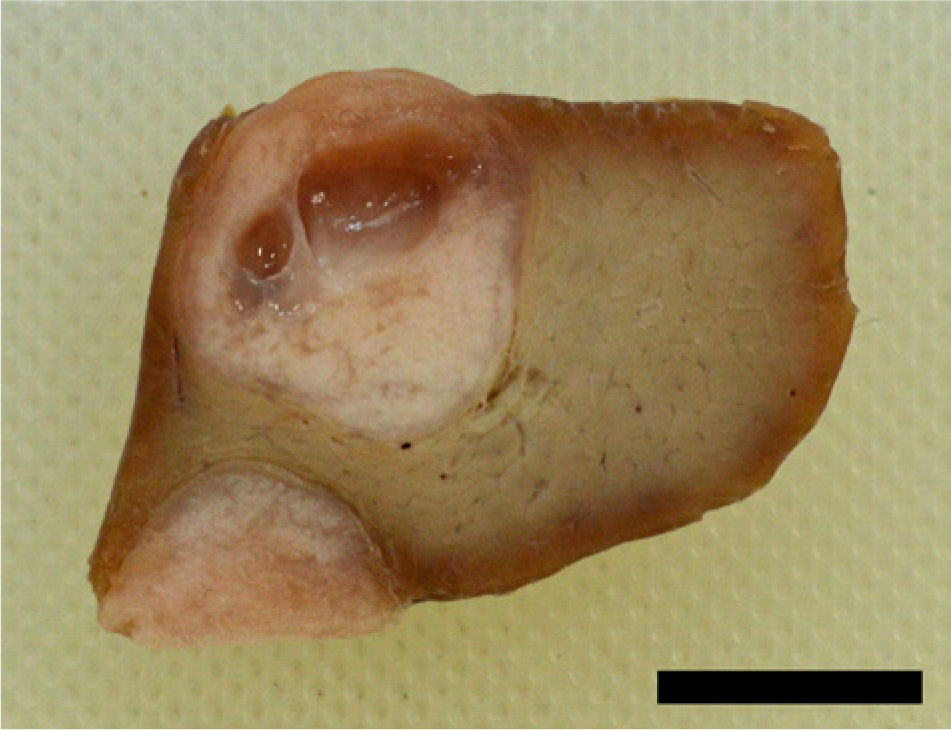
Macroscopic view of liver tumor nodules. Cut surface of 2 formalin-fixed porcine liver tumors. The tumors are spherical, well circumscribed, and pale. One tumor contains cystic spaces. Bar = 10 mm.
Histologically, tumor nodules were circumscribed, surrounded by thin connective tissue, including nonneoplastic hyperplastic foci of bile ductules at the periphery, giving rise to neoplastic cellular nests, and compressing adjacent liver parenchyma (Fig. 2A). The neoplastic cellular nests consisted of 3 cellular populations, hepatocytes, cholangiocytes, and polygonal to oval-shaped cells, separated by irregularly shaped connective tissue. Hepatocytes had small and round-to-oval nuclei with abundant, eosinophilic to clear and foamy cytoplasm. Cholangiocytes, columnar in shape, had oval-to-polygonal nuclei and eosinophilic cytoplasm, and formed multibranched ductular structures with focal multilayered cellular arrangements, adjacent to hepatocellular nests (Fig. 2B). A few, small polygonal-to-oval–shaped cells were observed within hepatocellular nests. All neoplastic cells had mild cellular atypia, and mitotic figures were rare. The tumors were without invasive growth, necrosis, or hemorrhage. With the exception of compression atrophy of liver cells, no other obvious changes in liver parenchyma surrounding the tumors was observed. The cytoplasm of hepatocytes and the goblet cells of cholangiocellular nests were positive for PAS. In addition, a small quantity of PAS-positive material was detected within tubular structures (Fig. 3A). Both hepatocytes and cholangiocytes were negative for Alcian blue staining.
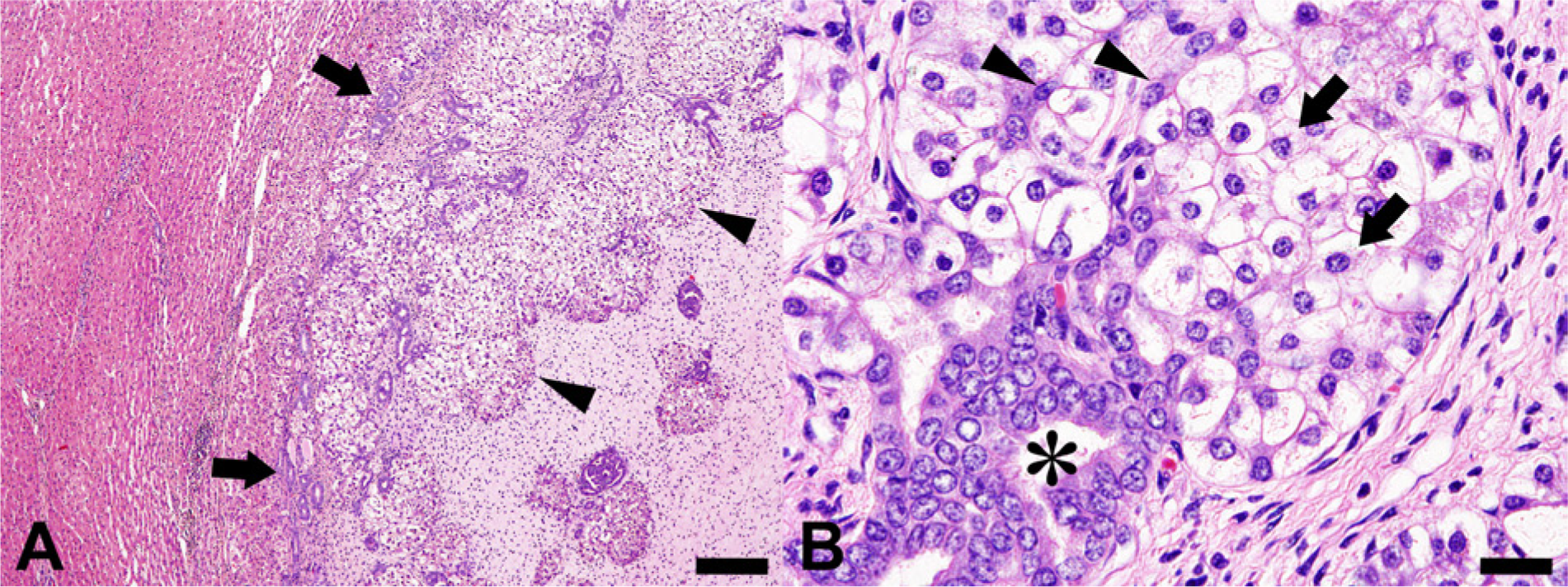
Light micrographs of a liver tumor nodule.
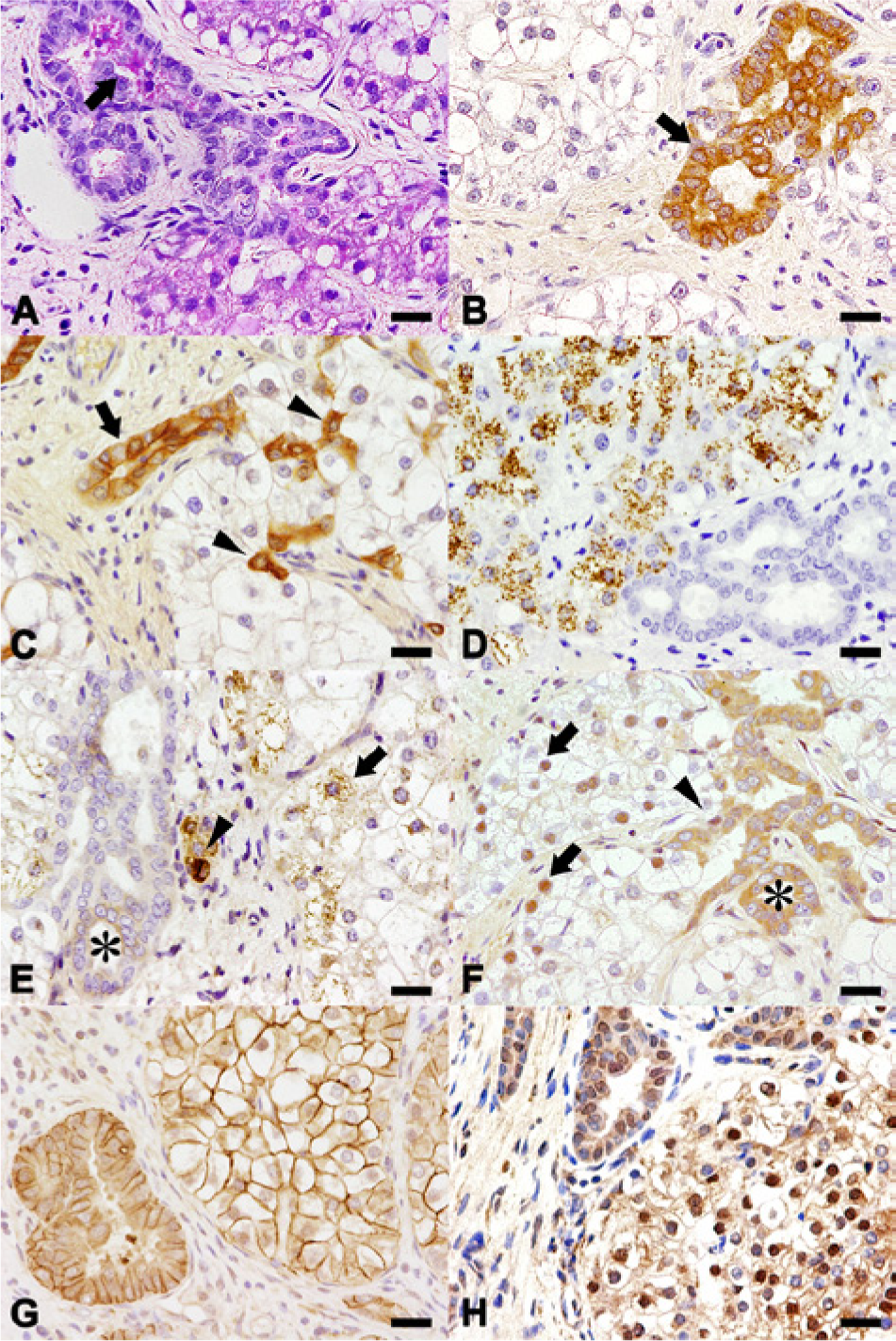
Histochemical (
Immunohistochemical analysis was performed using the avidin–biotin–peroxidase complex method a and primary antibodies against cytokeratins (CKs): CAM5.2 (undiluted solution), b AE1/AE3 (1:50), c and MNF116 (1:50) c ; hepatocyte paraffin 1 (Hep Par1; 1:50; clone OCH1E5) c ; α-1-fetoprotein (AFP; 1:400) c ; sal-like protein 4 (SALL4; 1:500) d ; epithelial cell adhesion molecule (EpCAM; 1:100) e ; carcinoembryonic antigen (CEA; 1:200) c ; p53 (1:50; clone sc-6243) e ; and proliferating cell nuclear antigens (PCNA; 1:100; clone PC10) c and Ki-67 (1:50; clone MIB-1). c
Prior to endogenous peroxidase blocking, sections were microwaved at 90°C for 10 min in citrate buffer (pH 6.0) for CAM5.2, AE1/AE3, MNF116, SALL4, EpCAM, and PCNA staining, or autoclaved at 121°C for 10 min in citrate buffer (pH 6.0) for Hep Par1, AFP, p53, and Ki-67 staining. For CEA staining, sections were pretreated with proteinase K. The chromogen was 3,3′-diaminobenzidine, and the counterstain was hematoxylin. Porcine pancreatic tissue was prepared as a positive control for CAM5.2, AE1/AE3, and MNF116. Porcine liver tissue was used as a control for Hep Par1, AFP, and CEA, and the lymph nodes were used as a control for p53 and Ki-67. Canine testicular tissue was used as a positive control for SALL4, the liver for EpCAM, and skin for PCNA. For negative controls, the primary antibody was replaced with nonimmunized sera.
With regard to the immunohistochemical analysis of CKs, only cholangiocytes were immunoreactive for CAM5.2 (Fig. 3B), cholangiocytes and oval-shaped cells were immunoreactive for AE1/AE3 and MNF116, and hepatocytes were very weakly immunoreactive to MNF116 (Fig. 3C). Most hepatocytes showed strong and granular cytoplasmic staining for Hep Par1, while cholangiocytes and oval-shaped cells were negative (Fig. 3D). The majority of hepatocytes and oval-shaped cells showed strong and granular cytoplasmic immunoreactivity to AFP, while cholangiocytes were only weakly immunoreactive, and displayed a diffuse cytoplasmic staining pattern (Fig. 3E). With regard to SALL4, hepatocytes and oval-shaped cells exhibited nuclear staining, while cytoplasmic staining was observed for cholangiocytes (Fig. 3F). All the hepatocellular, cholangiocellular, and oval-shaped cellular components in the tumors were positive for EpCAM (Fig. 3G), but negative for CEA. In addition, all tumor cells showed strong nuclear staining for p53 (Fig. 3H). The average ratios (%) of PCNA-positive cells counted in 10 randomly selected fields under 200× magnification were 1.97 in normal hepatocytes and 6.17 for the tumor cell population. Similarly, the average ratios (%) of Ki-67–positive cells were 2.17 in normal hepatocytes and 5.53 for the tumor cell population.
With regard to immunoreactivity to the CKs, only the cholangiocytes were immunoreactive to CAM5.2 in the current case study, recognizing CKs 7 and 8. Cholangiocytes were also immunoreactive to AE1/AE3 and MNF116. AE1/AE3 recognizes a broad spectrum of CKs, including CKs 1–8, 10, 14–16, and 19, while MNF116 recognizes CKs 5, 6, 8, 17, and 19. In human beings and rodents, both hepatic progenitor cells and cholangiocytes are known to express CKs 7 and 19.6,18 Therefore, it was reasonable that cholangiocellular components in the present case were immunoreactive to all CK clones used, and scattered immunoreactivity to AE1/AE3 and MNF116 for oval-shaped cells may be indicative of CK19 expression in hepatic progenitor cells. In contrast, hepatocellular differentiation is characterized by the progressive loss of biliary CKs, showing negativity to CK19 and the weak expression of CK7. 6 Positive hepatocellular immunoreactivity to MNF116 in the present case may be due to CK17 expression, but unfortunately at the time of writing there was no published reports to substantiate CK17 expression in either normal or neoplastic hepatocytes.
In the present case, granular immunoreactivity to Hep Par1 and AFP was observed, in addition to PAS positivity, in the cytoplasm of hepatocytes. Both Hep Par1 and AFP are hepatocellular markers, showing granular immunoreactivity to hepatocellular carcinoma (HCC) in human beings.4,9,21 Taken together, these results are suggestive of hepatocellular differentiation in the present case. The PAS-positive patterns may also support both the hepatocellular and cholangiocellular characteristics of this tumor. The presence of PAS-positive materials, as well as the sparse distribution of PAS-positive goblet cells within ductular structures, strongly supports the cholangiocellular nature of these tumors.
Sal-like protein 4 is a zinc finger transcription factor required for proliferation and maintenance of pluripotency, 3 and is considered a novel stem cell marker. 12 Sal-like protein 4 is exclusively expressed in human hepatobiliary tumors, inducing tumor-initiating cells to produce intermediate progenitor populations. 12 Epithelial cell adhesion molecule is also a known marker of hepatic progenitor cells in human beings and rodents,15,18,22 while α-1-fetoprotein is a marker for hepatoblasts, mouse oval cells, and hepatocytes.4,9,14 In the present case, immunoreactivity to AFP, SALL4, and EpCAM was observed for all types of neoplastic cells, suggesting that these proteins could be markers of hepatobiliary progenitor cells in pigs.
Based on the histologically benign features of hepatocellular and cholangiocellular components, and the absence of CEA expression, the present case was assumed to be that of hepatocholangiocellular adenomas. A mixed liver hamartoma presenting with similar morphology was reported for an equine foetus. 13 In the present case, the proliferation activities as estimated by PCNA and Ki-67 staining were higher for neoplastic cell populations than for the surrounding hepatocytes, indicating the exclusion of any developmental abnormality. Immunohistochemical analysis showed that p53 was highly expressed in all tumor cells. Studies have shown that p53 overexpression in hepatocellular, cholangiocellular, and stem cell and/or progenitor cell populations is accompanied with high proliferation activity in human cases of combined hepatocellular and cholangiocarcinoma (CHC). 19 The authors considered that p53 expression, in addition to high cell proliferation activity, may be caused by mutations in p53, which interfere with its role as gatekeeper in cell cycle regulations in CHC. The current adenoma case may also suggest dysfunctions in p53, most probably due to mutations, which alter cell proliferation. With regard to multiple occurrences, there have been 3 previously reported porcine cases of this type of tumor, diagnosed as benign.11,17 Consequently, porcine hepatocholangiocellular adenoma may arise multifocally.
In human beings, primary hepatic malignancies, namely combined hepatocellular and cholangiocarcinomas, were first reported in 1949. 1 The study further classified CHCs into the following 3 categories: 1) separate masses composed of either HCC or cholangiocarcinomas (CC); 2) contiguous masses with different characters that may mingle as they grow; or 3) mixed type showing individual masses displaying both features. 1 A 1985 study 7 proposed different criteria for the histological diagnosis of CHC, and classified CHC into 3 subtypes: 1) type I, collision tumors and double cancers, where the HCC and CC components are either completely separated or sharply demarcated; 2) type II, tumors with contiguous, independent masses of HCC and CC, separated by an intervening area of transition; or 3) type III, tumors consisting entirely of transitional areas, containing only very limited, ambiguous HCC and CC components. 7 Based on the observed histological features of cholangioductular formation adjacent to hepatocellular portions within tumor nests for the present case, it was identified as the benign counterpart of mixed type, according to the 1949 classification scheme, and type III according to the 1985 study.
There are 2 hypotheses on the cellular origin of CHC in human beings: 1) CHC originates from HCC through metaplasia to cholangiocytes; or 2) CHC originates from hepatic stem or progenitor cells, which have the potential to differentiate into both hepatocellular and cholangiocellular lineages.20,23 It has been hypothesized that hepatocholangiocellular adenomas in pigs arise from epithelial cells of the canals of Hering, which may retain a capacity to differentiate into liver cords or bile ducts.11,17 In rodents, oval cells, located at the canals of Hering, are reported to be bipotential progenitor cells expressing markers of both fetal hepatocytes and cholangiocytes. 8 Oval cells, undifferentiated oval-shaped small cells activated by hepatic damage to undertake liver cell regeneration, have also been considered to develop hepatic malignancies in animal models.5,16 In human beings, similar cells have been regarded as hepatic progenitor cells. 14 Considering that the oval-shaped cells in the present case exhibited characteristics of hepatic progenitor cells, it is plausible that the tumor may have originated from oval cells of the canals of Hering. However, regenerative changes in the hepatic parenchyma surrounding the tumors were notably absent for the present case, suggesting that hepatic damages were not responsible for oval cell activation. Because the tumor nodules were closely located within the same liver lobe, neoplastic transformation of hepatic progenitor cells might have occurred during development resulting in the formation of multiple nodules.
In summary, the benign tumors with both hepatobiliary differentiation were considered to be hepatocholangiocellular adenomas originating from hepatic progenitor cells. The multiple occurrences in close proximity to each other typical of this tumor may be due to neoplastic transformation during development in this case.
Footnotes
a.
VECTASTAIN Elite ABC kit, Vector Laboratories Inc., Burlingame, CA.
b.
BD, Franklin Lakes, NJ.
c.
Dako Denmark A/S, Glostrup, Denmark.
d.
Abcam Inc., Cambridge, United Kingdom.
e.
Santa Cruz Biotechnology Inc., Santa Cruz, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
