Abstract
A disintegrin-like and metalloprotease with thrombospondin type 1 repeat motifs 13 (ADAMTS13) is a von Willebrand factor (vWF)-cleaving protease. Deficiencies in ADAMTS13 activity are known to cause thrombotic diseases in human beings. The present study evaluated whether the human ADAMTS13 activity enzyme-linked immunosorbent assay (ELISA) kit containing human vWF73 (a minimal substrate) and anti-N10 antibody (which specifically recognizes the decapeptide of the C-terminal edge of cleaved vWF by human ADAMTS13) is applicable to the measurement of canine plasma ADAMTS13 activity. Human vWF73 fused with a GST-tag and a His-tag (GST-hvWF73-His) was reacted with recombinant canine (rc)ADAMTS13, canine plasma, and human plasma, and then used in Western blotting using anti-N10 antibody. Linearity and intra- and interassay reproducibility of the human ADAMTS13 activity ELISA kit in canine plasma were further evaluated. Finally, plasma ADAMTS13 activity was measured in 13 healthy dogs and 6 dogs with bacteremia using the human ADAMTS13 activity ELISA kit. Cleaved products with a 28-kDa GST-hvWF73-His were detected specifically in rcADAMTS13 as well as in human ADAMTS13, and also in canine plasma by anti-N10 antibody, showing excellent linearity. Intra-assay coefficient of variation (CV) was 3.0–12.4%, and interassay CV was 11.5–12.5%. The ADAMTS13 activity was significantly lower in dogs with bacteremia than in healthy dogs (P = 0.0025). The current study revealed that the human ADAMTS13 activity ELISA kit is applicable for measurement of canine plasma ADAMTS13 activity to elucidate the pathology of thrombotic diseases in dogs.
Keywords
Introduction
Thrombotic problems in dogs are frequently detected in various diseases, including immune-mediated hemolytic anemia, protein-losing diseases, neoplasms, hyperadrenocorticism, bacterial infection, viral infection, cardiac diseases, and pancreatitis. 6 The pathological mechanisms causing thrombus formation differ among underlying diseases. 6 More detailed explorations of the pathological mechanisms are thus required to establish more suitable management for each thrombotic disease.
A disintegrin-like and metalloprotease with thrombospondin type 1 motifs, number 13 (ADAMTS13) is a plasma zinc metalloprotease and is known as a von Willebrand factor (vWF)-cleaving protease.10,15 Unusually large vWF multimers (UL-vWFMs) have a high capacity for binding to platelets causing thrombosis,1,4,13 so ADAMTS13 deficiency is clearly a cause of such thrombotic diseases as thrombotic thrombocytopenic purpura (TTP) in human beings.5,10
Although several methods for measuring ADAMTS13 activity have been developed, 7 the human (h)ADAMTS13 activity enzyme-linked immunosorbent assay (ELISA) kit, which offers relatively rapid results and convenience, has been established as the principal clinical laboratory test in human medicine.7,8 The hADAMTS13 activity ELISA kit consists of recombinant human vWF73 peptide fused with a glutathione-S-transferase (GST)-tag at the N-terminus and a 6-histidine repeats (His)-tag at the C-terminus (GST-hvWF73-His), horseradish peroxidase (HRP)-conjugated anti-N10 antibody, and an anti-GST antibody precoated 96-well microplate. 8 von Willebrand factor 73 is the minimal substrate for hADAMTS13, containing 73 amino acid residues from the vWF A2 domain (from Asp1596 to Arg1668). 9 Anti-N10 antibody, a mouse monoclonal antibody, specifically recognizes the decapeptide (1596-DREQAPNLVY-1605 in human vWF) of the C-terminal edge of vWF cleaved by hADAMTS13. 8
Molecular cloning of canine ADAMTS13 and expression of this recombinant protein has already been reported, proving that canine ADAMTS13 is able to cleave human full-length mature vWF. 12 However, whether the hADAMTS13 activity ELISA kit can be used to measure plasma canine ADAMTS13 activity remains unclear.
The present study therefore evaluated whether canine ADAMTS13 offers GST-hvWF73-His–cleaving ability and whether anti-N10 antibody recognizes the product of GST-hvWF73-His cleaved by canine ADAMTS13 in Western blot analysis. Furthermore, the linearity, reproducibility, and clinical utility of using the hADAMTS13 activity ELISA kit for canine plasma were also assessed.
Materials and methods
Expression and purification of human vWF73
GST-hvWF73-His was expressed as described previously
9
with a slight modification. Human DNA was obtained from HeLa cells (human epithelial cervical cancer cells) with a DNA purification kit.
a
Polymerase chain reaction (PCR) was performed using primer pairs (5′-cgggatccGACCGGGAGCAGGCGCCCAACC-3′ and5′-cggaattc
Cleaving reaction of GST-hvWF73-His
Canine pooled plasma obtained by mixing citrated plasma collected from 5 clinically healthy dogs and the recombinant canine (rc)ADAMTS13 obtained using HeLa cells by the method reported previously 12 were used to evaluate the ability of cADAMTS13 to cleave GST-hvWF73-His. To obtain canine citrated plasma, blood samples were collected in tubes containing 3.8% trisodium citrate (9 parts blood to 1 part anticoagulant) and centrifuged at 2,000 × g for 10 min. Human pooled plasma m was used as a positive control. Heat-inactivated canine pooled plasma and the medium of HeLa cells that had been transfected with a no-template inserted plasmid (NT) were used as negative controls.
The cleaving reaction was performed as previously reported 9 with a slight modification. One microliter of each sample mentioned above was added to 39 µl of reaction buffer (150 ng GST-hvWF73-His, 5 mM Tris–HCl, 10 mM BaCl2, and 1 mM amidinophenyl-methanesulfonyl fluoride hydrochloride, pH 8.0) and then incubated at 37°C for 1 hr. The cleaving reaction was stopped by adding 8 µl of SDS sample buffer (0.35 M Tris–Cl [pH 6.8], 10% SDS, 30% glycerol, 9% dithiothreitol, 0.12% bromophenol blue).
Western blot using anti-GST antibody and anti-N10 antibody
Two sets of these cleaving reaction samples were electrophoresed on 15% SDS-PAGE and transferred onto a polyvinylidene difluoride membrane. After blocking with 1% bovine serum albumin, the membrane was divided into 2 sheets. One of the membranes was incubated with peroxidase-labeled anti-GST antibody n (1:15,000). The other transferred membrane was incubated with anti-N10 antibody o (1:1,000), and then incubated with HRP-conjugated anti-mouse immunoglobulin G antibody (1:15,000). Chemiluminescent detection was performed using a chemiluminescence reagent p with a chemiluminescence detection instrument. q
Evaluation of linearity and reproducibility of the human ADAMTS13 activity ELISA kit for measuring canine plasma ADAMTS13 activity
Linearity and reproducibility were evaluated for the commercially available hADAMTS13 activity ELISA kit, r which consists of GST-vWF73-His and HRP-conjugated anti-N10 antibody for measurement of canine plasma ADAMTS13 activity. Assay procedure was performed according to the instructions from the manufacturer. A standard curve was established using human pooled plasma in all assessments. Optimal absorbance was measured using a microplate reader. s
In a linearity assay, 3 different citrated canine plasma samples (high, 97.2%; medium, 51.3%; and low, 26.2%), which had been measured using the hADAMTS13 activity ELISA kit in advance, were serially diluted with heat-inactivated canine pooled plasma to obtain samples with 6 different activities (only undiluted plasma, 4:5, 3:5, 2:5, 1:5, only heat-inactivated canine pooled plasma). All sample activities were assayed in duplicate. Intra-assay reproducibility was determined by analyzing 3 canine plasma samples with different ADAMTS13 activity (high, 81.4%; medium, 41.5%; and low, 9.9%) with 6 wells for each sample on the same microplate. This assay was performed twice on other days. Inter-assay reproducibility was examined by running the same 3 samples used for intra-assay tests on 5 different days. Results of intra- and interassay reproducibility are represented as coefficients of variation (CVs).
Measurement of plasma ADAMTS13 activities in healthy dogs and dogs with bacteremia
To clarify the clinical utility of the hADAMTS13 activity ELISA kit in dogs, 19 citrated plasma samples obtained from 13 healthy dogs and 6 dogs with bacteremia referred to the Animal Medical Center of Nihon University (Japan) were used. Bacteremia was confirmed by blood culture. Measurement of plasma ADAMTS13 activity was performed in duplicate. Standard curves were established using human pooled plasma.
Statistical analysis
The Mann–Whitney U-test was performed using statistical software t to compare ADAMTS13 activity between healthy dogs and dogs with bacteremia. Data are represented as median and range. Values of P < 0.05 were considered statistically significant.
Results
Expression and purification of GST-hvWF73-His
Two bands of approximately 36 kDa and 28 kDa were mainly induced in E. coli after IPTG induction, while a single band of approximately 36 kDa corresponding to GST-hvWF73-His protein was observed after purification by nickel and glutathione chromatography in SDS-PAGE (Fig. 1).
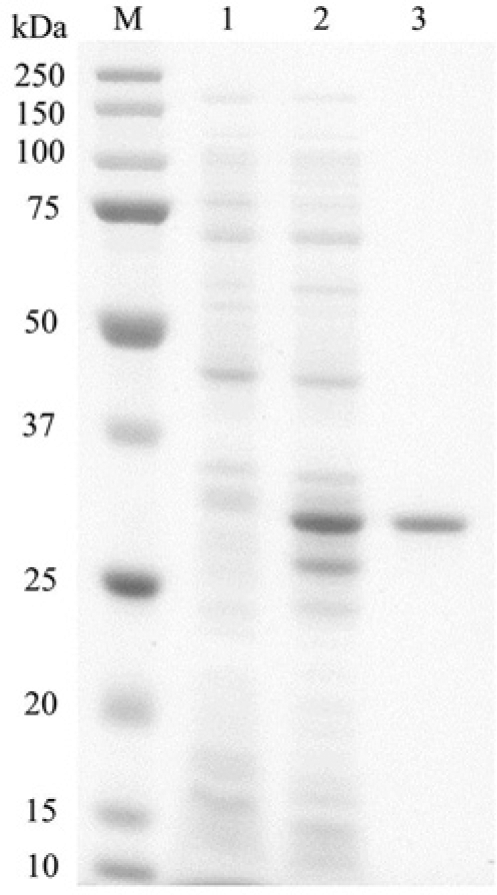
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis analysis of GST-hvWF73-His expressed in Escherichia coli. The E. coli pellet fraction with centrifugation before induction of expression (lane 1) and after isopropyl-β-
Confirming the ability of canine ADAMTS13 to cleave GST-hvWF73-His by Western blot
A band of cleaved products at approximately 28 kDa were observed from GST-hvWF73-His with rcADAMTS13, canine plasma, and human plasma in Western blot analysis using anti-GST antibody and anti-N10 antibody (Fig. 2A, 2B). However, no band of cleaved product was detected in heat-inactivated plasma or NT used as negative controls (Fig. 2A, 2B). In using anti-GST antibody, in addition to cleaved products, uncleaved GST-hvWF73-His at approximately 36 kDa was also observed in all samples (Fig. 2B).
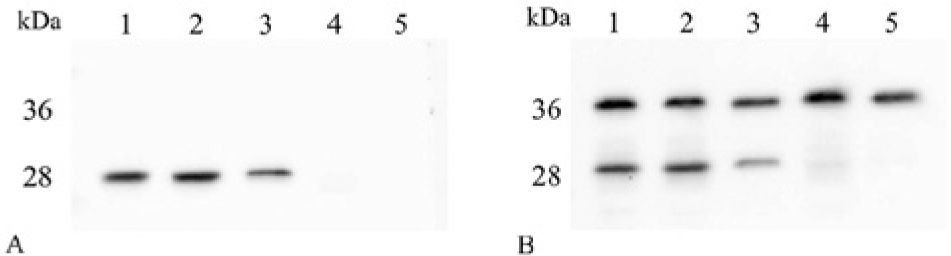
Profile of cleaved GST-hvWF73-His by recombinant canine (rc)ADAMTS13 and canine plasma in Western blot using anti-N10 antibody (
Linearity, reproducibility, and clinical utility of the human ADAMTS13 activity ELISA kit in measurement of canine plasma ADAMTS13 activity
Excellent linearity was observed in all samples with serial dilution (Fig. 3). Intra-assay CV was 3.0–12.4% in 3 samples with different activity (Table 1). Interassay CV was 11.5–12.5% in 3 samples with different activity (Table 2). Median plasma ADAMTS13 activity was significantly lower in dogs with bacteremia (51.0%; range: 37.7–68.8%) than in healthy dogs (76.6%; range: 56.8–110.5%; P = 0.0025;Fig. 4).
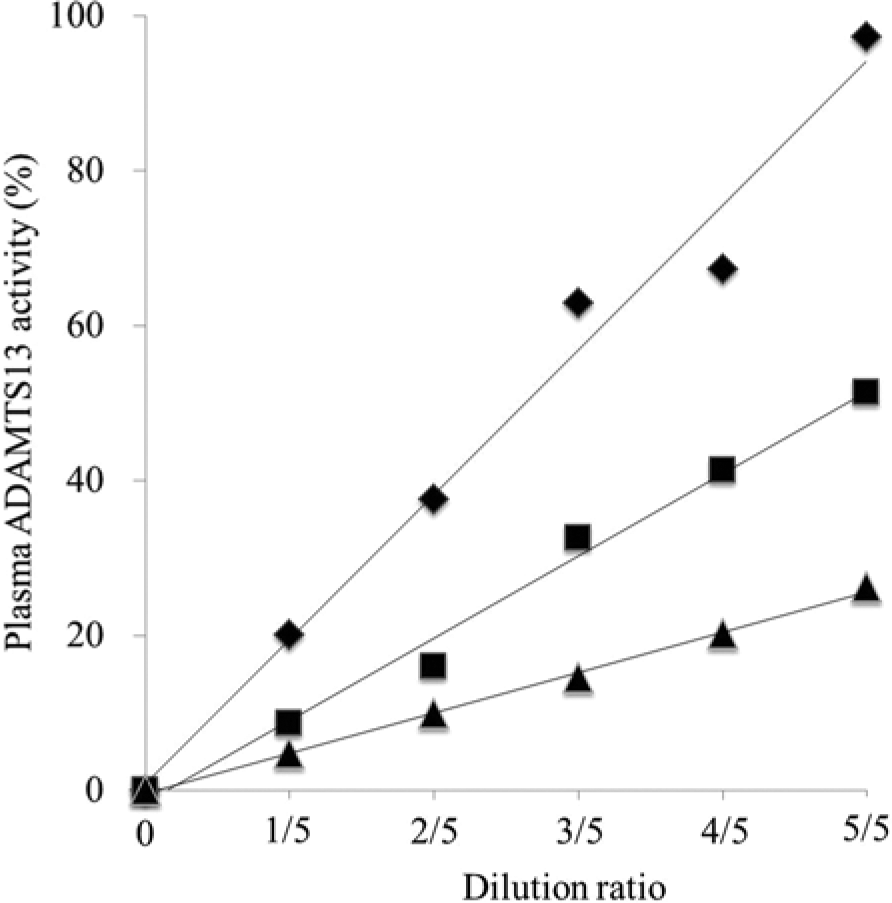
Dilution linearity of canine plasma ADAMTS13 activity. Three different plasma samples obtained from dogs with different plasma ADAMTS13 activity were serially diluted with heat-inactivated canine pooled plasma, and activity was assayed in duplicate. ♦: high, 97.2%; y = 0.93x + 0.86. ▲: medium, 51.3%;y = 0.53x − 1.56. ■: low, 26.2%; y = 0.26x − 0.39.
Intra-assay results using the human ADAMTS13 activity enzyme-linked immunosorbent assay kit for measurement of canine ADAMTS13 activity.*
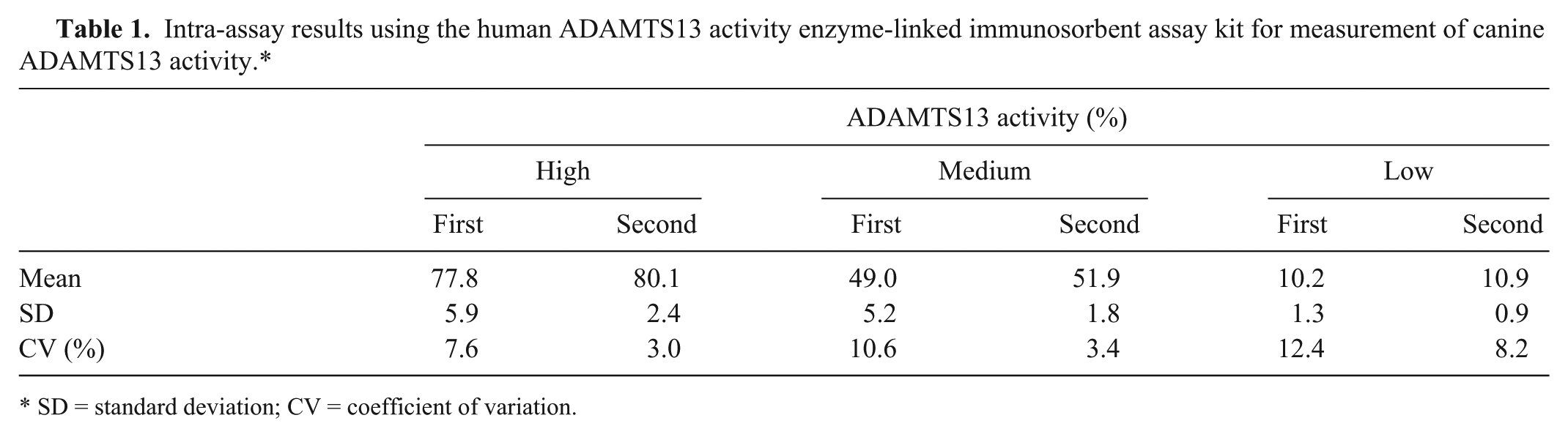
SD = standard deviation; CV = coefficient of variation.
Interassay result for human ADAMTS13 activity enzyme-linked immunosorbent assay kit in measurement of canine ADAMTS13 activity.*
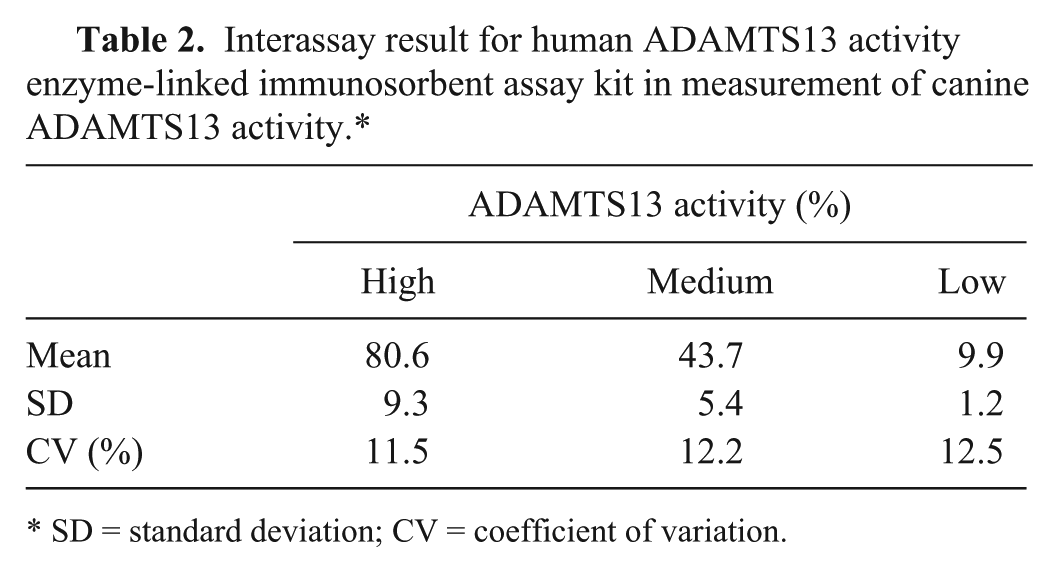
SD = standard deviation; CV = coefficient of variation.
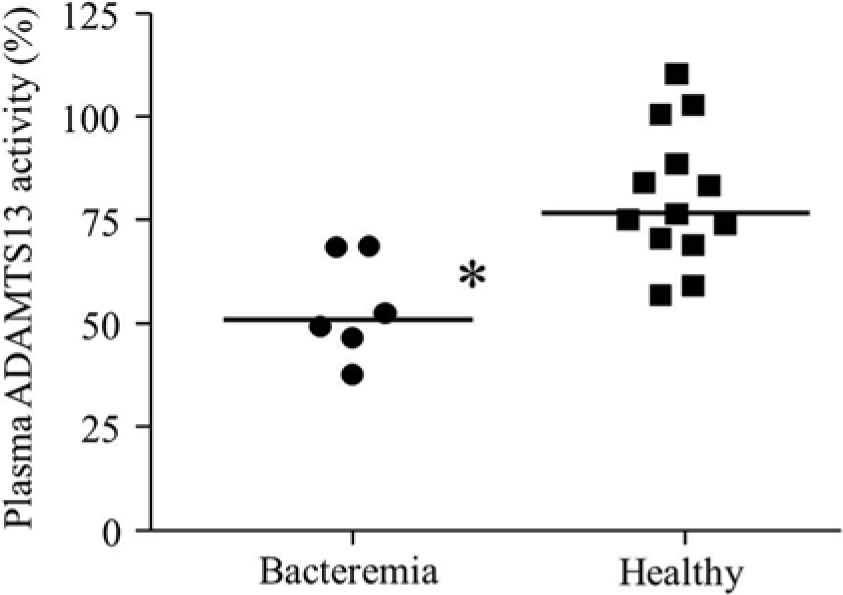
Distribution of plasma ADAMTS13 activity in 13 healthy dogs and 6 dogs with bacteremia. Horizontal bar represents median plasma ADAMTS13 activity. *P = 0.0025, compared with healthy dogs.
Discussion
Decreasing plasma ADAMTS13 activity has been identified not only as the cause of TTP but also as an exacerbating factor for thrombus formation in various human pathologies.7,11 However, whether ADAMTS13 is associated with thrombotic diseases in dogs has been unclear, as assay systems to specifically measure canine plasma ADAMTS13 activity have not been developed. The present study suggests that measurement of canine plasma ADAMTS13 activity may be useful to elucidate in detail the pathology of canine thrombotic diseases.
Several kinds of ADAMTS13 activity assays have been developed. 7 The assay method combining SDS–agarose gel electrophoresis and Western blotting was established first, but is extremely complex and time-consuming.3,7 The hADAMTS13 activity ELISA kit, which uses human vWF73, was established as a more rapid and convenient assay. 8 Evaluation of whether cADAMTS13 has the ability to cleave human vWF73 efficiently was needed, because the degree of human full-length mature vWF cleavage by canine plasma was weaker than that by human plasma.12,18 In Western blot analysis using anti-GST antibody in the current study, signal intensity and molecular size of the product of GST-hvWF73-His cleaved by canine plasma was the same as that cleaved by human plasma, indicating that cADAMTS13 has the capability to cleave GST-hvWF73-His as efficiently as hADAMTS13.
Anti-N10 antibody recognizes the decapeptide of the C-terminal edge of human vWF cleaved by hADAMTS13. 8 The fragments of GST-hvWF73-His cleaved by canine pooled plasma and rcADAMTS13 were detected by anti-N10 antibody, as were those from human pooled plasma in Western blot analysis, while no signal was detected in canine heat-inactivated plasma or NT used as negative controls. In addition, uncleaved substrate was not detected by anti-N10 antibody. These findings confirmed that the C-terminal edge of GST-hvWF73-His cleaved by canine ADAMTS13 was specifically recognized by anti-N10 antibody.
Linearity, precision, and reproducibility of hADAMTS13 activity ELISA kit in measurement of canine plasma ADAMTS13 activity were assessed. As results, excellent linearity was observed in serially diluted canine plasma samples with various ADAMTS13 activities, while intra- and interassay CVs were <12.4% and <12.5%, respectively. According to the information from the manufacturers of this hADAMTS13 activity ELISA kit, the CV of reproducibility is <10% in human beings. From the results, this hADAMTS13 activity ELISA kit offers reliable measurement of canine plasma ADAMTS13 activity, although reproducibility was slightly inferior in dogs than in human beings.
Plasma ADAMTS13 activity was reduced in dogs with bacteremia compared to healthy dogs. Plasma ADAMTS13 activity is reportedly decreased and UL-vWFMs are increased in human patients with sepsis causing disseminated intravascular coagulation, and the reason for decreased plasma ADAMTS13 is not only decreased synthesis of ADAMTS13 in the liver but also cleavage of ADAMTS13 by proteases such as thrombin, plasmin, and granulocyte elastase. 14 Plasma vWF antigen concentration was previously reported to be significantly higher in dogs with sepsis than in healthy dogs. 16 The present study was performed with a small number of cases, so the results might be limited. Further studies with increased cohort numbers are required to elucidate the relationships among plasma ADAMTS13 activity, vWF, systemic bacterial infection, and thrombus formation, because plasma vWF antigen concentration, UL-vWFMs, and degradation of ADAMTS13 by proteases have not been clearly evaluated.
Thrombotic thrombocytopenic purpura refers to a disorder in which widespread platelet- and vWF-rich microthrombi are observed in the small blood vessels of multiple organs, with clinical signs such as anemia, thrombocytopenia, renal failure, fever, and neurological dysfunction.2,10,17 Although dogs with clinical signs resembling human TTP are often encountered in clinical practice, no cases of canine TTP appear to have been reported to the authors’ knowledge. The reason that TTP has not been reported in dogs could be the absence of an assay system specifically for measuring canine plasma ADAMTS13 activity. The present results might thus facilitate the discovery of TTP in dogs.
In conclusion, this study revealed that cADAMTS13 efficiently cleaves human vWF73, and anti-N10 antibody recognizes the edge of vWF73 cleaved by cADAMTS13. The hADAMTS13 activity ELISA kit thus appears applicable for measurement of plasma ADAMTS13 activity in dogs. These results should facilitate elucidation of the detailed pathology of thrombotic disease in dogs.
Footnotes
a.
QIAamp DNA blood mini kit, Qiagen GmbH, Hilden, Germany.
b.
PrimerSTAR GXL DNA polymerase, Takara Bio Inc., Otsu, Shiga, Japan.
c.
pGEX-6P-1, GE Healthcare, Little Chalfont, Buckinghamshire, United Kingdom.
d.
BigDye Terminator v3.1 cycle sequencing kit, Applied Biosystems, Foster City, CA.
e.
ABI PRISM 310 genetic analyzer, Applied Biosystems, Foster City, CA.
f.
BL21 DE3 competent cells, Invitrogen Corp., Carlsbad, CA.
g.
CelLytic B, Sigma-Aldrich, St. Louis, MO.
h.
His Mag Sepharose Ni, GE Healthcare, Little Chalfont, Buckinghamshire, United Kingdom.
i.
MagneGST protein purification system, Promega Corp., Madison, WI.
j.
Amicon Ultra-10k, EMD Millipore Corp., Billerica, MA.
k.
DC Protein Assay, Bio-Rad Laboratories, Hercules, CA.
l.
Bio-Safe Coomassie stain, Bio-Rad Laboratories, Hercules, CA.
m.
Pooled normal plasma, George King Bio-Medical, Overland Park, KS.
n.
Anti-GST HRP conjugate, GE Healthcare, Little Chalfont, Buckinghamshire, United Kingdom.
o.
N10 (146), sc-101404; Santa Cruz Biotechnology Inc., Dallas, TX.
p.
ECL Prime Western blotting detection system, GE Healthcare, Little Chalfont, Buckinghamshire, United Kingdom.
q.
ChemiDoc XRS, Bio-Rad Laboratories, Hercules, CA.
r.
ADAMTS13 activity ELISA kit, KAINOS Laboratories, Tokyo, Japan.
s.
Multiskan GO microplate photometer, Thermo Fisher Scientific Inc., Waltham, MA.
t.
GraphPad Prism version 5.04 for Windows, GraphPad Software Inc., San Diego, CA.
Declaration of conflicting interests
The author(s) declare that there are no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by JSPS (Japan Society for the Promotion of Science) Grants-in-Aid for Scientific Research (grant 23780327).
