Abstract
A total of 5 psittacine birds in an enclosed zoological exhibit, including 2 princess parrots and 3 cockatoos of 2 different species, developed severe central nervous system clinical signs over a 2–3-month period and died or were euthanized. Histologically, all birds had a lymphoplasmacytic and histiocytic encephalitis with intralesional protozoa consistent with a Sarcocystis species in addition to intramuscular tissue sarcocysts. By immunohistochemical staining, merozoites in brain and tissue cysts in muscle did not react with polyclonal antisera against Sarcocystis falcatula, Sarcocystis neurona, Toxoplasma gondii, and Neospora caninum, or with a monoclonal antibody to S. neurona. Transmission electron microscopy on sarcocyst tissue cyst walls from 2 birds was morphologically consistent with Sarcocystis calchasi. Polymerase chain reaction (PCR) amplification and sequencing of partial 18S ribosomal RNA from muscle tissue cysts and brain schizonts from 3 birds was consistent with a clade containing S. calchasi and Sarcocystis columbae but could not distinguish these closely related Sarcocystis species. However, PCR amplification and sequencing of the internal transcribed spacer 1 RNA segment in the brain from 2 birds and muscle from 2 birds specifically identified the isolates as S. calchasi. The current report documents that multiple psittacine species are susceptible intermediate hosts of S. calchasi, and that infection can cause encephalitis resulting in significant morbidity and mortality in psittacine aviaries.
Keywords
There are isolated case reports of unidentified Sarcocystis species causing encephalitis in avian species,5,9,25,27 but until recently Sarcocystis falcatula has been the only known species to cause clinical encephalitis and/or pneumonia in numerous avian species, including psittacines.20,21,30 In 2010, Sarcocystis calchasi has also been identified as a cause of significant encephalitis in pigeons (order Columbiformes) in both Europe and North America.15–17,29,31 The closely related species Sarcocystis columbae also infects pigeons but there are no reports of clinically significant disease with S. columbae. The current report documents that psittacine birds (order Psittaciformes) are also susceptible intermediate hosts forS. calchasi and that infection can result in an outbreak of clinically significant encephalitis in a large group of mixed psittacine species.
Between April and June 2012, in a single exhibit aviary in California, 5 psittacine birds, including 2 princess parrots (or Alexandra parrots; Polytelis alexandrae, birds 1 and 5), a long-billed corella (Cacatua tenuirostris, bird 2), and 2 rose-breasted cockatoos (or galahs; Eolophus roseicapilla, birds 3 and 4), were necropsied with a history of either being found dead or humanely euthanized due to severe clinical neurological signs. When the disease outbreak began, the exhibit housed a total of 25 psittacines and 21 pigeons, including 4 long-billed corellas, 11 rose-breasted cockatoos, 3 greater sulfur-crested cockatoos (Cacatua galerita), a lesser sulfur-crested cockatoo (Cacatua sulphurea), 2 red-rumped parrots (Psephotus haematonotus), 3 princess parrots, a palm cockatoo (Probosciger aterrimus), 15 Australian pied imperial pigeons (Ducula bicolor), 4 Nicobar pigeons (Caloenas nicobarica), and 2 Victoria crowned pigeon (Goura victoria). The entire exhibit, including the roof, was enclosed in 12.7 mm × 25.4 mm wire mesh. The palm cockatoo was moved to a distant holding area shortly after the first bird showed clinical signs.
Clinically, the 5 affected psittacines developed central nervous system (CNS) signs that progressed rapidly (from approximately a week up to 11 days) and included torticollis, nystagmus, star-gazing, ataxia, and inability to stand, with accompanying anorexia prior to death or humane euthanasia. In contrast to the 5 affected psittacines, only 1 Australian pied imperial pigeon developed CNS signs during the same time period. Signs in this pigeon included a 6-week history of progressive neurologic disease with weakness, anorexia, decreased conscious proprioception of both pelvic limbs, and inability to fly, right itself, or walk. The pigeon survived, following several weeks of supportive care and treatment that included trimethoprim–sulfa and meloxicam, eventually regaining the ability to eat and partially ambulate, but remains neurologically compromised. Of the affected psittacines, the long-billed corella (bird 2) received a similar empirical treatment (meloxicam, trimethoprim–sulfa, and lactated ringers) but failed to improve and was euthanized after only 36 hr following onset of suspected aspiration pneumonia. In addition to these birds with CNS disease, 3 additional psittacines at the aviary died during this time period either of unrelated disease or were not necropsied.
Formalin-fixed sections of brain, kidney, lung, adrenal gland, intestines, pancreas, proventriculus, and ventriculus from bird 2 were submitted to the Zoo/Exotic Pathology Service (Sacramento, California); the bird was subsequently diagnosed with a type of protozoal encephalitis. The paraffin-embedded brain was subsequently submitted to the California Animal Health and Food Safety Laboratory System (CAHFS; Davis, California) for protozoal immunohistochemical evaluation. The 4 remaining affected psittacines that died or were euthanized (birds 1, 3–5) were submitted to the CAHFS laboratory in Tulare, California. At necropsy, these 4 psittacines were in fair to good body condition. No additional gross pathologic findings were noted. Multiple tissues, including liver, kidney, heart, lung, spleen, gonad, skin, brain, spinal cord, skeletal muscle, pancreas, multiple sections of gastrointestinal tract, adrenal gland, bone, bone marrow, thyroid, and sinus or conjunctival mucosa were collected from these birds, immersed in 10% buffered formalin, and routinely processed for histologic examination.
Microscopically, birds 1–5 all had a lymphoplasmacytic and histiocytic encephalitis that varied in extent, with single or multiple foci characterized by perivascular infiltrates of mixed macrophages, lymphocytes, and plasma cells, with variable accompanying parenchymal gliosis, edema, infrequent axon spheroid formation, rare individual cell necrosis, and variable hypertrophy of small vessels (see Table 1). These inflammatory foci were present randomly in brain stem, cerebellum, and cerebrum but were more prominent and consistently found in caudal brain stem and cerebellum. Immature to mature protozoal schizonts, the latter composed of clusters of small elliptical-shaped zoites approximately 4–6 µm in length, occasionally arranged in circular rosette-like patterns around central residual bodies consistent with a Sarcocystis species, were found within these foci (Fig. 1A, 1B).
Summarized findings in each bird, including optic microscopy, transmission electron microscopy, immunohistochemical staining, and polymerase chain reaction products.*
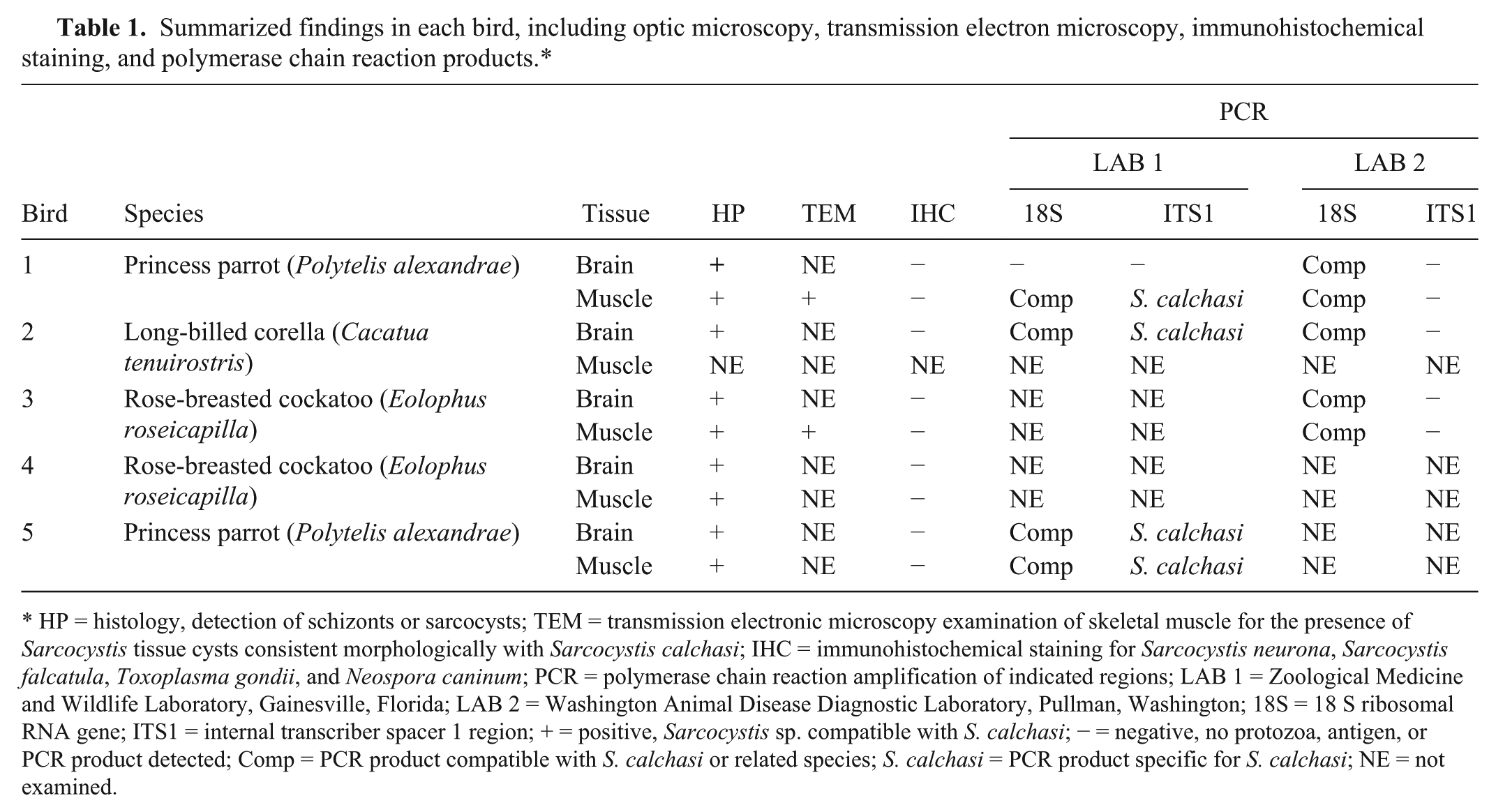
HP = histology, detection of schizonts or sarcocysts; TEM = transmission electronic microscopy examination of skeletal muscle for the presence of Sarcocystis tissue cysts consistent morphologically with Sarcocystis calchasi; IHC = immunohistochemical staining for Sarcocystis neurona, Sarcocystis falcatula, Toxoplasma gondii, and Neospora caninum; PCR = polymerase chain reaction amplification of indicated regions; LAB 1 = Zoological Medicine and Wildlife Laboratory, Gainesville, Florida; LAB 2 = Washington Animal Disease Diagnostic Laboratory, Pullman, Washington; 18S = 18 S ribosomal RNA gene; ITS1 = internal transcriber spacer 1 region; + = positive, Sarcocystis sp. compatible with S. calchasi; − = negative, no protozoa, antigen, or PCR product detected; Comp = PCR product compatible with S. calchasi or related species; S. calchasi = PCR product specific for S. calchasi; NE = not examined.
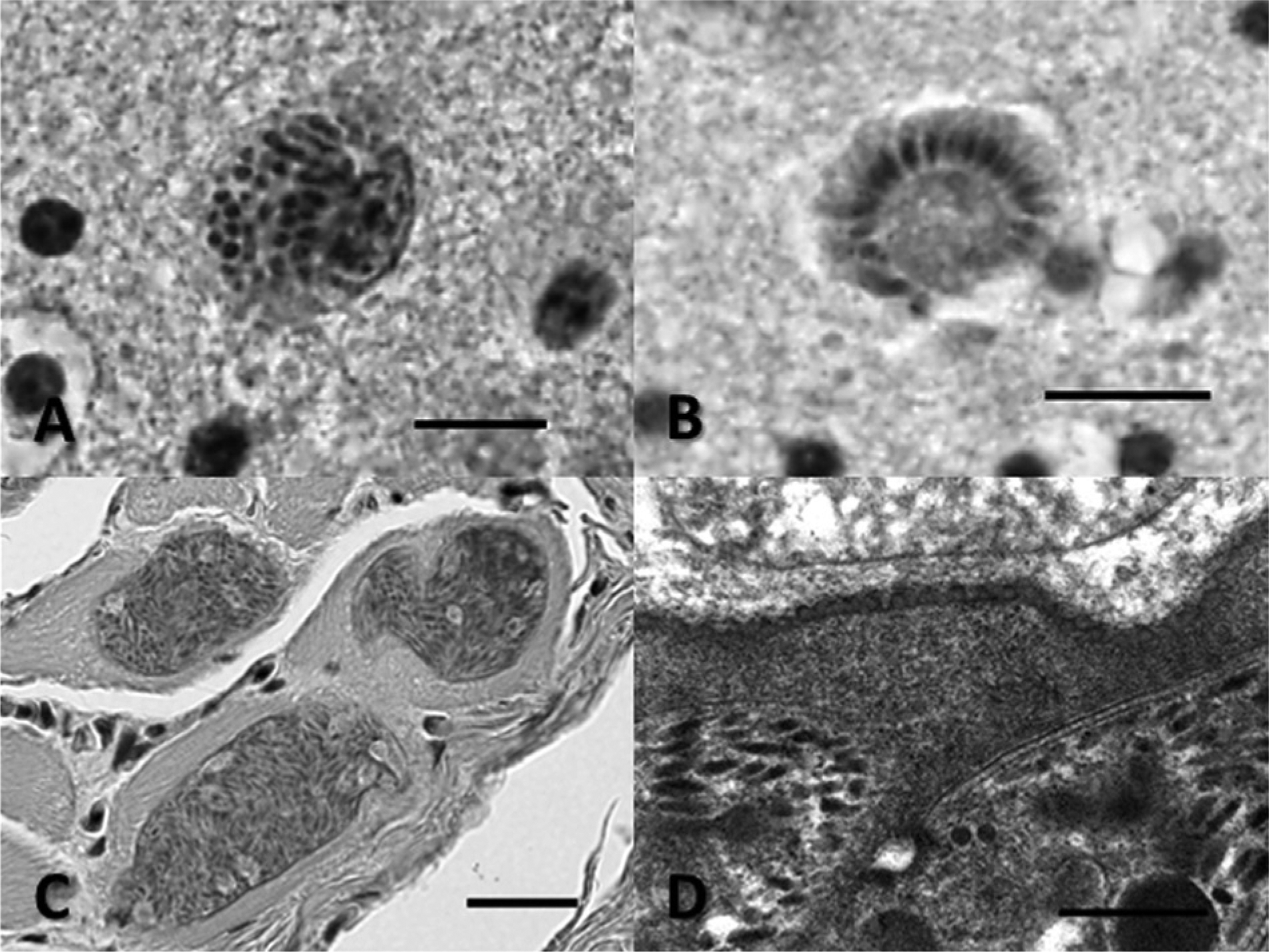
Additionally, in the skeletal muscle of all of the birds where skeletal muscle was available (birds 1, 3–5), there were similar appearing elongate tissue cysts present within myofibers that ranged approximately 10–22 µm in width and up to several hundred micrometers in length (single cysts measuring 265–476 µm in length were noted in birds 1 and 3). Tissue cyst walls were thin, less than 1 µm in width, with an apparently smooth outer surface. Tissue cyst septation was difficult to see but could be detected in some instances. The cysts contained closely packed bradyzoites and, occasionally, there were also isolated larger pale staining zoites, consistent with metrocytes, seen along the periphery or near presumed septa (Fig. 1C; Table 1). No inflammatory reaction associated with the cysts was detected in skeletal muscle. No tissue cysts were detected in the hearts of any birds. Additional histological findings from these birds included mild focal lymphocytic infiltrates in the kidneys of birds 3 and 4, mild lymphocytic infiltrates in the adrenal gland from bird 3, and an acute foreign material aspiration pneumonia in bird 2. No cysts were noted in surgically obtained pectoral muscle biopsies from the surviving Australian pied imperial pigeon.
Formalin-fixed skeletal muscle from birds 1 and 3 were processed for transmission electron microscopy (TEM) as previously described. 28 By TEM, protozoal tissue cysts from both birds were compatible with a Sarcocystis species and had a similar appearance. The tissue cysts were septate. Bradyzoites had features typical of the phylum Apicomplexa, with numerous anterior micronemes, an apical complex, small numbers of anterior rhoptries, and rare micropores. Bradyzoites were roughly 1.1–1.5 µm in width. There were no bradyzoite profiles seen that extended through the entire long axis of the organisms; the single longest bradyzoite profile measured 5.5 µm in length. The cyst walls were thin, approximately 0.3–0.6 µm in width. The outer portion of the cyst walls appeared smooth at low magnification but higher magnification revealed numerous small circular to ovoid protrusions along the outer cyst wall that were approximately 53–70 nm in diameter. The morphology of the cyst walls (Fig. 1D; Table 1) is identical to descriptions of the cyst wall ultrastructure described for both S. calchasi and S. columbae.14,18 Immunohistochemistry using polyclonal antisera raised in rabbits against S. falcatula, S. neurona, Toxoplasma gondii, and Neospora caninum, as well as a monoclonal antibody (2G5) against S. neurona was run on muscle containing tissue cysts from birds 1, 3, 4 and 5 and protozoal schizonts in brain sections from all birds (1–5) as previously described. 30 Neither the schizonts, in brain or the tissue cyst walls, nor the bradyzoites from tissue cysts in skeletal muscle reacted (bound) to any of these antibodies (see Table 1).
Unfixed frozen brain and muscle from birds 2 and 5, respectively, as well as 25-µm paraffin scrolls containing isolated sections of brain and muscle from bird 1 and brain from bird 5, were submitted to the Zoological Medicine Laboratory at the University of Florida (UF; Gainesville, Florida) for examination by polymerase chain reaction (PCR) and sequencing.
For the molecular work, DNA was extracted using a commercial kit. a In the brain of bird 2, initial pan-coccidial PCR methods previously described 8 were used that targeted an approximately 350–base pair (bp) segment of the small subunit ribosomal RNA (rRNA) gene. The PCR products were resolved in 1.5% agarose gels. The band was excised and purified using a commercial gel extraction kit. b Direct sequencing was performed using a commercial kit c and analyzed on an automated DNA sequencer. d All products were sequenced in both directions. Primer sequences were edited out prior to further analyses. The resultant 348-bp sequence clearly identified a Sarcocystis species but could not reliably distinguish members of a clade including S. calchasi, S. columbae, S. turdusi, and others. Hence, a differentSarcocystis-specific protocol targeting the internal transcribed spacer 1 (ITS1) region 12 was used on all tissues submitted to UF. Products were processed and sequenced as above. The brains of birds 2 and 5 and the muscles of birds 1 and 5, all resulted in identical 997-bp sequences. A BLASTN search 1 found that this sequence was 99% homologous to the reference S. calchasi sequence (GenBank accession number FJ232948), 93% homologous to Sarcocystis wobeseri (GU475112), and 83% homologous to S. columbae (GU253885). Sequences were submitted to GenBank under accession numbers KC733714–KC733718.
The predicted homologous ITS1 nucleotide sequences of related Sarcocystis species were aligned as previously described. 10 Bayesian analyses of each alignment were performed as described 19 on a public server (Miller MA, Pfeiffer W, Schwartz T: 2010, Creating the CIPRES Science Gateway for interference of large phylogenetic trees. In: Proceedings of the Gateway Computing Environments Workshop, pp. 1–8, November 14, 2010, New Orleans, LA) with gamma distributed rate variation and a proportion of invariant sites, and a general time reversible model. Sarcocystis felis was used as the outgroup. Four chains were run, and statistical convergence was assessed by looking at the standard deviation of split frequencies as well as potential scale reduction factors of parameters. The first 10% of 1,000,000 iterations were discarded as a burn in, based on examination of trends of the log probability against generation. Two independent analyses were run to avoid entrapment on local optima.
Maximum likelihood analysis of the alignment was performed as described 22 with gamma distributed rate variation and a proportion of invariant sites, and a general time reversible model. Again, S. felis was used as the outgroup. To test the strength of the tree topology, the alignment producing the most likely tree was then used for bootstrap analysis (1,000 resamplings). 7 The Bayesian tree is shown in Figure 2. Bayesian posterior probabilities are displayed at nodes on the left, and maximum likelihood values are to the left. The analysis clearly shows that the Sarcocystis sp. identified in the current report clusters strongly with the reference S. calchasi. The distance between this strain and the reference strain is similar to the intraspecies variation seen in other Sarcocystis spp. shown in Figure 2.
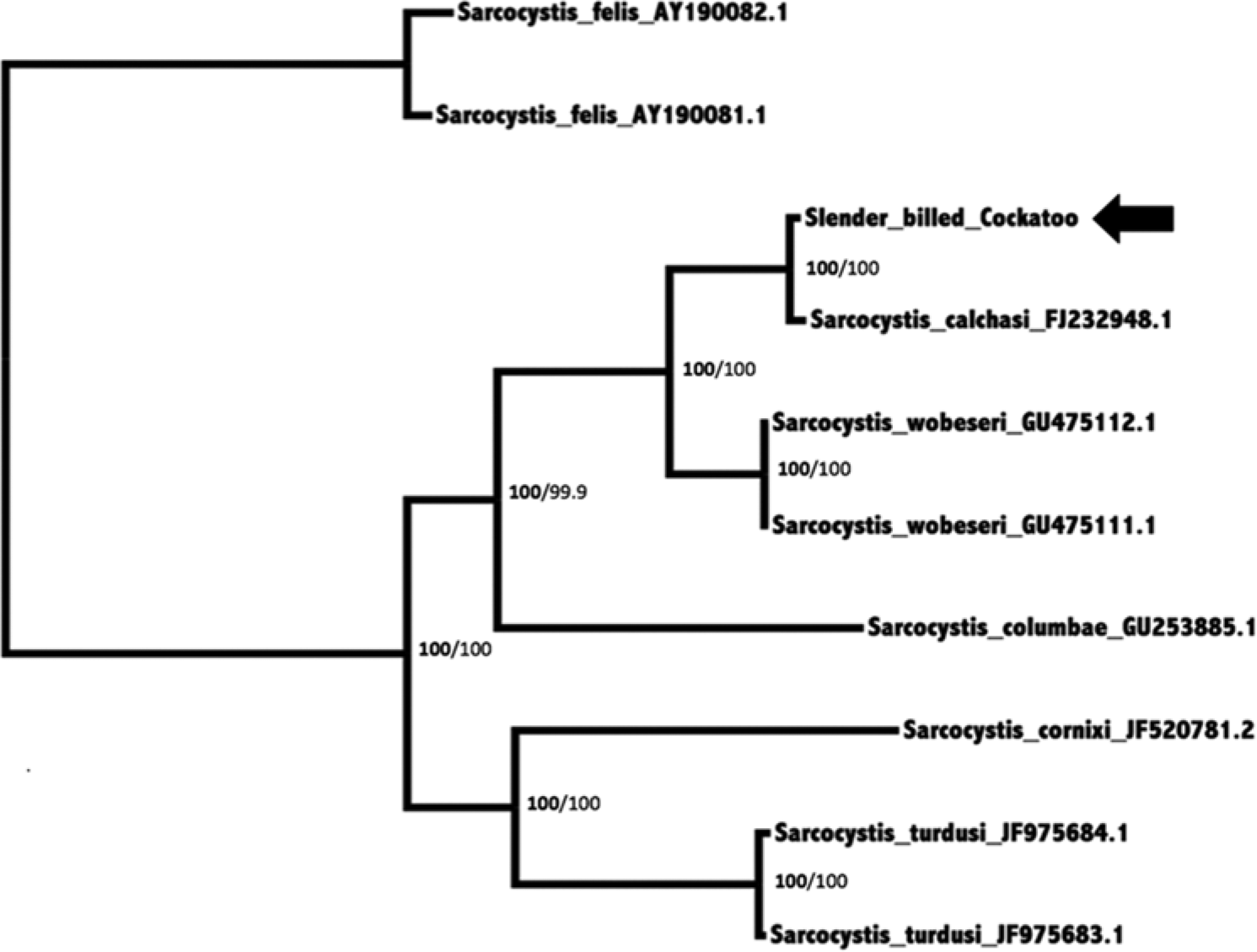
Bayesian phylogenetic tree of MAFFT alignment of homologous 802–876 base pairs of Sarcocystis ribosomal RNA internal transcribed spacer region 1 nucleotide sequences. The outgroup was Sarcocystis felis. Confidence of the tree topology obtained is shown by Bayesian posterior probabilities to the left of the slash in bold and maximum likelihood bootstrap values to the right. GenBank accession numbers are given to the right of the names. The Sarcocystis calchasi sequence obtained in the current study is marked with an arrow.
In addition, formalin-fixed, paraffin-embedded blocks of brain and muscle from birds 1 and 3, as well as brain from bird 2, were submitted to the Washington Animal Disease Diagnostic Laboratory (Pullman, Washington) for PCR detection and sequencing. The DNA was extracted from the tissue blocks, and a portion of the 18S rRNA gene was amplified by PCR and directly sequenced as previously described. 4 The 3 brain blocks and both skeletal muscle blocks yielded amplicons of the expected size and were directly sequenced. Forward and reverse sequences were aligned by a sequence alignment program, 26 and each sequence was compared using BLASTN to the GenBank nucleotide sequence database. Sequences from the 5 blocks were identical and most closely matched that of named species S. columbae andS. calchasi (100% sequence identity [457 bp/457 bp] to GenBank accession number GQ3253883 and GQ245670) when compared with sequences in GenBank. The sequences also matched that of unnamed species previously described 31 from hawks in Georgia (EU810395, EU810396, EU810397, EU810398) and from a great cormorant (unpublished, JQ733511).
Subsequently, the DNA extracted from these 5 blocks was subjected to PCR in an attempt to amplify the ITS1 region of the rRNA gene using a previously described method. 18 This assay was not successful. Results from histological evaluation (optic and electron microscopy), immunohistochemical staining, and PCR results from both laboratories are summarized in Table 1.
The combined findings indicate the presence of S. calchasi infection in all 5 psittacines. The histologic features of the organisms in the brains of all 5 cases and the muscle tissue cysts from the 4 birds where muscle was examined were all compatible with a Sarcocystis species.5,6 The Sarcocystis organisms did not react with antibodies directed against S. neurona, S. falcatula, T. gondii, or N. caninum, which rules these protozoa out as possible causes. The ultrastructure of the tissue cyst walls examined in 2 of the birds was consistent not only with S. calchasi but also with S. columbae. Finally, the ITS1 RNA gene sequence results from parasites in the brain (birds 2 and 5) and muscle (birds 1 and 5) definitively show that this parasite is S. calchasi. 18 In addition, 18S rRNA gene segment sequences from parasites found in the brains of 2 additional birds (birds 1 and 3) and muscle of 1 additional bird (bird 3) were also consistent with, but not specific for, S. calchasi infections. The results confirm S. calchasi infection in these multiple psittacine species and that infection can often result in clinically significant encephalitis and death. The presence of the S. calchasi muscle tissue cysts further indicates these psittacines are true susceptible intermediate hosts for S. calchasi. The current study also underlines the need for examination of genes beyond 18S rRNA for speciation of Sarcocystis sp. The situation with S. calchasi, S. columbae, and related organisms echoes previous findings with S. neurona and S. falcatula, which were synonymized based on 18S rRNA sequences but later shown to be differentiated with other genes.3,24
It should be noted that none of the pigeons sharing this aviary with the affected psittacines succumbed to protozoal encephalitis although 1 pigeon did develop significant persistent CNS signs during this outbreak but remains alive. The precise reason for the relative lack of clinical signs among the cohabitating pigeon population within the aviary is unknown. Possible explanations include a relative lack of susceptibility to either infection or clinical encephalitis among these particular species of pigeons, or possibly a lack of exposure to feed contaminated with sporulated sporocysts. The pigeons within the exhibit tended not to forage on the ground while some of the psittacines, especially the princess parrots and rose-breasted cockatoos, often foraged on the ground, potentially explaining the lack of clinical disease among this particular pigeon population. In addition to the affected psittacine birds within the exhibit, there was also a single wild pigeon that developed severe neurological signs outside the aviary collection; it was captured and euthanized for humane reasons. Necropsy findings in this free-ranging pigeon revealed it had a virtually identical appearing Sarcocystis encephalitis with similar muscle tissue cysts. The protozoa in the brain and muscle of this pigeon also did not react by immunohistochemical staining to any of the polyclonal antisera or the S. neurona monoclonal antibody, and TEM of the muscle tissue cyst walls also appeared identical to S. calchasi. However, PCR to amplify the ITS1 DNA gene segment was negative, so this diagnosis is not definitive. The negative PCR result and the additional negative PCR results on select paraffin-embedded samples may be related to use of formalin-fixed tissues, as formalin damages nucleic acids and can markedly reduce PCR assay sensitivity. 12
The short time sequence during which this disease outbreak occurred suggests that the affected birds were either infected simultaneously, or within a relatively short time period by ingesting sporulated fecal sporocysts shed by the definitive host for S. calchasi, which is reported to be the northern goshawk (Accipiter gentilis). 16 While it is certainly possible that the aviary could have been contaminated by fecal shedding from northern goshawks overflying this exhibit, the location of this aviary in Fresno, California within the California Central Valley is not reported to be within the habitat range for the northern goshawk. 11 Aside from direct fecal shedding by northern goshawks into the exhibit, other potential mechanisms for contamination of the exhibit with sporulated S. calchasi sporocysts include transfer of sporulated sporocysts by insect vectors or from introduction of contaminated soil into the exhibit, both of which have been proposed as alternate means for exposure to S. falcatula.2,13,23 Of these potential alternative modes of transmission, the northern goshawk explanation seems unlikely given the distance from the goshawk’s normal habitat range and the large number of affected birds within the exhibit. Furthermore, no new soil had been added to the exhibit, which rules out soil as a means for transmission.
In the absence of a known means for transmission of fecal sporulated sporocysts from northern goshawks, another related raptor species common within this region should be considered as potential definitive host candidates for S. calchasi. A previous study in Germany detected that a high percentage of free-ranging Eurasian sparrowhawks (Accipiter nisus) were positive for S. calchasi using a PCR assay targeting the ITS1 region. 17 In California, Cooper’s hawks (Accipiter cooperii) and sharp-shinned hawks (Accipiter striatus) should be considered as possible definitive hosts. The finding of a S. columbae/S. calchasi clade in the intestines of a red-tailed hawk (Buteo jamaicensis), a red-shouldered hawk (Buteo lineatus), and 2 Cooper’s hawks from Georgia also raise the possibility that additional raptor species may serve as definitive hosts for S. calchasi. 31 Unfortunately, only 18S rRNA was sequenced in those cases, which is insufficient for differentiation of species in this clade.
Footnotes
Acknowledgements
The authors wish to acknowledge and thank the California Animal Health and Food Safety (Davis) histology and electron microscopy laboratory staff for their excellent technical support.
a.
DNeasy tissue kit, Qiagen Inc., Valencia, CA.
b.
QIAquick gel extraction kit, Qiagen Inc., Valencia, CA.
c.
Big-Dye Terminator Kit, Applied Biosystems, Foster City, CA.
d.
ABI 3130 automated DNA sequencer, Applied Biosystems, Foster City, CA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
