Abstract
During an ongoing physiological ecology study on pups and adult female northern elephant seals (Mirounga angustirostris, Gill, 1866) on the mainland rookery at Año Nuevo State Reserve (California), an opportunity was afforded to collect fresh dead pups for parasitology and necropsy. The investigation was undertaken to delineate the causes of death of northern elephant seals recovered from Año Nuevo State Reserve. Prior to this study, there was no evidence of increased mortality or health problems on this rookery. Necropsies, histology, and ancillary diagnostic studies were conducted on 21 fresh dead preweaned pups. Ages ranged from 1 stillbirth to pups approximately 2 weeks of age. Gross lesions included varying degrees of bruising, hemorrhage, lacerations, and fractures attributed to blunt force trauma to the head, chest, and/or abdomen in 16 pups; starvation in 6 pups; bite wounds in 2 pups; generalized icterus in 2 pups; presumptive drowning in 2 pups; and 1 stillbirth. Most pups had multiple gross lesions. Following light microscopic examination, pups could be assigned into 4 general diagnostic categories: 1) trauma, 2) nutritional status, 3) infectious conditions, and 4) congenital anomalies. This investigation of preweaned pup mortality of northern elephant seals in California further refines diagnostic categories for perinatal pup mortality.
From the early 1800s to the 1860s, northern elephant seals (Mirounga angustirostris, Gill, 1866) were extensively harvested for blubber to render for oil. By the late 1870s, northern elephant seals were considered regionally extirpated. 10 In 1922, the Mexican Government designated Isla de Guadalupe a biological reserve, protecting the seals from further harvest. Following protection, the seals expanded and established new rookeries and haul-outs throughout this region and into southern California. At the time of publication, there are approximately 21 known breeding rookeries in Mexico and southern California with a population of approximately 166,000 animals.17,21
Northern elephant seals were first seen off the coast of southern California in 1925 on San Miguel Island, the most western of the Channel Islands, and were observed breeding there in the early 1950s. Northern elephant seals were also observed breeding on Santa Barbara Island and San Nicolas Island off the coast of southern California in 1950. Northern elephant seals were first observed on Año Nuevo State Reserve in 1955 and were breeding by 1961. 21 Northern elephant seals forage extensively throughout the northeastern Pacific Ocean from Baja to the Aleutian Islands of Alaska and are among the deepest diving marine mammals. 10
Trauma and starvation have been considered to be the primary cause of mortality of preweaned pups, and lack of successful weaning has been hypothesized to be related to mother–pup separation with subsequent starvation and/or inadvertent lethal injury inflicted by adults. 9 However, the history of a severe population bottleneck suggests a lack of genetic diversity in this population, which may predispose these animals to congenital anomalies and infectious disease processes.2,8,23
The aim of the current study was to perform necropsies on fresh dead northern elephant seal pups on 2 rookeries located in the Año Nuevo State Reserve, California (N 37.1086°, W 122.3378°) and determine the main causes of mortality. Between January 26 and February 11, 2012, 4–5 personnel searched all of the rookery locations once daily for fresh dead (code 1) pups. Animals were removed from the rookeries and necropsied according to standard necropsy procedures. Pups with moderate to advanced autolysis (codes 2 and 3) were not examined.
Ages of pups examined were estimated based on aeration within the lungs, meconium in the rectum, fresh umbilicus, and, to some degree, on standard body length. Pups were assigned to 1 of 3 categories (stillbirth, neonate, or pup). Stillbirths had no or only minimally aerated lungs; neonates (birth to 24 hr of age) had aerated lungs, fresh umbilicus, and meconium in the rectum; and pups (at least 1 day of age and older) had aerated lungs and the rectum was empty or had feces. Body condition was scored by mid-sternal blubber thickness. Animals also were assigned to 1 of 4 body condition categories: excellent (>15 mm blubber); good (10–15 mm); poor (5–10 mm); and emaciated due to starvation (<5 mm; Table 1).
Age and body condition categories of 21 preweaned northern elephant seals (Mirounga angustirostris, Gill, 1866) examined from Año Nuevo State Reserve, California. The measurements reflect midsternal blubber thickness.

Tissues from all major organs and any gross lesions were collected for histology and preserved in 10% neutral buffered formalin. Fixed tissues were trimmed, placed in cassettes, processed a , embedded in paraffin, sectioned at 5 μm, and stained with hematoxylin and eosin. Special stains including Grocott methenamine silver, periodic acid–Schiff, Ziehl–Neelsen acid-fast, Brown–Hopps modified Gram, Perls iron, Hall stain for bile, and von Kossa were conducted on selected tissues. All staining protocols followed procedures from the Armed Forces Institute of Pathology. 14 Viral and bacterial isolations, real-time polymerase chain reaction, and mineral analysis were all carried out by routine methods at the Colorado State Medical Diagnostic Center, College of Veterinary Medicine (Fort Collins, Colorado). 19 The digestive tract was collected and examined for parasites. 12 As funds were not available to pursue all diagnostic tests for each animal, only selected tests were performed on tissues from specific animals.
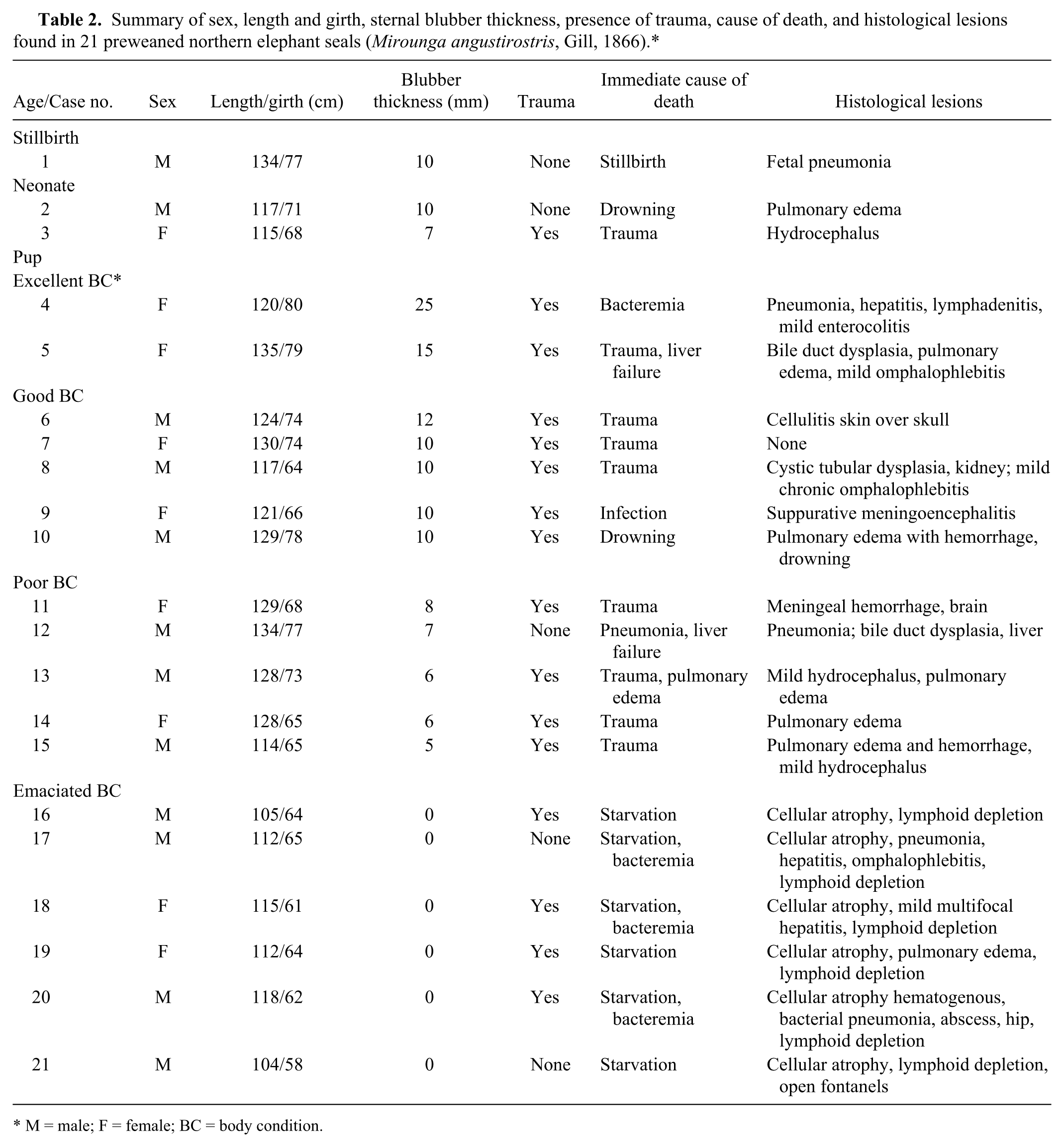
Twenty-one preweaned northern elephant seals (9 females and 12 males) were collected and necropsied. Estimated ages were from birth to approximately 2 weeks of age and included 1 stillbirth, 2 neonates, and 18 pups. Body conditions: excellent (2 pups), good (7), poor (6), and emaciated (6; Table 1, Fig. 1). Dead weaned pups (weaned at approximately 28 days of age), juvenile, subadult, or adult northern elephant seals were not found during the study period. The gross and histological lesions found in these 21 preweaned northern elephant seals have been presented in tabular form (Table 2).
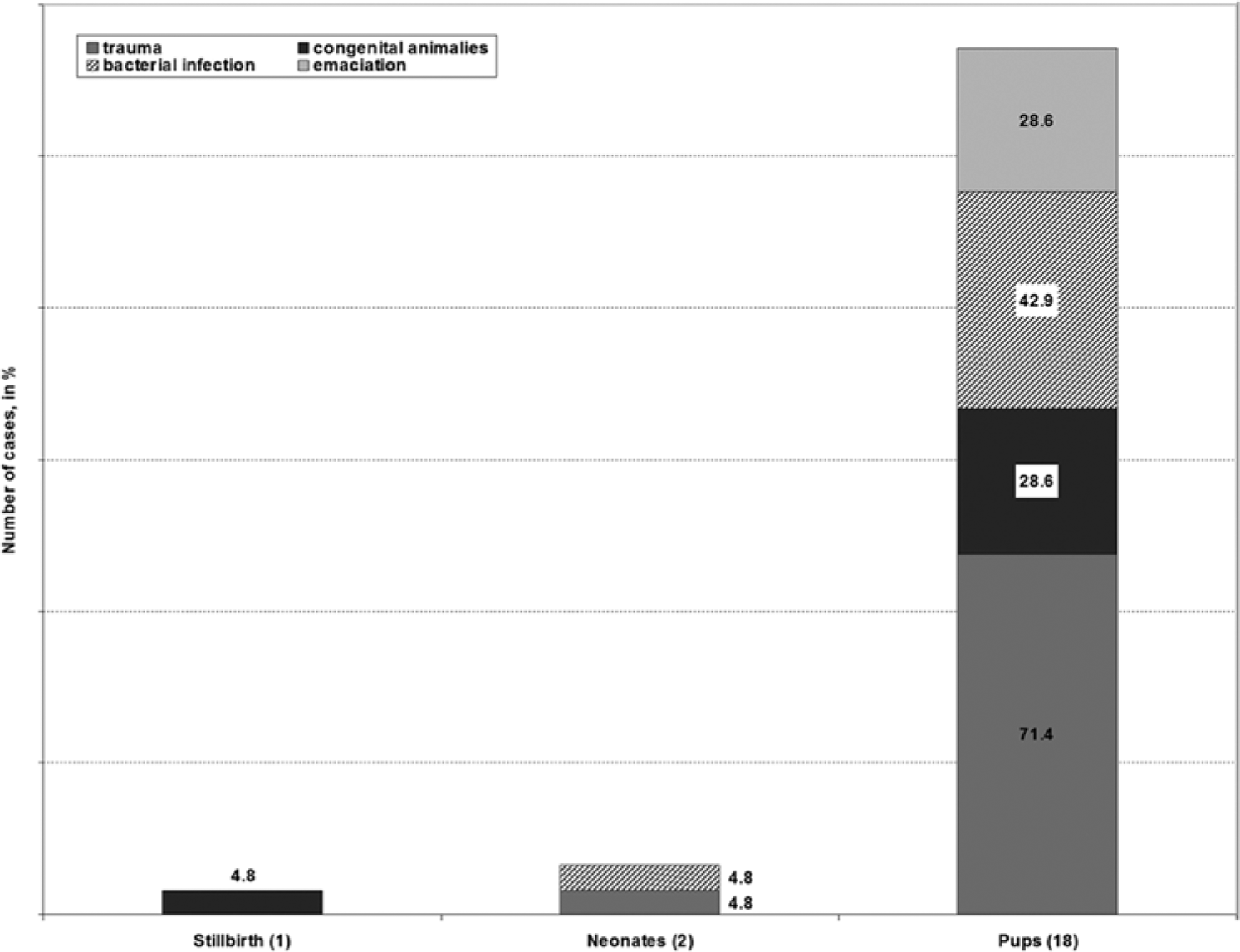
Categories of death found in preweaned northern elephant seal pups (Mirounga angustirostris, Gill, 1866).
Summary of sex, length and girth, sternal blubber thickness, presence of trauma, cause of death, and histological lesions found in 21 preweaned northern elephant seals (Mirounga angustirostris, Gill, 1866).*
M = male; F = female; BC = body condition.
Veterinary literature describing causes of death in juvenile northern elephant seals is available1,7,22; however, there is a paucity of information on the causes of death in preweaned northern elephant seal pups. A previous study 10 reported that mortality of preweaned northern elephant seal pups varied from 10% to 40% on crowded rookeries. 10 The primary causes of death were attributed to trauma, related to trampling and crushing by males during their agonistic activities, and starvation was reported to be due to prolonged maternal separation, usually due to storms washing pups away, aggression by other females, or male interference.9,17 A previous study 6 investigated a small population of southern elephant seals (Mirounga leonina) inhabiting the Falkland Islands and reported that mortality of preweaned pups was 1.74% in 1995 and 1.54% in 1996. The primary cause of death was attributed to stillbirths (58% in 1995; 60% in 1996). However, the diagnoses stated above were made by visual inspections of the dead pups, and comprehensive necropsies were not performed.
The breeding and pupping season for northern elephant seals usually spans from early January to early March; therefore, the present report provides insights into a subset of the possible causes of mortality in preweaned northern elephant seal pups. However, finding only 21 pups of approximately 2,100 pups born during this 3-week period in 2012 suggested that overall mortality was relatively low as compared to other reports, 21 but was comparable to previous reports with southern elephant seals on the Falkland Islands. 6
The primary categories for causes of death found in the northern elephant seal pups in the current study included trauma, perinatal mortality (stillbirths and neonatal mortality), congenital anomalies, starvation, and infections (Table 2). In most cases, multiple disease processes were identified in individual animals. The causes of death found in the present study were similar to those documented in northern fur seal pups (Callorhinus ursinus). 20 For consistency and in an attempt to standardize mortalities between these 2 species, postmortem findings will be discussed according to age and nutritional status.
The single most common lesion observed in pups was blunt force trauma, which was identified in 16 of the 21 (76%) pups from all age classes and body condition status. Of these 16 pups, trauma was the immediate cause of death in 9, but it was considered the primary cause of death in only 6. However, of those 6 pups, 3 also had mild hydrocephalus. There was only a single fatal bite wound to the head in 1 pup. For comparison, in northern fur seal pup mortality, trauma was the primary cause of death in 18% of 2,735 pups examined. 20 It was concluded that any condition that compromised mobility or activity predisposed young pups to trauma especially to the head and abdomen. Charging bulls sometimes reaching 1,700 kg, or aggressive females, were prime contributors to this type of injury. Therefore, findings from the current study indicate that while trauma was common in preweaned pups, physical injury was not the primary cause of death in many cases.
Perinatal mortality included stillbirths and animals dying within the first day of life. One stillbirth and 2 neonates were assigned to this category (Table 1). Stillbirths in pinnipeds are not uncommon. In a 20-year survey of northern fur seal pups, 9.8% were stillborn. 20 The reported percentage of southern elephant seal stillbirths varied from 58% to 60% between 1995 and 1996 on the Falkland Islands, 6 in contrast to the 4.7% rate found in the present study. However, the incidence of stillbirths on any northern elephant seal rookery has not yet been well defined or investigated. The single stillbirth found in the present study did have fetal pneumonia, and hemolytic Escherichia coli was isolated from the lung and liver (Fig. 2).
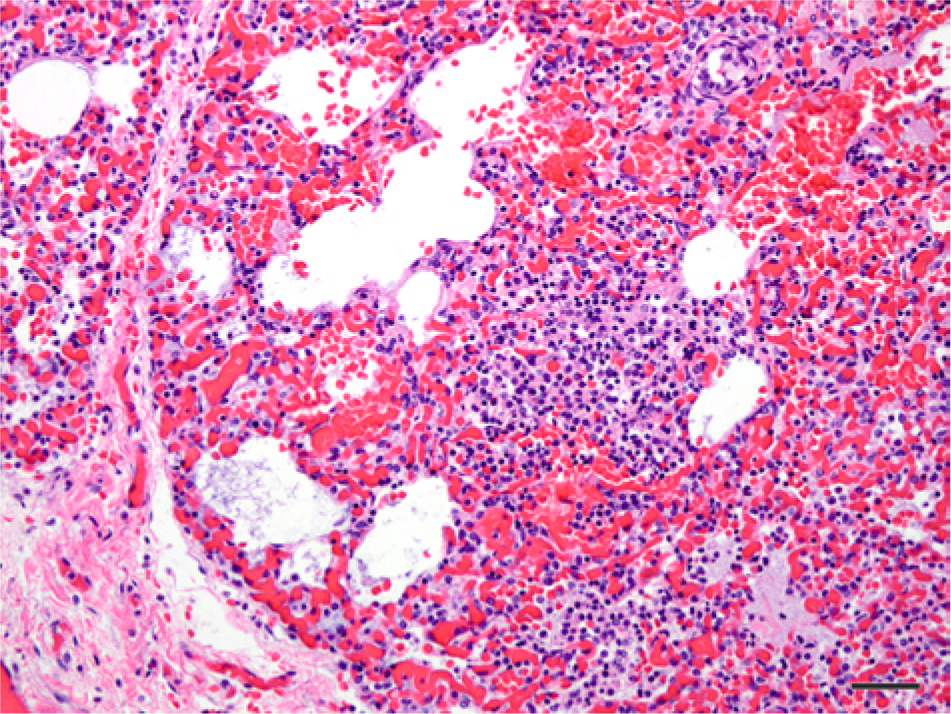
Photomicrograph of the lung of a stillborn northern elephant seal pup (Mirounga angustirostris, Gill, 1866; case 1) that had histological lesions characterized by multifocal, patchy areas of interstitial thickening of the alveolar walls; alveolar spaces partially filled with activated macrophages, neutrophils, edema, and fibrin. Hematoxylin and eosin. Bar = 50 µm.
Neonatal mortality is common for all species of pinnipeds16,20; however, an estimated percentage of neonatal deaths have yet to be reported for northern elephant seals. Two animals (9.5%) were considered to be neonatal deaths in the current study, comparable to 7.2% findings in northern fur seals. 20 Several disease processes were identified in these 2 neonates (Table 2). One neonatal pup (good body condition) with trauma to the head was recovered in the surf after a night of high tide and waves. Gross and microscopic findings were consistent with drowning as the immediate cause of death. Drowning of preweaned pups, especially neonates, has been previously observed in northern elephant seal pups by one of the authors (TR Spraker), who witnessed the drowning of several hundred northern elephant seal pups during a single storm on a high tide on San Miguel Island, Channel Islands, California, in January 2000. Therefore, finding 1 neonatal pup that had drowned during a high tide was not unusual. 16 Blunt force trauma to the chest and abdomen was the cause of death in the other neonate. The rookeries are extremely crowded, and it is reported that female elephant seals are not attentive mothers and trauma caused by territorial males and aggressive females is common. 9
Infections of bacterial etiology were documented in 9 (50%) of 18 pups and included pneumonias (Fig. 3) in 4 pups (22.2%), mild chronic omphalophlebitis in 3 pups (16.7%), mild multifocal necrotizing hepatitis in 3 pups (16.7%), acute suppurative meningoencephalitis in 1 pup (5.6%), enteritis in 1 pup (5.6%), multifocal necrotizing lymphadenitis in 1 pup (5.6%), and cellulitis over the top of the skull in 1 pup (5.6%). Several of these pups had multiple locations of infections suggesting bacteremia and multiple internal organ infections. Organisms isolated from these pups included E. coli, Streptococcus equi, Streptococcus sp., and Enterococcus sp., which are commonly recovered from marine mammals. 5 Influenza viruses, Coxiella sp., and Brucella sp. were searched for, but not detected. Bacterial infections were considered as the primary cause of death in only 4 pups, but evidence of bacterial infections were present to varying degrees in the other 5. The percentage of bacterial infections in preweaned northern elephant seals (50%) was much higher than those in northern fur seals (3%). 20 Nine of these northern elephant seals also had evidence of trauma.
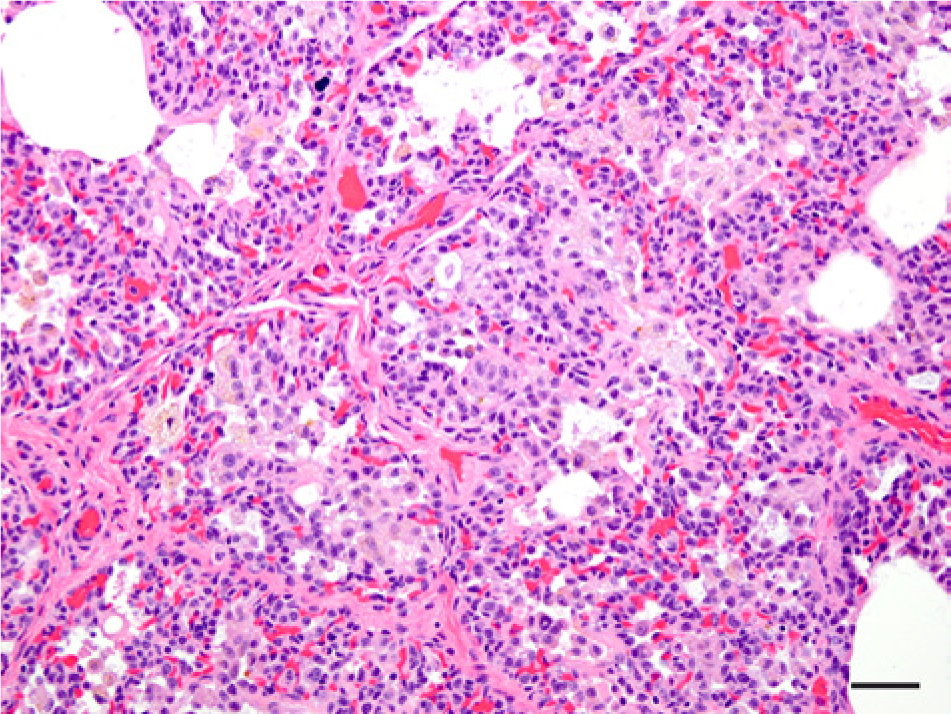
Photomicrograph of the lungs of a northern elephant seal pup (Mirounga angustirostris, Gill, 1866; case 4) with an interstitial pneumonia. Many of the alveolar macrophages contain an unidentified brown-yellow granular pigment. Hematoxylin and eosin. Bar = 50 µm.
Although congenital anomalies are commonly reported in domestic animals, 23 they are rarely reported in free-ranging marine mammals. An exception is in northern fur seals where 49 of 2,735 (1.8%) pups featured 33 different congenital anomalies involving 8 anatomical systems. 20 A previous study 11 published a list of 145 congenital anomalies from wild and zoological mammals, with only 7 marine mammals. 11 Congenital anomalies (n = 17) have been documented in juvenile elephant seals.2,3,13,24 One study 24 reported 12 cases (5.7%) of congenital anomalies from 210 juvenile northern elephant seals stranded along the central California coast from January 1, 1988 to December 31, 1995. Of the 12 cases, 7 individuals had mild-to-moderate hydrocephalus, 2 had severe cardiac anomalies, and single cases of hydronephrosis, focal pulmonary dysplasia, and congenital epidermal angiomatosis were identified. Common intercurrent disease processes found included verminous pneumonia and arteritis, verminous enteritis and colitis, and splenic and hepatic hemosiderosis. More severe anomalies resulted in debilitation, stranding, and death, whereas, milder malformations were found incidentally during routine gross necropsy and histology. 24
In the current study, 4 types of congenital anomalies were found in 7 (33.3%) of the 21 northern elephant seal pups examined including mild hydrocephalus in 3 pups, 2 with extensive and severe bile duct proliferation and dysplasia with periportal fibrosis (Fig. 4), 1 with cystic renal tubular dysplasia (Fig. 5), and 1 pup had open fontanels with osteopenia of the skull (Fig. 6). The immediate causes of death in these pups with congenital anomalies were either trauma (5 pups, 23.8%), trauma with secondary infection (1 pup, 4.8%), or starvation (1 pup, 4.8%); however, the congenital anomalies would probably eventually have been the cause of death in these pups.
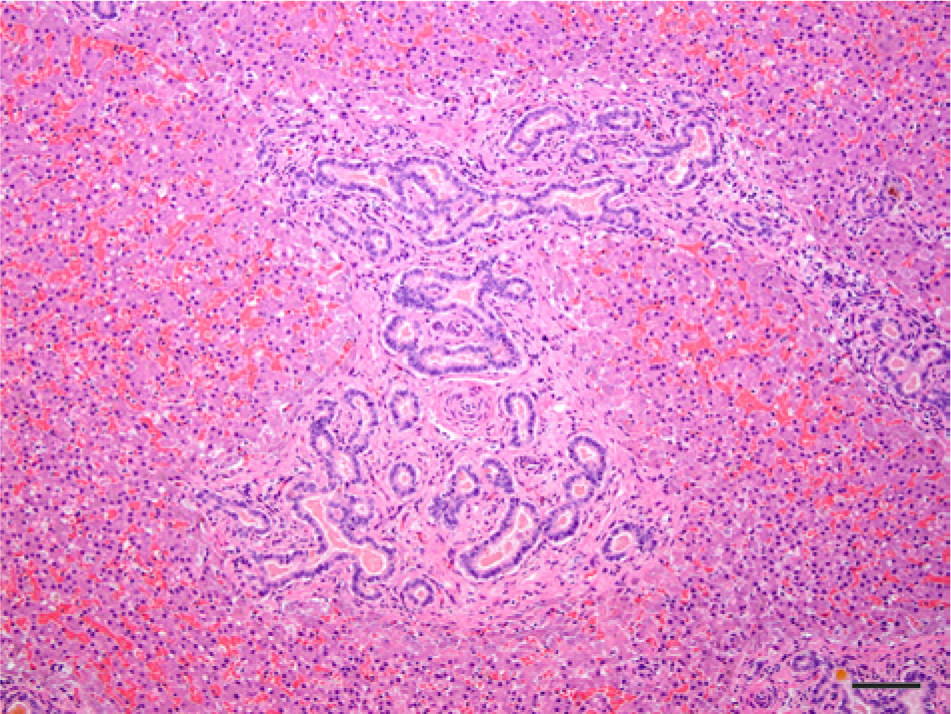
Photomicrograph of the liver of a northern elephant seal pup (Mirounga angustirostris, Gill, 1866; case no. 12) with lesions characterized by discrete multifocal regions of bile duct proliferation with the formation of new, irregular, and tortuous channels in the portal and periportal areas associated with extensive periductal fibrosis. Hematoxylin and eosin. Bar = 100 µm.
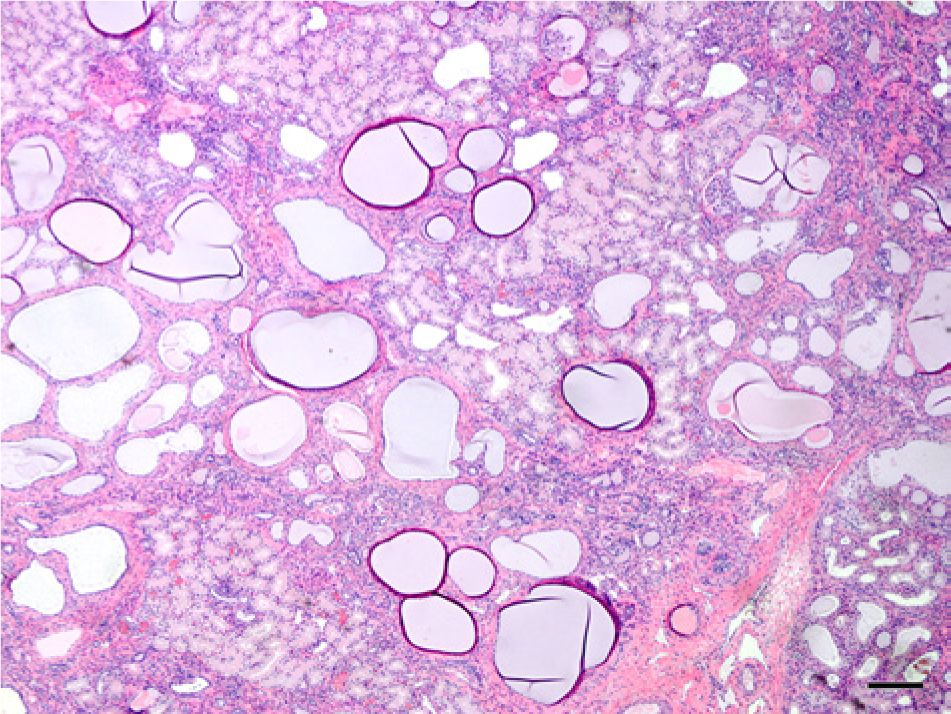
Photomicrograph of the cortex of the kidneys of a northern elephant seal pup (Mirounga angustirostris, Gill, 1866; case no. 8) with bilateral cystic renal cortical dysplasia characterized by numerous ectatic convoluted tubules, and a few dilated and cystic glomerular tufts. There were areas of patchy immature mesenchyme and a small degree of interstitial fibrosis associated with these ectatic convoluted tubules. Collecting tubules were mildly dilated. Hematoxylin and eosin. Bar = 200 µm.
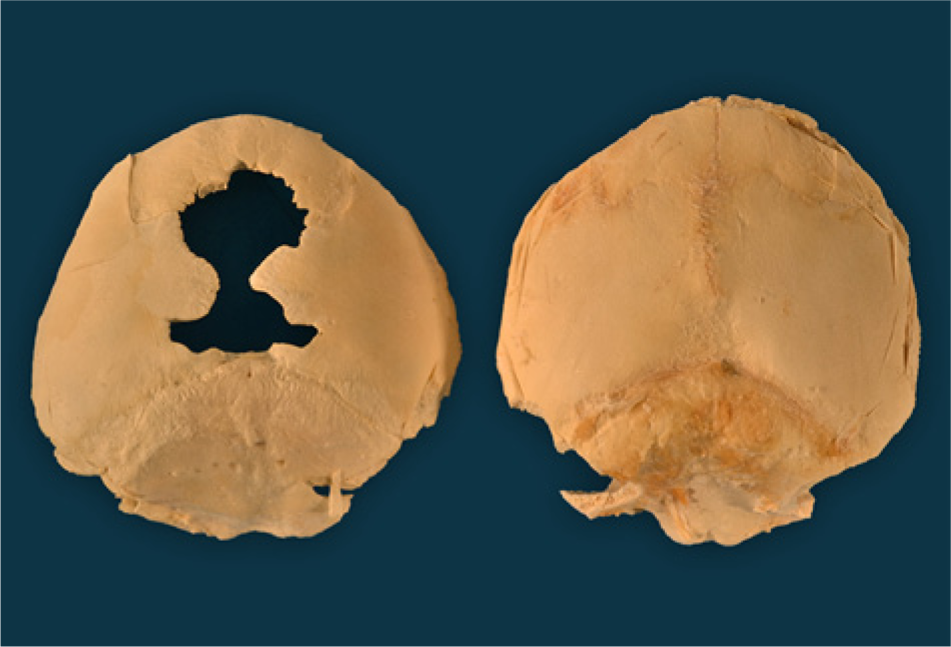
Dorsal view of the skulls from 2 northern elephant seal pups (Mirounga angustirostris, Gill, 1866) both approximately 1 week old. The one on the right is normal and the one on the left is osteopenic (thin and light in weight) and has widely open fontanels.
The percentage of congenital anomalies observed in the present study (33.3%) was extremely high as compared to the northern fur seal study where the percentage was 1.8% and the general average rate of 1.0% was reported.11,20 A reason for the elevated documentation of congenital anomalies could be attributed to the low genetic diversity documented in northern elephant seals.2,8,23
An unusual and interesting observation made by the authors in the present study was that California gulls (Larus californicus) and common ravens (Corvus corax) were often observed attacking or scavenging weak or small, recently born pups and would not only peck them in the eyes, which is extremely common, but would also strike them on the top of the skull, corresponding to the location of the open fontanels found in one of the pups.
Liver copper and mercury levels also were analyzed in 10 of the 21 animals to evaluate if these trace elements could be associated with any of the conditions found in these animals. Copper levels ranged from 17.5 to 209.0 ppm dry weight (DW; average = 89.8 ppm, median = 95.5 ppm) and mercury ranged from 0.75 to 2.05 ppm wet weight (WW; average = 1.12 ppm, median = 0.65 ppm). Normal levels of these elements in preweaned northern elephant seals were not found in the literature; however, levels in harbor seals (Phoca vitulina) have been reported (copper—liver: 16.0–100 ppm DW, kidney: 12.0–50 ppm DW; mercury—liver: <0.1–15.0 ppm WW, kidney: <0.1–2.9 ppm WW). 15 Therefore, these liver mercury and copper levels were considered to be within normal limits of other phocid seals in the same area. Specific lesions that could possibly be associated with these elements were not found in any of the tissues examined.
A confounding observation from the current study was that even with a proportionately higher incidence of congenital malformations, infections, and more restricted genetic diversity, the population of elephant seals is increasing; whereas, the northern fur seals, which feature a much lower rate of congenital anomalies and infections with a good genetic diversity, have been in decline for the past 40 years. 4 One possible explanation is that the northern elephant seals might have an adequate food supply because they forage at nearly half to a mile deep over an extremely large region (the North Pacific) and therefore they may not be nutritionally stressed. 18
Footnotes
Acknowledgements
This project was conducted under the University of California, Santa Cruz (UCSC), California NMFS permit 14636 and SSU IACUC protocol 2011. Appreciation is expressed for the assistance in collection of pups: Stephen Tavoni, Jeff Sharick, Sarah Peterson, Erin Pickett, John Harley, and many others at UCSC and SSU. Thanks are given to the rangers and docents at Año Nuevo State Reserve and to Dr. Daniel Costa for the use of his laboratory at the Long Marine Laboratory, Santa Cruz, California. The authors would like to thank Christina Weller and Kristy Pabilonia for the PCR for Influenza A virus, marine Coxiella and Brucella, and for the inoculation of lung tissues into embryonic chicken eggs for virus isolation for influenza viruses. The authors would like to thank Denise Bolte for the aerobic cultures; Michael Russell for mycoplasma cultures; David Hicks for mycoplasma PCR; Cathy Bedwell for the mercury and copper tissue level determinations; and Dr. Elise LaDouceur for aiding in the literature review for diseases of northern elephant seals.
a.
Tissue-Tek VIP 6, Sakura Finetek USA Inc., Torrance, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received private financial support for the research. Publication of this article was supported by Diagnostic Laboratory of the Colorado State University, Fort Collins, Colorado.
